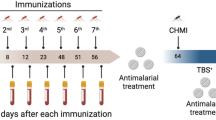
Abstract
While vaccines are central to eradicate malaria, they remain elusive, with numerous malaria vaccine candidates showing limited efficacy in Phase II and III studies. Controlled human malaria infection studies have showed that human volunteers, immunized with Plasmodium falciparum sporozoites under drug cover, were protected experimentally from a subsequent challenge. Here, to identify new targets associated with protection, we utilized a previously developed screening approach, where we screened sera from protected and non-protected individuals against newly included hypothetical antigens in a P. falciparum antigen library. PfVFT1 was found to be associated with protection, with antibodies against PfVFT1 being detected in all protected individuals. We found that vaccine-induced mouse anti-PfVFT sera inhibited parasite reinvasion into RBCs, promoted complement deposition to induce parasite lysis, and supported phagocytosis and antibody-dependent cellular inhibition of the parasite. Together, these data indicate that PfVFT1-specific antibodies can engage multiple effector mechanisms relevant to antimalarial immunity.
Similar content being viewed by others
Data Availability
The data generated in this study can be obtained upon reasonable request to the corresponding author. DNA sequences from 90 Thai field isolates are deposited in Genbank, and the Genbank accession numbers are PX636225 to PX636314, respectively.
References
WHO. World malaria report 2024. (2024).
Hemingway, J. et al. Averting a malaria disaster: Will insecticide resistance derail malaria control? Lancet 387, 1785–1788 (2016).
Rts, S. C. T. P. et al. First results of phase 3 trial of RTS,S/AS01 malaria vaccine in African children. N. Engl. J. Med. 365, 1863–1875 (2011).
Rts SCTP Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet 386, 31–45 (2015).
Datoo, M. S. et al. Safety and efficacy of malaria vaccine candidate R21/Matrix-M in African children: a multicentre, double-blind, randomised, phase 3 trial. Lancet 403, 533–544 (2024).
Ishizuka, A. S. et al. Protection against malaria at 1 year and immune correlates following PfSPZ vaccination. Nat. Med. 22, 614–623 (2016).
Roestenberg, M. et al. Protection against a malaria challenge by sporozoite inoculation. N. Engl. J. Med. 361, 468–477 (2009).
Seder, R. A. et al. Protection against malaria by intravenous immunization with a nonreplicating sporozoite vaccine. Science 341, 1359–1365 (2013).
Mwakingwe-Omari, A. et al. Two chemoattenuated PfSPZ malaria vaccines induce sterile hepatic immunity. Nature 595, 289–294 (2021).
Sulyok, Z. et al. Heterologous protection against malaria by a simple chemoattenuated PfSPZ vaccine regimen in a randomized trial. Nat. Commun. 12, 2518 (2021).
Doolan, D. L. et al. Profiling humoral immune responses to P. falciparum infection with protein microarrays. Proteomics 8, 4680–4694 (2008).
Felgner, P. L. et al. Pre-erythrocytic antibody profiles induced by controlled human malaria infections in healthy volunteers under chloroquine prophylaxis. Sci. Rep. 3, 3549 (2013).
Renia, L., Gruner, A. C., Mauduit, M. & Snounou, G. Vaccination against malaria with live parasites. Expert Rev. Vaccines 5, 473–481 (2006).
Peng, K. et al. Breadth of humoral response and antigenic targets of sporozoite-inhibitory antibodies associated with sterile protection induced by controlled human malaria infection. Cell Microbiol. 18, 1739–1750 (2016).
Aguiar, J. C. et al. Discovery of novel Plasmodium falciparum pre-erythrocytic antigens for vaccine development. PLoS One 10, e0136109 (2015).
Osier, F. H. et al. New antigens for a multicomponent blood-stage malaria vaccine. Sci. Transl. Med. 6, 247ra102 (2014).
Raj, D. K. et al. Antibodies to PfSEA-1 block parasite egress from RBCs and protect against malaria infection. Science 344, 871–877 (2014).
Khosh-Naucke, M. et al. Identification of novel parasitophorous vacuole proteins in P. falciparum parasites using BioID. Int. J. Med. Microbiol. 308, 13–24 (2018).
Nilsson Bark, S. K. et al. Quantitative proteomic profiling reveals novel plasmodium falciparum surface antigens and possible vaccine candidates. Mol. Cell Proteom. 17, 43–60 (2018).
Bijker, E. M. et al. Cytotoxic markers associate with protection against malaria in human volunteers immunized with Plasmodium falciparum sporozoites. J. Infect. Dis. 210, 1605–1615 (2014).
Sanders, P. R. et al. Identification of protein complexes in detergent-resistant membranes of Plasmodium falciparum schizonts. Mol. Biochem. Parasitol. 154, 148–157 (2007).
Ghorbal, M. et al. Genome editing in the human malaria parasite Plasmodium falciparum using the CRISPR-Cas9 system. Nat. Biotechnol. 32, 819–821 (2014).
Taborda, C. P., Rivera, J., Zaragoza, O. & Casadevall, A. More is not necessarily better: prozone-like effects in passive immunization with IgG. J. Immunol. 170, 3621–3630 (2003).
Jacobs, J. F., van der Molen, R. G., Bossuyt, X. & Damoiseaux, J. Antigen excess in modern immunoassays: to anticipate on the unexpected. Autoimmun. Rev. 14, 160–167 (2015).
Goh, Y. S. et al. Monoclonal antibodies of a diverse isotype induced by an o-antigen glycoconjugate vaccine mediate in vitro and in vivo killing of African invasive nontyphoidal Salmonella. Infect. Immun. 83, 3722–3731 (2015).
Gillet, P., Mori, M., Van Esbroeck, M., Van den Ende, J. & Jacobs, J. Assessment of the prozone effect in malaria rapid diagnostic tests. Malar. J. 8, 271 (2009).
Gillet, P. et al. Prozone in malaria rapid diagnostics tests: How many cases are missed? Malar. J. 10, 166 (2011).
Smith, G. & Holman, R. P. The prozone phenomenon with syphilis and HIV-1 co-infection. South Med. J. 97, 379–382 (2004).
Sidana, R., Mangala, H. C., Murugesh, S. B. & Ravindra, K. Prozone phenomenon in secondary syphilis. Indian J. Sex. Transm. Dis. AIDS 32, 47–49 (2011).
Sandoval, M. N. et al. Prozone masks elevated SARS-CoV-2 antibody level measurements. PLoS One 19, e0301232 (2024).
Byrne, J. et al. Specific thresholds of circulating antibody titers predict against infection and reduced disease severity in SARS-CoV-2 close contacts. J. Immunol. 214, 2238–2243 (2025).
Srivastava, K. et al. SARS-CoV-2-infection- and vaccine-induced antibody responses are long lasting with an initial waning phase followed by a stabilization phase. Immunity 57, 587–599.e584 (2024).
Llewellyn, D. et al. Standardization of the antibody-dependent respiratory burst assay with human neutrophils and Plasmodium falciparum malaria. Sci. Rep. 5, 14081 (2015).
Kerntke, C., Nimmerjahn, F. & Biburger, M. There is (scientific) strength in numbers: a comprehensive quantitation of Fc gamma receptor numbers on human and murine peripheral blood leukocytes. Front. Immunol. 11, 118 (2020).
Belambri, S. A. et al. NADPH oxidase activation in neutrophils: role of the phosphorylation of its subunits. Eur. J. Clin. Investig. 48, e12951 (2018).
Mayadas, T. N., Cullere, X. & Lowell, C. A. The multifaceted functions of neutrophils. Annu. Rev. Pathol. 9, 181–218 (2014).
Arama, C. et al. Genetic resistance to malaria is associated with greater enhancement of immunoglobulin (Ig)M than IgG responses to a broad array of Plasmodium falciparum antigens. Open forum Infect. Dis. 2, ofv118 (2015).
Boyle, M. J. et al. IgM in human immunity to Plasmodium falciparum malaria. Sci. Adv. 5, eaax4489 (2019).
Plasmodb.org. PlasmoDB : The Plasmodium Genomics Resource. Available at: http://plasmodb.org/plasmo/ [Accessed 25 Jan. 2018]. (2018).
Parker, M. L. et al. The structure of Plasmodium falciparum 3D7_0606800 reveals a bi-lobed architecture that supports re-annotation as a Venus Flytrap protein. Protein Sci. Publ. Protein Soc. 26, 1878–1885 (2017).
Gilson, P. R. et al. Identification and stoichiometry of glycosylphosphatidylinositol-anchored membrane proteins of the human malaria parasite Plasmodium falciparum. Mol. Cell. Proteom. MCP 5, 1286–1299 (2006).
Adams, J. H., Blair, P. L., Kaneko, O. & Peterson, D. S. An expanding EBL family of Plasmodium falciparum. Trends Parasitol. 17, 297–299 (2001).
Triglia, T., Duraisingh, M. T., Good, R. T. & Cowman, A. F. Reticulocyte-binding protein homologue 1 is required for sialic acid-dependent invasion into human erythrocytes by Plasmodium falciparum. Mol. Microbiol. 55, 162–174 (2005).
Duraisingh, M. T. et al. Phenotypic variation of Plasmodium falciparum merozoite proteins directs receptor targeting for invasion of human erythrocytes. EMBO J. 22, 1047–1057 (2003).
Gilberger, T. W. et al. A novel erythrocyte binding antigen-175 paralogue from Plasmodium falciparum defines a new trypsin-resistant receptor on human erythrocytes. J. Biol. Chem. 278, 14480–14486 (2003).
Rayner, J. C., Vargas-Serrato, E., Huber, C. S., Galinski, M. R. & Barnwell, J. W. A Plasmodium falciparum homologue of Plasmodium vivax reticulocyte binding protein (PvRBP1) defines a trypsin-resistant erythrocyte invasion pathway. J. Exp. Med. 194, 1571–1581 (2001).
Duraisingh, M. T., Maier, A. G., Triglia, T. & Cowman, A. F. Erythrocyte-binding antigen 175 mediates invasion in Plasmodium falciparum utilizing sialic acid-dependent and -independent pathways. Proc. Natl. Acad. Sci. USA 100, 4796–4801 (2003).
Bijker, E. M. et al. Protection against malaria after immunization by chloroquine prophylaxis and sporozoites is mediated by preerythrocytic immunity. Proc. Natl. Acad. Sci. USA 110, 7862–7867 (2013).
Belnoue, E. et al. Protective T cell immunity against malaria liver stage after vaccination with live sporozoites under chloroquine treatment. J. Immunol. 172, 2487–2495 (2004).
Hill, D. L. et al. Opsonising antibodies to P. falciparum merozoites associated with immunity to clinical malaria. PloS one 8, e74627 (2013).
Tiendrebeogo, R. W. et al. Antibody-dependent cellular inhibition is associated with reduced risk against Febrile malaria in a longitudinal cohort study involving Ghanaian children. Open forum Infect. Dis. 2, ofv044 (2015).
Kennedy, A. T. et al. Recruitment of factor H as a novel complement evasion strategy for blood-stage Plasmodium falciparum infection. J. Immunol. 196, 1239–1248 (2016).
Barfod, L. et al. Evasion of immunity to Plasmodium falciparum malaria by IgM masking of protective IgG epitopes in infected erythrocyte surface-exposed PfEMP1. Proc. Natl. Acad. Sci. USA 108, 12485–12490 (2011).
Boyle, M. J. et al. Human antibodies fix complement to inhibit Plasmodium falciparum invasion of erythrocytes and are associated with protection against malaria. Immunity 42, 580–590 (2015).
Bijker, E. M. et al. Sporozoite immunization of human volunteers under mefloquine prophylaxis is safe, immunogenic and protective: a double-blind randomized controlled clinical trial. PLoS One 9, e112910 (2014).
Douglas, A. D. et al. The blood-stage malaria antigen PfRH5 is susceptible to vaccine-inducible cross-strain neutralizing antibody. Nat. Commun. 2, 601 (2011).
Malleret, B. et al. A rapid and robust tri-color flow cytometry assay for monitoring malaria parasite development. Sci. Rep. 1, 118 (2011).
Boyle, M. J. et al. Isolation of viable Plasmodium falciparum merozoites to define erythrocyte invasion events and advance vaccine and drug development. Proc. Natl. Acad. Sci. USA 107, 14378–14383 (2010).
Tippett, E., Fernandes, L. A., Rogerson, S. J. & Jaworowski, A. A novel flow cytometric phagocytosis assay of malaria-infected erythrocytes. J. Immunol. Methods 325, 42–50 (2007).
Hill, D. L. et al. Efficient measurement of opsonising antibodies to Plasmodium falciparum merozoites. PloS One 7, e51692 (2012).
Stubbs, J. et al. Strain-transcending Fc-dependent killing of Plasmodium falciparum by merozoite surface protein 2 allele-specific human antibodies. Infect. Immun. 79, 1143–1152 (2011).
Acknowledgements
We thank Dr Jose-Juan Lopez-Rubio, University of Montpellier, France, for providing the CRISPR-Cas9 plasmids; Professor Zbynek Bozdech, Nanyang Technological University, Singapore, for providing the pBCam-3HA plasmid; Dr Simon Draper, Nuffield Department of Medicine, Jenner Institute, University of Oxford, UK, for providing the Rh5 antibodies; Professor Peter Preiser, School of Biological Sciences, Nanyang Technological University, Singapore, for providing the MSP1 antibodies. We also thank the FLOW facility at ID Labs and SIgN, A*STAR, Singapore for technical support with the cytometers. This work was funded by Agency for Science, Technology and Research (A*STAR) core grant. Shoklo Malaria Research Unit is part of the Mahidol Oxford University Research Unit, supported by the Wellcome Trust of Great Britain.
Author information
Authors and Affiliations
Contributions
Y.S.G. and L.R. conceived the study and wrote the paper. Y.S.G. designed the experiments. Y.S.G., H.M., P.X.H. and C.Y.L. performed the experiments. Y.S.G., H.M., P.X.H., C.Y.L. and L.R. analyzed the data. Z.W.C., C.C.H., R.W.S. and F.N. contributed the reagents and materials. All authors approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Goh, Y.S., Mao, H., Hor, P.X. et al. Vaccine-induced mouse antibodies targeting Plasmodium falciparum PfVFT antigen inhibit blood stages through multiple mechanisms. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01433-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01433-9