Abstract
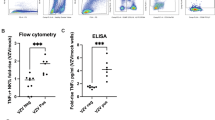
Assessing the functional activity of vaccine-induced antibodies is critical for evaluating immunogenicity. We developed and validated antibody-dependent cellular phagocytosis (ADCP) and antibody-dependent cellular cytotoxicity (ADCC) assays to quantify Fc-mediated antibody responses elicited by varicella and zoster vaccines. Both assays demonstrated robust performance and broad linearity. Antibody titers were measured using fluorescent antibody to membrane antigen (FAMA) and ELISA. ADCP and ADCC activities, along with FAMA and ELISA geometric mean titers (GMTs), were significantly increased in post- versus pre-vaccination sera (p < 0.0001). Strong correlations were observed between ADCP and ADCC activities and both FAMA and ELISA GMTs. Although children exhibited lower total varicella-zoster virus-specific IgG levels than adults, higher IgG3 subclass levels in children were associated with comparable Fc-mediated activities. These results highlight the utility of ADCP and ADCC as valuable assays for evaluating Fc-mediated antibody function and as potential surrogates of protective immunity to varicella and zoster vaccination.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request. This study did not generate or analyze datasets that require deposition in a public repository.
References
Haumont, M. et al. Neutralizing antibody responses induced by varicella-zoster virus gE and gB glycoproteins following infection, reactivation or immunization. J. Med. Virol. 53, 63–68 (1997).
Asada, H. VZV-specific cell-mediated immunity, but not humoral immunity, correlates inversely with the incidence of herpes zoster and the severity of skin symptoms and zoster-associated pain: The SHEZ study. Vaccine 37, 6776–6781 (2019).
Levin, M. J. et al. Varicella-zoster virus-specific immune responses in elderly recipients of a herpes zoster vaccine. J. Infect. Dis. 197, 825–835 (2008).
Good, R. A. & Zak, S. J. Disturbances in gamma globulin synthesis as experiments of nature. Pediatrics 18, 109–149 (1956).
Hayashida, K. et al. Evaluation of varicella zoster virus-specific cell-mediated immunity by using an interferon-gamma enzyme-linked immunosorbent assay. J. Immunol. Methods 426, 50–55 (2015).
Banovic, T. et al. Disseminated varicella infection caused by varicella vaccine strain in a child with low invariant natural killer T cells and diminished CD1d expression. J. Infect. Dis. 204, 1893–1901 (2011).
Etzioni, A. et al. Fatal varicella associated with selective natural killer cell deficiency. J. Pediatr. 146, 423–425 (2005).
Levy, O. et al. Disseminated varicella infection due to the vaccine strain of varicella-zoster virus, in a patient with a novel deficiency in natural killer T cells. J. Infect. Dis. 188, 948–953 (2003).
Vossen, M. T. et al. Absence of circulating natural killer and primed CD8+ cells in life-threatening varicella. J. Infect. Dis. 191, 198–206 (2005).
Goldblatt, D., Alter, G., Crotty, S. & Plotkin, S. A. Correlates of protection against SARS-CoV-2 infection and COVID-19 disease. Immunol. Rev. 310, 6–26 (2022).
Richardson, S. I. et al. SARS-CoV-2 Omicron triggers cross-reactive neutralization and Fc effector functions in previously vaccinated, but not unvaccinated, individuals. Cell Host Microbe 30, 880–886.e884 (2022).
Dufloo, J. et al. Asymptomatic and symptomatic SARS-CoV-2 infections elicit polyfunctional antibodies. Cell Rep. Med 2, 100275 (2021).
Felber, B. K. et al. Co-immunization of DNA and protein in the same anatomical sites induces superior protective immune responses against SHIV challenge. Cell Rep. 31, 107624 (2020).
Keshwara, R. et al. A recombinant Rabies virus expressing the Marburg virus glycoprotein is dependent upon antibody-mediated cellular cytotoxicity for protection against Marburg virus disease in a murine model. J. Virol. 93, e01865–01818 (2019).
Chen, D. et al. Reduced neutralization and Fc effector function to Omicron subvariants in sera from SARS-CoV-1 survivors after two doses of CoronaVac plus one dose subunit vaccine. J. Med. Virol. 95, e29136 (2023).
Bouayad, A. Multifaceted roles of Fcgamma receptors in COVID-19 and vaccine responses. Am. J. Transl. Res 15, 3040–3059 (2023).
Diez, J. M. et al. Anti-severe acute respiratory syndrome coronavirus 2 hyperimmune immunoglobulin demonstrates potent neutralization and antibody-dependent cellular cytotoxicity and phagocytosis through N and S proteins. J. Infect. Dis. 225, 938–946 (2022).
Park, S. Y. et al. Development of antibody-dependent cellular cytotoxicity in response to recombinant and live-attenuated herpes zoster vaccines. NPJ Vaccines 7, 123 (2022).
Zhang, Y., Hoppe, A. D. & Swanson, J. A. Coordination of Fc receptor signaling regulates cellular commitment to phagocytosis. Proc. Natl. Acad. Sci. USA. 107, 19332–19337 (2010).
Watzl, C. & Long, E. O. Signal transduction during activation and inhibition of natural killer cells. Curr. Protoc. Immunol. Chapter 11, 11–19B (2010).
Petro, C. D. et al. HSV-2 DeltagD elicits FcgammaR-effector antibodies that protect against clinical isolates. JCI Insight 1, e88529 (2016).
Dias, A. G. Jr. et al. Antibody Fc characteristics and effector functions correlate with protection from symptomatic dengue virus type 3 infection. Sci. Transl. Med. 14, eabm3151 (2022).
Zohar, T. et al. Upper and lower respiratory tract correlates of protection against respiratory syncytial virus following vaccination of nonhuman primates. Cell Host Microbe 30, 41–52.e45 (2022).
Bartsch, Y. C. et al. Antibody effector functions are associated with protection from respiratory syncytial virus. Cell 185, 4873–4886.e4810 (2022).
Richardson, S. I. et al. SARS-CoV-2 Beta and Delta variants trigger Fc effector function with increased cross-reactivity. Cell Rep. Med. 3, 100510 (2022).
Geers, D. et al. Ad26.COV2.S priming provided a solid immunological base for mRNA-based COVID-19 booster vaccination. iScience 26, 105753 (2023).
Adeniji, O. S. et al. COVID-19 severity is associated with differential antibody Fc-mediated innate immune functions. mBio 12, e00281–00221 (2021).
Paquin-Proulx, D. et al. Associations between antibody Fc-mediated effector functions and long-term sequelae in Ebola virus survivors. Front. Immunol. 12, 682120 (2021).
Henry Dunand, C. J. et al. Both neutralizing and non-neutralizing human H7N9 influenza vaccine-induced monoclonal antibodies confer protection. Cell Host Microbe 19, 800–813 (2016).
Richardson, S. I. et al. IgG3 enhances neutralization potency and Fc effector function of an HIV V2-specific broadly neutralizing antibody. PLoS Pathog. 15, e1008064 (2019).
Sundqvist, V. A., Linde, A. & Wahren, B. Virus-specific immunoglobulin G subclasses in herpes simplex and varicella-zoster virus infections. J. Clin. Microbiol. 20, 94–98 (1984).
Lee, Y. H. et al. Global varicella vaccination programs. Clin. Exp. Pediatr. 65, 555–562 (2022).
Kawai, K., Gebremeskel, B. G. & Acosta, C. J. Systematic review of incidence and complications of herpes zoster: towards a global perspective. BMJ Open 4, e004833 (2014).
de Jong, M. D., Weel, J. F., Schuurman, T., Wertheim-van Dillen, P. M. & Boom, R. Quantitation of varicella-zoster virus DNA in whole blood, plasma, and serum by PCR and electrochemiluminescence. J. Clin. Microbiol. 38, 2568–2573 (2000).
Arvin, A. M., Moffat, J. F. & Redman, R. Varicella-zoster virus: aspects of pathogenesis and host response to natural infection and varicella vaccine. Adv. Virus Res. 46, 263–309 (1996).
Burel, J. G., Apte, S. H., Groves, P. L., McCarthy, J. S. & Doolan, D. L. Polyfunctional and IFN-gamma monofunctional human CD4(+) T cell populations are molecularly distinct. JCI Insight 2, e87499 (2017).
Freen-van Heeren, J. J. et al. Assessing antigen-specific T cell responses through IFN-gamma Enzyme-Linked Immune Absorbent Spot (ELISpot). Methods Mol. Biol. 2782, 209–226 (2024).
Laing, K. J. et al. Zoster vaccination increases the breadth of CD4+ T cells responsive to varicella zoster virus. J. Infect. Dis. 212, 1022–1031 (2015).
Weinberg, A. et al. Varicella-zoster virus-specific cellular immune responses to the live attenuated zoster vaccine in young and older adults. J. Immunol. 199, 604–612 (2017).
Cunningham, A. L. et al. Immune responses to a recombinant glycoprotein E herpes zoster vaccine in adults aged 50 years or older. J. Infect. Dis. 217, 1750–1760 (2018).
Asanuma, H., Sharp, M., Maecker, H. T., Maino, V. C. & Arvin, A. M. Frequencies of memory T cells specific for varicella-zoster virus, herpes simplex virus, and cytomegalovirus by intracellular detection of cytokine expression. J. Infect. Dis. 181, 859–866 (2000).
Atyeo, C. et al. Distinct early serological signatures track with SARS-CoV-2 survival. Immunity 53, 524–532.e524 (2020).
Barouch, D. H. et al. Protective efficacy of a global HIV-1 mosaic vaccine against heterologous SHIV challenges in rhesus monkeys. Cell 155, 531–539 (2013).
Richardson, S. I. et al. HIV-specific Fc effector function early in infection predicts the development of broadly neutralizing antibodies. PLoS Pathog. 14, e1006987 (2018).
Ullah, I. et al. Live imaging of SARS-CoV-2 infection in mice reveals that neutralizing antibodies require Fc function for optimal efficacy. Immunity 54, 2143–2158.e2115 (2021).
Winkler, E. S. et al. Human neutralizing antibodies against SARS-CoV-2 require intact Fc effector functions for optimal therapeutic protection. Cell 184, 1804–1820.e1816 (2021).
Zohar, T. et al. Compromised humoral functional evolution tracks with SARS-CoV-2 mortality. Cell 183, 1508–1519.e1512 (2020).
Ihara, T. et al. Antibody response determined with antibody-dependent cell-mediated cytotoxicity (ADCC), neutralizing antibody, and varicella skin test in children with natural varicella and after varicella immunization. Acta Paediatr. Jpn. 33, 43–49 (1991).
Asano, Y. et al. Immunoglobulin subclass antibodies to varicella-zoster virus. Pediatrics 80, 933–936 (1987).
Jounai, N. et al. Age-specific profiles of antibody responses against respiratory syncytial virus infection. EBioMedicine 16, 124–135 (2017).
Bruhns, P. Properties of mouse and human IgG receptors and their contribution to disease models. Blood 119, 5640–5649 (2012).
Chung, A. W. et al. Polyfunctional Fc-effector profiles mediated by IgG subclass selection distinguish RV144 and VAX003 vaccines. Sci. Transl. Med. 6, 228–238 (2014).
Lim, J., Kim, Y., Hwang, J. Y., Lee, K. M. & Park, H. Evaluation of flow cytometry-based fluorescent antibody to membrane antigen test to measure the humoral immunity against the varicella-zoster virus. Heliyon 10, e36614 (2024).
Pollara, J. et al. Application of area scaling analysis to identify natural killer cell and monocyte involvement in the GranToxiLux antibody dependent cell-mediated cytotoxicity assay. Cytom. A 93, 436–447 (2018).
Parekh, B. S. et al. Development and validation of an antibody-dependent cell-mediated cytotoxicity-reporter gene assay. MAbs 4, 310–318 (2012).
Yang, F. et al. Validation of an IFN-gamma ELISpot assay to measure cellular immune responses against viral antigens in non-human primates. Gene Ther. 29, 41–54 (2022).
Butler, A. L., Fallon, J. K. & Alter, G. A Sample-Sparing Multiplexed ADCP Assay. Front. Immunol. 10, 1851 (2019).
European Medicines, A. ICH Q2(R2) Validation of analytical procedures scientific guideline. (2023).
Raimo, M., Zavoianu, A. G., Meijs, W., Scholten, P. & Spanholtz, J. Qualification of a flow cytometry-based method for the evaluation of in vitro cytotoxicity of GTA002 natural killer cell therapy. Heliyon 10, e24715 (2024).
Hwang, J. Y. et al. Cross-reactive humoral immunity of clade 2 Oka and MAV/06 strain-based varicella vaccines against different clades of varicella-zoster virus. Hum. Vaccin. Immunother. 19, 2210961 (2023).
Acknowledgements
This study was supported by grant 23202MFDS136 (2023) from the Ministry of Food and Drug Safety, Republic of Korea. The funding agency had no role in the study design; data collection, analysis, or interpretation; manuscript preparation; or the decision to publish.
Author information
Authors and Affiliations
Contributions
S.X., J.Y.H., H.S.L., O.S.S., J.Y.N., S.H.H., and H.P. conceived and designed the study. S.X., J.Y.H., Y.K., K.M.L., E.J.J., S.T.C., J.S.L., J.Y.B., K.K., B.Y., and J.H.L. performed the experiments and collected the data. S.X. and Y.K. analyzed the data. S.X., J.Y.H., Y.K., and H.P. drafted the manuscript, which was critically reviewed by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xayaheuang, S., Hwang, JY., Kim, Y. et al. Evaluating varicella-zoster virus vaccine immunogenicity through Fc-mediated antibody functions: the roles of ADCP and ADCC. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01424-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01424-w