Abstract
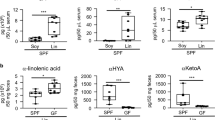
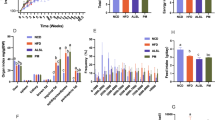
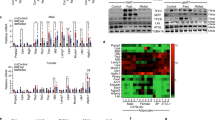
Enhanced α-linolenic acid (ALA) intake effectively alleviates population sub-health status through dietary pattern modification, demonstrating substantial implications for promoting national health advancement. In this study, the multi-targeted regulation on ALA micellization, absorption, and lymphatic transport by natural flaxseed lignan macromolecules (FLM) and its thermal treatment products (FLM 150) was systematically investigated in an emulsion delivery system. In vitro Caco-2 transport assays revealed that ALA absorption was enhanced by 95% with the addition of FLM 150. Moreover, in vivo rat mesenteric lymph cannulation proved that FLM 150-enriched emulsion elevated ALA absorption by 493%, while significantly reducing lipid oxidation. Besides, lipidomic profiling indicated that FLM 150 enhanced triglyceride resynthesis during lipid resynthesis in intestinal epithelial cells, as well as promoted metabolism of ALA. Therefore, these findings established FLM 150 as a multi-target regulator optimizing ALA bioavailability through concurrent mucosal barrier reinforcement, oxidative stress mitigation, and lipid metabolic pathway redirection. These insights highlighted the potential of engineered emulsion systems to lipid absorption kinetics and intestinal metabolic outcomes, offering novel strategies for enhancing the bioavailability of ALA.
Similar content being viewed by others
Data availability
The lipidomics were analyzed using the Public Database MSDAIL and Peakview 2.0 software. Global lipidomic principal component analysis (PCA), heatmap, and correlation analysis were performed using the software of MetaboAnalyst 4.0.
References
Lai, H. T. et al. D. S., Serial circulating omega 3 polyunsaturated fatty acids and healthy ageing among older adults in the Cardiovascular Health Study: prospective cohort study. BMJ 363, https://doi.org/10.1136/bmj.k4067 (2018).
Lister, N. B. et al. Child and adolescent obesity. Nat. Rev. Dis. Prim. 9, 1 (2023).
Barceló-Coblijn, G. & Murphy, E. J. Alpha-linolenic acid and its conversion to longer chain n-3 fatty acids: benefits for human health and a role in maintaining tissue n-3 fatty acid levels. Prog. Lipid Res. 48, 355–374 (2009).
D’Aquila, T., Hung, Y. H., Carreiro, A. & Buhman, K. K. Recent discoveries on absorption of dietary fat: Presence, synthesis, and metabolism of cytoplasmic lipid droplets within enterocytes. Biochim. Biophys Acta (BBA) - Mol. Cell Biol. Lipids 1861, 730–747 (2016).
Damianos, J., Abdelnaem, N. & Camilleri, M. Gut Goo: physiology, diet, and therapy of intestinal mucus and biofilms in gastrointestinal health and disease. Clin. Gastroenterol. Hepatol. 23, 205–215 (2025).
Murgia, X. et al. Size-limited penetration of nanoparticles into porcine respiratory mucus after aerosol deposition. Biomacromolecules 17, 1536–1542 (2016).
Bao, C., Liu, B., Li, B., Chai, J. & Li, Y. Enhanced transport of shape and rigidity-tuned α-Lactalbumin nanotubes across intestinal mucus and cellular barriers. Nano Lett. 20, 1352–1361 (2020).
Wang, D. et al. Nanocarriers transport across the gastrointestinal barriers: The contribution to oral bioavailability via blood circulation and lymphatic pathway. Adv. Drug Deliv. Rev. 203, 115130 (2023).
Cheng, C., Yu, X., Huang, F., Peng, D. & Deng, Q. Effect of different structural flaxseed lignans on the stability of flaxseed oil-in-water emulsion: an interfacial perspective. Food Chem. 357, 129522 (2021).
Sugasini, D., Devaraj, V. C., Ramesh, M. & Lokesh, B. R. Lymphatic transport of α-Linolenic acid and its conversion to long chain n-3 fatty acids in rats fed microemulsions of linseed oil. Lipids 49, 225–233 (2013).
Feng, N. et al. Physical and oxidative stability of flaxseed oil-in-water emulsions prepared by natural lignin-carbohydrate complex. Int. J. Biol. Macromol. 270, 132154 (2024).
Wang, X., Chen, H., Xu, Y. & Deng, Q. The role of micro-structures in the aqueous phase of emulsion in lipid oxidation process. Food Chem. 464, 141760 (2025).
Feng, G. et al. Interaction of Pyrogallol-containing polyphenols with mucin reinforces intestinal mucus barrier properties. J. Agric. Food Chem. 70, 9536–9546 (2022).
Adolphe, J. L., Whiting, Juurlink, S. J., Thorpe, B. H. J. & Alcorn, L. U. J., Health effects with consumption of the flax lignan secoisolariciresinol diglucoside. Br. J. Nutr. 103, 929–938 (2010).
Habtemariam, S. The what and who of dietary lignans in human health: Special focus on prooxidant and antioxidant effects. Trends Food Sci. Technol. 106, 382–390 (2020).
Wang, L. et al. Conversion of α-linolenic acid into n-3 long-chain polyunsaturated fatty acids: bioavailability and dietary regulation. Crit. Rev. Food Sci. Nutr. 1-33. https://doi.org/10.1080/10408398.2024.2442064 (2024).
Adlercreutz, H. Lignans and human health. Crit. Rev. Clin. Lab. Sci. 44, 483–525 (2008).
Yang, X., Guo, Y., Tse, T. J., Purdy, S. K. & Reaney, M. J. T. Oral Pharmacokinetics of enriched Secoisolariciresinol Diglucoside and Its Polymer in Rats. J. Nat. Prod. 84, 1816–1822 (2021).
Cheng, C., Wang, L., Yu, X., Huang, F. & Deng, Q. Structural identification and antioxidative activity evaluation of flaxseed lignan macromolecules: structure-activity correlation. Food Sci. Hum. Wellness 13, 3224–3235 (2024).
Cheng, C. et al. Effect of heat-treated flaxseed lignan macromolecules on the interfacial properties and physicochemical stability of α-linolenic acid-enriched O/W emulsions. Food Funct. 15, 9524–9540 (2024).
Abdulkarim, M. et al. Nanoparticle diffusion within intestinal mucus: Three-dimensional response analysis dissecting the impact of particle surface charge, size and heterogeneity across polyelectrolyte, pegylated and viral particles. Eur. J. Pharm. Biopharm. 97, 230–238 (2015).
Floor, E. et al. Development of a Caco-2-based intestinal mucosal model to study intestinal barrier properties and bacteria–mucus interactions. Gut Microbes 17, 2434685 (2024).
Wilson, F. A., Sallee, V. L. & Dietschy, J. M. Unstirred water layers in intestine: rate determinant of fatty acid absorption from micellar solutions. Science 174, 1031–1033 (1971).
Chloé, R. et al. Rapeseed Lecithin increases lymphatic lipid output and α-linolenic acid bioavailability in rats. J. Nutr. 150, 2900–2911 (2020).
Takechi, R., Hiramatsu, N., Mamo, J. C. L. & Pal, S. Red wine polyphenolics suppress the secretion and the synthesis of Apo B48 from human intestinal Caco-2 cells. BioFactors 22, 181–183 (2004).
Pang, J. et al. Resveratrol intervention attenuates chylomicron secretion via repressing intestinal FXR-induced expression of scavenger receptor SR-B1. Nat. Commun. 14, 2656 (2023).
Gobert, M. et al. Fruits, vegetables and their polyphenols protect dietary lipids from oxidation during gastric digestion. Food Funct. 5, 2166–2174 (2014).
Jia, W., Wu, X., Zhang, R., Wang, X. & Shi, L. Novel insight into the resilient drivers of bioaccumulation perchlorate on lipid nutrients alterations in goat milk by spatial multi-omics. Lwt 165, 113717 (2022).
Zhou, Z. et al. Metal ion-mediated pro-oxidative reactions of different lipid molecules: revealed by nontargeted lipidomic approaches. J. Agric. Food Chem. 70, 10284–10295 (2022).
Trevaskis, N. L. The mesenteric lymph duct cannulated rat model: application to the assessment of intestinal lymphatic drug transport. J. Vis. Exp. 97, 52389 (2015).
Zhang, P. et al. Lipin 2/3 phosphatidic acid phosphatases maintain phospholipid homeostasis to regulate chylomicron synthesis. J. Clin. Investig. 129, 281–295 (2019).
Zembroski, A. S., Xiao, C. & Buhman, K. K. The roles of cytoplasmic lipid droplets in modulating intestinal uptake of dietary fat. Annu. Rev. Nutr. 41, 79–104 (2021).
Brodkorb, A. et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 14, 991–1014 (2019).
Tan, Y., Zhang, Z., Zhou, H., Xiao, H. & McClements, D. J. Factors impacting lipid digestion and β-carotene bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): oil droplet concentration. Food Funct. 11, 7126–7137 (2020).
Gong, S. et al. Taxifolin ameliorates lipopolysaccharide-induced intestinal epithelial barrier dysfunction via attenuating NF-kappa B/MLCK pathway in a Caco-2 cell monolayer model. Food Res. Int. 158, 111502 (2022).
Wang, L. et al. Flax lignans regulate the conversion of α-linolenic acid into n-3 LCPUFAs in mice ingesting sunflower phospholipid-stabilized nanoemulsions. Food Sci. Hum. Wellness https://doi.org/10.26599/FSHW.2024.9250371 (2024).
Acknowledgements
The authors gratefully acknowledge the financial support from National Natural Science Foundation of China (32402168 and 32072267), Research Funding of Wuhan Polytechnic University NO.2025R2050, and China Agriculture Research System of CRAS-14. We extend our gratitude to WETRYBIO Biotechnology (Shanghai) Co., Ltd. for providing rat thoracic lymphatic duct cannulation experimental technique and platform (WTPZ20221212001).
Author information
Authors and Affiliations
Contributions
Chen Cheng was responsible for the writing of the original draft, methodology, and visualization. Xiao Yu was responsible for the writing of the original draft and visualization. Lei Wang was responsible for the conceptualization and data analysis. Shuyi Li was responsible for conceptualization and formal analysis. Xu Chen was responsible for conceptualization, writing of the original draft, and visualization. Qianchun Deng was responsible for supervision, conceptualization, and funding acquisition. Zhenzhou Zhu was responsible for conceptualization and visualization. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cheng, C., Yu, X., Wang, L. et al. The regulation mechanism of α-linolenic acid bioavailability by flaxseed lignan macromolecules in O/W emulsions. npj Sci Food (2026). https://doi.org/10.1038/s41538-026-00814-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41538-026-00814-7