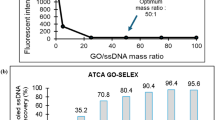
Abstract
Based on the specific aptamer-functionalized magnetic beads and the hybrid chain reaction (HCR), a rapid detection method for Bacillus cereus was established. B. cereus competes with carboxylfluorescein-labeled cDNA (FAM-cDNA) to bind to biotin-labeled aptamer fixed on streptavidin magnetic beads. After magnetic separation, the released FAM-cDNA was used as an initiator to trigger HCR amplification, significantly enhancing the fluorescence signal. In the absence of B. cereus, the initiator binds to the aptamer, generating only a weak fluorescent signal. More importantly, graphene oxide (GO) can quench the fluorescence of unreacted FAM-cDNA, achieving a more accurate detection effect and thus enabling the detection of B. cereus at lower concentrations. This method has good specificity and can detect B. cereus as low as 5.4×101 CFU/mL. A linear response was observed across the concentration range of 5.4×105 CFU/mL-5.4×101 CFU/mL. The feasibility of this method was verified by detecting B. cereus in artificially contaminated milk samples.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Osimani, A., Aquilanti, L. & Clementi, F. Bacillus cereus foodborne outbreaks in mass catering. Int. J. Hosp. Manag. 72, 145–153 (2018).
Yan, J. et al. Dual recognition strategy for the rapid and precise detection of Bacillus cereus using post-modified nano-MOF and aptamer. Sens. Actuators B: Chem. 386, 133745 (2023).
Buchacher, T., Digruber, A., Kranzler, M., Del Favero, G. & Ehling-Schulz, M. Bacillus cereus extracellular vesicles act as shuttles for biologically active multicomponent enterotoxins. Cell Commun. Signal. 21, 112 (2023).
Lotte, R., Chevalier, A., Boyer, L. & Ruimy, R. Bacillus cereus Invasive Infections in Preterm Neonates: an Up-to-Date Review of the Literature. Clin. Microbiol. Rev. 35, e00088-21 (2022).
Carlson, C. J. et al. The global distribution of Bacillus anthracis and associated anthrax risk to humans, livestock and wildlife. Nat. Microbiol. 4, 1337–1343 (2019).
Schulten, S. M. et al. Evaluation of the ISO 7932 standard for the enumeration of Bacillus cereus in foods. Int. J. Food Microbiol. 57, 53–61 (2000).
Ramarao, N., Tran, S.-L., Marin, M. & Vidic, J. Advanced Methods for Detection of Bacillus cereus and Its Pathogenic Factors. Sensors 20, 2667–2689 (2020).
Law, J. W., Ab Mutalib, N. S., Chan, K. G. & Lee, L. H. Rapid methods for the detection of foodborne bacterial pathogens: Principles, applications, advantages and limitations. Front. Microbiol. 5 https://doi.org/10.3389/fmicb.2014.00770 (2015).
Kaur, M., Harpaz, D. & Eltzov, E. Development of a portable colorimetric point-of-care device for the detection of Bacillus cereus in food specimens. Sens. Actuators B Chem. 356, 131354 (2022).
Xiong, Q. et al. Blocker-tailed PCR coupled with rolling circle amplification for fluorescent detection of emetic Bacillus cereus in milk. LWT 162, 113462 (2022).
Wei, S. et al. Development of a multiplex real-time PCR for simultaneous detection of Bacillus cereus, Listeria monocytogenes, and Staphylococcus aureus in food samples. J. Food Saf. 39, e12558 (2019).
Fischbach, J., Xander, N., Frohme, M. & Glökler, J. Shining a light on LAMP assays’ A comparison of LAMP visualization methods including the novel use of berberine. BioTechniques 58, 189–194 (2015).
Zhu, L. et al. Development of a double-antibody sandwich ELISA for rapid detection of Bacillus Cereus in food. Sci. Rep. 6, 16092 (2016).
He, J. et al. Strong and oriented conjugation of nanobodies onto magnetosomes for the development of a rapid immunomagnetic assay for the environmental detection of tetrabromobisphenol-A. Anal. Bioanal. Chem. 410, 6633–6642 (2018).
Qiao, Z., Cai, Q., Fu, Y., Lei, C. & Yang, W. Visual and quantitative detection of E. coli O157:H7 by coupling immunomagnetic separation and quantum dot-based paper strip. Anal. Bioanal. Chem. 413, 4417–4426 (2021).
Wang, Z., Cai, R., Gao, Z., Yuan, Y. & Yue, T. Immunomagnetic separation: An effective pretreatment technology for isolation and enrichment in food microorganisms detection. Compr. Rev. Food Sci. SaF. 19, 3802–3824 (2020).
Liu, Y. et al. Colorimetric immunoassay for rapid detection of Vibrio parahaemolyticus. Microchim Acta 184, 4785–4792 (2017).
Park, J. Y. et al. Detection of Escherichia coli O157:H7 using automated immunomagnetic separation and enzyme-based colorimetric assay. Sens 20, 1395 (2020).
Jia, F. et al. A magnetic relaxation switch aptasensor for the rapid detection of Pseudomonas aeruginosa using superparamagnetic nanoparticles. Microchim. Acta 184, 1539–1545 (2017).
Pang, L. et al. G-triplex/hemin DNAzyme mediated colorimetric aptasensor for Escherichia coli O157:H7 detection based on exonuclease III-assisted amplification and aptamers-functionalized magnetic beads. Talanta 269, 125457 (2024).
Vinayaka, A. C. et al. Rapid detection of Salmonella enterica in food samples by a novel approach with combination of sample concentration and direct PCR. Biosens. Bioelectron. 129, 224–230 (2019).
Lan, L., Yao, Y., Ping, J. & Ying, Y. Recent progress in nanomaterial-based optical aptamer assay for the detection of food chemical contaminants. ACS Appl. Mater. Interfaces 9, 23287–23301 (2017).
Chang, H., Tang, L., Wang, Y., Jiang, J. & Li, J. Graphene fluorescence resonance energy transfer aptasensor for the thrombin detection. Anal. Chem. 82, 2341–2346 (2010).
Wei, M. et al. A simple and sensitive electrochemiluminescence aptasensor for determination of ochratoxin A based on a nicking endonuclease-powered DNA walking machine. Food Chem. 282, 141–146 (2019).
Wang, Y. et al. Construction and bioapplications of aptamer-based dual recognition strategy. Biosens. Bioelectron. 195, 113661 (2022).
Chen, L. et al. Probing cellular molecules with polyA-based engineered aptamer nanobeacon. ACS Appl. Mater. Interfaces 9, 8014–8020 (2017).
Vajhadin, F. et al. MXene-based cytosensor for the detection of HER2-positive cancer cells using CoFe2O4@Ag magnetic nanohybrids conjugated to the HB5 aptamer. Biosens. Bioelectron. 195, 113626 (2022).
Yang, T. et al. A novel smartphone-based colorimetric aptasensor for on-site detection of Escherichia coli O157:H7 in milk. J. Dairy Sci. 104, 8506–8516 (2021).
Mahmoudpour, M. et al. Aptamer functionalized nanomaterials for biomedical applications: Recent advances and new horizons. Nano Today 39, 101177 (2021).
Naeeminejad, S., Abnous, K. & Taghdisi, S. M. A simple and label-free fluorescent aptasensor for detection of tobramycin: Appropriate for on-site antibiotic monitoring. Microchem. J. 165, 106128 (2021).
Ma, Q., Wang, Y., Jia, J. & Xiang, Y. Colorimetric aptasensors for determination of tobramycin in milk and chicken eggs based on DNA and gold nanoparticles. Food Chem. 249, 98–103 (2018).
Rowe, A. A., Miller, E. A. & Plaxco, K. W. Reagentless measurement of aminoglycoside antibiotics in blood serum via an electrochemical, ribonucleic acid aptamer-based biosensor. Anal. Chem. 82, 7090–7095 (2010).
Zhao, Y. et al. Locking-fluorescence signals regulated CRISPR/Cas12a biosensor based on metal–organic framework for sensitive detection of Salmonella typhimurium. J. Agric. Food Chem. https://doi.org/10.1021/acs.jafc.4c07681 (2024).
Augspurger, E. E., Rana, M. & Yigit, M. V. Chemical and biological sensing using hybridization chain reaction. ACS Sens 3, 878–902 (2018).
Zhao, J. et al. The application of hybridization chain reaction in the detection of foodborne pathogens. Foods 12, 4067 (2023).
Bi, S., Yue, S. & Zhang, S. Hybridization chain reaction: A versatile molecular tool for biosensing, bioimaging, and biomedicine. Chem. Soc. Rev. 46, 4281–4298 (2017).
Duan, Y. et al. Mechanically triggered hybridization chain reaction. Angew. Chem. Int. Ed. 60, 19974–19981 (2021).
Sun, M. et al. Toehold strand displacement-mediated exponential HCR for highly sensitive and specific analysis of miRNA in living cells. Anal. Chem. 96, 9078–9087 (2024).
Zhai, Q. et al. A simple and ultrasensitive electrochemical biosensor for detection of microRNA based on hybridization chain reaction amplification. J. Electroanal. Chem. 758, 20–25 (2015).
Li, L. et al. Extracellular ATP-activated hybridization chain reaction for accurate and sensitive detection of cancer cells. Chin. Chem. Lett. 34, 108399 (2023).
Xie, J. et al. Aptamer-based DNA-catalyzed amplification strategy for sensitive fluorescence resonance energy transfer detection of Acinetobacter baumannii. Talanta 255, 124212 (2023).
Wang, J. et al. Structure-switching aptamer triggering signal amplification strategy for tobramycin detection based on hybridization chain reaction and fluorescence synergism. Talanta 243, 123318 (2022).
Yu, B. et al. Hybridization chain reaction-based flow cytometric bead sensor for the detection of emetic Bacillus cereus in milk. Sens. Actuators B: Chem. 256, 624–631 (2018).
Xu, Y. et al. Ultrasensitive hairpin mediated upconversion fluorescence biosensor for Staphylococcus aureus detection in foods and waters exploiting g–C3N4–assisted catalysis. Anal. Chim. Acta 1239, 340738 (2023).
Panphut, W. et al. A superhydrophobic magneto-flow system for semi-automated electrochemical detection of Salmonella Typhimurium in food samples utilizing an aptamer-based technique. Sens. Actuators, B 446, 138624 (2026).
Lu, C., Liu, Y., Ying, Y. & Liu, J. Comparison of MoS2, WS2, and graphene oxide for DNA adsorption and sensing. Langmuir 33, 630–637 (2017).
Hashemzadeh, H. et al. A novel label-free graphene oxide nano-wall surface decorated with gold nano-flower biosensor for electrochemical detection of brucellosis antibodies in human serum. Talanta Open 7, 100215 (2023).
He, S. et al. A graphene nanoprobe for rapid, sensitive, and multicolor fluorescent DNA Analysis. Adv. Funct. Mater. 20, 453–459 (2010).
Zheng, H., Sheng, R., Li, H., ahmad, W. & Chen, Q. Rapid and selective detection of Bacillus cereus in food using cDNA-based up-conversion fluorescence spectrum copy and aptamer modified magnetic separation. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 267, 120618 (2022).
Zhou, Z. et al. Portable dual-aptamer microfluidic chip biosensor for Bacillus cereus based on aptamer tailoring and dumbbell-shaped probes. J. Hazard. Mater. 445, 130545 (2023).
Li, D. et al. Sensitive fluorescent aptasensing of tobramycin on graphene oxide coupling strand displacement amplification and hybridization chain reaction. Int. J. Biol. Macromol. 220, 1287–1293 (2022).
Saikia, L. et al. An outbreak of Bacillus cereus emetic toxin mediated food poisoning after consumption of fried rice in Assam. Indian J. Microbiol 3, 957–962 (2023).
Yuan, H. et al. Picoinjection-enabled multitarget loop-mediated isothermal amplification for detection of foodborne pathogens. Anal. Chem. 90, 13173–13177 (2018).
Zhan, Z. et al. Catalytic hairpin assembly combined with graphene oxide for the detection of emetic Bacillus cereus in milk. J. Dairy Sci. 102, 4945–4953 (2019).
Tieu, M.-V. et al. Impedimetric gram-positive bacteria biosensor using vancomycin-coated silica nanoparticles with a gold nanocluster-deposited electrode. Anal. Chem. 96, 16658–16667 (2024).
Park, J. et al. Novel high-throughput electrochemical detection of staphylococcus aureus, bacillus cereus, or micrococcus luteus using AuNPs@Ti3C2Tz functionalized with sandwich peptides. Small (Weinh. Bergstr. Ger.) 21, 2411486 (2025).
Li, P.-R. et al. An RPA-assisted CRISPR/Cas12a assay combining fluorescence and lateral flow strips for the rapid detection of enterotoxigenic Bacillus cereus. J. Agric. Food Chem. 72, 14000–14010 (2024).
Bao, H. et al. Rapid and simple detection of Bacillus cereus in milk by real-time competitive annealing mediated isothermal amplification. Analyst 145, 6677–6682 (2020).
Zhong, Z., Gao, X., Gao, R. & Jia, L. Selective capture and sensitive fluorometric determination of Pseudomonas aeruginosa by using aptamer modified magnetic nanoparticles. Microchim Acta 185, 377 (2018).
Dong, H., Gao, W., Yan, F., Ji, H. & Ju, H. Fluorescence Resonance Energy Transfer between Quantum Dots and Graphene Oxide for Sensing Biomolecules. Anal. Chem. 82, 5511–5517 (2010).
Tang, J., Wang, Z., Zhou, J., Lu, Q. & Deng, L. Enzyme-free hybridization chain reaction-based signal amplification strategy for the sensitive detection of Staphylococcus aureus. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 215, 41–47 (2019).
Yun, W. et al. Graphene oxide-based fluorescent “turn-on” strategy for Hg2+ detection by using catalytic hairpin assembly for amplification. Sens. Actuators B: Chem. 249, 493–498 (2017).
Kim, H.-S. et al. Two-stage label-free aptasensing platform for rapid detection of Cronobacter sakazakii in powdered infant formula. Sens. Actuators B: Chem. 239, 94–99 (2017).
Ye, S. et al. Colorimetric aptasensor based on magnetic beads and gold nanoparticles for detecting mucin 1. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 315, 124236 (2024).
Zhang, F., Wang, S. & Liu, J. Gold nanoparticles adsorb DNA and aptamer probes too strongly and a comparison with graphene oxide for biosensing. Anal. Chem. 91, 14743–14750 (2019).
Chen, S. et al. A novel AuNPs colorimetric sensor for sensitively detecting viable Salmonella typhimurium based on dual aptamers. Food Control 115, 107281 (2020).
Wang, L., Lyu, S., Gu, G. & Bolten, S. Selection of aptamers targeted to food-borne pathogenic bacteria Vibrio parahaemolyticus. Food Sci. Nutr. 8, 3835–3842 (2020).
Wang, C. et al. An integrated magnetic separation enzyme-linked colorimetric sensing platform for field detection of Escherichia coli O157: H7 in food. Microchim Acta 191, 454 (2024).
Xu, Y., & Zheng, Z. Hybridization chain reaction for direct mRNA detection without nucleic acid purification, in: I. Gaspar (Ed.), RNA detection: Methods and protocols, Springer, New York, NY, 2018: 187–196.
Zhu, W. et al. A fluorescence aptasensor based on hybridization chain reaction for simultaneous detection of T-2 toxins and zearalenone. Talanta 255, 124249 (2023).
Acknowledgements
The authors express sincere gratitude to the Enzyme Engineering Laboratory of the College of Food Science and Engineering at Nanjing Agricultural University for the equipment and assistance provided. This study was supported by grants Key Laboratory of Detection and Traceability Technology of Foodborne Pathogenic Microorganisms, State Administration forMarketRegulation (GrantNo.KFW202503), and Agricultural Independent InnovationProgramin JiangsuProvince (GrantNo.CX(23)3043).
Author information
Authors and Affiliations
Contributions
**X.S.:** Conceptualization, Methodology, Formal analysis, Investigation, Writing-Original draft. **C.S.:** Conceptualization, Methodology. **Y.L.:** Resources, Data curation. **X.L.:** Resources. **X.B.:** Project administration, Funding acquisition, Supervision, Writing-Review & Editing. **J.Y.:** **Funding acquisition**.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial or non-financial interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Song, X., Shi, C., Lv, Y. et al. Aptamer-functionalized magnetic beads combined with hybridization chain reaction for detection of Bacillus cereus. npj Sci Food (2026). https://doi.org/10.1038/s41538-026-00751-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41538-026-00751-5