Abstract
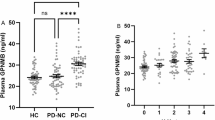
Osteokines, primarily secreted by bone, have been implicated in brain function and Parkinson’s disease (PD) pathogenesis, yet their circulating levels in PD and potential role in the relationship between bone mineral density (BMD) and PD remain unclear. 80 participants (40 PD patients and 40 controls) were enrolled to measure plasma levels of eight osteokines (GPNMB, OPN, SOST, DKK1, RANKL, FGF23, BMP2, and BMP4) and assess their associations with clinical scales. Mendelian randomization (MR), SMR, and colocalization analyses were performed to evaluate causal relationships between osteokines and PD. Restricted cubic spline (RCS) models were applied to explore nonlinear associations between BMD, osteokines, and PD. GPNMB levels were significantly elevated in PD patients and showed a linear association with PD risk. Higher GPNMB levels were associated with worse cognitive performance and clinical severity, while higher SOST levels correlated with milder symptoms. Genetic analyses consistently supported a causal and colocalized relationship between GPNMB and PD. Total coxa BMD and T-score were lower in PD, but not statistically significant. RCS analysis revealed an “n-shaped” association between total coxa T-score and both PD and GPNMB levels. Overall, GPNMB appears causally linked to PD risk and may mediate the bone-brain axis connecting BMD with PD susceptibility.
Similar content being viewed by others
Data availability
The GWAS summary statistics data used in this MR study are publicly available which are included in the additional files. Clinical data are available from the corresponding author upon reasonable request, subject to scientific review and the completion of a material transfer agreement.
References
Morris, H. R., Spillantini, M. G., Sue, C. M. & Williams-Gray, C. H. The pathogenesis of Parkinson’s disease. Lancet 403, 293–304 (2024).
Ortega, R. A. et al. Association of dual LRRK2 G2019S and GBA variations with parkinson disease progression. JAMA Netw. Open 4, e215845 (2021).
Landoulsi, Z. et al. Large-scale copy number variant analysis in genes linked to Parkinson´s disease. npj Parkinsons Dis. 11, 225 (2025).
Fiorini, M. R., Li, J., Fon, E. A., Farhan, S. M. K. & Thomas, R. A. Neural networks reveal novel gene signatures in Parkinson disease from single-nuclei transcriptomes. npj Parkinsons Dis. 11, 304 (2025).
Sharma, K. et al. A Novel PINK1 p.F385S loss-of-function mutation in an indian family with Parkinson’s disease. Mov. Disord. 39, 1217–1225 (2024).
Yang, J., Wu, X. & Song, Y. Recent advances in novel mutation genes of Parkinson’s disease. J. Neurol. 270, 3723–3732 (2023).
Torsney, K. M. et al. Bone health in Parkinson’s disease: a systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 85, 1159–1166 (2014).
Koo, H. Y. et al. Fracture risk in Parkinson’s disease according to its severity and duration. Osteoporos. Int. 34, 81–89 (2023).
Liu, B. et al. Bone mineral density and related scores in Parkinson’s disease: a systematic review and meta-analysis. World Neurosurg. 146, e1202–e1218 (2021).
Park, K. Y. et al. Bone mineral density and the risk of Parkinson’s disease in postmenopausal women. Mov. Disord. 38, 1606–1614 (2023).
Van den Bos, F. et al. Bone mineral density and vitamin D status in Parkinson’s disease patients. J. Neurol. 260, 754–760 (2013).
Kwon, M. J. et al. The occurrence of Alzheimer’s disease and Parkinson’s disease in individuals with osteoporosis: a longitudinal follow-up study using a National Health screening database in Korea. Front Aging Neurosci. 13, 786337 (2021).
Feng, S. H., Huang, Y. P., Yeh, K. C. & Pan, S. L. Osteoporosis and the risk of Parkinson’s disease: a nationwide, propensity score-matched, longitudinal follow-up study. J. Clin. Endocrinol. Metab. 106, e763–e771 (2021).
Lima, D. P. et al. Osteoporosis in Parkinson’s disease and the role of lean body mass: a cross-sectional study in a Brazilian tertiary center. Front. Endocrinol. 15, 1326212 (2024).
Zaidi, M., Zaidi, S. & Yuen, T. Understanding osteokine biology. Cell Metab. 36, 888–890 (2024).
Liang, W. et al. An integrated multi-omics analysis reveals osteokines involved in global regulation. Cell Metab. 36, 1144–1163.e1147 (2024).
Gerosa, L. & Lombardi, G. Bone-to-brain: a round trip in the adaptation to mechanical stimuli. Front. Physiol. 12, 623893 (2021).
Lin, Y. et al. Bone-derived factors as potential biomarkers for Parkinson’s disease. Front. Aging Neurosci. 13, 634213 (2021).
Maetzler, W. et al. Osteopontin is elevated in Parkinson’s disease and its absence leads to reduced neurodegeneration in the MPTP model. Neurobiol. Dis. 25, 473–482 (2007).
Cappellano, G. et al. The Yin-Yang of osteopontin in nervous system diseases: damage versus repair. Neural Regen. Res. 16, 1131–1137 (2021).
Guo, X. et al. Causal effect of blood osteocalcin on the risk of Alzheimer’s disease and the mediating role of energy metabolism. Transl. Psychiatry 14, 205 (2024).
Kahles, F., Findeisen, H. M. & Bruemmer, D. Osteopontin: a novel regulator at the cross roads of inflammation, obesity and diabetes. Mol. Metab. 3, 384–393 (2014).
Shi, T. et al. Osteocyte-derived sclerostin impairs cognitive function during ageing and Alzheimer’s disease progression. Nat. Metab. 6, 531–549 (2024).
Jiang, Y. L. et al. The Protective effects of osteocyte-derived extracellular vesicles against Alzheimer’s disease diminished with aging. Adv. Sci. 9, e2105316 (2022).
Lajoie, J. M. & Shusta, E. V. Targeting receptor-mediated transport for delivery of biologics across the blood-brain barrier. Annu. Rev. Pharm. Toxicol. 55, 613–631 (2015).
Guo, X. Z. et al. Osteocalcin ameliorates motor dysfunction in a 6-hydroxydopamine-induced Parkinson’s disease rat model through AKT/GSK3β signaling. Front Mol. Neurosci. 11, 343 (2018).
Gillett, D. A., Wallings, R. L., Uriarte Huarte, O. & Tansey, M. G. Progranulin and GPNMB: interactions in endo-lysosome function and inflammation in neurodegenerative disease. J. Neuroinflammation 20, 286 (2023).
Diaz-Ortiz, M. E. et al. GPNMB confers risk for Parkinson’s disease through interaction with α-synuclein. Science 377, eabk0637 (2022).
Pingault, J. B. et al. Using genetic data to strengthen causal inference in observational research. Nat. Rev. Genet. 19, 566–580 (2018).
Hemani, G., Tilling, K. & Davey Smith, G. Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 13, e1007081 (2017).
Qi, X. et al. Increased plasma GPNMB levels in patients with parkinson’s disease and cognitive impairment. Sci. Rep. 15, 20684 (2025).
Zhu, X. C. et al. CSF GPNMB in Parkinson’s disease: a potential association with age and microglial activation. J. Parkinsons Dis. 14, 1533–1542 (2024).
Moloney, E. B., Moskites, A., Ferrari, E. J., Isacson, O. & Hallett, P. J. The glycoprotein GPNMB is selectively elevated in the substantia nigra of Parkinson’s disease patients and increases after lysosomal stress. Neurobiol. Dis. 120, 1–11 (2018).
Zimmermann, M. et al. The longevity gene Klotho and its cerebrospinal fluid protein profiles as a modifier for Parkinson´s disease. Eur. J. Neurol. 28, 1557–1565 (2021).
Kjaergard, L. L., Villumsen, J. & Gluud, C. Reported methodologic quality and discrepancies between large and small randomized trials in meta-analyses. Ann. Intern Med 135, 982–989 (2001).
Liu, Z. T., Liu, M. H., Xiong, Y., Wang, Y. J. & Bu, X. L. Crosstalk between bone and brain in Alzheimer’s disease: mechanisms, applications, and perspectives. Alzheimers Dement 20, 5720–5739 (2024).
Hu, X., Zhang, P., Xu, Z., Chen, H. & Xie, X. GPNMB enhances bone regeneration by promoting angiogenesis and osteogenesis: potential role for tissue engineering bone. J. Cell Biochem. 114, 2729–2737 (2013).
Huang, B. et al. Extracellular vesicles from GPNMB-modified bone marrow mesenchymal stem cells attenuate bone loss in an ovariectomized rat model. Life Sci. 272, 119208 (2021).
Sheng, M. H., Wergedal, J. E., Mohan, S. & Lau, K. H. Osteoactivin is a novel osteoclastic protein and plays a key role in osteoclast differentiation and activity. FEBS Lett. 582, 1451–1458 (2008).
Frara, N. et al. Transgenic expression of osteoactivin/gpnmb enhances bone formation in vivo and osteoprogenitor differentiation ex vivo. J. Cell Physiol. 231, 72–83 (2016).
Postuma, R. B. et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 30, 1591–1601 (2015).
Martínez-Martín, P. et al. Parkinson’s disease severity levels and MDS-unified Parkinson’s disease rating scale. Parkinsonism Relat. Disord. 21, 50–54 (2015).
Goetz, C. G. et al. Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov. Disord. 23, 2129–2170 (2008).
Eldjarn, G. H. et al. Large-scale plasma proteomics comparisons through genetics and disease associations. Nature 622, 348–358 (2023).
GTEx Consortium. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science 369, 1318–1330 (2020).
Nalls, M. A. et al. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: a meta-analysis of genome-wide association studies. Lancet Neurol. 18, 1091–1102 (2019).
Kurki, M. I. et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature 613, 508–518 (2023).
Desquilbet, L. & Mariotti, F. Dose-response analyses using restricted cubic spline functions in public health research. Stat. Med. 29, 1037–1057 (2010).
de Winter, J. C., Gosling, S. D. & Potter, J. Comparing the Pearson and Spearman correlation coefficients across distributions and sample sizes: a tutorial using simulations and empirical data. Psychol. Methods 21, 273–290 (2016).
Schober, P., Boer, C. & Schwarte, L. A. Correlation coefficients: appropriate use and interpretation. Anesth. Analg. 126, 1763–1768 (2018).
Schlomer, G. L., Bauman, S. & Card, N. A. Best practices for missing data management in counseling psychology. J. Couns. Psychol. 57, 1–10 (2010).
Acknowledgements
This work was supported by the Natural Science Basic Research Plan in Shaanxi Province of China (No. 2024JC-YBQN-0799) and the Shaanxi Provincial Innovation of Healthcare Program (No. 2024PT-02). The authors thank the IPDGC, FinnGen, deCODE, and GTEx consortium, and all the participants and investigators who made the summary-level data publicly available.
Author information
Authors and Affiliations
Contributions
X.G. and R.L. conceived and designed the project. P.W., W.S., R.Z., and X.G. collected samples and clinical data. X.G. and W.S. analyzed the data. X.G. drafted the manuscript. R.L. revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, X., Wei, P., Shi, W. et al. Integrative clinical and genomic analyses reveal a causal role of GPNMB in the bone-brain axis of Parkinson’s disease. npj Parkinsons Dis. (2026). https://doi.org/10.1038/s41531-026-01325-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-026-01325-8