Abstract
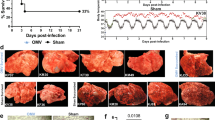
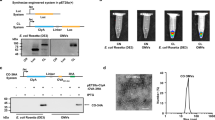
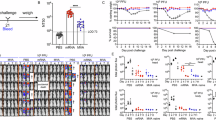
Outer membrane vesicle (OMV)-based vaccines elicit strong immune responses and have emerged as a versatile platform for targeting multiple pathogens, yet the mechanisms underlying their efficacy remain incompletely understood. Here, we investigated the early immune response following intranasal administration of an OMV-based pneumococcal vaccine candidate in mice. Using in vivo bioluminescence imaging, spectral flow cytometry, and high-resolution microscopy, we tracked OMV biodistribution and immune activation throughout the respiratory tract from 1 to 72 h post-immunization. OMVs persisted in the nasal cavity for up to 48 h and rapidly recruited Ly6Ghi neutrophils and myeloid-derived suppressor cells, followed by activation of local T cells. MHCII expression was significantly upregulated on Ly6Ghi neutrophils in nasal tissue and coincided with a marked expansion at 24 h. In the lungs, alveolar macrophages and plasmacytoid dendritic cells emerged as early responders. OMV exposure also induced costimulatory molecule expression across multiple myeloid cell subsets. Together, these findings reveal distinct spatio-temporal patterns of innate and adaptive immune activation at mucosal sites, providing mechanistic insight into OMV-induced mucosal immunity and underscoring their potential as a versatile vaccine platform.

Similar content being viewed by others
Data availability
All data supporting the findings of this study have been deposited in the project specific data collection in Radboud Data Repository and are accessible via the following DOI: (https://doi.org/10.34973/f82t-nt54). The dataset associated with this manuscript will be made publicly available upon publication.
References
Zahid, A., Ismail, H., Wilson, J. C. & Grice, I. D. Bioengineering outer-membrane vesicles for vaccine development: strategies, advances, and perspectives. Vaccines 13, https://doi.org/10.3390/vaccines13070767 (2025).
Kulp, A. & Kuehn, M. J. Biological functions and biogenesis of secreted bacterial outer membrane vesicles. Annu. Rev. Microbiol. 64, 163–184 (2010).
Ellis, T. N. & Kuehn, M. J. Virulence and immunomodulatory roles of bacterial outer membrane vesicles. Microbiol. Mol. Biol. Rev. 74, 81–94 (2010).
van der Pol, L., Stork, M. & van der Ley, P. Outer membrane vesicles as platform vaccine technology. Biotechnol. J. 10, 1689–1706 (2015).
Tan, K., Li, R., Huang, X. & Liu, Q. Outer membrane vesicles: current status and future direction of these novel vaccine adjuvants. Front. Microbiol. 9, 783 (2018).
Micoli, F. & MacLennan, C. A. Outer membrane vesicle vaccines. Semin Immunol. 50, 101433 (2020).
Iwasaki, A. & Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 16, 343–353 (2015).
Iwasaki, A. Exploiting mucosal immunity for antiviral vaccines. Annu. Rev. Immunol. 34, 575–608 (2016).
Mettelman, R. C., Allen, E. K. & Thomas, P. G. Mucosal immune responses to infection and vaccination in the respiratory tract. Immunity 55, 749–780 (2022).
Janeway, C. A. Jr, Immunobiology: The Immune System in Health and Disease. 5th edn, (Garland Science, 2001).
Alu, A. et al. Intranasal COVID-19 vaccines: from bench to bed. EBioMedicine 76, 103841 (2022).
Sui, Y. et al. Adjuvanted subunit intranasal vaccine reduces SARS-CoV-2 onward transmission in hamsters. Front Immunol. 16, 1514845 (2025).
Hall, M. P. et al. Engineered luciferase reporter from a deep sea shrimp utilizing a novel imidazopyrazinone substrate. ACS Chem. Biol. 7, 1848–1857 (2012).
England, C. G., Ehlerding, E. B. & Cai, W. NanoLuc: a small luciferase is brightening up the field of bioluminescence. Bioconjug Chem. 27, 1175–1187 (2016).
Voss, F. et al. Lipidation of pneumococcal antigens leads to improved immunogenicity and protection. Vaccines 8, https://doi.org/10.3390/vaccines8020310 (2020).
van Beek, L. F. et al. Intranasal vaccination with protein bodies elicit strong protection against Streptococcus pneumoniae colonization. Vaccine 39, 6920–6929 (2021).
van Beek, L. F. et al. Exploring metal availability in the natural niche of Streptococcus pneumoniae to discover potential vaccine antigens. Virulence, 11, 1310–1328 (2020).
Van, H. B. V. D. B., Jong, W. S. P. & Luirink, J. (Google Patents, 2021).
Kotecha, N., Krutzik, P. O. & Irish, J. M. Web-based analysis and publication of flow cytometry experiments. Curr. Protoc. Cytom., Unit10 17 (2010).
Holmgren, J. & Czerkinsky, C. Mucosal immunity and vaccines. Nat. Med. 11, S45–S53 (2005).
Kuipers, K. et al. Salmonella outer membrane vesicles displaying high densities of pneumococcal antigen at the surface offer protection against colonization. Vaccine 33, 2022–2029 (2015).
Kuipers, K. et al. Antigen-independent restriction of pneumococcal density by mucosal adjuvant cholera toxin subunit B. J. Infect. Dis. 214, 1588–1596 (2016).
Kuipers, K. et al. Th17-mediated cross protection against pneumococcal carriage by vaccination with a variable antigen. Infect. Immun. 85, https://doi.org/10.1128/IAI.00281-17 (2017).
van der Ley, P. & van den Dobbelsteen, G. Next-generation outer membrane vesicle vaccines against Neisseria meningitidis based on nontoxic LPS mutants. Hum. Vaccin 7, 886–890 (2011).
Ballesteros, I. et al. Co-option of neutrophil fates by tissue environments. Cell 183, 1282–1297 e1218 (2020).
Mancini, F., Rossi, O., Necchi, F. & Micoli, F. OMV vaccines and the role of TLR agonists in immune response. Int. J. Mol. Sci. 21, https://doi.org/10.3390/ijms21124416 (2020).
Gabrilovich, D. I. & Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 9, 162–174 (2009).
Ostrand-Rosenberg, S. & Fenselau, C. Myeloid-derived suppressor cells: immune-suppressive cells that impair antitumor immunity and are sculpted by their environment. J. Immunol. 200, 422–431 (2018).
Park, C. et al. Murine alveolar macrophages rapidly accumulate intranasally administered SARS-CoV-2 Spike protein leading to neutrophil recruitment and damage. Elife 12, https://doi.org/10.7554/eLife.86764 (2024).
Yusuf, H. & Kett, V. Current prospects and future challenges for nasal vaccine delivery. Hum. Vaccin Immunother. 13, 34–45 (2017).
Zhang, Z. et al. Nanotechnology-driven advances in intranasal vaccine delivery systems against infectious diseases. Front. Immunol. 16, 1573037 (2025).
Blank, F. et al. Size-dependent uptake of particles by pulmonary antigen-presenting cell populations and trafficking to regional lymph nodes. Am. J. Respir. Cell Mol. Biol. 49, 67–77 (2013).
Prochetto, E., Borgna, E., Jimenez-Cortegana, C., Sanchez-Margalet, V. & Cabrera, G. Myeloid-derived suppressor cells and vaccination against pathogens. Front. Cell Infect. Microbiol. 12, 1003781 (2022).
Hussell, T. & Bell, T. J. Alveolar macrophages: plasticity in a tissue-specific context. Nat. Rev. Immunol. 14, 81–93 (2014).
Kopf, M., Schneider, C. & Nobs, S. P. The development and function of lung-resident macrophages and dendritic cells. Nat. Immunol. 16, 36–44 (2015).
Swiecki, M. & Colonna, M. The multifaceted biology of plasmacytoid dendritic cells. Nat. Rev. Immunol. 15, 471–485 (2015).
Takashima, A. & Yao, Y. Neutrophil plasticity: acquisition of phenotype and functionality of antigen-presenting cell. J. Leukoc. Biol. 98, 489–496 (2015).
Beauvillain, C. et al. Neutrophils efficiently cross-prime naive T cells in vivo. Blood 110, 2965–2973 (2007).
Hagen, C. et al. Structural basis of vesicle formation at the inner nuclear membrane. Cell 163, 1692–1701 (2015).
Jones, E. J. et al. The uptake, trafficking, and biodistribution of Bacteroides thetaiotaomicron generated outer membrane vesicles. Front Microbiol, 11, 57 (2020).
Rompikuntal, P. K. et al. Perinuclear localization of internalized outer membrane vesicles carrying active cytolethal distending toxin from Aggregatibacter actinomycetemcomitans. Infect. Immun. 80, 31–42 (2012).
Yoshida, K. et al. Porphyromonas gingivalis outer membrane vesicles in cerebral ventricles activate microglia in mice. Oral. Dis. 29, 3688–3697 (2023).
Mueller, S. N. & Mackay, L. K. Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol. 16, 79–89 (2016).
Ruane, D. T. & Lavelle, E. C. The role of CD103(+) dendritic cells in the intestinal mucosal immune system. Front. Immunol. 2, 25 (2011).
Sallusto, F., Lanzavecchia, A., Araki, K. & Ahmed, R. From vaccines to memory and back. Immunity 33, 451–463 (2010).
Plotkin, S. A. Correlates of protection induced by vaccination. Clin. Vaccin. Immunol. 17, 1055–1065 (2010).
Ge, C. et al. Bystander activation of pulmonary trm cells attenuates the severity of bacterial pneumonia by enhancing neutrophil recruitment. Cell Rep. 29, 4236–4244 e4233 (2019).
Maurice, N. J., Taber, A. K. & Prlic, M. The ugly duckling turned to swan: a change in perception of bystander-activated memory CD8 T Cells. J. Immunol. 206, 455–462 (2021).
Paprckova, D., Salyova, E., Michalik, J. & Stepanek, O. Bystander activation in memory and antigen-inexperienced memory-like CD8 T cells. Curr. Opin. Immunol. 82, 102299 (2023).
Pizzolla, A. et al. Resident memory CD8(+) T cells in the upper respiratory tract prevent pulmonary influenza virus infection. Sci. Immunol. 2, https://doi.org/10.1126/sciimmunol.aam6970 (2017).
Sueyoshi, K. et al. Lipopolysaccharide suppresses T cells by generating extracellular ATP that impairs their mitochondrial function via P2Y11 receptors. J. Biol. Chem. 294, 6283–6293 (2019).
Godfrey, D. I., Koay, H. F., McCluskey, J. & Gherardin, N. A. The biology and functional importance of MAIT cells. Nat. Immunol. 20, 1110–1128 (2019).
Chien, Y. H., Meyer, C. & Bonneville, M. γδ T cells: first line of defense and beyond. Annu. Rev. Immunol. 32, 121–155 (2014).
Hartwell, B. L. et al. Intranasal vaccination with lipid-conjugated immunogens promotes antigen transmucosal uptake to drive mucosal and systemic immunity. Sci. Transl. Med. 14, eabn1413 (2022).
Balmelli, C., Demotz, S., Acha-Orbea, H., De Grandi, P. & Nardelli-Haefliger, D. Trachea, lung, and tracheobronchial lymph nodes are the major sites where antigen-presenting cells are detected after nasal vaccination of mice with human papillomavirus type 16 virus-like particles. J. Virol. 76, 12596–12602 (2002).
Heyder, J. Deposition of inhaled particles in the human respiratory tract and consequences for regional targeting in respiratory drug delivery. Proc. Am. Thorac. Soc. 1, 315–320 (2004).
Acknowledgements
We thank the Radboudumc Technology Center—Animal research facility for their immense support in animal experiments. We are grateful to the Imaging Core Facility of Radboud University for assistance with imaging. We are also thankful to Xuehui He and Laurien Waaijer for their support in data analysis via R and flow cytometry panel design, respectively. A special thanks to Tieke Kuijpers and Tan Zhan Ber for their support in construct purification and in vivo experiments. This work was supported by Horizon2020 Marie Curie Individual Fellowship under the name of VacTrack.
Author information
Authors and Affiliations
Contributions
S.K. conceived the study, designed and performed experiments, analysed data, and wrote the manuscript. S.V. processed FIB-SEM images, assisted with 3D segmentation, correlative image registration, and subsequent analysis. R.U. assisted with the design of the flow cytometry panel. B.J. and R.R. contributed to confocal and FIB-SEM image acquisition, respectively. B.v.C. assisted with flow cytometry data analysis and interpretation. F.J.v.O. conducted the in vivo experiments. B.v.D.B.v.S. and J.L. produced OMVs displaying nLuc-fused antigens. C.E.v.d.G.d.J., E.v.R., and L.F.v.B were involved in tissue processing experiments. D.A.D., L.F.v.B., and M.I.d.J. provided conceptual input and supervision and critically revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
Conflicts of interest are in Abera Bioscience AB that aims to exploit the presented OMV technology and has patented this vaccine platform18. The vaccine candidate and the immunization studies conducted in this study are based on information available in this patent application. However, the insights gained through this manuscript are not part of the patent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kanwal, S., To, S.V., Uijen, R. et al. Tracking spatio-temporal dynamics of early immune responses to an intranasal OMV-based pneumococcal vaccine candidate in mice. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01430-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01430-y