Abstract
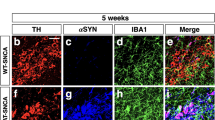
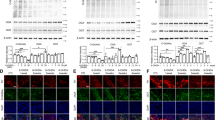
O-GlcNAcylation, a nutrient-sensitive post-translational modification, has emerged as a key regulator of immune and inflammatory processes. However, its role in neuroinflammation and neurodegenerative disease progression remains poorly defined. In this study, we explored how reduced O-GlcNAcylation contributes to neuroinflammatory signaling in Parkinson’s disease (PD), a disorder increasingly recognized to involve dysregulated immune–metabolic interactions. Analysis of postmortem PD substantia nigra (SN) revealed a marked reduction in global O-GlcNAcylation levels, concomitant with enhanced neuroinflammatory signatures and a predominance of pro-inflammatory microglial activation states. In a lipopolysaccharide (LPS)-induced PD mouse model, pharmacological elevation of O-GlcNAcylation through glucosamine (GlcN) or the Thiamet-G significantly ameliorated motor deficits, preserved tyrosine hydroxylase (TH)-positive dopaminergic neurons, and attenuated neuroinflammatory responses, including glial activation and inflammasome assembly. In primary microglial cultures, enhanced O-GlcNAcylation suppressed LPS-induced pro-inflammatory gene expression while promoting anti-inflammatory and homeostatic phenotypes. Mechanistically, increased O-GlcNAcylation dampened NF-κB signaling activity and reduced the production of pro-inflammatory cytokines, thereby reprogramming microglial functional states. Collectively, these findings identify O-GlcNAcylation as a critical modulator of microglial-mediated neuroinflammation and highlight its therapeutic potential for inflammation-associated neurodegenerative disorders such as PD.
Similar content being viewed by others
Data availability
The data and materials used in this research are available upon request from the corresponding author.
References
Poewe, W. et al. Parkinson disease. Nat. Rev. Dis. Prim. 3, 17013 (2017).
Sarrafchi, A., Bahmani, M., Shirzad, H. & Rafieian-Kopaei, M. Oxidative stress and Parkinson’s disease: New hopes in treatment with herbal antioxidants. Curr. Pharm. Des. 22, 238–246 (2016).
Rocha, E. M., De Miranda, B. & Sanders, L. H. Alpha-synuclein: Pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurobiol. Dis. 109, 249–257 (2018).
Gelders, G., Baekelandt, V. & Van der Perren, A. Linking Neuroinflammation and Neurodegeneration in Parkinson’s Disease. J. Immunol. Res. 2018, 4784268 (2018).
Biswas, K. Microglia mediated neuroinflammation in neurodegenerative diseases: A review on the cell signaling pathways involved in microglial activation. J. Neuroimmunol. 383, 578180 (2023).
Hickman, S., Izzy, S., Sen, P., Morsett, L. & El Khoury, J. Microglia in neurodegeneration. Nat. Neurosci. 21, 1359–1369 (2018).
Salter, M. W. & Stevens, B. Microglia emerge as central players in brain disease. Nat. Med. 23, 1018–1027 (2017).
Orihuela, R., McPherson, C. A. & Harry, G. J. Microglial M1/M2 polarization and metabolic states. Br. J. Pharm. 173, 649–665 (2016).
Colonna, M. & Butovsky, O. Microglia Function in the Central Nervous System During Health and Neurodegeneration. Annu. Rev. Immunol. 35, 441–468 (2017).
Tang, Y. & Le, W. Differential Roles of M1 and M2 Microglia in Neurodegenerative Diseases. Mol. Neurobiol. 53, 1181–1194 (2016).
Pajares, M., A, I. R., Manda, G., Bosca, L. & Cuadrado, A. Inflammation in Parkinson’s Disease: Mechanisms and Therapeutic Implications. Cells 9, https://doi.org/10.3390/cells9071687 (2020).
Sharma, N. & Nehru, B. Characterization of the lipopolysaccharide induced model of Parkinson’s disease: Role of oxidative stress and neuroinflammation. Neurochem Int 87, 92–105 (2015).
Liu, M. & Bing, G. Lipopolysaccharide animal models for Parkinson’s disease. Parkinsons Dis. 2011, 327089 (2011).
Fu, S. P. et al. Anti-inflammatory effects of BHBA in both in vivo and in vitro Parkinson’s disease models are mediated by GPR109A-dependent mechanisms. J. Neuroinflamm. 12, 9 (2015).
Deng, I., Corrigan, F., Zhai, G., Zhou, X. F. & Bobrovskaya, L. Lipopolysaccharide animal models of Parkinson’s disease: Recent progress and relevance to clinical disease. Brain Behav. Immun. Health 4, 100060 (2020).
Heidari, A., Yazdanpanah, N. & Rezaei, N. The role of Toll-like receptors and neuroinflammation in Parkinson’s disease. J. Neuroinflamm. 19, 135 (2022).
Yildirim-Balatan, C. et al. Parkinson’s disease-derived alpha-synuclein assemblies combined with chronic-type inflammatory cues promote a neurotoxic microglial phenotype. J. Neuroinflamm. 21, 54 (2024).
Beier, E. E. et al. Alternative microglial activation is associated with cessation of progressive dopamine neuron loss in mice systemically administered lipopolysaccharide. Neurobiol. Dis. 108, 115–127 (2017).
Wang, Q., Liu, Y. & Zhou, J. Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl. Neurodegener. 4, 19 (2015).
Mosley, R. L. et al. Neuroinflammation, oxidative stress and the pathogenesis of Parkinson’s disease. Clin. Neurosci. Res 6, 261–281 (2006).
Yu, H. et al. Metabolic reprogramming and polarization of microglia in Parkinson’s disease: Role of inflammasome and iron. Ageing Res Rev. 90, 102032 (2023).
Zhao, Y. et al. TRAF6 promotes spinal microglial M1 polarization to aggravate neuropathic pain by activating the c-JUN/NF-kB signaling pathway. Cell Biol. Toxicol. 40, 54 (2024).
Liu, Q. et al. Cathepsin C promotes microglia M1 polarization and aggravates neuroinflammation via activation of Ca(2+)-dependent PKC/p38MAPK/NF-kappaB pathway. J. Neuroinflamm. 16, 10 (2019).
Zachara, N. E. & Hart, G. W. Cell signaling, the essential role of O-GlcNAc!. Biochim Biophys. Acta 1761, 599–617 (2006).
Hardiville, S. & Hart, G. W. Nutrient regulation of signaling, transcription, and cell physiology by O-GlcNAcylation. Cell Metab. 20, 208–213 (2014).
Yang, X. & Qian, K. Protein O-GlcNAcylation: emerging mechanisms and functions. Nat. Rev. Mol. Cell Biol. 18, 452–465 (2017).
Liu, Y. et al. O-GlcNAcylation: the “stress and nutrition receptor” in cell stress response. Cell Stress Chaperones 26, 297–309 (2021).
Hart, G. W. Nutrient regulation of signaling and transcription. J. Biol. Chem. 294, 2211–2231 (2019).
Zhang, D. et al. OGT-mediated O-GlcNAcylation promotes NF-kappaB activation and inflammation in acute pancreatitis. Inflamm. Res. 64, 943–952 (2015).
Li, X. et al. O-GlcNAc Transferase Suppresses Inflammation and Necroptosis by Targeting Receptor-Interacting Serine/Threonine-Protein Kinase 3. Immunity 50, 1115 (2019).
Hwang, S. Y. et al. Glucosamine exerts a neuroprotective effect via suppression of inflammation in rat brain ischemia/reperfusion injury. Glia 58, 1881–1892 (2010).
Hwang, J. S. et al. Glucosamine improves survival in a mouse model of sepsis and attenuates sepsis-induced lung injury and inflammation. J. Biol. Chem. 294, 608–622 (2019).
Kim, D. Y., Kim, S. M., Cho, E. J., Kwak, H. B. & Han, I. O. Protective effect of increased O-GlcNAc cycling against 6-OHDA-induced Parkinson’s disease pathology. Cell Death Dis. 15, 287 (2024).
Taetzsch, T. et al. Redox regulation of NF-kappaB p50 and M1 polarization in microglia. Glia 63, 423–440 (2015).
Liu, J. Q. et al. Rg1 improves LPS-induced Parkinsonian symptoms in mice via inhibition of NF-kappaB signaling and modulation of M1/M2 polarization. Acta Pharm. Sin. 41, 523–534 (2020).
Lind-Holm Mogensen, F., Seibler, P., Grunewald, A. & Michelucci, A. Microglial dynamics and neuroinflammation in prodromal and early Parkinson’s disease. J. Neuroinflamm. 22, 136 (2025).
Croisier, E., Moran, L. B., Dexter, D. T., Pearce, R. K. & Graeber, M. B. Microglial inflammation in the Parkinsonian substantia nigra: relationship to alpha-synuclein deposition. J. Neuroinflamm. 2, 14 (2005).
Chen, L., Wang, C., Qin, L. & Zhang, H. Parkinson’s disease and glucose metabolism impairment. Transl. Neurodegener. 14, 10 (2025).
Dai, C. et al. Glucose metabolism impairment in Parkinson’s disease. Brain Res Bull. 199, 110672 (2023).
Zhu, J. H., Kulich, S. M., Oury, T. D. & Chu, C. T. Cytoplasmic aggregates of phosphorylated extracellular signal-regulated protein kinases in Lewy body diseases. Am. J. Pathol. 161, 2087–2098 (2002).
Gordon, R. et al. Inflammasome inhibition prevents alpha-synuclein pathology and dopaminergic neurodegeneration in mice. Sci. Transl. Med. 10, https://doi.org/10.1126/scitranslmed.aah4066 (2018).
Hunot, S. et al. Nuclear translocation of NF-kappaB is increased in dopaminergic neurons of patients with Parkinson disease. Proc. Natl. Acad. Sci. USA 94, 7531–7536 (1997).
Miao, Y. et al. Modulation of O-GlcNAc cycling influences alpha-synuclein amplification, degradation, and associated neuroinflammatory pathology. Mol. Neurodegener. 20, 113 (2025).
Zhu, S., Wang, N., Chen, S., Zou, J. & Tan, S. Thiamet-G ameliorates Parkinson’s disease-associated cognitive impairment via increasing O-GlcNAcylation of STING in the microglia. Neurochem. Int. 188, 106014 (2025).
Cheng, J. et al. Early glycolytic reprogramming controls microglial inflammatory activation. J. Neuroinflamm. 18, 129 (2021).
Wellen, K. E. et al. The hexosamine biosynthetic pathway couples growth factor-induced glutamine uptake to glucose metabolism. Genes Dev. 24, 2784–2799 (2010).
Kelly, B. & O’Neill, L. A. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 25, 771–784 (2015).
Acknowledgements
This work was supported by National Research Foundation (NRF) of Korea Grants (RS-2024-00346770). The brain research resource human brain tissues were provided by Korea Brain Bank Network (Asan Medical Center Brain Bank, Catholic Medical College Catholic Brain Bank, Inje University Brain Bank, and Severance Brain Bank) operated through the National Brain Bank Project (25-BR-09-01) and the Korean Brain cluster promotion project (RS-2021-NR057633) funded by the Ministry of Science and ICT. We would like to express our sincerest gratitude to Sun Ju Chung, Soo Jeong Nam, Joong-Seok Kim, In-Beom Kim, Sang Jin Kim, Eun Joo Chung, Byung Seok Ye, and Ji Woong Oh for providing human brain research resources.
Author information
Authors and Affiliations
Contributions
D.Y.K. designed and conducted the experiments, analyzed the data, prepared the figures, and wrote the main manuscript text. S.M.K. and C.L. contributed to technical assistance. I.O.H. designed the experiments, analyzed the data, and contributed to writing the main manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, D.Y., Kim, SM., Lee, C. et al. O-GlcNAcylation regulates microglial neuroinflammation in Parkinson’s disease. npj Parkinsons Dis. (2026). https://doi.org/10.1038/s41531-026-01319-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-026-01319-6