Abstract
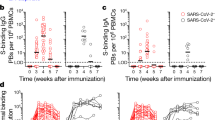
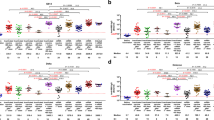
The SARS-CoV-2 mRNA vaccine provides effective protection against viral infection and severe disease by inducing efficient adaptive immunity. However, vaccine efficacy is decreased against emerging variants, and immune memory is relatively short-lived. Here, we added new T cell epitopes to the RBD (receptor-binding domain) mRNA vaccine and identified a SARS-CoV-2 membrane epitope that significantly improved vaccine-induced immunity and protection in vivo. That new vaccine, designated G1-C, induced 8.2-fold higher levels of RBD-specific antibodies than did RBD and enhanced spike-specific T cell and B cell responses. Remarkably, the G1-C modulated hematopoietic stem cell (HSC) differentiation and increased levels of B and NK cells by regulating multiple signaling pathways in bone marrow potentially via Fos, Klf4, and Klf6 transcription factors. Altogether, these findings identify a new vaccine candidate to control viral infection by affecting the lymphoid-myeloid lineage bias and suggest the potential role of T cell epitopes in vaccine design and development.

Similar content being viewed by others
Data availability
Single-cell RNA sequencing data are deposited on GEO: GSE281662. The code used for data analysis is available on GitHub: https://github.com/jingw1072/mice-BM-scRNAseq-analysis-code.
Code availability
Single-cell RNA sequencing data are deposited on GEO: GSE281662. The code used for data analysis is available on GitHub: https://github.com/jingw1072/mice-BM-scRNAseq-analysis-code.
References
Hu, B., Guo, H., Zhou, P. & Shi, Z. L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 19, 141–154 (2021).
Jackson, C. B., Farzan, M., Chen, B. & Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 23, 3–20 (2022).
Almanza, G. et al. Structure-selected RBM immunogens prime polyclonal memory responses that neutralize SARS-CoV-2 variants of concern. PLoS Pathog. 18, e1010686 (2022).
Fiolet, T., Kherabi, Y., MacDonald, C. J., Ghosn, J. & Peiffer-Smadja, N. Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: a narrative review. Clin. Microbiol. Infect. 28, 202–221 (2022).
Tregoning, J. S., Flight, K. E., Higham, S. L., Wang, Z. & Pierce, B. F. Progress of the COVID-19 vaccine effort: viruses, vaccines and variants versus efficacy, effectiveness and escape. Nat. Rev. Immunol. 21, 626–636 (2021).
Alter, G. et al. Immunogenicity of Ad26.COV2.S vaccine against SARS-CoV-2 variants in humans. Nature 596, 268–272 (2021).
Emary, K. R. W. et al. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): an exploratory analysis of a randomised controlled trial. Lancet 397, 1351–1362 (2021).
Jones, I. & Roy, P. Sputnik V COVID-19 vaccine candidate appears safe and effective. Lancet 397, 642–643 (2021).
Baden, L. R. et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 384, 403–416 (2021).
Lyke, K. E. et al. Immunogenicity of NVX-CoV2373 heterologous boost against SARS-CoV-2 variants. NPJ Vaccines 8, 98 (2023).
Ryzhikov, A. B. et al. Assessment of safety and prophylactic efficacy of the EpiVacCorona Peptide vaccine for COVID-19 prevention (Phase III). Vaccines 11, https://doi.org/10.3390/vaccines11050998 (2023).
Hadj Hassine, I. Covid-19 vaccines and variants of concern: a review. Rev. Med. Virol. 32, e2313 (2022).
Chi, W. Y. et al. COVID-19 vaccine update: vaccine effectiveness, SARS-CoV-2 variants, boosters, adverse effects, and immune correlates of protection. J. Biomed. Sci. 29, 82 (2022).
Ying, B. et al. Protective activity of mRNA vaccines against ancestral and variant SARS-CoV-2 strains. Sci. Transl. Med. 14, eabm3302 (2022).
Scheaffer, S. M. et al. Bivalent SARS-CoV-2 mRNA vaccines increase breadth of neutralization and protect against the BA.5 Omicron variant in mice. Nat. Med. 29, 247–257 (2023).
Netea, M. G., Schlitzer, A., Placek, K., Joosten, L. A. B. & Schultze, J. L. Innate and adaptive immune memory: an evolutionary continuum in the host’s response to pathogens. Cell Host Microbe 25, 13–26 (2019).
Primorac, D. et al. Adaptive immune responses and immunity to SARS-CoV-2. Front. Immunol. 13, 848582 (2022).
Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 23, 186–193 (2022).
Sette, A., Sidney, J. & Crotty, S. T cell responses to SARS-CoV-2. Annu. Rev. Immunol. 41, 343–373 (2023).
Almendro-Vazquez, P., Laguna-Goya, R. & Paz-Artal, E. Defending against SARS-CoV-2: the T cell perspective. Front. Immunol. 14, 1107803 (2023).
Altmann, D. M. & Boyton, R. J. SARS-CoV-2 T cell immunity: Specificity, function, durability, and role in protection. Sci. Immunol. 5, https://doi.org/10.1126/sciimmunol.abd6160 (2020).
Reynolds, C. J. et al. Strong CD4 T cell responses to Zika virus antigens in a cohort of Dengue virus immune mothers of congenital Zika virus syndrome infants. Front. Immunol. 11, 185 (2020).
Yin, K. et al. Long COVID manifests with T cell dysregulation, inflammation and an uncoordinated adaptive immune response to SARS-CoV-2. Nat. Immunol. 25, 218–225 (2024).
Sette, A. & Crotty, S. Adaptive immunity to SARS-CoV-2 and COVID-19. Cell 184, 861–880 (2021).
Vardhana, S., Baldo, L., Morice, W. G. 2nd & Wherry, E. J. Understanding T cell responses to COVID-19 is essential for informing public health strategies. Sci. Immunol. 7, eabo1303 (2022).
Grifoni, A. et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 181, 1489–1501.e1415 (2020).
Tarke, A. et al. Comprehensive analysis of T cell immunodominance and immunoprevalence of SARS-CoV-2 epitopes in COVID-19 cases. Cell Rep. Med. 2, 100204 (2021).
Heitmann, J. S. et al. A COVID-19 peptide vaccine for the induction of SARS-CoV-2 T cell immunity. Nature 601, 617–622 (2022).
Addetia, A. et al. Neutralization, effector function and immune imprinting of Omicron variants. Nature 621, 592–601 (2023).
Arieta, C. M. et al. The T-cell-directed vaccine BNT162b4 encoding conserved non-spike antigens protects animals from severe SARS-CoV-2 infection. Cell 186, 2392–2409.e2321 (2023).
Arts, R. J. W. et al. BCG vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe 23, 89–100.e105 (2018).
Walk, J. et al. Outcomes of controlled human malaria infection after BCG vaccination. Nat. Commun. 10, 874 (2019).
Ciarlo, E. et al. Trained immunity confers broad-spectrum protection against bacterial infections. J. Infect. Dis. 222, 1869–1881 (2020).
Kalafati, L. et al. Innate immune training of granulopoiesis promotes anti-tumor activity. Cell 183, 771–785.e712 (2020).
Koeken, V., Verrall, A. J., Netea, M. G., Hill, P. C. & van Crevel, R. Trained innate immunity and resistance to Mycobacterium tuberculosis infection. Clin. Microbiol. Infect. 25, 1468–1472 (2019).
Sun, W. et al. The self-assembled nanoparticle-based trimeric RBD mRNA vaccine elicits robust and durable protective immunity against SARS-CoV-2 in mice. Signal Transduct. Target Ther. 6, 340 (2021).
Tai, W. et al. A novel receptor-binding domain (RBD)-based mRNA vaccine against SARS-CoV-2. Cell Res. 30, 932–935 (2020).
Yang, J. et al. A vaccine targeting the RBD of the S protein of SARS-CoV-2 induces protective immunity. Nature 586, 572–577 (2020).
Liang, Q. et al. RBD trimer mRNA vaccine elicits broad and protective immune responses against SARS-CoV-2 variants. iScience 25, 104043 (2022).
Zhao, H. et al. Long-term stability and protection efficacy of the RBD-targeting COVID-19 mRNA vaccine in nonhuman primates. Signal Transduct. Target Ther. 6, 438 (2021).
Uraki, R. et al. An mRNA vaccine encoding the SARS-CoV-2 receptor-binding domain protects mice from various Omicron variants. NPJ Vaccines 9, 4 (2024).
Yarmarkovich, M., Warrington, J. M., Farrel, A. & Maris, J. M. Identification of SARS-CoV-2 vaccine epitopes predicted to induce long-term population-scale immunity. Cell Rep. Med. 1, 100036 (2020).
Pan, K. et al. Mass spectrometric identification of immunogenic SARS-CoV-2 epitopes and cognate TCRs. Proc. Natl Acad. Sci. USA 118, https://doi.org/10.1073/pnas.2111815118 (2021).
Saini, S. K. et al. SARS-CoV-2 genome-wide T cell epitope mapping reveals immunodominance and substantial CD8(+) T cell activation in COVID-19 patients. Sci. Immunol. 6, https://doi.org/10.1126/sciimmunol.abf7550 (2021).
Hare, J. et al. Direct identification of HLA-presented CD8 T cell epitopes from transmitted founder HIV-1 variants. Proteomics 21, e2100142 (2021).
Mohan, T., Sharma, C., Bhat, A. A. & Rao, D. N. Modulation of HIV peptide antigen specific cellular immune response by synthetic alpha- and beta-defensin peptides. Vaccine 31, 1707–1716 (2013).
Naz, A. et al. Designing multi-epitope vaccines to combat emerging coronavirus disease 2019 (COVID-19) by employing immuno-informatics approach. Front. Immunol. 11, 1663 (2020).
Verbeke, R., Lentacker, I., De Smedt, S. C. & Dewitte, H. The dawn of mRNA vaccines: the COVID-19 case. J. Control Rel. 333, 511–520 (2021).
Park, J. W., Lagniton, P. N. P., Liu, Y. & Xu, R. H. mRNA vaccines for COVID-19: what, why and how. Int. J. Biol. Sci. 17, 1446–1460 (2021).
Tarke, A. et al. SARS-CoV-2 vaccination induces immunological T cell memory able to cross-recognize variants from Alpha to Omicron. Cell 185, 847–859.e811 (2022).
Steiner, S. et al. Reactive T cells in convalescent COVID-19 patients with negative SARS-CoV-2 antibody serology. Front. Immunol. 12, 687449 (2021).
Thieme, C. J. et al. Robust T cell response toward spike, membrane, and nucleocapsid SARS-CoV-2 proteins is not associated with recovery in critical COVID-19 patients. Cell Rep. Med. 1, 100092 (2020).
Zhang, Z. et al. Humoral and cellular immune memory to four COVID-19 vaccines. Cell 185, 2434–2451.e2417 (2022).
Laidlaw, B. J. & Ellebedy, A. H. The germinal centre B cell response to SARS-CoV-2. Nat. Rev. Immunol. 22, 7–18 (2022).
Inoue, T., Shinnakasu, R. & Kurosaki, T. Generation of high quality memory B cells. Front. Immunol. 12, 825813 (2021).
Akkaya, M., Kwak, K. & Pierce, S. K. B cell memory: building two walls of protection against pathogens. Nat. Rev. Immunol. 20, 229–238 (2020).
Baccin, C. et al. Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 22, 38–48 (2020).
Blank, U. & Karlsson, S. TGF-beta signaling in the control of hematopoietic stem cells. Blood 125, 3542–3550 (2015).
Molnarfi, N., Bjarnadottir, K., Benkhoucha, M., Juillard, C. & Lalive, P. H. Activation of human B cells negatively regulates TGF-beta1 production. J. Neuroinflammation 14, 13 (2017).
Regis, S., Dondero, A., Caliendo, F., Bottino, C. & Castriconi, R. NK cell function regulation by TGF-beta-induced epigenetic mechanisms. Front. Immunol. 11, 311 (2020).
Tamayo, E., Alvarez, P. & Merino, R. TGFbeta superfamily members as regulators of B cell development and function-implications for autoimmunity. Int. J. Mol. Sci. 19, https://doi.org/10.3390/ijms19123928 (2018).
Staal, F. J. & Sen, J. M. The canonical Wnt signaling pathway plays an important role in lymphopoiesis and hematopoiesis. Eur. J. Immunol. 38, 1788–1794 (2008).
Haseeb, M., Pirzada, R. H., Ain, Q. U. & Choi, S. Wnt signaling in the regulation of immune cell and cancer therapeutics. Cells 8, https://doi.org/10.3390/cells8111380 (2019).
Bertoli, C., Skotheim, J. M. & de Bruin, R. A. Control of cell cycle transcription during G1 and S phases. Nat. Rev. Mol. Cell Biol. 14, 518–528 (2013).
Duronio, R. J. & Xiong, Y. Signaling pathways that control cell proliferation. Cold Spring Harb. Perspect. Biol. 5, a008904 (2013).
Dalton, S. Linking the cell cycle to cell fate decisions. Trends Cell Biol. 25, 592–600 (2015).
Liu, L., Michowski, W., Kolodziejczyk, A. & Sicinski, P. The cell cycle in stem cell proliferation, pluripotency and differentiation. Nat. Cell Biol. 21, 1060–1067 (2019).
Whately, K. M. et al. Spon1+ inflammatory monocytes promote collagen remodeling and lung cancer metastasis through lipoprotein receptor 8 signaling. JCI Insight 9, https://doi.org/10.1172/jci.insight.168792 (2024).
Soriano-Romani, L., Contreras-Ruiz, L., Lopez-Garcia, A., Diebold, Y. & Masli, S. Topical application of TGF-beta-activating peptide, KRFK, prevents inflammatory manifestations in the TSP-1-deficient mouse model of chronic ocular inflammation. Int. J. Mol. Sci. 20, https://doi.org/10.3390/ijms20010009 (2018).
Wu, H. et al. The development of a novel transforming growth factor-beta (TGF-beta) inhibitor that disrupts ligand-receptor interactions. Eur. J. Med. Chem. 189, 112042 (2020).
Wen, J. et al. Pharmacological suppression of glycogen synthase kinase-3 reactivates HIV-1 from latency via activating Wnt/beta-catenin/TCF1 axis in CD4(+) T cells. Emerg. Microbes Infect. 11, 391–405 (2022).
Huang, X., Zhong, L., Hendriks, J., Post, J. N. & Karperien, M. The effects of the WNT-signaling modulators BIO and PKF118-310 on the chondrogenic differentiation of human mesenchymal stem cells. Int. J. Mol. Sci. 19, https://doi.org/10.3390/ijms19020561 (2018).
Hwang, S. Y. et al. Direct targeting of beta-catenin by a small molecule stimulates proteasomal degradation and suppresses oncogenic Wnt/beta-catenin signaling. Cell Rep. 16, 28–36 (2016).
Netea, M. G. et al. Defining trained immunity and its role in health and disease. Nat. Rev. Immunol. 20, 375–388 (2020).
Almas, T. et al. Epidemiology, clinical ramifications, and cellular pathogenesis of COVID-19 mRNA-vaccination-induced adverse cardiovascular outcomes: a state-of-the-heart review. Biomed. Pharmacother. 149, 112843 (2022).
Trougakos, I. P. et al. Adverse effects of COVID-19 mRNA vaccines: the spike hypothesis. Trends Mol. Med. 28, 542–554 (2022).
Ndeupen, S. et al. The mRNA-LNP platform’s lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. bioRxiv, https://doi.org/10.1101/2021.03.04.430128 (2021).
Cosentino, M. & Marino, F. The spike hypothesis in vaccine-induced adverse effects: questions and answers. Trends Mol. Med. 28, 797–799 (2022).
Kuhn, C. C. et al. Direct Cryo-ET observation of platelet deformation induced by SARS-CoV-2 spike protein. Nat. Commun. 14, 620 (2023).
Amini, A. et al. MAIT and other innate-like T cells integrate adaptive immune responses to modulate interval-dependent reactogenicity to mRNA vaccines. Sci. Immunol. 10, eadu3337 (2025).
Acknowledgements
We thank Dr. Kristen Jepsen of the Institute of Genomic Medicine at UCSD for help with scRNA-seq, Dr. Neal Sekiya and Ms. Tara Rambled at the Center for AIDS Research at UCSD for flow cytometry analysis, and members of the Rana lab for helpful discussions and advice. This publication includes data generated at the UC San Diego IGM Genomics Center utilizing an Illumina NovaSeq 6000 purchased with funding from a National Institutes of Health SIG grant (#S10 OD026929). This work was supported in part by institutional funds and from grants from the National Institutes of Health (AI125103, CA177322, CA030199, DA046171).
Author information
Authors and Affiliations
Contributions
J.W. designed and performed experiments, analyzed data, and wrote the manuscript draft; J.M. designed and performed experiments, and analyzed data; L.T. and S.B. analyzed the scRNA-seq data; A.S., R.M., J.T., and L.W. performed experiments; D.M.S. provided reagents, S.S. and K.G. participated in experimental design, data analysis and interpretation; T.M.R. conceived and planned the project and participated in experimental design, data analysis, data interpretation, and manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wen, J., Moon, J., Tucciarone, L. et al. A new mRNA antigen vaccine induces potent B and T cell responses and in vivo protection against SARS-CoV-2. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01421-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01421-z