Abstract
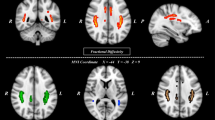
Symptom severity in schizophrenia has been repeatedly associated with thinner cortical gray matter. While global and regional white matter microstructure alterations in schizophrenia are well-documented, their association with clinical symptom severity remains unclear. As this is likely due to methodological heterogeneity across studies, we tested whether symptom severity in schizophrenia was associated with regional and global white matter alterations using standardized methods. We hypothesized that positive symptom severity would be associated with temporal white matter changes and that negative symptom severity would be associated with alterations in frontal white matter. Using a standardized fractional anisotropy (FA) analysis pipeline developed by the ENIGMA consortium, we conducted a meta-analysis of the association between white matter microstructure and symptom severity in n = 1025 (ages 16–68 years; 369 women/656 men) across 19 ENIGMA sites. Where significant heterogeneity was detected across sites, we examined whether variation in association strength between white matter microstructure and symptom severity was explained by duration of illness and/or current antipsychotic use. Positive symptom severity was significantly associated with white matter microstructure as measured using temporal lobe FA and global FA. Negative symptom severity showed no significant association with white matter microstructure as measured using frontal lobe FA or global FA. Significant heterogeneity across sites was observed for the negative symptom analysis, explained partly by duration of illness. Post-hoc exploratory analyses identified one site as disproportionately contributing to this heterogeneity, and when removed, negative symptom severity was significantly associated with both global and frontal FA. These findings support the view of schizophrenia as a disorder of brain connectivity, in a manner relevant to understanding variation in clinical symptom severity.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (G.D.) upon reasonable request.
Code availability
The codes supporting the findings of this study are available from the first author (A.W.) upon reasonable request.
References
Bora, E. et al. Neuroanatomical abnormalities in schizophrenia: a multimodal voxelwise meta-analysis and meta-regression analysis. Schizophr. Res. 127, 46–57 (2011).
Honea, R., Crow, T. J., Passingham, D. & Mackay, C. E. Regional deficits in brain volume in schizophrenia: a meta-analysis of voxel-based morphometry studies. Am. J. Psychiatry 162, 2233–2245 (2005).
Rimol, L. M. et al. Cortical thickness and subcortical volumes in schizophrenia and bipolar disorder. Biol. Psychiatry 68, 41–50 (2010).
van Erp, T. G. M. et al. Cortical brain abnormalities in 4474 individuals with schizophrenia and 5098 control subjects via the Enhancing Neuro Imaging Genetics Through Meta Analysis (ENIGMA) Consortium. Biol. Psychiatry 84, 644–654 (2018).
Karlsgodt, K. H. White matter microstructure across the psychosis spectrum. Trends Neurosci. 43, 406–416 (2020).
Kelly, S. et al. Widespread white matter microstructural differences in schizophrenia across 4322 individuals: results from the ENIGMA Schizophrenia DTI Working Group. Mol. Psychiatry 23, 1261–1269 (2018).
Kubicki, M., McCarley, R. W. & Shenton, M. E. Evidence for white matter abnormalities in schizophrenia. Curr. Opin. Psychiatry 18, 121–134 (2005).
Narr, K. L. et al. Mapping cortical thickness and gray matter concentration in first episode schizophrenia. Cereb. Cortex 15, 708–719 (2005).
Walton, E. et al. Positive symptoms associate with cortical thinning in the superior temporal gyrus via the ENIGMA Schizophrenia consortium. Acta Psychiatr. Scand. 135, 439–447 (2017).
Cui, Y. et al. Auditory verbal hallucinations are related to cortical thinning in the left middle temporal gyrus of patients with schizophrenia. Psychol. Med. 48, 115–122 (2018).
Pettersson-Yeo, W., Allen, P., Benetti, S., McGuire, P. & Mechelli, A. Dysconnectivity in schizophrenia: Where are we now?. Neurosci. Biobehav. Rev. 35, 1110–1124 (2011).
Knöchel, C. et al. Association between white matter fiber integrity and subclinical psychotic symptoms in schizophrenia patients and unaffected relatives. Schizophr. Res. 140, 129–135 (2012).
Seitz, J. et al. Tractography analysis of 5 white matter bundles and their clinical and cognitive correlates in early-course schizophrenia. Schizophr. Bull. 42, 762–771 (2016).
Whitford, T. J. et al. Corpus callosum abnormalities and their association with psychotic symptoms in patients with schizophrenia. Biol. Psychiatry 68, 70–77 (2010).
Abdul-Rahman, M. F. et al. Arcuate fasciculus abnormalities and their relationship with psychotic symptoms in schizophrenia. PLoS ONE 7, e29315 (2012).
Ćurčić-Blake, B. et al. Not on speaking terms: hallucinations and structural network disconnectivity in schizophrenia. Brain Struct. Funct. 220, 407–418 (2015).
Asami, T. et al. Cerebral white matter abnormalities and their associations with negative but not positive symptoms of schizophrenia. Psychiatry Res. 222, 52–59 (2014).
Choi, H. et al. Diffusion tensor imaging of anterior commissural fibers in patients with schizophrenia. Schizophr. Res. 130, 78–85 (2011).
Rotarska-Jagiela, A. et al. Anatomical brain connectivity and positive symptoms of schizophrenia: a diffusion tensor imaging study. Psychiatry Res. Neuroimaging 174, 9–16 (2009).
Seok, J.-H. et al. White matter abnormalities associated with auditory hallucinations in schizophrenia: a combined study of voxel-based analyses of diffusion tensor imaging and structural magnetic resonance imaging. Psychiatry Res. 156, 93–104 (2007).
Allen, P. et al. Neuroimaging auditory hallucinations in schizophrenia: from neuroanatomy to neurochemistry and beyond. Schizophr. Bull. 38, 695–703 (2012).
Hubl, D. et al. Pathways that make voices: white matter changes in auditory hallucinations. Arch. Gen. Psychiatry 61, 658–668 (2004).
Shergill, S. S. et al. A diffusion tensor imaging study of fasciculi in schizophrenia. Am. J. Psychiatry 164, 467–473 (2007).
Szeszko, P. R. et al. Clinical and neuropsychological correlates of white matter abnormalities in recent onset schizophrenia. Neuropsychopharmacology 33, 976–984 (2008).
Rabinowitz, J. et al. Negative symptoms have greater impact on functioning than positive symptoms in schizophrenia: analysis of CATIE data. Schizophr. Res. 137, 147–150 (2012).
Venkatasubramanian, G., Jayakumar, P. N., Gangadhar, B. N. & Keshavan, M. S. Automated MRI parcellation study of regional volume and thickness of prefrontal cortex (PFC) in antipsychotic-naïve schizophrenia. Acta Psychiatr. Scand. 117, 420–431 (2008).
Walton, E. et al. Prefrontal cortical thinning links to negative symptoms in schizophrenia via the ENIGMA Consortium. Psychol. Med. 48, 82–94 (2018).
Automated MRI parcellation study of regional volume and thickness of prefrontal cortex (PFC) in antipsychotic-naïve schizophrenia - Venkatasubramanian - 2008 - Acta Psychiatrica Scandinavica - Wiley Online Library. https://onlinelibrary-wiley-com.nuigalway.idm.oclc.org/doi/10.1111/j.1600-0447.2008.01198.x.
Adamczyk, P. et al. On the relation of white matter brain abnormalities and the asociality symptoms in schizophrenia outpatients—a DTI study. Acta Neurobiol. Exp. 81, 80–95 (2021).
Kristensen, T. D. et al. Changes in negative symptoms are linked to white matter changes in superior longitudinal fasciculus in individuals at ultra-high risk for psychosis. Schizophr. Res. 237, 192–201 (2021).
Wolkin, A. et al. Inferior frontal white matter anisotropy and negative symptoms of schizophrenia: a diffusion tensor imaging study. AJP 160, 572–574 (2003).
Wang, X. et al. Frontal white matter abnormalities reveal the pathological basis underlying negative symptoms in antipsychotic-naïve, first-episode patients with adolescent-onset schizophrenia: evidence from multimodal brain imaging. Schizophr. Res. 222, 258–266 (2020).
Seitz-Holland, J. et al. Elucidating the relationship between white matter structure, demographic, and clinical variables in schizophrenia—a multicenter harmonized diffusion tensor imaging study. Mol. Psychiatry 26, 5357–5370 (2021).
Steinmann, S., Leicht, G. & Mulert, C. The interhemispheric miscommunication theory of auditory verbal hallucinations in schizophrenia. Int. J. Psychophysiol. 145, 83–90 (2019).
Holleran, L. et al. The relationship between white matter microstructure and general cognitive ability in patients with schizophrenia and healthy participants in the ENIGMA Consortium. Am. J. Psychiatry 177, 537–547 (2020).
Castle, D. J. et al. The diagnostic interview for psychoses (DIP): development, reliability and applications. Psychol. Med. 36, 69–80 (2006).
Ferstl, E. C., Neumann, J., Bogler, C. & von Cramon, D. Y. The extended language network: a meta-analysis of neuroimaging studies on text comprehension. Hum. Brain Mapp. 29, 581–593 (2008).
Tyler, L. K. & Marslen-Wilson, W. Fronto-temporal brain systems supporting spoken language comprehension. Philos. Trans. R. Soc. Lond. B Biol. Sci. 363, 1037–1054 (2008).
Asami, T. et al. Abnormalities of middle longitudinal fascicle and disorganization in patients with schizophrenia. Schizophr. Res. 143, 253–259 (2013).
Bijanki, K. R., Hodis, B., Magnotta, V. A., Zeien, E. & Andreasen, N. C. Effects of age on white matter integrity and negative symptoms in schizophrenia. Schizophr. Res. 161, 29–35 (2015).
Holleran, L. et al. Altered interhemispheric and temporal lobe white matter microstructural organization in severe chronic schizophrenia. Neuropsychopharmacology 39, 944–954 (2014).
Male, A. G. et al. Structural white matter abnormalities in Schizophrenia and associations with neurocognitive performance and symptom severity. Psychiatry Res. Neuroimaging 342, 111843 (2024).
Viher, P. V. et al. Cerebral white matter structure is associated with DSM-5 schizophrenia symptom dimensions. NeuroImage Clin. 12, 93–99 (2016).
Bartzokis, G. et al. In vivo evidence of differential impact of typical and atypical antipsychotics on intracortical myelin in adults with schizophrenia. Schizophr. Res. 113, 322–331 (2009).
Chen, A. T. & Nasrallah, H. A. Neuroprotective effects of the second generation antipsychotics. Schizophr. Res. 208, 1–7 (2019).
Vita, A., De Peri, L., Deste, G., Barlati, S. & Sacchetti, E. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol. Psychiatry 78, 403–412 (2015).
Christensen, J., Holcomb, J. & Garver, D. L. State-related changes in cerebral white matter may underlie psychosis exacerbation. Psychiatry Res. Neuroimaging 130, 71–78 (2004).
Huang, J.-Y. et al. Shared and distinct alterations of white matter tracts in remitted and nonremitted patients with schizophrenia. Hum. Brain Mapp. 39, 2007–2019 (2018).
Serpa, M. H. et al. State-dependent microstructural white matter changes in drug-naïve patients with first-episode psychosis. Psychol. Med. 47, 2613–2627 (2017).
Kay, S. R., Fiszbein, A. & Opler, L. A. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Andreasen NC (1984) The scale for the assessment of positive symptoms (SAPS). University of Iowa, Iowa City.
Andreasen NC (1983) The scale for the assessment of negative symptoms (SANS). University of Iowa, Iowa City.
van Erp, T. G. M. et al. Converting positive and negative symptom scores between PANSS and SAPS/SANS. Schizophr. Res. 152, 289–294 (2014).
Smith, S. M. et al. Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. Neuroimage 31, 1487–1505 (2006).
Winklewski, P. J. et al. Understanding the physiopathology behind axial and radial diffusivity changes—What do we know? Front. Neurol. 9, 92 (2018).
Wheeler-Kingshott, C. A. M. & Cercignani, M. About “axial” and “radial” diffusivities. Magn. Reson. Med. 61, 1255–1260 (2009).
Jones, D. K. & Cercignani, M. Twenty-five pitfalls in the analysis of diffusion MRI data. NMR Biomed. 23, 803–820 (2010).
Vollmar, C. et al. Identical, but not the same: intra-site and inter-site reproducibility of fractional anisotropy measures on two 3.0 T scanners. Neuroimage 51, 1384–1394 (2010).
Mori, S., Oishi, K. & Faria, A. V. White matter atlases based on diffusion tensor imaging. Curr. Opin. Neurol. 22, 362–369 (2009).
Cox, S. R. et al. Ageing and brain white matter structure in 3,513 UK Biobank participants. Nat. Commun. 7, 13629 (2016).
Penke, L. et al. A general factor of brain white matter integrity predicts information processing speed in healthy older people. J. Neurosci. 30, 7569–7574 (2010).
R Core Team (2024). _R: A Language and Environment for Statistical Computing_. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Balduzzi S, Rücker G, Schwarzer G (2019). “How to perform a meta-analysis with R: a practical tutorial.” Evidence-Based Mental Health, 153–160.
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J Stat Softw 36, 1–48, https://doi.org/10.18637/jss.v036.i03 (2010).
Funding
This research was generously supported by grants from the European Research Council (grant no. 677467), Science Foundation Ireland (16/ERCS/3787), and the Health Research Board (RL-2020-007) to G.D., and by the Ministry of Health of the Czech Republic (grant no. NU22-04-00143) to F.S. The ENIGMA Schizophrenia Working Group gratefully acknowledges support from the NIH BD2K award, U54EB020403.
Author information
Authors and Affiliations
Consortia
Contributions
All authors are members of the ENIGMA Schizophrenia Working Group and contributed data following established protocols (https://doi.org/10.1038/mp.2017.170; https://doi.org/10.1176/appi.ajp.2019.19030225). Each participating site performed site-specific preliminary analyses to assess associations between white matter tract microstructure and symptom severity, accounting for scanner and acquisition differences. G.D. supervised the study. A.W. and L.H. carried out the pooled data analysis, with guidance from G.D. A.W. and G.D. wrote the manuscript. T.v.E. and J.T. currently lead the ENIGMA Schizophrenia Working Group. All authors contributed to manuscript review and editing and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest related to this work. N.J. and P.T. received a research grant from Biogen, Inc., for research unrelated to this manuscript. O.A.A. is a consultant for HealthLytix.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Warren, A., Holleran, L., Agartz, I. et al. An ENIGMA Consortium study of the relationship between white matter microstructure and positive and negative symptom severity in patients with schizophrenia. Schizophr (2026). https://doi.org/10.1038/s41537-026-00728-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-026-00728-z