Abstract
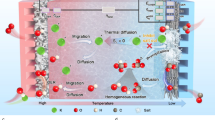
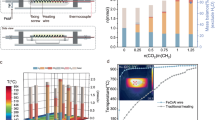
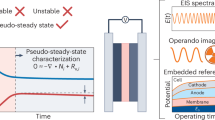
Low-temperature carbon dioxide electrolysis (CO2E) provides a one-step means of converting CO2 into carbon-based fuels using electrical inputs at temperatures below 100 °C. Over the past decade, an abundance of work has been carried out at ambient temperature, and high CO2E rates and product selectivities have been achieved. With scaling of CO2E technologies underway, greater discourse surrounding heat management and the viable operating temperatures of larger systems is important. In this Perspective we argue that, owing to the energy inefficiency of electrolysers, heat generation in CO2E stacks will favour operating temperatures of between 40 and 70 °C, far from the ambient temperatures used so far. Such elevated temperatures put further pressure on catalyst and membrane stability and on the stack design. On the other hand, elevated temperatures could alleviate challenges in salt precipitation, water management and high cell voltages, aiding the technology. We reflect on these aspects and discuss the opportunities for waste heat valorization to increase the economic feasibility of the process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barecka, M. H. & Ager, J. W. Towards an accelerated decarbonization of the chemical industry by electrolysis. Energy Adv. 2, 268–279 (2023).
Segets, D., Andronescu, C. & Apfel, U.-P. Accelerating CO2 electrochemical conversion towards industrial implementation. Nat. Commun. 14, 7950 (2023).
Burdyny, T. & Mulder, F. M. Scale-up of CO2 and CO electrolyzers. Joule 8, 2449–2452 (2024).
Petrov, K. V. et al. Bipolar membranes for intrinsically stable and scalable CO2 electrolysis. Nat. Energy https://doi.org/10.1038/s41560-024-01574-y (2024).
Ozden, A. et al. Energy- and carbon-efficient CO2/CO electrolysis to multicarbon products via asymmetric ion migration–adsorption. Nat. Energy 8, 179–190 (2023).
Reichbauer, T. et al. Electrical energy input efficiency limitations in CO2-to-CO electrolysis and attempts for improvement. Electrochem. Sci. Adv. 4, e2300024 (2024).
Endrődi, B. et al. High carbonate ion conductance of a robust PiperION membrane allows industrial current density and conversion in a zero-gap carbon dioxide electrolyzer cell. Energy Environ. Sci. 13, 4098–4105 (2020).
Technical targets for proton exchange membrane electrolysis. US Department of Energy (accessed 20 March 2025); https://www.energy.gov/eere/fuelcells/technical-targets-proton-exchange-membrane-electrolysis
Giron Rodriguez, C. A. et al. Insights into zero-gap CO2 electrolysis at elevated temperatures. EES Catal. 2, 850–861 (2024).
Vos, R. E. et al. How temperature affects the selectivity of the electrochemical CO2 reduction on copper. ACS Catal. 13, 8080–8091 (2023).
Schellekens, M. P., Raaijman, S. J., Koper, M. T. M. & Corbett, P. J. Temperature-dependent selectivity for CO electroreduction on copper-based gas-diffusion electrodes at high current densities. Chem. Eng. J. 483, 149105 (2024).
Krause, R. et al. Industrial application aspects of the electrochemical reduction of CO2 to CO in aqueous electrolyte. Chem. Ing. Tech. 92, 53–61 (2020).
Quentmeier, M., Schmid, B., Tempel, H. & Eichel, R.-A. Modular CO2-to-CO electrolysis short-stack design—impact of temperature gradients and insights into position-dependent cell behavior. ACS Sustain. Chem. Eng. 12, 3876–3885 (2024).
Crandall, B. S. et al. Kilowatt-scale tandem CO2 electrolysis for enhanced acetate and ethylene production. Nat. Chem. Eng. 1, 421–429 (2024).
Wakerley, D. et al. Gas diffusion electrodes, reactor designs and key metrics of low-temperature CO2 electrolysers. Nat. Energy 7, 130–143 (2022).
Lees, E. W., Mowbray, B. A. W., Parlane, F. G. L. & Berlinguette, C. P. Gas diffusion electrodes and membranes for CO2 reduction electrolysers. Nat. Rev. Mater. 7, 55–64 (2022).
Iglesias van Montfort, H.-P. et al. An advanced guide to assembly and operation of CO2 electrolyzers. ACS Energy Lett. 8, 4156–4161 (2023).
Kibria, M. G. et al. Electrochemical CO2 reduction into chemical feedstocks: from mechanistic electrocatalysis models to system design. Adv. Mater. 31, 1807166 (2019).
Weng, L.-C., Bell, A. T. & Weber, A. Z. Towards membrane-electrode assembly systems for CO2 reduction: a modeling study. Energy Environ. Sci. 12, 1950–1968 (2019).
Hansen, K. U., Cherniack, L. H. & Jiao, F. Voltage loss diagnosis in CO2 electrolyzers using five-electrode technique. ACS Energy Lett. 7, 4504–4511 (2022).
Garg, S., Giron Rodriguez, C. A., Rufford, T. E., Varcoe, J. R. & Seger, B. How membrane characteristics influence the performance of CO2 and CO electrolysis. Energy Environ. Sci. 15, 4440–4469 (2022).
Incropera, F. P., DeWitt, D. P., Bergman, T. L. & Lavine, A. S. Fundamentals of Heat and Mass Transfer 6th edn (Wiley, 2007).
Wijaya, G. H. A., Im, K. S. & Nam, S. Y. Advancements in commercial anion exchange membranes: a review of membrane properties in water electrolysis applications. Desalin. Water Treat. 320, 100605 (2024).
Industrial Cooling Systems (Callens, accessed 19 September 2024); https://www.callens.eu/en/air-technology/industrial-cooling
Mokhatab, S., Poe, W. A. & Mak, J. Y. in Handbook of Natural Gas Transmission and Processing: Principles and Practices 537–578 (Elsevier, 2019); https://doi.org/10.1016/B978-0-12-815817-3.00018-6
Toghyani, S., Afshari, E., Baniasadi, E. & Atyabi, S. A. Thermal and electrochemical analysis of different flow field patterns in a PEM electrolyzer. Electrochim. Acta 267, 234–245 (2018).
Rossen, A., Daems, N., Choukroun, D. & Breugelmans, T. Differential pressure across a gas diffusion electrode controls efficiency of liquid-fed electrolyzers for CO2 electroreduction at elevated temperatures. ACS Sustain. Chem. Eng. https://doi.org/10.1021/acssuschemeng.4c01908 (2024).
Deng, X., Yang, F., Li, Y., Dang, J. & Ouyang, M. Quantitative study on gas evolution effects under large current density in zero-gap alkaline water electrolyzers. J. Power Sources 555, 232378 (2023).
Belsa, B. et al. Materials challenges on the path to gigatonne CO2 electrolysis. Nat. Rev. Mater. https://doi.org/10.1038/s41578-024-00696-9 (2024).
Moss, A. B. et al. In operando investigations of oscillatory water and carbonate effects in MEA-based CO2 electrolysis devices. Joule 7, 350–365 (2023).
Disch, J. et al. High-resolution neutron imaging of salt precipitation and water transport in zero-gap CO2 electrolysis. Nat. Commun. 13, 6099 (2022).
Joensen, B. Ó. et al. Unveiling transport mechanisms of cesium and water in operando zero-gap CO2 electrolyzers. Joule https://doi.org/10.1016/j.joule.2024.02.027 (2024).
Sisler, J. et al. Ethylene electrosynthesis: a comparative techno-economic analysis of alkaline vs membrane electrode assembly vs CO2–CO–C2H4 tandems. ACS Energy Lett. 6, 997–1002 (2021).
García De Arquer, F. P. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Nesbitt, N. T. et al. Liquid–solid boundaries dominate activity of CO2 reduction on gas-diffusion electrodes. ACS Catal. 10, 14093–14106 (2020).
Biemolt, J., Singh, J., Prats Vergel, G., Pelzer, H. M. & Burdyny, T. Preventing salt formation in zero-gap CO2 electrolyzers by quantifying cation accumulation. ACS Energy Lett. 10, 807–814 (2025).
Nesbitt, N. T. & Smith, W. A. Water and solute activities regulate CO2 reduction in gas-diffusion electrodes. J. Phys. Chem. C 125, 13085–13095 (2021).
Zhang, H., Gao, J., Raciti, D. & Hall, A. S. Promoting Cu-catalysed CO2 electroreduction to multicarbon products by tuning the activity of H2O. Nat. Catal. 6, 807–817 (2023).
Luo, X., Rojas-Carbonell, S., Yan, Y. & Kusoglu, A. Structure–transport relationships of poly(aryl piperidinium) anion-exchange membranes: effect of anions and hydration. J. Membr. Sci. 598, 117680 (2020).
Wheeler, D. G. et al. Quantification of water transport in a CO2 electrolyzer. Energy Environ. Sci. 13, 5126–5134 (2020).
Hurkmans, J.-W., Pelzer, H. M., Burdyny, T., Peeters, J. & Vermaas, D. A. Heating dictates the scalability of CO2 electrolyzer types. EES Catal. https://doi.org/10.1039/D4EY00190G (2025)
Jensen, J. K. et al. Heat pump COP, part 2: generalized COP estimation of heat pump processes. In Proc. 13th IIR Gustav Lorentzen Conference on Natural Refrigerants (GL2018) (International Institute of Refrigeration, 2018); https://doi.org/10.18462/IIR.GL.2018.1386
Siegmund, D. et al. Crossing the valley of death: from fundamental to applied research in electrolysis. JACS Au 1, 527–535 (2021).
Garg, S. et al. How alkali cations affect salt precipitation and CO2 electrolysis performance in membrane electrode assembly electrolyzers. Energy Environ. Sci. 16, 1631–1643 (2023).
Solubility Table for Water at Temperature (Merck, accessed 10 September 2024); https://www.sigmaaldrich.com/NL/en/support/calculators-and-apps/solubility-table-compounds-water-temperature
Sustainion® Anion Exchange Membranes (Dioxide Materials, accessed 11 September 2024); https://dioxidematerials.com/technology/sustainion-membranes/
Vaughan, G. L. & Carrington, C. G. Psychrometric properties of a moist carbon dioxide atmosphere. Int. J. Food Prop. 1, 77–87 (1998).
Zheng, Y. et al. Energy related CO2 conversion and utilization: advanced materials/nanomaterials, reaction mechanisms and technologies. Nano Energy 40, 512–539 (2017).
Acknowledgements
This project received funding from project “e-Heat: Understanding and controlling heat to enable large scale electrolysers” (NWO OTP 19757).
Author information
Authors and Affiliations
Contributions
Conceptualization was by H.M.P., D.V. and T.B. Visualization was by H.M.P., N.K., D.V. and T.B. The original draught was written by H.M.P., N.K., D.V. and T.B., and the manuscript was reviewed and edited by H.M.P., D.V. and T.B. Funding acquisition was by D.V. and T.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Feng Jiao, Guenter Schmid and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pelzer, H.M., Kolobov, N., Vermaas, D.A. et al. Scaling and heating will drive low-temperature CO2 electrolysers to operate at higher temperatures. Nat Energy 10, 549–556 (2025). https://doi.org/10.1038/s41560-025-01745-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01745-5
This article is cited by
-
Synergistic electrode design for efficient CO2 electrolysis to multicarbon products at elevated temperatures
Nature Communications (2026)
-
A cation-functionalized layer for ethylene electrosynthesis via CO reduction paired with H2 oxidation in a pure-water-fed solid-state electrolyser
Nature Energy (2026)
-
Water dissociation efficiencies control the viability of reverse-bias bipolar membranes for CO2 electrolysis
Nature Chemical Engineering (2025)