Abstract
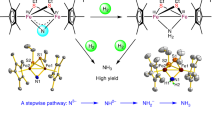
Living systems carry out the reduction of N2 to ammonia (NH3) through a series of protonation and electron transfer steps under ambient conditions using the enzyme nitrogenase. In the chemical industry, the Haber–Bosch process hydrogenates N2 but requires high temperatures and pressures. Both processes rely on iron-based catalysts, but molecular iron complexes that promote the formation of NH3 on addition of H2 to N2 have remained difficult to devise. Here, we isolate the tri(iron)bis(nitrido) complex [(Cp′Fe)3(μ3-N)2] (in which Cp′ = η5-1,2,4-(Me3C)3C5H2), which is prepared by reduction of [Cp′Fe(μ-I)]2 under an N2 atmosphere and comprises three iron centres bridged by two μ3-nitrido ligands. In solution, this complex reacts with H2 at ambient temperature (22 °C) and low pressure (1 or 4 bar) to form NH3. In the solid state, it is converted into the tri(iron)bis(imido) species, [(Cp′Fe)3(μ3-NH)2], by addition of H2 (10 bar) through an unusual solid–gas, single-crystal-to-single-crystal transformation. In solution, [(Cp′Fe)3(μ3-NH)2] further reacts with H2 or H+ to form NH3.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The authors declare that all of the data supporting the findings of this study are available within the paper and the Supplementary Information, and also from the corresponding authors upon reasonable request. This includes experimental details, NMR studies in solution, solid-state NMR studies, crystallographic details, X-band EPR studies, solid-state magnetic susceptibility studies, zero-field 57Fe Mössbauer studies, and computational details. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers 1939746 (1) and 1939747 (2). Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures.
References
Haber, F. The production of ammonia from nitrogen and hydrogen. Naturwissenschaften 10, 1041–1049 (1922).
Schlögl, R. Catalytic synthesis of ammonia—a ‘never-ending story’? Angew. Chem. Int. Ed. 42, 2004–2008 (2003).
Kandemir, T., Schuster, M. E., Senyshyn, A., Behrens, M. & Schlögl, R. The Haber–Bosch process revisited: on the real structure and stability of ‘ammonia iron’ under working conditions. Angew. Chem. Int. Ed. 52, 12723–12726 (2013).
Tuczek, F. Synthetic vs biological nitrogen fixation. Nachr. Chem. 54, 1190–1194 (2006).
Lancaster, K. M. et al. X-ray emission spectroscopy evidences a central carbon in the nitrogenase iron–molybdenum cofactor. Science 334, 974–977 (2011).
Spatzal, T. et al. Evidence for interstitial carbon in nitrogenase FeMo cofactor. Science 334, 940 (2011).
Hoffman, B. M., Lukoyanov, D., Yang, Z.-Y., Dean, D. R. & Seefeldt, L. C. Mechanism of nitrogen fixation by nitrogenase: the next stage. Chem. Rev. 114, 4041–4062 (2014).
Walter, M. D. in Advances in Organometallic Chemistry Vol. 65 (ed. Pérez, P. J.) Ch. 5 (Academic Press, 2016).
Nishibayashi, Y. (ed.) Transition Metal–Dinitrogen Complexes (Wiley, 2019).
Légaré, M.-A. et al. Nitrogen fixation and reduction at boron. Science 359, 896–900 (2018).
Del Castillo, T. J., Thompson, N. B. & Peters, J. C. A synthetic single-site Fe nitrogenase: high turnover, freeze–quench 57Fe Mössbauer data, and a hydride resting state. J. Am. Chem. Soc. 138, 5341–5350 (2016).
Kuriyama, S. et al. Direct transformation of molecular dinitrogen into ammonia catalyzed by cobalt dinitrogen complexes bearing anionic PNP pincer ligands. Angew. Chem. Int. Ed. 55, 14291–14295 (2016).
Yandulov, D. V. & Schrock, R. R. Catalytic reduction of dinitrogen to ammonia at a single molybdenum center. Science 301, 76–78 (2003).
Ashida, Y., Arashiba, K., Nakajima, K. & Nishibayashi, Y. Molybdenum-catalysed ammonia production with samarium diiodide and alcohols or water. Nature 568, 536–540 (2019).
Falcone, M., Chatelain, L., Scopelliti, R., Zivkovic, I. & Mazzanti, M. Nitrogen reduction and functionalization by a multimetallic uranium nitride complex. Nature 547, 332–335 (2017).
Falcone, M. & Mazzanti, M. Four-electron reduction and functionalization of N2 by a uranium(iii) bridging nitride. Chimia 72, 199–202 (2018).
Falcone, M., Poon, L. N., Fadaei Tirani, F. & Mazzanti, M. Reversible dihydrogen activation and hydride transfer by a uranium nitride complex. Angew. Chem. Int. Ed. 57, 3697–3700 (2018).
Pool, J. A., Bernskoetter, W. H. & Chirik, P. J. On the origin of dinitrogen hydrogenation promoted by [(η5-C5Me4H)2Zr]2(μ2,η2,η2-N2). J. Am. Chem. Soc. 126, 14326–14327 (2004).
Pool, J. A., Lobkovsky, E. & Chirik, P. J. Hydrogenation and cleavage of dinitrogen to ammonia with a zirconium complex. Nature 427, 527–530 (2004).
Ohki, Y. & Fryzuk, M. D. Dinitrogen activation by group 4 metal complexes. Angew. Chem. Int. Ed. 46, 3180–3183 (2007).
Fryzuk, M. D. Side-on end-on bound dinitrogen: an activated bonding mode that facilitates functionalizing molecular nitrogen. Acc. Chem. Res. 42, 127–133 (2009).
Rodriguez, M. M., Bill, E., Brennessel, W. W. & Holland, P. L. N2 reduction and hydrogenation to ammonia by a molecular iron-potassium complex. Science 334, 780–783 (2011).
MacLeod, K. C. & Holland, P. L. Recent developments in the homogeneous reduction of dinitrogen by molybdenum and iron. Nat. Chem. 5, 559 (2013).
MacLeod, K. C., McWilliams, S. F., Mercado, B. Q. & Holland, P. L. Stepwise N–H bond formation from N2-derived iron nitride, imide and amide intermediates to ammonia. Chem. Sci. 7, 5736–5746 (2016).
Lee, Y. et al. Dinitrogen activation upon reduction of a triiron(ii) complex. Angew. Chem. Int. Ed. 54, 1499–1503 (2015).
Anderson, J. S., Rittle, J. & Peters, J. C. Catalytic conversion of nitrogen to ammonia by an iron model complex. Nature 501, 84–87 (2013).
Reiners, M., Baabe, D., Zaretzke, M.-K., Freytag, M. & Walter, M. D. Reversible dinitrogen binding to [Cp′Fe(NHC)] associated with an N2-induced spin state change. Chem. Commun. 53, 7274–7277 (2017).
Ferreira, R. B. et al. Catalytic silylation of dinitrogen by a family of triiron complexes. ACS Catal. 8, 7208–7212 (2018).
Bozso, F., Ertl, G. & Weiss, M. Interaction of nitrogen with iron surfaces: II. Fe(110). J. Catal. 50, 519–529 (1977).
Somorjai, G. A. & Materer, N. Surface structures in ammonia synthesis. Top. Catal. 1, 215–231 (1994).
Spencer, N. D., Schoonmaker, R. C. & Somorjai, G. A. Iron single crystals as ammonia synthesis catalysts: effect of surface structure on catalyst activity. J. Catal. 74, 129–135 (1982).
Weatherburn, M. W. Phenol-hypochlorite reaction for determination of ammonia. Anal. Chem. 39, 971–974 (1967).
Chiang, K. P., Bellows, S. M., Brennessel, W. W. & Holland, P. L. Multimetallic cooperativity in activation of dinitrogen at iron–potassium sites. Chem. Sci. 5, 267–274 (2014).
Bhutto, S. M. & Holland, P. L. Dinitrogen activation and functionalization using β-diketiminate iron complexes. Eur. J. Inorg. Chem. 2019, 1861–1869 (2019).
Reiners, M. et al. Reactivity studies on [Cp′Fe(μ-I)]2: nitrido-, sulfido- and diselenide iron complexes derived from pseudohalide activation. Chem. Sci. 8, 4108–4122 (2017).
Huang, Z., White, P. S. & Brookhart, M. Ligand exchanges and selective catalytic hydrogenation in molecular single crystals. Nature 465, 598–601 (2010).
Pike, S. D. et al. Synthesis and characterization of a rhodium(i) σ-alkane complex in the solid state. Science 337, 1648–1651 (2012).
Pike, S. D. et al. Solid-state synthesis and characterization of σ-alkane complexes, [Rh(L2)(η2,η2-C7H12)][BArF4] (L2 = bidentate chelating phosphine). J. Am. Chem. Soc. 137, 820–833 (2015).
Chadwick, F. M. et al. Selective C–H activation at a molecular rhodium sigma-alkane complex by solid/gas single-crystal to single-crystal H/D exchange. J. Am. Chem. Soc. 138, 13369–13378 (2016).
Chadwick, F. M. et al. A rhodium-pentane sigma-alkane complex: characterization in the solid state by experimental and computational techniques. Angew. Chem. Int. Ed. 55, 3677–3681 (2016).
Walter, M. D., Grunenberg, J. & White, P. S. Reactivity studies on [Cp′FeI]2: from iron hydrides to P4-activation. Chem. Sci. 2, 2120–2130 (2011).
Reiners, M. et al. Monomeric Fe(iii) half-sandwich complexes [Cp′FeX2] - synthesis, properties and electronic structure. Dalton Trans. 47, 10517–10526 (2018).
Moore, W. J. & Hummel, D. O. (eds) Physikalische Chemie 4th edn (Walter de Gruyter, 1986).
Argouarch, G., Hamon, P., Toupet, L., Hamon, J.-R. & Lapinte, C. [(η5-C5Me5)Fe(Ph2PCH2CH2CH2PPh2)][SO3CF3], a stable 16-electron complex with a coordinating counteranion and without agostic interaction: the dramatic role of a trivial methylene group. Organometallics 21, 1341–1348 (2002).
Gütlich, P., Bill, E. & Trautwein, A. X. (eds) Mössbauer Spectroscopy and Transition Metal Chemistry (Springer, 2011).
Spasyuk, D. M. et al. Facile hydrogen atom transfer to iron(iii) imido radical complexes supported by a dianionic pentadentate ligand. Chem. Sci. 7, 5939–5944 (2016).
Brown, S. D., Mehn, M. P. & Peters, J. C. Heterolytic H2 activation mediated by low-coordinate L3Fe-(μ-N)-FeL3 complexes to generate Fe(μ-NH)(μ-H)Fe species. J. Am. Chem. Soc. 127, 13146–13147 (2005).
Kefalidis, C. E. et al. Can a pentamethylcyclopentadienyl ligand act as a proton-relay in f-element chemistry? Insights from a joint experimental/theoretical study. Dalton Trans. 44, 2575–2587 (2015).
Chalkley, M. J., Del Castillo, T. J., Matson, B. D., Roddy, J. P. & Peters, J. C. Catalytic N2 -to-NH3 conversion by Fe at lower driving force: a proposed role for metallocene-mediated PCET. ACS Cent. Sci. 3, 217–223 (2017).
Chalkley, M. J., Oyala, P. H. & Peters, J. C. Cp* noninnocence leads to a remarkably weak C–H bond via metallocene protonation. J. Am. Chem. Soc. 141, 4721–4729 (2019).
Drover, M. W., Schild, D. J., Oyala, P. H. & Peters, J. C. Snapshots of a migrating H-atom: characterization of a reactive iron(iii) indenide hydride and its nearly isoenergetic ring-protonated iron(i) isomer. Angew. Chem. Int. Ed. 58, 15504–15511 (2019).
Walter, M. D. & White, P. S. [Cp′FeI]2 as convenient entry into iron-modified pincer complexes: bimetallic η6,κ1-POCOP-pincer iron iridium compounds. New J. Chem. 35, 1842–1854 (2011).
Schwindt, M. A., Lejon, T. & Hegedus, L. S. Improved synthesis of (aminocarbene)chromium(0) complexes with use of C8K-generated Cr(CO)5 2−. Multivariant optimization of an organometallic reaction. Organometallics 9, 2814–2819 (1990).
Massiot, D. et al. Modelling one- and two-dimensional solid-state NMR spectra. Magn. Reson. Chem. 40, 70–76 (2002).
Sheldrick, G. Crystal structure refinement with SHELXL. Acta Crystallogr. C 71, 3–8 (2015).
Stoll, S. & Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 178, 42–55 (2006).
Walter, M. D., Schultz, M. & Andersen, R. A. Weak paramagnetism in compounds of the type Cp′2Yb(bipy). New J. Chem. 30, 238–246 (2006).
Bain, G. A. & Berry, J. F. Diamagnetic corrections and Pascal’s constants. J. Chem. Educ. 85, 532–536 (2008).
Brand, R. A. WinNormos-for-Igor v. 3.0 (Wissenschaftliche Elektronik GmbH, 2009).
Gaussian 16, Revisions B.01v and B.01 (Gaussian Inc., 2016).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Burke, K., Perdew, J. P. & Wang, Y. in Electronic Density Functional Theory: Recent Progress and New Directions (eds Dobson, J. F., Vignale, G. et al.) Ch. II (Plenum, 1997).
Dolg, M., Wedig, U., Stoll, H. & Preuss, H. Energy‐adjusted ab initio pseudopotentials for the first row transition elements. J. Chem. Phys. 86, 866–872 (1987).
Ehlers, A. W. et al. A set of f-polarization functions for pseudo-potential basis sets of the transition metals Sc-Cu, Y-Ag and La-Au. Chem. Phys. Lett. 208, 111–114 (1993).
Hariharan, P. C. & Pople, J. A. The influence of polarization functions on molecular orbital hydrogenation energies. Theor. Chem. Acc. 28, 213–222 (1973).
Hehre, W. J., Ditchfield, R. & Pople, J. A. Self-consistent molecular orbital methods. XII. Further extensions of Gaussian-type basis sets for use in molecular orbital studies of organic molecules. J. Chem. Phys. 56, 2257–2261 (1972).
Reed, A. E., Curtiss, L. A. & Weinhold, F. Intermolecular interactions from a natural bond orbital, donor–acceptor viewpoint. Chem. Rev. 88, 899–926 (1988).
Walter, M. D. & White, P. S. [{Cp′Fe(μ-OH)}3: the synthesis of a unique organometallic iron hydroxide. Dalton Trans. 41, 8506–8508 (2012).
Acknowledgements
We thank P. Schweyen for EPR data collection on complex 1, and M. Bröring and F. Jochen Litterst for providing access to the SQUID magnetometer and 57Fe Mössbauer spectrometer. We acknowledge the help of S. Schneider and J. Abbenseth in the studies of solution 15N and 2H NMR. This work was supported by the Emmy Noether (WA 2513/2) and Heisenberg (WA 2513/6-8) programs of the Deutsche Forschungsgemeinschaft (DFG) (for M.D.W.); a fellowship for experienced researchers of the Humboldt Foundation and the Chinese Academy of Science (L.M.); a computational grant from CalMip; and CNRS (S.B.). This contribution is dedicated to the memory of Richard A. Andersen.
Author information
Authors and Affiliations
Contributions
M.R., D.B., Y.C., S.B. and M.D.W. conceived the experiments; M.R., D.B., K.M., M.-K.Z., M.F., P.G.J., Y.C., S.B. and M.D.W. designed and performed experiments and analysed the data; I.d.R. and L.M. performed computational studies; M.R., D.B., S.B., L.M. and M.D.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–26, Supplementary Tables 1–5 and supplementary references 1–14.
Supplementary Data 1
Crystallographic data (CIF) for complex 1
Supplementary Data 2
Crystallographic data (CIF) for complex 1
Supplementary Data 3 (download ZIP )
Cartesian coordinates for optimized computed structures as .xyz files
Rights and permissions
About this article
Cite this article
Reiners, M., Baabe, D., Münster, K. et al. NH3 formation from N2 and H2 mediated by molecular tri-iron complexes. Nat. Chem. 12, 740–746 (2020). https://doi.org/10.1038/s41557-020-0483-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-020-0483-7
This article is cited by
-
Ammonia synthesis by photocatalytic hydrogenation of a N2-derived molybdenum nitride
Nature Synthesis (2022)
-
Ammonia formation revisited
Nature Chemistry (2022)
-
A thiolate-bridged FeIVFeIV μ-nitrido complex and its hydrogenation reactivity toward ammonia formation
Nature Chemistry (2022)
-
Visible light enables catalytic formation of weak chemical bonds with molecular hydrogen
Nature Chemistry (2021)