Abstract
Cholelithiasis affects 10–20% of adults globally, and while cheese consumption may influence risk, underlying biological pathways remain unclear. In this prospective cohort study of 399,467 UK Biobank participants without prior cholelithiasis, we examined cheese intake frequency (never to ≥1/day) via baseline food-frequency questionnaires and identified incident cholelithiasis through hospital records, primary care data, and self-reports. Multivariable COX regression and causal mediation analyses assessed risk associations and mediation by cholesterol subtypes. Over follow-up, 15,897 participants developed cholelithiasis. Higher cheese intake showed a dose-dependent inverse association, with daily consumers having 26.3% lower odds (adjusted OR = 0.737, 95% CI: 0.653–0.832) versus non-consumers. HDL-C significantly mediated this association (proportion mediated: 4.03–6.98%). Frequent cheese consumption was associated with significantly lower cholelithiasis risk, partially mediated by HDL-C, suggesting a potential dietary strategy for prevention, though residual confounding and mechanisms require further investigation.
Similar content being viewed by others
Introduction
Cholelithiasis, or gallstone disease, is a common hepatobiliary disorder characterized by the formation of solid calculi (stones) in the gallbladder, primarily classified into cholesterol and pigment types1. With a global prevalence of 10–20% in adults1,2,3, cholelithiasis poses a substantial public health burden. Complications such as cholecystitis, pancreatitis, and gallbladder cancer further amplify its clinical and economic impact. Cholelithiasis is also a well-established risk factor for gallbladder cancer4. Therefore, understanding the influencing factors of cholelithiasis is of great significance for disease prevention and improving quality of life.
Dietary factors significantly contribute to the pathogenesis of gallstone formation5, making nutritional interventions a key area of interest for understanding and mitigating this disease risk. Cheese, a nutrient-dense dairy product containing calcium, vitamins, conjugated linoleic acid, and bioactive peptides6,7,8, may modulate gallstone formation. Previous Mendelian randomization studies9,10,11 provided preliminary evidence for an inverse association between cheese consumption and cholelithiasis risk, yet failed to further examine potential non-linear relationships, thereby substantially limiting the robustness of these findings.
To further validate the relationship between cheese consumption and the risk of cholelithiasis, we conducted this prospective study. This design not only enables the comparison of associations between different frequencies of cheese intake and cholelithiasis risk, but also facilitates long-term dynamic surveillance of incident cholelithiasis cases and the adjustment of potential confounding factors. These features collectively enhance the reliability of causal inferences regarding the cheese-cholelithiasis association. Therefore, using the UK Biobank cohort, encompassing >500,000 participants with dietary assessment, longitudinal clinical data, and multi-omics biomarker profiles12, we established two distinct objectives for the present study: the first is to prospectively evaluate the association between cheese consumption frequency and cholelithiasis risk; the second is to investigate the potential mediating effects of serum cholesterol subtypes (total cholesterol, HDL-C, and LDL-C) using causal mediation analysis. This dual-objective design not only deepens the understanding of the cheese-cholelithiasis relationship but also provides insights into the potential role of serum cholesterol, thereby informing targeted dietary and clinical strategies for cholelithiasis prevention.
Results
Participant characteristics
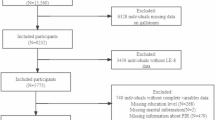
The study participants were drawn from the UK Biobank cohort, recruited between 2006 and 2010, and have been under continuous follow-up until the present. As shown in Fig. 1, from 501,940 UK Biobank participants, we excluded 102,473 individuals with missing data (e.g., deprivation index, biomarkers, lifestyle factors) or prior cholelithiasis, leaving 399,467 for analysis. The baseline characteristics of participants, stratified by cholelithiasis occurrence during follow-up, are presented in Table 1. Of 399,467 total participants, 15,897 (4.0%) developed incident cholelithiasis over a median follow-up of 85 months (interquartile range [IQR]: 46–126 months), while 383,570 (96.0%) remained non-incident (median follow-up: 184 months, IQR: 174–193 months); the overall cohort had a median follow-up of 183 months (IQR: 173–192 months). Demographically, the cohort was 53.1% female with an overall median age of 58.0 years (IQR: 50.0–63.0) (incident group: 60.0 years, IQR: 52.0–64.0). Most (94.6%) were White, with a higher proportion in the incident group (96.1% vs. 94.6% in non-incident). The overall median Townsend deprivation index was −2.18 (IQR: −3.67 to 0.44), and median BMI was 26.7 (IQR: 24.1–29.8) (incident group: 28.8, IQR: 25.9–32.3). For lifestyle factors: 7.5% never drank alcohol, 11.1% drank only on special occasions, 11.0% drank 1–3 times monthly, 25.9% 1–2 times weekly, 23.5% 3–4 times weekly, and 20.8% daily/almost daily. The incident group had more non-drinkers (10.7%) and occasional drinkers (16.2%), and fewer frequent drinkers. Smoking status: 54.7% never smoked, 34.7% were former smokers, 10.6% current smokers. The incident group had more former smokers (37.8%) and fewer never smokers (50.9%). Biochemically, overall median total cholesterol was 5.7 mmol/L (IQR: 4.9–6.4), HDL cholesterol 1.4 mmol/L (IQR: 1.2–1.7), and direct LDL 3.5 mmol/L (IQR: 3.0–4.1); the incident group had slightly lower total cholesterol (5.6 mmol/L, IQR: 4.8–6.4) and HDL cholesterol (1.3 mmol/L, IQR: 1.1–1.6). All characteristics differed significantly between groups (p < 0.001) except direct LDL (p = 0.6).
This table presents the flowchart of participant selection in the UK Biobank study. A total of 501,940 participants from the UK Biobank were initially considered. Participants were excluded based on the following criteria: unknown Townsend deprivation index (n = 621), unknown alcohol intake frequency (n = 1497), unknown smoking status (n = 1883), unknown BMI (n = 2655), unknown cheese intake (n = 13,009), prior diagnosis of cholelithiasis (n = 15,732), unknown total cholesterol (n = 29,383), unknown HDL-C (n = 37,046), and unknown direct LDL-C (n = 647). After applying these exclusion criteria, 399,467 participants were finally included in the study.
Cheese and cholelithiasis risk
Multivariable COX regression analysis indicated a significant inverse correlation between cheese consumption frequency and cholelithiasis risk. As shown in Fig. 2, we analyzed the association between cheese intake frequency and cholelithiasis risk, using “never consuming cheese” as the reference category. Models were compared under two analytical scenarios: one including all cholelithiasis cases (Fig. 2a–d) and the other excluding 540 self-reported cholelithiasis cases (Fig. 2e–h). In the multivariable COX regression model including all cholelithiasis cases (Fig. 2a), a significant dose-response relationship was observed: cheese intake frequency was inversely associated with cholelithiasis risk, with HRs gradually decreasing from 0.871 (95%CI: 0.797–0.952) for less than once a week to 0.737 (95%CI: 0.653–0.832) for once or more daily. In the multivariable COX regression model excluding self-reported cholelithiasis cases (Fig. 2e), an inverse association between cheese intake frequency and cholelithiasis risk remained significant: the HR was 0.872 (95%CI: 0.797–0.954) for <1/week, 0.838 (95%CI: 0.766–0.916) for 1/week, 0.799 (95%CI: 0.732–0.871) for 2–4/week, 0.734 (95%CI: 0.663–0.812) for 5–6/week, and 0.745 (95%CI: 0.658–0.842) for ≥1/day. All multivariable Cox regression models in Fig. 2 were adjusted for key covariates, including age, sex, Townsend deprivation index, ethnicity, smoking status, BMI, and alcohol intake frequency. Full information on covariate adjustments and additional statistical metrics is provided in Supplementary Table 1 (incorporating all cholelithiasis cases) and Supplementary Table 2 (excluding 540 self-reported cholelithiasis cases).
This figure illustrates the association between cheese intake frequency and cholelithiasis risk, quantified by hazard ratios (HRs) with 95% confidence intervals. It employs multiple analytical models, accounts for cholesterol levels, and examines both all cholelithiasis diagnoses and cases excluding 540 self-reported ones. Panels (a–d) focus on all cholelithiasis diagnoses: Panel (a) contrasts univariable and multivariable models for the association, while (b–d) display multivariable models without and with additional adjustment for total cholesterol, HDL cholesterol, and LDL cholesterol, respectively. Panels (e–h) analyze cases excluding self-reported ones: Panel (e) compares univariable and multivariable models for the association, and (f–h) present multivariable models without and with additional adjustment for total cholesterol, HDL cholesterol, and LDL cholesterol, respectively. All models include covariates such as age, sex, Townsend deprivation index, ethnic background, smoking status, body mass index (BMI), and alcohol intake frequency, with “Never” cheese intake as the reference category. Notably, the blue double arrows across Panels (a–h) all signify multivariable analysis. The orange double arrows represent univariable analysis in Panels (a, e); in (b–d), they denote multivariable analysis for all cholelithiasis diagnoses with additional adjustment for total cholesterol, HDL cholesterol, and LDL cholesterol, respectively; and in (f–h), they denote multivariable analysis for cases excluding 540 self-reported ones with additional adjustment for total cholesterol, HDL cholesterol, and LDL cholesterol, respectively.
In summary, higher cheese consumption frequency demonstrated an inverse relationship with cholelithiasis risk.
Cholesterol in cheese-cholelithiasis association
To further investigate whether the inverse association between cheese consumption and cholelithiasis risk was mediated by cholesterol levels, we conducted additional multivariable COX regression analyses with separate adjustments for total cholesterol, LDL-C, and HDL-C. After adjusting for cholesterol profiles, the inverse association between cheese consumption and cholelithiasis risk showed slight attenuation, potentially reflecting a partial mediating role of cholesterol. This trend is evident in Fig. 2b–d (incorporating all cholelithiasis cases), which present multivariable models with additional adjustment for total cholesterol, HDL cholesterol, and LDL cholesterol respectively; it is also observed in Fig. 2f–h (excluding 540 self-reported cholelithiasis cases), which display the same set of multivariable models (with adjustments for total, HDL, and LDL cholesterol) in the scenario of excluding self-reported cases. Notably, this attenuation was most pronounced after adjusting for HDL-C, as illustrated in Fig. 2c, g. Notably, all three cholesterol metrics exhibited independent associations with a reduced risk of cholelithiasis, as shown in Supplementary Table 1 (incorporating all cholelithiasis cases) and Supplementary Table 2 (excluding 540 self-reported cholelithiasis cases). This association was consistently observed in both the total cholelithiasis cohort (total cholesterol: OR = 0.950, 95% CI = 0.937–0.963; HDL-C: OR = 0.519, 95% CI = 0.491–0.548; LDL-C: OR = 0.970, 95% CI = 0.953–0.988) and the cholelithiasis cohort excluding self-reported cases (total cholesterol: OR = 0.949, 95% CI = 0.936–0.962; HDL-C: OR = 0.513, 95% CI = 0.485–0.542; LDL-C: OR = 0.969, 95% CI = 0.951–0.986). Among the three metrics, HDL-C demonstrated the most pronounced protective effect against cholelithiasis.
Multicollinearity assessment using generalized variance inflation factors (GVIF) showed all values were below 2 (Supplementary Table 3), indicating no substantial collinearity among predictors in any of the four models. We performed multivariable linear regression analyses to examine the associations between cheese intake frequency and cholesterol profiles (total cholesterol, LDL-C, and HDL-C). The results demonstrated significant correlations between cheese consumption and all three cholesterol parameters, with detailed findings presented in Supplementary Table 4. Notably, multivariable linear regression analyses revealed a strict dose-response relationship between cheese intake frequency and HDL-C, with the following group-specific results relative to the never intake reference group: <1/week (Beta = 0.0119, SE = 0.0032, P = 0.00022), once a week (Beta = 0.0166, SE = 0.0032, P < 0.0001), 2–4 times a week (Beta = 0.0233, SE = 0.0031, P < 0.0001), 5-6 times a week (Beta = 0.0306, SE = 0.0034, P < 0.0001), and once or more daily (Beta=0.0373, SE = 0.0040, P < 0.0001).
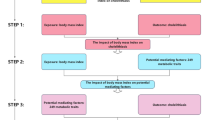
To assess the mediating effect of cholesterol, we performed bootstrap mediation analyses with 5,000 resamples, as summarized in Supplementary Table 5. Specifically, HDL-C consistently acted as a significant mediator in the association between cheese consumption (across all frequency categories compared to never) and cholelithiasis risk, with all relevant average causal mediation effect (ACME) estimates showing statistical significance (p < 0.05) and 95% CIs not crossing zero. To better present the mediation ratios of HDL-C across different cheese intake frequencies, the mediating effect of HDL-C is visualized in Fig. 3. We found that HDL-C accounting for 4.03–6.98% of the protective association between cheese consumption and lower cholelithiasis risk. Total cholesterol exhibited a partial mediating role only in two cheese consumption groups, namely less than once a week (proportion mediated = 1.0%; 95% CI = 0.28%–6.7%) and 2–4 times a week (proportion mediated = 2.9%; 95% CI = 1.6%–6.5%), while its mediating effect was non-significant in other frequency categories. In contrast, direct LDL exerted no mediating effect in this relationship.
This figure illustrates the mediation effect of high-density lipoprotein cholesterol (HDL-C) in the relationship between various cheese intake frequencies (compared with never consuming cheese: <1/week, 1/week, 2–4/week, 5–6/week, and ≥1/day) and the risk of cholelithiasis. For each cheese intake category, the mediation ratio (with 95% confidence interval, CI) represents the proportion of the effect of cheese intake on cholelithiasis risk that is mediated by HDL-C.
Discussion
In this large prospective cohort of 399,467 UK Biobank participants, we observed a significant inverse dose-response relationship between cheese consumption and cholelithiasis risk, with up to 26.3% lower odds in daily consumers compared to non-consumers. Notably, HDL-C emerged as a partial mediator, explaining 4.03–6.98% of this protective association, while total cholesterol and LDL-C demonstrated only partial or nonsignificant mediation effects. These findings suggest that cheese intake may influence gallstone prevention through pathways beyond lipid modulation, warranting further investigation into potential mechanisms (e.g., anti-inflammatory effects, gut microbiota modulation).
Our study aligns with prior evidence linking cheese intake to a reduced risk of cholelithiasis. A Mendelian randomization study9 found that cheese intake reduces gallstone risk. Observational studies also support a positive association between cheese consumption and higher HDL-C levels. A cross-sectional study of 11,377 participants (aged 40–99) in the Tromsø Study13 found that cheese intake was positively associated with HDL-C levels (+0.02 mmol/L; 95% CI: 0.01, 0.03). A cross-sectional study14 of 1752 Iranian adults found that higher cheese consumption frequency (7–14 vs. <7 times/week) was significantly associated with increased HDL-C levels and 13% lower odds of low HDL-C (OR = 0.87, 95%CI:0.79–0.96). A NHANES III study15 of 10,872 U.S. adults demonstrated that higher cheese consumption frequency was consistently associated with increased HDL-C levels. Several previous studies have demonstrated an inverse association between HDL-C levels and the risk of cholelithiasis16,17. A meta-analysis of 104 studies identified a robust protective association between HDL-C levels and gallstone disease (OR = 0.636, P = 5.97 × 10−16), confirming low HDL-C as an independent risk factor in gallstone pathogenesis16. A recent prospective study17 utilizing UK Biobank data further corroborated the inverse association between HDL-C and cholelithiasis. This study prospectively examined the cheese-cholelithiasis association using data from nearly 400,000 UK Biobank participants, establishing both a dose-response relationship and demonstrating HDL-C’s mediating role in this association. It is important to note that in the UK Biobank, cheese consumption18 was assessed retrospectively, whereas HDL-C and other lipid profiles were measured at enrollment. The prospective follow-up of participants for incident cholelithiasis, beginning at baseline assessment, provides critical temporal evidence supporting a scientifically acceptable causal sequence: habitual cheese consumption may modulate circulating lipid profiles (e.g., elevating HDL-C levels), which could subsequently alter long-term cholelithiasis risk. Although evidence directly linking cheese consumption to cholelithiasis remains scarce, our large-scale prospective analysis substantiates this association by demonstrating a robust inverse dose-response relationship and identifying HDL-C as a significant partial mediator. Moreover, the prospective cohort design enhances causal inference, reinforcing the potential role of cheese intake as a modifiable dietary determinant in gallstone prevention.
As with all observational studies, our findings should be interpreted in the context of certain inherent limitations. First, our assessment of cheese consumption and serum cholesterol relied solely on baseline measurements from UK Biobank enrollment. Dietary habits and lipid profiles may have changed during follow-up, potentially introducing misclassification bias. Second, cheese intake was assessed via a single-item food-frequency questionnaire with broad frequency categories. This method may introduce measurement error and overlook differences in cheese types (e.g., fat content, processing methods), which could differentially impact lipid metabolism and cholelithiasis risk. Third, while we adjusted for major confounders (e.g., BMI, smoking), residual confounding from unmeasured or imperfectly characterized factors (e.g., physical activity, detailed dietary patterns beyond cheese, genetic predisposition) may persist. Fourth, the UK Biobank cohort is predominantly White (95%) and healthier than the general UK population (“healthy volunteer” bias19), which may limit the generalizability of our findings to other ethnic groups or individuals with higher comorbidity burdens. Replication in more diverse populations is warranted.
This large prospective study of 399,467 UK Biobank participants demonstrated a significant inverse dose-response relationship between cheese consumption and cholelithiasis risk, with HDL-C mediating 4.03–6.98% of this protective association. These findings highlight the potential role of cheese as a modifiable dietary factor in gallstone prevention, partly through HDL-C pathways. Further research should explore additional biological mechanisms and validate these associations in more diverse populations to strengthen causal inference and support public health recommendations.
Methods
Design and variables
This prospective cohort study utilized data from the UK Biobank (UKB) to examine the association between cheese consumption and the risk of cholelithiasis (ICD-10: K80), with further evaluation of the potential mediating role of serum cholesterol markers. Cheese consumption was categorized into six frequency groups based on food-frequency questionnaires: never, <1 time/week, 1 time/week, 2–4 times/week, 5–6 times/week, and ≥1 time/day. Incident cases of cholelithiasis were identified through three primary sources: hospital admissions, primary care records, and patient self-reports. All cases were initially recorded at their first reported occurrence using ICD-10 diagnostic codes (K80). Recognizing that self-reported cholelithiasis cases may be unreliable and could introduce bias into the analysis, we excluded 540 participants with self-reported cholelithiasis and then re-conducted the analysis to ensure more robust and accurate results. Serum lipids (including total cholesterol, LDL-C, and HDL-C) were measured at baseline and considered as potential mediating factors in the analysis. To minimize confounding bias, we controlled for sociodemographic factors (age, sex, Townsend deprivation index, ethnicity), lifestyle factors (smoking status, alcohol intake frequency), and body mass index (BMI). Ethical approval for the UK Biobank was granted by the Northwest Multicentre Research Ethics Committee (approval number: 21/NW/0157). Prior to study participation, all individuals provided written informed consent in accordance with ethical guidelines. This research was conducted using access to the UK Biobank Resource, which was approved under application number 456,996.
Statistical analysis
All statistical analyses were performed using R software (version 4.4.0), with statistical significance set at a two-sided p-value < 0.05. Baseline characteristics were compared using Pearson’s Chi-squared test for categorical variables and the Kruskal-Wallis rank sum test for continuous variables. The association between cheese intake and risk of cholelithiasis was evaluated using multivariable Cox proportional hazards regression models, adjusted for relevant covariates. Results are presented as adjusted hazard ratios (HRs) with 95% confidence intervals (CIs). To assess potential mediating effects, multivariable linear regression models were first used to examine relationships between cheese intake and lipid biomarkers (total cholesterol, LDL, HDL-C). Subsequent bootstrap-based mediation analyses (5,000 iterations) were performed using the ‘mediation’ package (version 4.5.1) to quantify indirect effects, with significance determined if 95% bootstrap CIs excluded zero. Multicollinearity was assessed using variance inflation factors (VIFs) calculated with the ‘car’ package (version 3.1-3), with a VIF > 5 considered indicative of meaningful multicollinearity.
Data availability
This research was conducted using the UK Biobank resource under Application Number [456996]. The data are available to approved researchers through the UK Biobank access process (https://www.ukbiobank.ac.uk/). Restrictions apply to the availability of these data, which were used under license for the current study.
Code availability
Code supporting this study is available from the corresponding author upon reasonable request.
References
Lammert, F. et al. Gallstones. Nat. Rev. Dis. Prim. 2, 16024 (2016).
Shaffer, E. A. Epidemiology and risk factors for gallstone disease: has the paradigm changed in the 21st century? Curr. Gastroenterol. Rep. 7, 132–140 (2005).
Schirmer, B. D., Winters, K. L. & Edlich, R. F. Cholelithiasis and cholecystitis. J. long.-term. Eff. Med. Implants 15, 329–338 (2005).
Wistuba, I. I. & Gazdar, A. F. Gallbladder cancer: lessons from a rare tumour. Nat. Rev. Cancer 4, 695–706 (2004).
Cuevas, A., Miquel, J. F., Reyes, M. S., Zanlungo, S. & Nervi, F. Diet as a risk factor for cholesterol gallstone disease. J. Am. Coll. Nutr. 23, 187–196 (2004).
Gherghina, E. et al. Assessment of vitamin content in different types of Romanian cheese. Rom. Biotechnological Lett. 26, 2375–2383 (2021).
Agarwal, S., Powers, J. R., Swanson, B. G., Chen, S. & Clark, S. Cheese pH, protein concentration, and formation of calcium lactate crystals. J. Dairy Sci. 89, 4144–4155 (2006).
Santurino, C. et al. Consumption of Goat Cheese Naturally Rich in Omega-3 and Conjugated Linoleic Acid Improves the Cardiovascular and Inflammatory Biomarkers of Overweight and Obese Subjects: A Randomized Controlled Trial. Nutrients.12, 1315 (2020).
Alyahyawi, K. O. et al. Exploration of the Causal Association Between Behavioral Risk Factors and Gallstone Disease Development in Two European Ancestry Populations. Cureus 15, e37110 (2023).
Xie, L., Xu, M., Lei, Y., Li, J. & Xie, J. The causal relationship between diet habits and cholelithiasis: a comprehensive Mendelian randomization (MR) study. Front. Nutr. 11, 1377631 (2024).
Zheng, E., Cai, B. & Huang, X. Novel Insights Into the Genetic Basis Between Cholelithiasis and Four Dietary Patterns. Food Sci. Nutr. 13, e71052 (2025).
Sudlow, C. et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Machlik, M. L., Hopstock, L. A., Wilsgaard, T. & Hansson, P. Associations Between Intake of Fermented Dairy Products and Blood Lipid Concentrations Are Affected by Fat Content and Dairy Matrix - The Tromsø Study: Tromsø7. Front. Nutr. 8, 773468 (2021).
Sadeghi, M. et al. Cheese consumption in relation to cardiovascular risk factors among Iranian adults- IHHP Study. Nutr. Res. Pract. 8, 336–341 (2014).
Houston, D. K., Driver, K. E., Bush, A. J. & Kritchevsky, S. B. The association between cheese consumption and cardiovascular risk factors among adults. J. Hum. Nutr. dietetics: Off. J. Br. Dietetic Assoc. 21, 129–140 (2008).
Zhang, M. et al. Blood lipid metabolism and the risk of gallstone disease: a multi-center study and meta-analysis. Lipids health Dis. 21, 26 (2022).
Chen, L. et al. Novel insights into causal effects of serum lipids and lipid-modifying targets on cholelithiasis. Gut 73, 521–532 (2024).
UK-Biobank. Field 1408: Cheese intake. 2025; https://biobank.ctsu.ox.ac.uk/crystal/field.cgi?id=1408. Accessed 24 April 2025.
Fry, A. et al. Comparison of Sociodemographic and Health-Related Characteristics of UK Biobank Participants With Those of the General Population. Am. J. Epidemiol. 186, 1026–1034 (2017).
Acknowledgements
We are grateful to the UK Biobank participants and the UK Biobank team for their dedication and contribution to this research. This research was conducted using the UK Biobank resource under Application Number 456996. Funding was provided by the Central Incentive Fund for Regional S&T Development (grant no. 25ZYJA024).
Author information
Authors and Affiliations
Contributions
The study was designed by Wenwen Yang and Rui Ji. Statistical analyses were performed by Wenwen Yang and Rui Ji. The manuscript was written by Wenwen Yang, Zhixian Bao, Yuhua Chen, Jie Gao, and Rui Ji. All authors contributed to the interpretation of data and commented on the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, W., Bao, Z., Chen, Y. et al. Cheese consumption and lower cholelithiasis risk a prospective UK biobank study with HDL-C mediation. npj Sci Food 10, 28 (2026). https://doi.org/10.1038/s41538-025-00644-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41538-025-00644-z