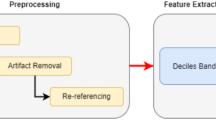
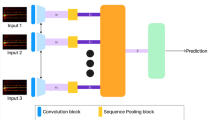
Abstract
Electroencephalography (EEG) connectomes offer powerful tools for studying brain connectivity and advancing our understanding of brain function and dysfunction in both healthy and pathological conditions. Celebrating the 100th anniversary of EEG discovery, this Perspective explores the frontiers of EEG-based brain connectivity in basic and translational neuroscience research. We review new concepts, emerging analysis frameworks and significant advances in harnessing EEG connectomes. We suggest that leveraging machine learning approaches may offer promising paths to maximize the strengths of EEG connectomes. We also discuss how combined EEG connectome and neuromodulation provide a personalized and adaptive closed-loop paradigm to promote neuroplasticity and treat dysfunctional brains. We further address the limitations and challenges of the current methodology and touch on important issues regarding research rigour and clinical viability for translational impact.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Code availability
The software for producing some figures in this article will be available on request.
References
Biasiucci, A., Franceschiello, B. & Murray, M. M. Electroencephalography. Curr. Biol. 29, R80–R85 (2019).
Cao, J. et al. Brain functional and effective connectivity based on electroencephalography recordings: a review. Hum. Brain Mapp. 43, 860–879 (2022).
Sadahiani, S., Brookes, M. J. & Baillet, S. Connectomis of human electrophysiology. Neuroimage 247, 118788 (2022).
Miljevic, A., Brailey, N. W., Vila-Rodriguez, F., Herring, S. E. & Fitzgerald, P. B. Electroencephalographic connectivitiy: a fundamental guide and checklist for optimal study design and evaluation. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 7, 546–554 (2022).
Glasser, M. et al. The Human Connectome Project’s neuroimaging approach. Nat. Neurosci. 19, 1175–1187 (2016).
Parvizi, J. & Kastner, S. Promises and limitations of human intracranial electroencephalography. Nat. Neurosci. 21, 474–483 (2018).
Michelmann, S. et al. Moment-by-moment tracking of naturalistic learning and its underlying hippocampo–cortical interactions. Nat. Commun. 12, 5394 (2021).
Metzger, S. L. et al. Generalizable spelling using a speech neuroprosthesis in an individual with severe limb and vocal paralysis. Nat. Commun. 13, 6510 (2022).
Metzger, S. L. et al. A high-performance neuroprosthesis for speech decoding and avatar control. Nature 620, 1037–1046 (2023).
Zrenner, C., Desideri, D., Belardinelli, P. & Ziemann, U. Real-time EEG-defined excitability states determine efficacy of TMS-induced plasticity in human motor cortex. Brain Stimul. 11, 374–389 (2018).
Sporns, O. The human connectome: a complex network. Ann. NY Acad. Sci. 1224, 109–125 (2011).
Wu, W. et al. An electroencephalographic signature predicts antidepressant response in major depression. Nat. Biotechnol. 38, 439–447 (2020).
Toll, R. et al. An electroencephalography connectomic profile of post-traumatic stress disorder. Am. J. Psych. 177, 233–243 (2020).
Kabbara, A. et al. An electroencephalography connectome predictive model of major depressive disorder severity. Sci. Rep. 12, 6816 (2022).
Deligianni, F., Centeno, M., Carmichael, D. W. & Clayden, J. D. Relating resting-state fMRI and EEG whole-brain connectomes across frequency bands. Front. Neurosci. 8, 258 (2014).
Wirsich, J., Giraud, A.-L. & Sadaghiani, S. Concurrent EEG- and fMRI-derived functional connectomes exhibit linked dynamics. Neuroimage 219, 116998 (2020).
Tóth, B. et al. Dynamics of EEG functional connectivity during statistical learning. Neurobiol. Learn. Mem. 144, 216–229 (2017).
He, B. et al. Electrophysiological brain connectivity: theory and implementation. IEEE Trans. Biomed. Eng. 66, 2115–2137 (2019).
Stoyell, S. M. et al. High-density EEG in current clinical practice and opportunities for the future. J. Clin. Neurophysiol. 38, 112–123 (2021).
Kaiju, T. et al. High spatiotemporal resolution ECoG recording of somatosensory evoked potentials with flexible microelectrode arrays. Front. Neural Circuits 11, 20 (2017).
Michel, C. M. & Brunet, D. EEG source imaging: a practical review of the analysis steps. Front. Neurol. 10, 325 (2019).
Seeber, M. et al. Subcortical electrophysiological activity is detectable with high-density EEG source imaging. Nat. Commun. 10, 753 (2019).
Hnazaee, M. F. et al. Localization of deep brain activity with scalp and subdural EEG. Neuroimage 223, 117344 (2020).
Piastra, M. C. et al. A comprehensive study on electroencephalography and magnetoencephalography sensitivity to cortical and subcortical sources. Hum. Brain Mapp. 42, 978–992 (2021).
Hecker, L., Rupprecht, R., Tebartz Van Elst, L. & Kornmeier, J. ConvDip: a convolutional neural network for better EEG source imaging. Front. Neurosci. 15, 569918 (2021).
Sun, R., Sohrabpour, A., Worrell, G. A. & He, B. Deep neural networks constrained by neural mass models improve electrophysiological source imaging of spatiotemporal brain dynamics. Proc. Natl Acad. Sci. USA 119, e2201128119 (2022).
Huang, G. et al. Electromagnetic source imaging via a data-synthesis-based convolutional encoder-decoder network. IEEE Trans. Neural Netw. Learn. Syst. 35, 6423–6437 (2024).
Pezzulo, G., Zorzi, M. & Corbetta, M. The secret life of predictive brains: what’s spontaneous activity for? Trends Cogn. Sci. 25, 730–743 (2021).
Kaushik, P. et al. Comparing resting state and task-based EEG using machine learning to predict vulnerability to depression in a non-clinical population. Sci. Rep. 13, 7467 (2023).
Lynch, L. K. et al. Task-evoked functional connectivity does not explain functional connectivity differences between rest and task conditions. Hum. Brain Mapp. 39, 4939–4948 (2018).
Abid, A. et al. Exploring patterns enriched in a dataset with contrastive principal component analysis. Nat. Commun. 9, 2134 (2018).
Jann, K. et al. Linking brain connectivity across different time scales with electroencephalogram, functional magnetic resonance imaging, and diffusion tensor imaging. Brain Connect. 2, 11–20 (2012).
Glomb, K. et al. Connectome spectral analysis to track EEG task dynamics on a subsecond scale. Neuroimage 221, 117137 (2020).
Tu T., Paisle J., Haufe S. & Sajda P. A state-space model for inferring effective connectivity of latent neural dynamics from simultaneous EEG/fMRI. Adv. Neural Inform. Proc. Syst. 32, 4662–4671 (2019).
Warbrick, T. Simultaneous EEG–fMRI: what have we learned and what does the future hold? Sensors 22, 2262 (2022).
Menon V. & Crottaz-Herbette S. Combined EEG and fMRI studies of human brain function. Int. Rev. Neurobiol. 66, 291–321 (2005).
Wirsich, J., Amico, E., Giraud, A. L., Goñi, J. & Sadaghiani, S. Multi-timescale hybrid components of the functional brain connectome: a bimodal EEG–fMRI decomposition. Netw. Neurosci. 4, 658–677 (2020).
Wirsich, J. et al. The relationship between EEG and fMRI connectomes is reproducible across simultaneous EEG–fMRI studies from 1.5T to 7T. Neuroimage 231, 117864 (2021).
Shu, H. et al. Disturbed temporal dynamics of episodic retrieval activity with preserved spatial activity pattern in amnestic mild cognitive impairment: a simultaneous EEG–fMRI study. Neuroimage Clin. 30, 102572 (2021).
Shah, P. et al. Characterizing the role of the structural connectome in seizure dynamics. Brain 142, 1955–1972 (2019).
Mostame, P. & Sadaghiani, S. Phase- and amplitude-coupling are tied by an intrinsic spatial organization but show divergent stimulus-related changes. Neuroimage 219, 117051 (2020).
Zhang, Y. et al. Multiview feature learning with multiatlas-based functional connectivity networks for MCI diagnosis. IEEE Trans. Cyber. 52, 6822–6833 (2022).
Phang, C. R., Noman, F., Hussain, H., Ting, C. M. & Ombao, H. A multi-domain connectome convolutional neural network for identifying schizophrenia from EEG connectivity patterns. IEEE J. Biomed. Health Inform. 24, 1333–1343 (2020).
Cai, L. et al. Functional integration and segregation in multiplex brain networks for Alzheimer’s disease. Front. Neurosci. 14, 51 (2020).
Vecchio, F., Miraglia, F. & Maria Rossini, P. Connectome: graph theory application in functional brain network architecture. Clin. Neurophysiol. Pract. 2, 206–213 (2017).
Sun, S. et al. Graph theory analysis of functional connectivity in major depression disorder with high-density resting state EEG data. IEEE Trans. Neural Syst. Rehabil. Eng. 27, 429–439 (2019).
Zhang, Z., Cui, P. & Zhu, W. Deep learning on graphs: a survey. IEEE Trans. Knowl. Data Eng. 34, 249–270 (2020).
Bessadok, A., Mahjoub, M. A. & Rekik, I. Graph neural networks in network neuroscience. IEEE Trans. Pattern Anal. Mach. Intell. 45, 5833–5848 (2023).
Tang, S. et al. Self-supervised graph neural networks for improved electroencephalographic seizure analysis. in Proc. Int. Conf. Learning Representations (ICLR’22) https://iclr.cc/Conferences/2022 (ICLR, 2022).
Battaglia P. et al. Relational inductive biases, deep learning, and graph networks. Preprint at https://doi.org/10.48550/arXiv.1806.01261 (2018).
Kim, B.-H., Ye, J. C. & Kim, J. J. Learning dynamic graph representation of brain connectome with spatio-temporal attention. Adv. Neural Inform. Proc. Syst. 35, 4314–4327 (2021).
Rutherford, S. et al. The normative modeling framework for computational psychiatry. Nat. Protoc. 17, 1711–1734 (2022).
Chen, Z. S., Galatzer-Levy, I. R., Bigio, B., Nasca, C. & Zhang, Y. Modern views of machine learning for precision psychiatry. Patterns 3, 100602 (2022).
Janiukstyte, V. et al. Normative brain mapping using scalp EEG and potential clinical application. Sci. Rep. 13, 13442 (2023).
Tong, X. et al. Individual deviations from normative electroencephalographic connectivity predict antidepressant response. J. Affect. Disord. 351, 220–230 (2024).
Tong, X. et al. Symptom dimensions of resting-state electroencephalographic functional connectivity in austim. Nat. Ment. Health 2, 287–298 (2024).
Aglinskas, A., Hartshorne, J. K. & Anzellotti, S. Contrastive machine learning reveals the structure of neuroanatomical variation within autism. Science 376, 1070–1074 (2022).
Zhou, G., Cichocki, A., Zhang, Y. & Mandic, D. P. Group component analysis for multiblock data: common and individual feature extraction. IEEE Trans. Neural Netw. Learn. Syst. 27, 2426–2439 (2016).
Cao, B. et al. t-BNE: tensor-based brain network embedding. In Proc. 2017 SIAM Int. Conf. Data Mining (SDM’17) 189–197 (Society for Industrial and Applied Mathematics, 2017).
Yang Y., Cai G., Ye C., Xiang Y. & Ma T. Tensor-based complex-valued graph neural network for dynamic coupling multimodal brain networks. in Proc. ICASSP’23 https://doi.org/10.1109/ICASSP49357.2023.10095707 (IEEE, 2023).
Kostas, D., Aroca-Ouellette, S. & Rudzicz, F. BENDR: using transformers and a contrastive self-supervised learning task to learn from massive amounts of EEG data. Front. Hum. Neurosci. 15, 653659 (2021).
Tang, J., LeBel, A., Jain, S. & Huth, A. G. Semantic reconstruction of continuous language from non-invasive brain recordings. Nat. Neurosci. 26, 858–866 (2023).
Defossez, A., Caucheteux, C., Rapin, J., Kabeli, O. & King, J.-R. Decoding speech perception from non-invasive brain recordings. Nat. Mach. Intell. 5, 1097–1107 (2023).
Liu, X. et al. Self-supervised learning: generative or contrastive. IEEE Trans. Knowl. Data Eng. 35, 857–876 (2023).
Rafiei, M. H., Gauthier, L. V., Adeli, H. & Takabi, D. Self-supervised learning for electroencephalography. IEEE Trans. Neural Netw. Learn. Syst. 35, 1457–1471 (2024).
Tang, Y., Huang, W., Liu, R. & Yu, Y. Learning interpretable brain functional connectivity via self-supervised triplet network with depth-wise attention. IEEE J. Biomed. Health Inform. https://doi.org/10.1109/JBHI.2024.3429169 (2024).
Thomas, A. W., Re, C. & Poldrack, R. A. Self-supervised learning of brain dynamics from broad neuroimaging data. Adv. Neural Inform. Proc. Syst. 36, 21255–21269 (2022).
Geng, D., Alkhachroum, A., Bicchi, M. A. M., Cajigas, I. & Chen, Z. S. Deep learning for robust detection of interictal epileptiform discharges. J. Neural Eng. 18, 056015 (2021).
Rasheed, K., Qadir, J., O’Brien, T. J., Kuhlmann, L. & Razi, A. A generative model to synthesize EEG data for epileptic seizure prediction. IEEE Trans. Neural Syst. Rehabil. Eng. 29, 2322–2332 (2021).
Bird, J. J., Pritchard, M., Fratini, A., Ekárt, A. & Faria, D. R. Synthetic biological signals machine-generated by GPT-2 improve the classification of EEG and EMG through data augmentation. IEEE Robot. Autom. Lett. 6, 3498–3504 (2021).
Wang, Y. et al. DiffMDD: a diffusion-based deep learning framework for MDD diagnosis using EEG. IEEE Trans. Neural Syst. Rehabil. Eng. 32, 728–738 (2024).
Ahmad, W. et al. A new generative adversarial network for medical images super resolution. Sci. Rep. 12, 9533 (2022).
Kwon, M., Han, S., Kim, K. & Jun, S. C. Super-resolution for improving EEG spatial resolution using deep convolutional neural network-feasibility study. Sensors 19, 5317 (2019).
Li, Z. et al. Graph-generative neural network for EEG-based epileptic seizure detection via discovery of dynamic brain functional connectivity. Sci. Rep. 12, 18998 (2022).
Zhang, Y. et al. Identification of psychiatric disorder subtypes from functional connectivity patterns in resting-state electroencephalography. Nat. Biomed. Eng. 5, 309–323 (2021).
Zhang, Y. et al. Machine learning-based identification of a psychotherapy-predictive electroencephalographic signature in PTSD. Nat. Ment. Health 1, 284–294 (2023).
Duan, F. et al. Topological network analysis of early Alzheimer’s disease based on resting-state EEG. IEEE Trans. Neural Syst. Rehab. Eng. 28, 2164–2172 (2020).
Ciprian, C., Masychev, K., Ravan, M., Reilly, J. P. & Maccrimmon, D. A machine learning approach using effective connectivity to predict response to clozapine treatment. IEEE Trans. Neural Syst. Rehab. Eng. 28, 2598–2607 (2020).
Ntolkeras, G. et al. Interictal EEG source connectivity to localize the epileptogenic zone in patients with drug‐resistant epilepsy: a machine learning approach. Epilepsia 65, 944–960 (2024).
Nickel, M. M. et al. Neural oscillations and connectivity characterizing the state of tonic experimental pain in humans. Hum. Brain Mapp. 41, 17–29 (2020).
Marquand, A. F. et al. Conceptualizing mental disorders as deviations from normative functioning. Mol. Psychiatry 24, 1415–1424 (2019).
Maron-Katz, A. et al. Individual patterns of abnormality in resting-state functional connectivity reveal two data-driven PTSD subgroups. Am. J. Psychiatry 177, 244–253 (2020).
Bergmann, T. O., Karabanov, A., Hartwigsen, G., Thielscher, A. & Siebner, H. R. Combining non-invasive transcranial brain stimulation with neuroimaging and electrophysiology: current approaches and future perspectives. Neuroimage 140, 4–19 (2016).
Miniussi, C., Brignani, D. & Pellicciari, M. C. Combining transcranial electrical stimulation with electroencephalography: a multimodal approach. Clin. EEG Neurosci. 43, 184–191 (2012).
Hubbard, R. J. et al. Brain connectivity alterations during sleep by closed-loop transcranial neurostimulation predict metamemory sensitivity. Netw. Neurosci. 5, 734–756 (2021).
Ferrarelli, F. & Phillips, M. L. Examining and modulating neural circuits in psychiatric disorders with transcranial magnetic stimulation and electroencephalography: present practices and future developments. Am. J. Psychiatry 178, 400–413 (2021).
Mueller, J., Legon, W., Opitz, A., Sato, T. F. & Tyler, W. J. Transcranial focused ultrasound modulates intrinsic and evoked EEG dynamics. Brain Stimul. 7, 900–908 (2014).
Yuksel, M. M. et al. Low-intensity focused ultrasound neuromodulation for stroke recovery: a novel deep brain stimulation approach for neurorehabilitation? IEEE Open J. Eng. Med. Biol. 4, 300–318 (2023).
Rogasch, N. C. & Fitzgerald, P. B. Assessing cortical network properties using TMS-EEG. Hum. Brain Mapp. 34, 1652–1669 (2013).
Ozdemir, R. A. et al. Individualized perturbation of the human connectome reveals reproducible biomarkers of network dynamics relevant to cognition. Proc. Natl Acad. Sci. USA 117, 8115–8125 (2020).
Roy, A., Baxter, B. & He, B. High-definition transcranial direct current stimulation induces both acute and persistent changes in broadband cortical synchronization: a simultaneous tDCS–EEG study. IEEE Trans. Biomed. Eng. 61, 1967–1978 (2014).
Klooster, D. C., Ferguson, M. A., Boon, P. A. & Baeken, C. Personalizing repetitive transcranial magnetic stimulation parameters for depression treatment using multimodal neuroimaging. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 7, 536–545 (2022).
Christiansen, L., Liu, M. L. & Siebner, H. R. in Transcranial Direct Current Stimulation in Neuropsychiatric Disorders: Clinical Principles and Management (eds Brunoni, A. R. et al.) 251–263 (Springer, 2021).
Luppi, J. J., Stam, C. J., Scheltens, P. & de Haan, W. Virtual neural network-guided optimization of non-invasive brain stimulation in Alzheimer’s disease. PLoS Comput. Biol. 20, e1011164 (2024).
Keller, C. J. et al. Mapping human brain networks with cortico-cortical evoked potentials. Philos. Trans. R. Soc. B 369, 20130528 (2014).
Crocker, B. et al. Local and distant responses to single pulse electrical stimulation reflect different forms of connectivity. Neuroimage 237, 118094 (2021).
Hasan, M. A., Fraser, M., Conway, B. A., Allan, D. B. & Vučković, A. Reversed cortical over-activity during movement imagination following neurofeedback treatment for central neuropathic pain. Clin. Neurophysiol. 127, 3118–3127 (2016).
Ros T. & Gruzelier J. H. in Neurofeedback and Neuromodulation Techniques and Applications (eds Coben R. & Evans J. R.) 381–402 (Academic Press, 2011).
Tervo, A. E. et al. Closed-loop optimization of transcranial magnetic stimulation with electroencephalography feedback. Brain Stimul. 15, 523–531 (2022).
Lubianiker, N., Paret, C., Dayan, P. & Hendler, T. Neurofeedback through the lens of reinforcement learning. Trends Neurosci. 45, 579–593 (2022).
Pineau, J., Guez, A., Vincent, R., Panuccio, G. & Avoli, M. Treating epilepsy via adaptive neurostimulation: a reinforcement learning approach. Int. J. Neural Syst. 19, 227–240 (2009).
Ozdemir, R. A. et al. Cortical responses to noninvasive perturbations enable individual brain fingerprinting. Brain Stimul. 14, 391–403 (2021).
Tăuƫan, A. M. et al. TMS-EEG perturbation biomarkers for Alzheimer’s disease patients classification. Sci. Rep. 13, 7667 (2023).
Casula, E. P. et al. Regional precuneus cortical hyperexcitability in Alzheimer’s disease patients. Ann. Neurol. 93, 371–383 (2023).
Harquel, S. et al. Stroke recovery-related changes in cortical reactivity based on modulation of intracortical inhibition. Stroke 55, 1629–1640 (2024).
Naim-Feil, J. et al. Anomalies in global network connectivity associated with early recovery from alcohol dependence: A network transcranial magnetic stimulation and electroencephalography study. Addict. Biol. 27, e13146 (2022).
Dang, Y. et al. Deep brain stimulation improves electroencephalogram functional connectivity of patients with minimally conscious state. CNS Neurosci. Ther. 29, 344–353 (2023).
Lepage, K. Q., Ching, S. & Kramer, M. A. Inferring evoked brain connectivity through adaptive perturbation. J. Comput. Neurosci. 34, 303–318 (2013).
Tang, E. & Bassett, D. S. Control of dynamics in brain networks. Rev. Mod. Phys. 90, 031003 (2018).
Alexander, L. M. et al. An open resource for transdiagnostic research in pediatric mental health and learning disorders. Sci. Data 4, 170181 (2017).
Van Dijk, H. et al. The two decades brainclinics research archive for insights in neurophysiology (TDBRAIN) database. Sci. Data 9, 333 (2022).
Obeid, I. & Picone, J. The temple university hospital EEG data corpus. Front. Neurosci. 10, 195498 (2016).
Cavanagh, J. F., Napolitano, A., Wu, C. & Mueen, A. The patient repository for EEG data + computational tools (PRED+CT). Front. Neuroinform. 11, 67 (2017).
Prado, P. et al. The BrainLat project, a multimodal neuroimaging dataset of neurodegeneration from underrepresented backgrounds. Sci. Data 10, 889 (2023).
Babayan, A. et al. A mind-brain-body dataset of MRI, EEG, cognition, emotion, and peripheral physiology in young and old adults. Sci. Data 6, 180308 (2019).
Valdes-Sosa, P. A. et al. The Cuban human brain mapping project, a young and middle age population-based EEG, MRI, and cognition dataset. Sci. Data 8, 45 (2021).
Trivedi, M. H. et al. Establishing moderators and biosignatures of antidepressant response in clinical care (EMBARC): rationale and design. J. Psychiatr. Res. 78, 11–23 (2016).
McPartland, J. C. et al. The autism biomarkers consortium for clinical trials (ABC-CT): scientific context, study design, and progress toward biomarker qualification. Front. Integr. Neurosci. 14, 16 (2020).
Sosulski, J. & Tangermann, M. Introducing block-Toeplitz covariance matrices to remaster linear discriminant analysis for event-related potential brain-computer interfaces. J. Neural Eng. 19, 066001 (2022).
Wang, C., Du, J. & Fan, X. High-dimensional correlation matrix estimation for general continuous data with Bagging technique. Mach. Learn. 111, 2905–2927 (2022).
Li, Y. et al. Targeting EEG/LFP synchrony with neural nets. Adv. Neural Inform. Proc. Syst. 30, 4621–4631 (2017).
Shan, X. et al. Spatial–temporal graph convolutional network for Alzheimer classification based on brain functional connectivity imaging of electroencephalogram. Hum. Brain Mapp. 43, 5194–5209 (2022).
Sauseng, P. et al. The interplay between theta and alpha oscillations in the human electroencephalogram reflects the transfer of information between memory systems. Neurosci. Lett. 324, 121–124 (2002).
Zhang, H., Watrous, A. J., Patel, A. & Jacobs, J. Theta and alpha oscillations are traveling waves in the human neocortex. Neuron 98, 1269–1281 (2018).
Alamia, A., Terral, L., D’ambra, M. R. & Van Rullen, R. Distinct roles of forward and backward alpha-band waves in spatial visual attention. eLife 12, e85035 (2023).
Hayashi, M. et al. Spatially bivariate EEG-neurofeedback can manipulate interhemispheric inhibition. eLife 11, e76411 (2022).
Momi, D. et al. Perturbation of resting-state network nodes preferentially propagates to structurally rather than functionally connected regions. Sci. Rep. 11, 12458 (2021).
Alves, C. L. et al. Analysis of functional connectivity using machine learning and deep learning in different data modalities from individuals with schizophrenia. J. Neural Eng. 20, 056025 (2023).
Kiebel, S. J., Garrido, M. I., Moran, R. J. & Friston, K. J. Dynamic causal modelling for EEG and MEG. Cogn. Neurodyn 2, 121–136 (2008).
Van de Steen, F., Almgren, H., Razi, A., Friston, K. & Marinazzo, D. Dynamic causal modelling of fluctuating connectivity in resting-state EEG. Neuroimage 189, 476–484 (2019).
Qiao, L. et al. Estimating functional brain networks by incorporating a modularity prior. Neuroimage 141, 399–407 (2016).
Li, J., Hao, Y., Zhang, W., Li, X. & Hu, B. Effective connectivity based EEG revealing the inhibitory deficits for distracting stimuli in major depression disorders. IEEE Trans. Affect. Comput. 14, 694–705 (2023).
Brunner, C., Billinger, M., Seeber, M., Mullen, T. R. & Makeig, S. Volume conduction influences scalp-based connectivity estimates. Front. Comput. Neurosci. 10, 121 (2016).
Szabó, C. Á. et al. Baboon model of generalized epilepsy: continuous intracranial video-EEG monitoring with subdural electrodes. Epilepsy Res. 101, 46–55 (2012).
Sandhaeger, F., von Nicolai, C., Miller, E. K. & Siegel, M. Monkey EEG links neuronal color and motion information across species and scales. eLife 8, e45645 (2019).
Chen, Z. S. & Pesaran, B. Improving scalability in systems neuroscience. Neuron 109, 1776–1790 (2021).
Barron, H. C., Mars, R. B., Dupret, D., Lerch, J. P. & Sampaio-Baptista, C. Cross-species neuroscience: closing the explanatory gap. Philos. Trans. R. Soc. B 376, 20190633 (2021).
Rolle, C. E. et al. Functional connectivity using high density EEG shows competitive reliability and agreement across test/retest sessions. J. Neurosci. Methods 367, 109424 (2022).
Rosenblatt, M. et al. Connectome-based machine learning models are vulnerable to subtle data manipulations. Patterns 4, 100756 (2023).
Fernandez, L. et al. Assessing cerebellar–cortical connectivity using concurrent TMS-EEG: a feasibility study. J. Neurophysiol. 125, 1768–1787 (2021).
Mahjoory, K. et al. Consistency of EEG source localization and connectivity estimates. Neuroimage 152, 590–601 (2017).
O’Shaughnessy, M., Canal, G., Connor, M., Davenport, M. & Rozell, C. Generative causal explanations of black-box classifiers. Adv. Neural Inform. Proc. Syst. 33, 5453–5467 (2020).
Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat. Mach. Intell. 1, 206–215 (2019).
Ying, R., Bourgeois, D., You, J., Zitnik, M. & Leskovec, J. GNNExplainer: generating explanations for graph neural networks. Adv. Neural Inform. Proc. Syst. 32, 9240–9251 (2019).
Van Gerven, M. A. J., Hesse, C., Jensen, O. & Heskes, T. Interpreting single trial data using groupwise regularisation. Neuroimage 46, 665–676 (2009).
Mamashli, F. et al. Permutation statistics for connectivity analysis between regions of interest in EEG and MEG data. Sci. Rep. 9, 7942 (2019).
Niso, G., Romero, E., Moreau, J. T., Araujo, A. & Krol, L. R. Wireless EEG: a survey of systems and studies. Neuroimage 269, 119774 (2023).
Song, S. & Nordin, A. D. Mobile electroencephalography for studying neural control of human locomotion. Front. Hum. Neurosci. 15, 749017 (2021).
Cole, M. W., Ito, T., Cocuzza, C. & Sanchez-Romero, R. The functional relevance of task-state functional connectivity. J. Neurosci. 41, 2684–2702 (2021).
Anjum, M. F. et al. Resting-state EEG measures cognitive impairment in Parkinson’s disease. npj Parkinson’s Dis. 10, 6 (2024).
Phalen, H., Coffman, B. A., Ghuman, A., Sejdić, E. & Salisbury, D. F. Non-negative matrix factorization reveals resting-state cortical alpha network abnormalities in the first-episode schizophrenia spectrum. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 5, 961–970 (2020).
Xie, W., Toll, R. T. & Nelson, C. A. EEG functional connectivity analysis in the source space. Dev. Cogn. Neurosci. 56, 101119 (2022).
Schoffelen, J. M. & Gross, J. Source connectivity analysis with MEG and EEG. Hum. Brain Mapp. 30, 1857–1865 (2009).
Bastos, A. M. & Schoffelen, J. M. A tutorial review of functional connectivity analysis methods and their interpretational pitfalls. Front. Syst. Neurosci. 9, 175 (2016).
Balasubramani, P. P. et al. Simultaneous gut-brain electrophysiology shows cognition and satiety specific coupling. Sensors 22, 9242 (2022).
Mayeli, A. et al. Parieto-occipital ERP indicators of gut mechanosensation in humans. Nat. Commun. 14, 3398 (2023).
Candia-Rivera, D., Catrambone, V. & Valenza, G. The role of electroencephalography electrical reference in the assessment of functional brain-heart interplay: from methodology to user guidelines. J. Neurosci. Methods 360, 109269 (2021).
Abdalbari, H. et al. Brain and brain–heart Granger causality during wakefulness and sleep. Front. Neurosci. 16, 927111 (2022).
Chen, P. C., Zhang, J., Thayer, J. F. & Mednick, S. C. Understanding the roles of central and autonomic activity during sleep in the improvement of working memory and episodic memory. Proc. Natl Acad. Sci. USA 119, e2123417119 (2022).
Rho, G. et al. EEG cortical activity and connectivity correlates of early sympathetic response during cold pressor test. Sci. Rep. 13, 1338 (2023).
Acknowledgements
This study was funded by grants from the US National Institutes of Health (grant numbers RF1-DA056394, R01-MH118928, R01-NS123928, RF1-NS121776, P50-MH132642 and UG3NS135170 to Z.S.C.; and R21-MH130956, R01-MH129694 and R21-AG080425 to Y.Z.), the Alzheimer’s Association (grant number AARG-22-972541 to Y.Z.), and Lehigh University’s FIG (grant number FIGAWD35) and CORE grants to Y.Z. The views, opinions, and findings expressed are those of the authors and should not be interpreted as representing the official views or policies of the US Government.
Author information
Authors and Affiliations
Contributions
Z.S.C. and Y.Z. conceived this research and wrote the paper; both authors participated in the editing and revisions of the manuscript.
Corresponding authors
Ethics declarations
Citation diversity statement
The authors acknowledge that research authored by scholars from historically excluded groups is systematically under-cited. Every attempt has been made to reference relevant research in a manner that is equitable in terms of racial, ethnic, gender and geographical representation.
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Friedhelm Hummel, Enzo Tagliazucchi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Chen, Z.S. Harnessing electroencephalography connectomes for cognitive and clinical neuroscience. Nat. Biomed. Eng 9, 1186–1201 (2025). https://doi.org/10.1038/s41551-025-01442-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-025-01442-4