Abstract
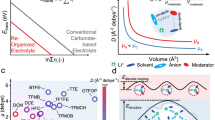
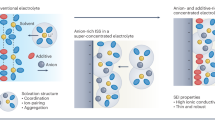
Ion association in highly concentrated electrolytes and localized highly concentrated electrolytes facilitates solid electrolyte interphase formation but compromises the thermal stability. Here we investigated the thermal behaviours of 20 electrolytes and uncovered that ion association lowers exothermic onset temperature by ~94 °C. To enhance the thermal stability without impairing solid electrolyte interphase formation, we developed a solvent-relay strategy that promotes ion association at ambient temperature while inducing dissociation at elevated temperatures. This approach enabled 4.5-V graphite-NCM811 pouch cells (1.1 Ah) to deliver 1,000 cycles under 0.45 C over 4,100 h with ~81.9% capacity retention and exceptional thermal safety, with a temperature rise lower than 3.5 °C during nail penetration, compared with 555.2 °C for commercial carbonate-based electrolytes. These findings elucidate the pivotal role of ion association in thermal runaway and offer a viable strategy to simultaneously achieve long cycle life, high-voltage operation and enhanced safety in ampere-hour-scale lithium-ion batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets analysed and generated in this study are included in the paper and its Supplementary Information.
References
Goodenough, J. B. & Park, K.-S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 135, 1167–1176 (2013).
Xie, J. & Lu, Y.-C. A retrospective on lithium-ion batteries. Nat. Commun. 11, 2499 (2020).
Li, M., Lu, J., Chen, Z. & Amine, K. 30 years of lithium-ion batteries. Adv. Mater. 30, 1800561 (2018).
An, Y., Zeng, Y., Luan, D. & Lou, X. W. D. Materials design for high-energy-density anode-free batteries. Matter 7, 1466–1502 (2024).
Manthiram, A. An outlook on lithium-ion battery technology. ACS Cent. Sci. 3, 1063–1069 (2017).
Wang, Z. et al. Thermally rearranged covalent organic framework with flame-retardancy as a high safety Li-ion solid electrolyte. eScience 2, 311–318 (2022).
Zhang, H. et al. A polymer electrolyte with a thermally induced interfacial ion-blocking function enables safety-enhanced lithium metal batteries. eScience 2, 201–208 (2022).
Hobold, G. M. et al. Moving beyond 99.9% coulombic efficiency for lithium anodes in liquid electrolytes. Nat. Energy 6, 951–960 (2021).
Efaw, C. M. et al. Localized high-concentration electrolytes get more localized through micelle-like structures. Nat. Mater. 22, 1531–1539 (2023).
Feng, X., Ren, D., He, X. & Ouyang, M. Mitigating thermal runaway of lithium-ion batteries. Joule 4, 743–770 (2020).
Gond, R., van Ekeren, W., Mogensen, R., Naylor, A. J. & Younesi, R. Non-flammable liquid electrolytes for safe batteries. Mater. Horiz. 8, 2913–2928 (2021).
Davies, C. W. Ion Association (Butterworths, 1962).
Yamada, Y., Wang, J., Ko, S., Watanabe, E. & Yamada, A. Advances and issues in developing salt-concentrated battery electrolytes. Nat. Energy 4, 269–280 (2019).
Jiao, S. et al. Stable cycling of high-voltage lithium metal batteries in ether electrolytes. Nat. Energy 3, 739–746 (2018).
Wang, Y. et al. Challenges and opportunities to mitigate the catastrophic thermal runaway of high-energy batteries. Adv. Energy Mater. 13, 2203841 (2023).
Wang, W. et al. Deciphering advanced sensors for life and safety monitoring of lithium batteries. Adv. Energy Mater. 14, 2304173 (2024).
Jia, H. et al. Is nonflammability of electrolyte overrated in the overall safety performance of lithium ion batteries? A sobering revelation from a completely nonflammable electrolyte. Adv. Energy Mater. 13, 2203144 (2023).
Hou, J. et al. Thermal runaway of lithium-ion batteries employing LiN(SO2F)2-based concentrated electrolytes. Nat. Commun. 11, 5100 (2020).
Jie, Y. et al. Towards long-life 500 Wh kg−1 lithium metal pouch cells via compact ion-pair aggregate electrolytes. Nat. Energy 9, 987–998 (2024).
Huang, J. et al. Operando decoding of chemical and thermal events in commercial Na(Li)-ion cells via optical sensors. Nat. Energy 5, 674–683 (2020).
Zhao, L., Inoishi, A. & Okada, S. Thermal risk evaluation of concentrated electrolytes for Li-ion batteries. J. Power Sources Adv. 12, 100079 (2021).
Ren, D. et al. Model-based thermal runaway prediction of lithium-ion batteries from kinetics analysis of cell components. Appl. Energy 228, 633–644 (2018).
Ko, S. et al. Electrode potential influences the reversibility of lithium-metal anodes. Nat. Energy 7, 1217–1224 (2022).
Wu, J. et al. In situ detecting thermal stability of solid electrolyte interphase (SEI). Small 19, 2208239 (2023).
Lu, Z. et al. Conformational isomerism breaks the electrolyte solubility limit and stabilizes 4.9 V Ni-rich layered cathodes. Nat. Commun. 15, 9108 (2024).
Hobold, G. M., Wang, C., Steinberg, K., Li, Y. & Gallant, B. M. High lithium oxide prevalence in the lithium solid–electrolyte interphase for high coulombic efficiency. Nat. Energy 9, 580–591 (2024).
Gillespie, R. J. & Robinson, E. A. The raman spectra of sulphuric, deuterosulphuric, fluorosulphuric, chlorosulphuric, and methanesulphonic acids and their anions. Can. J. Chem. 40, 644–657 (1962).
McLain, S. E., Benmore, C. J. & Turner, J. F. C. The structure of liquid fluorosulfuric acid investigated by neutron diffraction. J. Chem. Phys. 117, 3816–3821 (2002).
Kerner, M., Plylahan, N., Scheers, J. & Johansson, P. Thermal stability and decomposition of lithium bis(fluorosulfonyl)imide (LiFSI) salts. RSC Adv. 6, 23327–23334 (2016).
Zhou, S. et al. Direct evidences for bis(fluorosulfonyl)imide anion hydrolysis in industrial production: pathways based on thermodynamics analysis and theoretical simulation. J. Power Sources 577, 233249 (2023).
Li, Z. et al. Non-polar ether-based electrolyte solutions for stable high-voltage non-aqueous lithium metal batteries. Nat. Commun. 14, 868 (2023).
Chen, X., Zhang, X., Li, H. & Zhang, Q. Cation–solvent, cation–anion, and solvent–solvent interactions with electrolyte solvation in lithium batteries. Batteries Supercaps 2, 128–131 (2019).
Borodin, O., Smith, G. D. & Henderson, W. Li+ cation environment, transport, and mechanical properties of the LiTFSI doped N-methyl-N-alkylpyrrolidinium+TFSI− ionic liquids. J. Phys. Chem. B 110, 16879–16886 (2006).
Xu, J. et al. Electrolyte design for Li-ion batteries under extreme operating conditions. Nature 614, 694–700 (2023).
Kwon, H. et al. Borate–pyran lean electrolyte-based Li-metal batteries with minimal Li corrosion. Nat. Energy 9, 57–69 (2023).
Ko, S. et al. Electrolyte design for lithium-ion batteries with a cobalt-free cathode and silicon oxide anode. Nat. Sustain. 6, 1705–1714 (2023).
Ubaldi, S. et al. Suppression capacity and environmental impact of three extinguishing agents for lithium-ion battery fires. Energy Environ. Sci. 10, 100810 (2024).
Wu, Y. et al. High-voltage and high-safety practical lithium batteries with ethylene carbonate-free electrolyte. Adv. Energy Mater. 11, 2102299 (2021).
Yi, X. et al. Safe electrolyte for long-cycling alkali-ion batteries. Nat. Sustain. 7, 326–337 (2024).
Fang, M., Yue, X., Dong, Y., Chen, Y. & Liang, Z. A temperature-dependent solvating electrolyte for wide-temperature and fast-charging lithium metal batteries. Joule 8, 91–103 (2024).
Li, L. et al. Large-scale current collectors for regulating heat transfer and enhancing battery safety. Nat. Chem. Eng. 1, 542–551 (2024).
Lu, D. et al. Ligand-channel-enabled ultrafast Li-ion conduction. Nature 627, 101–107 (2024).
Xu, N. et al. In situ cross-linked F- and P-containing solid polymer electrolyte for long-cycling and high-safety lithium metal batteries with various cathode materials. Angew. Chem. Int. Ed. 63, e202404400 (2024).
Zhang, S. et al. In situ-polymerized lithium salt as a polymer electrolyte for high-safety lithium metal batteries. Energy Environ. Sci. 16, 2591–2602 (2023).
Meng, Y. et al. Designing phosphazene-derivative electrolyte matrices to enable high-voltage lithium metal batteries for extreme working conditions. Nat. Energy 8, 1023–1033 (2023).
Song, I. T. et al. Thermal runaway prevention through scalable fabrication of safety reinforced layer in practical Li-ion batteries. Nat. Commun. 15, 8294 (2024).
Cui, Z., Liu, C., Wang, F. & Manthiram, A. Navigating thermal stability intricacies of high-nickel cathodes for high-energy lithium batteries. Nat. Energy 10, 490–501 (2025).
Acknowledgements
The work described in this paper was supported by grants from the Research Grant Council (RGC) of the Hong Kong Special Administrative Region, China (project numbers RFS2223-4S03, R5019-22 and C1002-21G) and a grant from the Innovation and Technology Commission of the Hong Kong Special Administrative Region, China (project number ITS/219/21FP). Y.-C.L. acknowledges the support from Xplorer Prize by New Cornerstone Science Foundation.
Author information
Authors and Affiliations
Contributions
Y.S. and Y.-C.L. designed and conceived the experiments. C.Z. conducted pouch cell testing. W.W. conducted XPS tests. J.X. and L.J. conducted thermal analysis. H.W. conducted NMR, IR measurements and abuse tests. Y.S. and Y.-C.L. wrote the paper. Y.-C.L. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Fei Wang, Guy Marlair and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–37, Notes 1–4 and Tables 1–7.
Supplementary Video 1 (download MP4 )
Videos showing nail penetration tests of 1.1 Ah graphite-NCM811 pouch cells using commercial electrolytes (1 M LiPF₆ in, 1:1 vol%) and GBF-D2 electrolyte.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, Y., Zuo, C., Wang, H. et al. Designing safe and long-life lithium-ion batteries via a solvent-relay strategy. Nat Energy 10, 1450–1457 (2025). https://doi.org/10.1038/s41560-025-01888-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01888-5
This article is cited by
-
Safe, long-life lithium batteries via solvent-relay
Science China Materials (2026)