Abstract
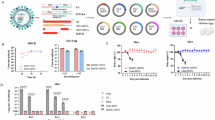
Seasonal influenza viruses evade immunity through antigenic drift, enabling escape from inhibitory antibodies targeting hemagglutinin (HA) and neuraminidase (NA). In this study, we evaluated a non-propagating vesicular stomatitis virus (VSV) vector encoding HA and NA antigens of A/Hamburg/4/2009 (H1N1) in a porcine animal model to assess induction of cross-reactive immunity. A single intramuscular immunization elicited high titers of H1N1-neutralizing antibodies and potent N1-sialidase inhibition. A boost with the same single-cycle VSV-vectored H1/N1 antigens or with a live-attenuated influenza vaccine (LAIV) enhanced inhibitory activity against the antigen-drifted A/Victoria/2570/2019 (H1N1) strain. Vaccination induced N1-specific antibodies that also inhibited avian N1 sialidase and suppressed replication of a bovine-derived H5N1 virus in vitro. Following nasal challenge with a 6:2 reassortant virus encoding drifted HA and NA antigens, vaccinated pigs showed no detectable virus shedding, whereas control animals shed infectious virus. Homologous prime-boost vaccination with the VSV-vectored H1/N1 antigens conferred protection comparable to the heterologous VSV/LAIV regimen in the upper respiratory tract. These findings demonstrate that a single-cycle VSV vector encoding both HA and NA induces cross-protective immunity against antigen-drifted influenza viruses, reduces the risk of vaccine mismatch, and may limit infection by zoonotic H5N1 viruses.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files. The datasets generated and/or analyzed during the current study are available in the Zenodo repository [https://doi.org/10.5281/zenodo.17302034]. The nucleotide sequences of the A/cattle/Texas/063224-24-1/2024 (H5N1) genome segments are deposited at the GenBank database under the following accession numbers: PX831491, PX831492, PX831493, [PX831494], [PX831495], [PX831496], [PX831497], [PX831498]. The cDNAs encoding genome segments 4 and 6 of A/Victoria/2570/2019 (H1N1) are deposited at the GenBank data bank under accession numbers [OQ719015] and OQ719014, respectively. The sequences of the eight genome segments of A/bovine/Texas/24-029328-02/2024 (H5N1) are deposited at GenBank under the following accession numbers: [PP599470], [PP599471], [PP599472], [PP599473], [PP599474], [PP599475], [PP599476], [PP599477]. The HA amino acid sequences of A/Hamburg/4/2009 (H1N1) and A/Victoria/2570/2019 (H1N1) are deposited at the GenBank database under the accession numbers ACR10223 and WEY08940, respectively. The NA amino acid sequences of A/Hamburg/4/2009 (H1N1) and A/Victoria/2570/2019 (H1N1) are deposited at the GenBank database under the accession numbers ACR10227 and WEY08939, respectively.
References
Wu, N. C. & Wilson, I. A. A perspective on the structural and functional constraints for immune evasion: insights from influenza virus. J. Mol. Biol. 429, 2694–2709 (2017).
Wu, N. C. & Wilson, I. A. Influenza hemagglutinin structures and antibody recognition. Cold Spring Harb. Perspect. Med. 10, ea038778 (2020).
Kim, H., Webster, R. G. & Webby, R. J. Influenza virus: dealing with a drifting and shifting pathogen. Viral Immunol. 31, 174–183 (2018).
Morimoto, N. & Takeishi, K. Change in the efficacy of influenza vaccination after repeated inoculation under antigenic mismatch: a systematic review and meta-analysis. Vaccine 36, 949–957 (2018).
Russell, C. A. et al. Seasonal influenza vaccine performance and the potential benefits of mRNA vaccines. Hum. Vaccin. Immunother. 20, 2336357 (2024).
Palese, P., Tobita, K., Ueda, M. & Compans, R. W. Characterization of temperature sensitive influenza virus mutants defective in neuraminidase. Virology 61, 397–410 (1974).
Matrosovich, M. N., Matrosovich, T. Y., Gray, T., Roberts, N. A. & Klenk, H. D. Neuraminidase is important for the initiation of influenza virus infection in human airway epithelium. J. Virol. 78, 12665–12667 (2004).
Tan, J. et al. Human anti-neuraminidase antibodies reduce airborne transmission of clinical influenza virus isolates in the guinea pig model. J. Virol. 96, e0142121 (2022).
Stadlbauer, D. et al. Broadly protective human antibodies that target the active site of influenza virus neuraminidase. Science 366, 499–504 (2019).
Halbherr, S. J. et al. Biological and protective properties of immune sera directed to the influenza virus neuraminidase. J. Virol. 89, 1550–1563 (2015).
Gao, J. et al. Antigenic drift of the influenza A(H1N1)pdm09 virus neuraminidase results in reduced effectiveness of A/California/7/2009 (H1N1pdm09)-specific antibodies. mBio 10, e00307-19 (2019).
Sandbulte, M. R. et al. Discordant antigenic drift of neuraminidase and hemagglutinin in H1N1 and H3N2 influenza viruses. Proc. Natl. Acad. Sci. USA 108, 20748–20753 (2011).
Wan, H. et al. The neuraminidase of A(H3N2) influenza viruses circulating since 2016 is antigenically distinct from the A/Hong Kong/4801/2014 vaccine strain. Nat. Microbiol. 4, 2216–2225 (2019).
Giurgea, L. T., Morens, D. M., Taubenberger, J. K. & Memoli, M. J. Influenza neuraminidase: a neglected protein and its potential for a better influenza vaccine. Vaccines 8, 409 (2020).
Avanthay, R., Garcia-Nicolas, O., Zimmer, G. & Summerfield, A. NS1 and PA-X of H1N1/09 influenza virus act in a concerted manner to manipulate the innate immune response of porcine respiratory epithelial cells. Front Cell Infect. Microbiol 13, 1222805 (2023).
Avanthay, R. et al. Evaluation of a novel intramuscular prime/intranasal boost vaccination strategy against influenza in the pig model. PLoS Pathog. 20, e1012393 (2024).
World Health Organization (WHO). Recommended composition of influenza virus vaccines for use in the 2021 southern hemisphere influenza season. Available at: https://cdn.who.int/media/docs/default-source/emergency-preparedness/global-influenza-programme/recommended-composition-of-influenza-virus-vaccines/202009-recommendation.pdf?sfvrsn=1331fa94_2&download=true (2020).
European Medicines Agency (EMA). EU recommendations for 2022-2023 seasonal flu vaccine composition. Available at: https://www.ema.europa.eu/en/news/eu-recommendations-2022-2023-seasonal-flu-vaccine-composition?utm_source=chatgpt.com (2022).
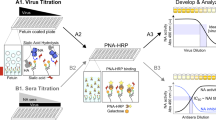
Couzens, L. et al. An optimized enzyme-linked lectin assay to measure influenza A virus neuraminidase inhibition antibody titers in human sera. J. Virol. Methods 210, 7–14 (2014).
Lambre, C. R., Terzidis, H., Greffard, A. & Webster, R. G. Measurement of anti-influenza neuraminidase antibody using a peroxidase-linked lectin and microtitre plates coated with natural substrates. J. Immunol. Methods 135, 49–57 (1990).
Straus, M. R. & Whittaker, G. R. A peptide-based approach to evaluate the adaptability of influenza A virus to humans based on its hemagglutinin proteolytic cleavage site. PLoS ONE 12, e0174827 (2017).
Holzer, B., Martini, V., Edmans, M. & Tchilian, E. T and B Cell Immune Responses to Influenza Viruses in Pigs. Front. Immunol. 10, 98 (2019).
Vandoorn, E. et al. Pathobiology of an NS1-truncated H3N2 swine influenza virus strain in pigs. J. Virol. 96, e0051922 (2022).
Hatta, M. et al. An influenza mRNA vaccine protects ferrets from lethal infection with highly pathogenic avian influenza A(H5N1) virus. Sci. Transl. Med. 16, eads1273 (2024).
Henry, B. & Laidlaw, B. J. Functional heterogeneity in the memory B-cell response. Curr. Opin. Immunol. 80, 102281 (2023).
Lam, N., Lee, Y. & Farber, D. L. A guide to adaptive immune memory. Nat. Rev. Immunol. 24, 810–829 (2024).
Palgen, J. L. et al. Optimize prime/boost vaccine strategies: trained immunity as a new player in the game. Front. Immunol. 12, 612747 (2021).
Renegar, K. B., Small, P. A. Jr., Boykins, L. G. & Wright, P. F. Role of IgA versus IgG in the control of influenza viral infection in the murine respiratory tract. J. Immunol. 173, 1978–1986 (2004).
Spiekermann, G. M. et al. Receptor-mediated immunoglobulin G transport across mucosal barriers in adult life: functional expression of FcRn in the mammalian lung. J. Exp. Med. 196, 303–310 (2002).
Graaf-Rau, A. et al. Reassortment incompetent live attenuated and replicon influenza vaccines provide improved protection against influenza in piglets. NPJ Vaccines 9, 127 (2024).
Cline, B. L. Ecological associations of vesicular stomatitis virus in rural Central America and Panama. Am. J. Trop. Med. Hyg. 25, 875–883 (1976).
Haynes, E. et al. Surveillance of feral swine (Sus scrofa) in the Western USA for antibodies to vesicular stomatitis virus, 2013-21. J. Wildl. Dis. 60, 1011–1015 (2024).
Creytens, S., Pascha, M. N., Ballegeer, M., Saelens, X. & de Haan, C. A. M. Influenza neuraminidase characteristics and potential as a vaccine target. Front. Immunol. 12, 786617 (2021).
Palese, P. & Compans, R. W. Inhibition of influenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA): mechanism of action. J. Gen. Virol. 33, 159–163 (1976).
Air, G. M. Influenza neuraminidase. Influenza Other Respir. Viruses 6, 245–256 (2012).
Ehre, C. et al. Overexpressing mouse model demonstrates the protective role of Muc5ac in the lungs. Proc. Natl. Acad. Sci. USA 109, 16528–16533 (2012).
Zanin, M., Baviskar, P., Webster, R. & Webby, R. The Interaction between Respiratory Pathogens and Mucus. Cell Host Microbe 19, 159–168 (2016).
Schulman, J. L., Khakpour, M. & Kilbourne, E. D. Protective effects of specific immunity to viral neuraminidase on influenza virus infection of mice. J. Virol. 2, 778–786 (1968).
Kilbourne, E. D. et al. Protection of mice with recombinant influenza virus neuraminidase. J. Infect. Dis. 189, 459–461 (2004).
Sandbulte, M. R. et al. Cross-reactive neuraminidase antibodies afford partial protection against H5N1 in mice and are present in unexposed humans. PLoS Med. 4, e59 (2007).
Krammer, F., Li, L. & Wilson, P. C. Emerging from the Shadow of Hemagglutinin: Neuraminidase Is an Important Target for Influenza Vaccination. Cell Host Microbe 26, 712–713 (2019).
Walz, L., Kays, S. K., Zimmer, G. & von Messling, V. Neuraminidase-inhibiting antibody titers correlate with protection from heterologous influenza virus strains of the same neuraminidase subtype. J. Virol. 92, e01006-18 (2018).
Peacock, T. P. et al. The global H5N1 influenza panzootic in mammals. Nature 637, 304–313 (2025).
Mostafa, A. et al. Avian influenza A (H5N1) virus in dairy cattle: origin, evolution, and cross-species transmission. mBio 15, e0254224 (2024).
Garg, S. et al. Highly pathogenic avian influenza A(H5N1) virus infections in humans. N. Engl. J. Med. 392, 843–854 (2025).
Belser, J. A. et al. Ocular infectivity and replication of a clade 2.3.4.4b A(H5N1) influenza virus associated with human conjunctivitis in a dairy farm worker in the USA: an in-vitro and ferret study. Lancet Microbe 6, 101070 (2025).
Chen, Y. Q. et al. Influenza infection in humans induces broadly cross-reactive and protective neuraminidase-reactive antibodies. Cell 173, 417–429.e410 (2018).
Hansen, L. et al. Human anti-N1 monoclonal antibodies elicited by pandemic H1N1 virus infection broadly inhibit HxN1 viruses in vitro and in vivo. Immunity 56, 1927–1938.e1928 (2023).
Le Sage, V. et al. Influenza A(H5N1) immune response among ferrets with influenza A(H1N1)pdm09 immunity. Emerg. Infect. Dis. 31, 477–487 (2025).
Sun, X. et al. Effect of prior influenza A(H1N1)pdm09 virus infection on pathogenesis and transmission of human influenza A(H5N1) Clade 2.3.4.4b virus in ferret model. Emerg. Infect. Dis. 31, 458–466 (2025).
Doyon-Plourde, P., Przepiorkowski, J., Young, K., Zhao, L. & Sinilaite, A. Intraseasonal waning immunity of seasonal influenza vaccine - a systematic review and meta-analysis. Vaccine 41, 4462–4471 (2023).
Sasaki, S. et al. Limited efficacy of inactivated influenza vaccine in elderly individuals is associated with decreased production of vaccine-specific antibodies. J. Clin. Invest. 121, 3109–3119 (2011).
Hanika, A. et al. Use of influenza C virus glycoprotein HEF for generation of vesicular stomatitis virus pseudotypes. J. Gen. Virol. 86, 1455–1465 (2005).
Caserta, L. C. et al. Spillover of highly pathogenic avian influenza H5N1 virus to dairy cattle. Nature 634, 669–676 (2024).
Hoffmann, E., Neumann, G., Kawaoka, Y., Hobom, G. & Webster, R. G. A DNA transfection system for generation of influenza A virus from eight plasmids. Proc. Natl. Acad. Sci. USA 97, 6108–6113 (2000).
Burrough, E. R. et al. Highly pathogenic avian influenza A(H5N1) Clade 2.3.4.4b virus infection in domestic dairy cattle and cats, United States, 2024. Emerg. Infect. Dis. 30, 1335–1343 (2024).
Ramakrishnan, M. A. Determination of 50% endpoint titer using a simple formula. World J. Virol. 5, 85–86 (2016).
Halbherr, S. J. et al. Vaccination with recombinant RNA replicon particles protects chickens from H5N1 highly pathogenic avian influenza virus. PLoS ONE 8, e66059 (2013).
Ricklin, M. E. et al. Partial protection against porcine influenza A virus by a hemagglutinin-expressing virus replicon particle vaccine in the absence of neutralizing antibodies. Front. Immunol. 7, 253 (2016).
Berger Rentsch, M. & Zimmer, G. A vesicular stomatitis virus replicon-based bioassay for the rapid and sensitive determination of multi-species type I interferon. PLoS ONE 6, e25858 (2011).
Kalhoro, N. H., Veits, J., Rautenschlein, S. & Zimmer, G. A recombinant vesicular stomatitis virus replicon vaccine protects chickens from highly pathogenic avian influenza virus (H7N1). Vaccine 27, 1174–1183 (2009).
Thompson, D., Cismaru, C. V., Rougier, J. S., Schwemmle, M. & Zimmer, G. The M2 proteins of bat influenza A viruses reveal atypical features compared to conventional M2 proteins. J. Virol. 97, e0038823 (2023).
Lenz-Ajuh, N. et al. Impact of pH and temperature in dairy processing on the infectivity of H5N1 avian influenza viruses. Int J. Food Microbiol 441, 111328 (2025).
Hofmann, M. A., Renzullo, S. & Baumer, A. Phylogenetic characterization of H5N1 highly pathogenic avian influenza viruses isolated in Switzerland in 2006. Virus Genes 37, 407–413 (2008).
Spackman, E. et al. Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. J. Clin. Microbiol. 40, 3256–3260 (2002).
Hedges, L. V. Effect sizes for experimental research. Br. J. Math. Stat. Psychol. 79, 31–45 (2026).
Acknowledgements
This work has received funding from the Swiss National Research Foundation (SNSF), grant no. 189903 (A.S., G.Z.) and was co-funded by the European Union’s Horizon Europe Project 101136346 EUPAHW by a grant to A.S. The funders had no role in study design, data collection and analysis, decisions to publish, or preparation of the manuscript. We are grateful to Daniel Brechbühl and Katarzyna Sliz for their support in animal experimentation. We like to thank Martin Schwemmle (University of Freiburg, Germany) and Yoshihiro Sakoda (University of Sapporo, Japan) for providing plasmids and Diego Diel (Cornell University, USA) for providing the bovine H5N1 virus isolate.
Author information
Authors and Affiliations
Contributions
A.S. and G.Z. conceived the study and were responsible for funding acquisition; N.R., O.G., and G.Z. conceptualized and conducted the animal experiments; R.A., L.B., and G.Z. generated recombinant influenza viruses and VSV-derived VRP vaccines; L.B. performed RT-qPCR analysis, virus-neutralization tests, virus release inhibition tests, and sialidase inhibition tests; L.B. and G.Z. analyzed and evaluated the data. G.Z. wrote the manuscript draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The Institute of Virology and Immunology IVI, Bern, Switzerland, and the University of Bern, Bern, Switzerland, filed a patent application “Prime-Boost Immunization against Influenza” (application no. WO2025133030A1), indicating A.S., G.Z., and R.A. as inventors. The invention relates to the heterologous prime-boost vaccine strategy also described in this work. The other authors do not have any competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
García-Nicolás, O., Butticaz, L., Avanthay, R. et al. Non-propagating RNA virus-vectored HA/NA vaccine prevents shedding of antigen-drifted H1N1 influenza virus in pigs. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01428-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01428-6