Abstract
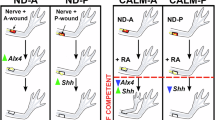
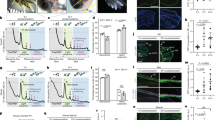
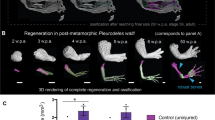
Axolotl digits offer an experimentally versatile model for studying complex tissue regeneration. Here, we provide a comprehensive morphological and molecular characterization of digit regeneration, revealing both conserved features and notable divergences from classical limb regeneration. Digit blastemas progress through similar morphological stages, are nerve-dependent, contain key regenerative cell populations, and express many canonical morphogens and mitogens. However, they exhibit minimal expression of the A–P patterning genes Shh, Fgf8, and Grem1; suggesting distal outgrowth and patterning occur independently of these signals. Joint regenerative fidelity varies significantly across digits and cannot be explained by differences in nerve supply, cell proliferation, or differential expression of any patterning genes assessed in this study. Furthermore, functional experiments reveal Hedgehog signaling is essential for interphalangeal joint regeneration, but activation alone is insufficient to improve fidelity in less robust digits. This system combines experimental accessibility with intrinsic variation in regenerative outcomes, making it an ideal platform to identify critical determinants of successful tissue regeneration and refine models of appendage patterning.
Similar content being viewed by others
Data availability
All data generated and analyzed during this study are included in this published article and its supplementary source data files.
Code availability
All custom code generated for the analysis of datasets within this manuscript is available for public access on GitHub via this link: https://github.com/Monaghan-Lab/digitPaper.
References
Gerber, T. et al. Single-cell analysis uncovers convergence of cell identities during axolotl limb regeneration. Science 362, eaaq0681 (2018).
Muneoka, K., Fox, W. F. & Bryant, S. V. Cellular contribution from dermis and cartilage to the regenerating limb blastema in axolotls. Dev. Biol. 116, 256–260 (1986).
Lin, T.-Y. et al. Fibroblast dedifferentiation as a determinant of successful regeneration. Dev. Cell 56, 1541–1551.e6 (2021).
Yamamoto, S., Kashimoto, R., Furukawa, S., Ohashi, A. & Satoh, A. Lmx1b activation in axolotl limb regeneration. Dev. Dyn. 251, 1509–1523 (2022).
Oliveira, C. R. et al. Tig1 regulates proximo-distal identity during salamander limb regeneration. Nat. Commun. 13, 1141 (2022).
Nacu, E., Gromberg, E., Oliveira, C. R., Drechsel, D. & Tanaka, E. M. FGF8 and SHH substitute for anterior–posterior tissue interactions to induce limb regeneration. Nature 533, 407–410 (2016).
Duerr, T. J. et al. Retinoic acid breakdown is required for proximodistal positional identity during axolotl limb regeneration. Nat. Commun. 16, 4798 (2025).
Lovely, A. M. et al. Wnt signaling coordinates the expression of limb patterning genes during axolotl forelimb development and regeneration. Front. Cell Dev. Biol. 10, 814250 (2022).
Tanaka, E. M. The molecular and cellular choreography of appendage regeneration. Cell 165, 1598–1608 (2016).
Laufer, E., Nelson, C. E., Johnson, R. L., Morgan, B. A. & Tabin, C. Sonic hedgehog and Fgf-4 act through a signaling cascade and feedback loop to integrate growth and patterning of the developing limb bud. Cell 79, 993–1003 (1994).
Smith, A. R. Digit regeneration in the amphibian—Triturus cristatus. J. Embryol. Exp. Morphol. 44, 105–112 (1978).
Koussoulakos, S. & Kiortsis, V. Digit regeneration in Triturus cristatus after pattern transforming interventions. In Recent Trends in Regeneration Research (eds Kiortsis, V. et al.) 465–469 (Springer US, 1989).
Riquelme-Guzmán, C. et al. Osteoclast-mediated resorption primes the skeleton for successful integration during axolotl limb regeneration. Elife 11, e79966 (2022).
Currie, J. D. et al. Live imaging of axolotl digit regeneration reveals spatiotemporal choreography of diverse connective tissue progenitor pools. Dev. Cell 39, 411–423 (2016).
Satoh, A., Gardiner, D. M., Bryant, S. V. & Endo, T. Nerve-induced ectopic limb blastemas in the axolotl are equivalent to amputation-induced blastemas. Dev. Biol. 312, 231–244 (2007).
Mescher, A. L. Effects on adult newt limb regeneration of partial and complete skin flaps over the amputation surface. J. Exp. Zool. 195, 117–127 (1976).
Tsai, S. L., Baselga-Garriga, C. & Melton, D. A. Midkine is a dual regulator of wound epidermis development and inflammation during the initiation of limb regeneration. Elife 9, e50765 (2020).
Thornton, C. S. The effect of apical cap removal on limb regeneration in Amblystoma larvae. J. Exp. Zool. 134, 357–381 (1957).
Goss, R. J. Regenerative inhibition following limb amputation and immediate insertion into the body cavity. Anat. Rec. 126, 15–27 (1956).
Leigh, N. D. et al. Transcriptomic landscape of the blastema niche in regenerating adult axolotl limbs at single-cell resolution. Nat. Commun. 9, 5153 (2018).
Godwin, J. W., Pinto, A. R. & Rosenthal, N. A. Macrophages are required for adult salamander limb regeneration. Proc. Natl. Acad. Sci. USA 110, 9415–9420 (2013).
Bryant, D. M. et al. A tissue-mapped axolotl de novo transcriptome enables identification of limb regeneration factors. Cell Rep. 18, 762–776 (2017).
Vincent, E. et al. BMP signaling is essential for sustaining proximo-distal progression in regenerating axolotl limbs. Develop. 147, dev170829 (2020).
Sader, F. & Roy, S. Tgf-β superfamily and limb regeneration: Tgf-β to start and Bmp to end. Dev. Dyn. 251, 973–987 (2022).
Guimond, J.-C. et al. BMP-2 functions independently of SHH signaling and triggers cell condensation and apoptosis in regenerating axolotl limbs. BMC Dev. Biol. 10, 15 (2010).
Glotzer, G. L., Tardivo, P. & Tanaka, E. M. Canonical Wnt signaling and the regulation of divergent mesenchymal Fgf8 expression in axolotl limb development and regeneration. Elife 11, e79762 (2022).
Han, M. J., An, J. Y. & Kim, W. S. Expression patterns of Fgf-8 during development and limb regeneration of the axolotl. Dev. Dyn. 220, 40–48 (2001).
Tickle, C. & Towers, M. Sonic hedgehog signaling in limb development. Front. Cell Dev. Biol. 5 (2017).
Torok, M. A., Gardiner, D. M., Izpisùa-Belmonte, J.-C. & Bryant, S. V. Sonic Hedgehog (shh) expression in developing and regenerating axolotl limbs. J. Exp. Zool. 284, 197–206 (1999).
Nelson, C. E. et al. Analysis of Hox gene expression in the chick limb bud. Development 122, 1449–1466 (1996).
Chiang, C. et al. Manifestation of the limb prepattern: limb development in the absence of sonic hedgehog function. Dev. Biol. 236, 421–435 (2001).
Satoh, A. et al. Characterization of Xenopus digits and regenerated limbs of the froglet. Dev. Dyn. 235, 3316–3326 (2006).
Otsuki, L., Plattner, S. A., Taniguchi-Sugiura, Y., Falcon, F. & Tanaka, E. M. Molecular basis of positional memory in limb regeneration. Nature https://doi.org/10.1038/s41586-025-09036-5 (2025).
Singer, M. & Craven, L. The growth and morphogenesis of the regenerating forelimb of adult Triturus following denervation at various stages of development. J. Exp. Zool. 108, 279–308 (1948).
Duerr, T. J. et al. 3D visualization of macromolecule synthesis. Elife 9, 1–34 (2020).
Geraudie, J. & Singer, M. Nerve dependent macromolecular synthesis in the epidermis and blastema of the adult newt regenerate. J. Exp. Zool. 203, 455–460 (1978).
Maden, M. Neurotrophic control of the cell cycle during amphibian limb regeneration. Development 48, 169–175 (1978).
Singer, M. The influence of the nerve in regeneration of the amphibian extremity. Q. Rev. Biol. 27, 169–200 (1952).
Duerr, T. J. et al. A constitutively expressed fluorescent ubiquitination-based cell-cycle indicator (FUCCI) in axolotls for studying tissue regeneration. Development 149, 1–13 (2022).
Roy, S. & Gardiner, D. M. Cyclopamine induces digit loss in regenerating axolotl limbs. J. Exp. Zool. 293, 186–190 (2002).
Purushothaman, S., Lopez Aviña, B. B. & Seifert, A. W. Sonic hedgehog is essential for proximal-distal outgrowth of the limb bud in salamanders. Front. Cell Dev. Biol 10, 797352 (2022).
Koyama, E. et al. Synovial joint formation during mouse limb skeletogenesis. Ann. N. Y. Acad. Sci. 1116, 100–112 (2007).
Zhu, J., Patel, R., Trofka, A., Harfe, B. D. & Mackem, S. Sonic hedgehog is not a limb morphogen but acts as a trigger to specify all digits in mice. Dev. Cell 57, 2048–2062.e4 (2022).
St-Jacques, B., Hammerschmidt, M. & McMahon, A. P. Indian hedgehog signaling regulates proliferation and differentiation of chondrocytes and is essential for bone formation. Genes Dev. 13, 2072–2086 (1999).
Bonnet, C. Memoire sur la reproduction des Membres de la Salamandre aquatique. J. Phys. 10, 385–405 (1777).
Endo, T., Bryant, S. V. & Gardiner, D. M. A stepwise model system for limb regeneration. Dev. Biol. 270, 135–145 (2004).
Márquez-Flórez, K. M., Monaghan, J. R., Shefelbine, S. J., Ramirez-Martínez, A. & Garzón-Alvarado, D. A. A computational model for the joint onset and development. J. Theor. Biol. 454, 345–356 (2018).
Khattak, S. et al. Germline transgenic methods for tracking cells and testing gene function during regeneration in the axolotl. Stem Cell Rep. 1, 90–103 (2013).
Yandulskaya, A. S. & Monaghan, J. R. Establishing a new research axolotl colony. Methods Mol. Biol. 27–39. https://doi.org/10.1007/978-1-0716-2659-7_2 (2023).
Lovely, A. M., Duerr, T. J., Stein, D. F., Mun, E. T. & Monaghan, J. R. Hybridization chain reaction fluorescence in situ hybridization (HCR-FISH) in Ambystoma mexicanum tissue. Methods Mol. Biol. 2562, 109–122 (2023).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25, 402–408 (2001).
Riquelme-Guzmán, C. & Sandoval-Guzmán, T. Methods for studying appendicular skeletal biology in axolotls. Methods Mol. Biol. 2562, 155–163 (2023).
Acknowledgements
The authors thank Guoxin Rong and the Institute for Chemical Imaging of Living Systems (RRID:SCR_022681) at Northeastern University for consultation and instrument support. We also appreciate Alex Lovely and the Harvard Center for Biological Imaging (RRID:SCR_018673) for infrastructure and support. The Ambystoma Genetic Stock Center at the University of Kentucky, supported by the NIH (P40-OD019794), is recognized for providing animals for this study. Funding for this work was provided by NIH grant R01HD099174 and NSF grants 2318594 and DGE-1938052.
Author information
Authors and Affiliations
Contributions
J.R.G. and J.R.M. conceived the project. J.R.M. secured funding and supervised the project. J.R.G. performed all experiments and analyses, with assistance from T.J.D. for qRT-PCR data. A.E.O. assisted in cell cycle phase analysis. M.M. assisted in data analysis and visualization. J.R.G. led the writing of the manuscript with input and approval from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Griffiths, J.R., Miller, M., Duerr, T.J. et al. A characterization of axolotl digit regeneration: conserved mechanisms, divergent patterning, and a critical role for hedgehog signaling. npj Regen Med (2026). https://doi.org/10.1038/s41536-026-00461-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41536-026-00461-2