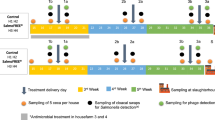
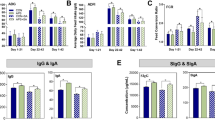
Abstract
Salmonella enterica serovar Enteritidis (S. Enteritidis) is one of most common Salmonella serovars associated with human illness in the U.S. and worldwide. Surveillance from the U.S. National Antimicrobial Resistance Monitoring System indicates an increase in both chicken and human isolates of S. Enteritidis with decreased susceptibility to ciprofloxacin (DSC), a critical antibiotic prescribed for complicated human salmonellosis infections. S. Enteritidis reduction in chickens is a priority of poultry producers and public health agencies to improve food safety. In the current study, efficacy assessment of a live Salmonella vaccine (BBS 1134) revealed significant reduction of cecal and splenic colonization, and prevention of dissemination to the bone marrow by DSC S. Enteritidis in broiler chickens. Microbiome analysis indicated the cecal microbiota of vaccinated chickens is distinct compared to mock-vaccinated birds. The IDEXX SE Ab X2 Test did not detect antibodies to S. Enteritidis in vaccinated chicken serum, thereby permitting differentiation of infected from vaccinated animals (DIVA). Altogether, the Salmonella vaccine is a DIVA vaccine, afforded cross-protection, and significantly reduced intestinal colonization and dissemination to the spleen and bone marrow by DSC S. Enteritidis in chickens, thereby offering a prospective intervention for animal production to reduce food product contamination and improve food safety.
Similar content being viewed by others
Data availability
Raw 16S reads were uploaded to the NCBI Sequence Read Archive under BioProject PRJNA1309157.
Code availability
R scripts and full R session information with loaded libraries and their versions are located on GitHub (https://github.com/USDA-FSEPRU/bbs_1134_vaccine_vs_dsc_enteritidis_challenge.git).
References
Majowicz, S. E. et al. The global burden of nontyphoidal Salmonella gastroenteritis. Clin. Infect. Dis. 50, 882–889 (2010).
Ford, L. et al. Salmonella outbreaks associated with not ready-to-eat breaded, stuffed chicken products - United States, 1998-2022. MMWR Morb. Mortal. Wkly Rep. 72, 484–487 (2023).
Altekruse, S. F. et al. Salmonella Enteritidis in broiler chickens, United States, 2000-2005. Emerg. Infect. Dis. 12, 1848–1852 (2006).
Sher, A. A., Mustafa, B. E., Grady, S. C., Gardiner, J. C. & Saeed, A. M. Outbreaks of foodborne Salmonella Enteritidis in the United States between 1990 and 2015: an analysis of epidemiological and spatial-temporal trends. Int J. Infect. Dis. 105, 54–61 (2021).
Andino, A. & Hanning, I. Salmonella enterica: survival, colonization, and virulence differences among serovars. ScientificWorldJournal 2015, 520179 (2015).
Williams, M. S., Ebel, E. D., Robertson-Hale, K., Shaw, S. L. & Kissler, B. W. Differences in Salmonella serotypes in broiler chickens within and between slaughter establishments in the United States. J. Food Prot. 88, 100506 (2025).
Haro, J. et al. Increase in the Frequency of Salmonella Enteritidis with Decreased Susceptibility to Ciprofloxacin, <https://www.fsis.usda.gov/news-events/news-press-releases/increase-frequency-salmonella-enteritidis-decreased-susceptibility> (2024).
CDC. Antibiotic Resistance Threats in the United States, 2019 (Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2019).
Bearson, S. M. D. et al. Cross-protective Salmonella vaccine reduces cecal and splenic colonization of multidrug-resistant Salmonella enterica serovar Heidelberg. Vaccine 37, 1255–1259 (2019).
Bearson, B. L., Bearson, S. M. & Kich, J. D. A DIVA vaccine for cross-protection against Salmonella. Vaccine 34, 1241–1246 (2016).
Bearson, B. L. et al. Salmonella DIVA vaccine reduces disease, colonization and shedding due to virulent S. Typhimurium infection in swine. J. Med. Microbiol. 66, 651–661 (2017).
Bearson, B. L. & Bearson, S. M. Mutated Salmonella enterica. US 9868769, B2 (2018).
Bearson, B. L. et al. Iron regulated genes of Salmonella enterica serovar Typhimurium in response to norepinephrine and the requirement of fepDGC for norepinephrine-enhanced growth. Microbes Infect. 10, 807–816 (2008).
Gast, R. K. & Beard, C. W. Isolation of Salmonella Enteritidis from internal organs of experimentally infected hens. Avian Dis. 34, 991–993 (1990).
Alali, W. Q. et al. The relationship between levels in chicken spleen and mechanically separated ground chicken. Food Control 66, 250–255 (2016).
Velaudapillai, T. Salmonellae in bones of slaughtered cattle and poultry. Z. Hyg. Infektionskr. 150, 10–12 (1964).
Wu, D. Z., Alali, W. Q., Harrison, M. A. & Hofacre, C. L. Prevalence of in neck skin and bone of chickens. J. Food Prot. 77, 1193–1197 (2014).
Kassem, I. I., Sanad, Y. M., Stonerock, R. & Rajashekara, G. An evaluation of the effect of sodium bisulfate as a feed additive on Salmonella enterica serotype Enteritidis in experimentally infected broilers. Poult. Sci. 91, 1032–1037 (2012).
Jones-Ibarra, A. M., Alvarado, C. Z., Caldwell, D. H., Byrd, J. A. & Taylor, T. M. Salmonella recovery from chicken bone marrow and cecal counts differ by pathogen challenge method. Poult. Sci. 98, 4104–4112 (2019).
Brooks, L. A., Bailey, M. A., Krehling, J. T., Chasteen, K. S. & Macklin, K. S. A Comparison of colonizing ability between Salmonella Enteritidis and Salmonella Heidelberg in broiler chickens challenged through feed administration. Foodborne Pathog. Dis. 18, 784–789 (2021).
Jan, T. R., Lin, C. S., Wang, S. Y. & Yang, W. Y. Cytokines and cecal microbiome modulations conferred by a dual vaccine in Salmonella-infected layers. Poult. Sci. 102, 102373 (2023).
Park, S. H., Kim, S. A., Rubinelli, P. M., Roto, S. M. & Ricke, S. C. Microbial compositional changes in broiler chicken cecal contents from birds challenged with different Salmonella vaccine candidate strains. Vaccine 35, 3204–3208 (2017).
Lyimu, W. M., Leta, S., Everaert, N. & Paeshuyse, J. Influence of live attenuated Salmonella vaccines on cecal microbiome composition and microbiota abundances in young broiler chickens. Vaccines 11, https://doi.org/10.3390/vaccines11061116 (2023).
Khan, S. et al. Vaccine protection of broilers against various doses of wild-type Salmonella Typhimurium and changes in gut microbiota. Vet. Q. 45, 1–14 (2025).
Lu, J. et al. Diversity and succession of the intestinal bacterial community of the maturing broiler chicken. Appl. Environ. Microbiol. 69, 6816–6824 (2003).
Burrows, P. B. et al. Decoding the chicken gastrointestinal microbiome. BMC Microbiol. 25, 35 (2025).
Kers, J. G. & Saccenti, E. The power of microbiome studies: some considerations on which alpha and beta metrics to use and how to report results. Front. Microbiol. 12, 796025 (2021).
Jan, T. R., Lin, C. S. & Yang, W. Y. Differential cytokine profiling and microbial species involved in cecal microbiota modulations in SPF chicks immunized with a dual vaccine against Salmonella Typhimurium infection. Poult. Sci. 103, 103334 (2024).
Maki, J. J., Bobeck, E. A., Sylte, M. J. & Looft, T. Eggshell and environmental bacteria contribute to the intestinal microbiota of growing chickens. J. Anim. Sci. Biotechnol. 11, 60 (2020).
Martin, R. et al. Faecalibacterium: a bacterial genus with promising human health applications. FEMS Microbiol. Rev. 47, https://doi.org/10.1093/femsre/fuad039 (2023).
Fan, Y., Ju, T., Bhardwaj, T., Korver, D. R. & Willing, B. P. Week-old chicks with high Bacteroides abundance have increased short-chain fatty acids and reduced markers of gut inflammation. Microbiol Spectr. 11, e0361622 (2023).
Ameer, A. et al. Temporal stability and community assembly mechanisms in healthy broiler cecum. Front. Microbiol. 14, 1197838 (2023).
Zhang, L. et al. Rosemary leaf powder improves egg quality, antioxidant status, gut barrier function, and cecal microbiota and metabolites of late-phase laying hens. Anim. Nutr. 17, 325–334 (2024).
Bearson, S. M. et al. Prophylactic administration of vector-encoded porcine granulocyte-colony stimulating factor reduces Salmonella shedding, tonsil colonization, and microbiota alterations of the gastrointestinal tract in Salmonella-challenged swine. Front. Vet. Sci. 3, 66 (2016).
Bearson, S. M. D. et al. Commercial vaccine provides cross-protection by reducing colonization of Salmonella enterica serovars Infantis and Hadar in turkeys. Vaccine 42, 727–731 (2024).
Bearson, B. L. et al. The role of Salmonella Genomic Island 4 in metal tolerance of Salmonella enterica serovar i 4,[5],12:i:- pork outbreak isolate USDA15WA-1. Genes 11 https://doi.org/10.3390/genes11111291 (2020).
Kozich, J. J., Westcott, S. L., Baxter, N. T., Highlander, S. K. & Schloss, P. D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl Environ. Microbiol 79, 5112–5120 (2013).
R: a language and environment for statistical computing. (R Foundation for Statistical Computing, Vienna, Austria, 2024).
RStudio: integrated development environment for R v. 2024.09.0 (Boston, MA, 2024).
Davis, N. M., Proctor, D. M., Holmes, S. P., Relman, D. A. & Callahan, B. J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 6, 226 (2018).
Wickham, H. et al. Welcome to the tidyverse. J. Open Source Softw. 4, 1686 (2019).
McMurdie, P. J. & Holmes, S. phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Lin, H. & Peddada, S. D. Analysis of compositions of microbiomes with bias correction. Nat. Commun. 11, 3514 (2020).
Lin, H., Eggesbo, M. & Peddada, S. D. Linear and nonlinear correlation estimators unveil undescribed taxa interactions in microbiome data. Nat. Commun. 13, 4946 (2022).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag New York, 2016).
Acknowledgements
The authors are greatly appreciative of the outstanding technical support of Margaret Walker and Kellie Winter. We thank the Animal Research Unit staff for providing animal care. This work was supported by the United States Department of Agriculture (USDA), Agricultural Research Service (ARS) appropriated funds from CRIS projects 5030-32000-227-000D and 5030-31000-007-000-D. Funding for open access charge: USDA, ARS. This research utilized an appointment to the Agricultural Research Service Research Participation Program administered by the Oak Ridge Institute for Science and Education (ORISE) through an interagency agreement between the U.S. Department of Energy (DOE) and the USDA. ORISE is managed by ORAU under DOE contract number DE-SC0014664. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendations or endorsement by the U.S. Department of Agriculture. The U.S. Department of Agriculture is an equal opportunity provider and employer.
Author information
Authors and Affiliations
Contributions
Conceptualization—S.M.D.B. and B.L.B; Investigation—B.L.B, S.J.W., M.P.N.E., D.P.N., M.S.M., C.L.A., and S.M.D.B.; Formal analysis—B.L.B, M.P.N.E., D.J.B., M.S.M., and S.M.D.B.; Methodology—B.L.B, S.J.W., M.P.N.E., D.J.B., M.S.M., C.L.A. and S.M.D.B.; Validation—B.L.B, M.P.N.E., D.J.B., M.S.M., C.L.A. and S.M.D.B.; Visualization—B.L.B, D.J.B., and M.S.M.; Data curation—B.L.B, D.J.B., S.J.W., M.P.N.E., and M.S.M.; Funding acquisition—S.M.D.B. and B.L.B; Project administration—S.M.D.B. and B.L.B; Resources—S.M.D.B.; Supervision—S.M.D.B. and B.L.B; Writing—original draft—B.L.B, S.J.W., M.P.N.E., D.J.B., M.S.M., and S.M.D.B.; Writing—review and editing—B.L.B, S.J.W., M.P.N.E., D.P.N., D.J.B., M.S.M., C.L.A., and S.M.D.B.
Corresponding author
Ethics declarations
Competing interests
Two patents (US 9,868,769 B2 and US 10,351,606 B2) have been issued for vaccine strain BBS 1134 with B.L.B. and S.M.D.B. indicated as inventors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bearson, B.L., Whelan, S.J., Encinosa, M.P.N. et al. Chicken vaccination reduces colonization and dissemination of Salmonella serovar Enteritidis with decreased susceptibility to ciprofloxacin. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01414-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01414-y