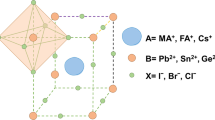
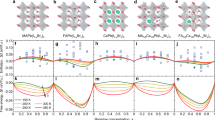
Abstract
Lead halide perovskites containing organic–inorganic cations are commonly used as absorber materials in high-performance perovskite solar cells. However, these materials are susceptible to degradation under device operational conditions, where illumination and/or bias trigger photo/electrochemical redox reactions that drive halide segregation, ion migration and ultimately perovskite decomposition and device failure. Here we discuss the effect of these photo/electrochemical redox reactions, taking into account not only commonly discussed halide oxidation but also irreversible reactions involving organic cations. We summarize possible oxidation and reduction reactions and outline key degradation pathways of organic cations under illumination and bias, highlighting their critical impact on the long-term stability of perovskite solar cells and the existing gaps in understanding. Finally, we discuss approaches to inhibit these undesirable reactions through organic cation design, additive incorporation and device architecture optimization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yu, X., Wang, Y. & Gao, P. The effect of redox reactions on the stability of perovskite solar cells. ChemPhotoChem 7, e202200311 (2023).
Xu, Z., Kerner, R. A., Kronik, L. & Rand, B. P. Beyond ion migration in metal halide perovskites: toward a broader photoelectrochemistry perspective. ACS Energy Lett. 9, 4645–4654 (2024).
DuBose, J. T. & Kamat, P. V. Hole trapping in halide perovskites induces phase segregation. Acc. Mater. Res. 3, 761–771 (2022).
Xu, Z., Kerner, R. A., Berry, J. J. & Rand, B. P. Iodine electrochemistry dictates voltage-induced halide segregation thresholds in mixed-halide perovskite devices. Adv. Funct. Mater. 32, 2203432 (2022).
Jiang, F. et al. Improved reverse bias stability in p–i–n perovskite solar cells with optimized hole transport materials and less reactive electrodes. Nat. Energy 9, 1275–1284 (2024).
Xu, Z. et al. Halogen redox shuttle explains voltage-induced halide redistribution in mixed-halide perovskite devices. ACS Energy Lett. 8, 513–520 (2023).
Frolova, L. A. et al. Reversible Pb2+/Pb0 and I−/I3− redox chemistry drives the light-induced phase segregation in all-inorganic mixed halide perovskites. Adv. Energy Mater. 11, 2002934 (2021).
Ni, Z. et al. Evolution of defects during the degradation of metal halide perovskite solar cells under reverse bias and illumination. Nat. Energy 7, 65–73 (2022).
Hu, J. et al. Triiodide attacks the organic cation in hybrid lead halide perovskites: mechanism and suppression. Adv. Mater. 35, 2303373 (2023).
Yamilova, O. R. et al. Reduction of methylammonium cations as a major electrochemical degradation pathway in MAPbI3 perovskite solar cells. J. Phys. Chem. Lett. 11, 221–228 (2020).
Akbulatov, A. F. et al. Light or heat: what is killing lead halide perovskites under solar cell operation conditions? Chem. Lett. 11, 333–339 (2020).
Juarez-Perez, E. J., Ono, L. K. & Qi, Y. Thermal degradation of formamidinium based lead halide perovskites into sym-triazine and hydrogen cyanide observed by coupled thermogravimetry-mass spectrometry analysis. J. Mater. Chem. A 7, 16912–16919 (2019).
Juarez-Perez, E. J. et al. Photodecomposition and thermal decomposition in methylammonium halide lead perovskites and inferred design principles to increase photovoltaic device stability. J. Mater. Chem. A 6, 9604–9612 (2018).
Juarez-Perez, E. J., Hawash, Z., Raga, S. R., Ono, L. K. & Qi, Y. Thermal degradation of CH3NH3PbI3 perovskite into NH3 and CH3I gases observed by coupled thermogravimetry-mass spectrometry analysis. Energy Environ. Sci. 9, 3406–3410 (2016).
Juarez-Perez, E. J., Ono, L. K., Uriarte, I., Cocinero, E. J. & Qi, Y. Degradation mechanism and relative stability of methylammonium halide based perovskites analyzed on the basis of acid-base theory. ACS Appl. Mater. Interfaces 11, 12586–12593 (2019).
Shi, L. et al. Gas chromatography-mass spectrometry analyses of encapsulated stable perovskite solar cells. Science 368, eaba2412 (2020).
Cira, S. G. et al. Light-induced degradation of mixed-cation, mixed-halide perovskite: observed rates and influence of oxygen. J. Mater. Chem. A 13, 5033–5044 (2025).
Ren, Z. et al. Increased resistance to photooxidation in Dion–Jacobson lead halide perovskites: implication for perovskite device stability. Matter 8, 101937 (2025).
Nickel, N. H., Lang, F., Brus, V. V., Shargaieva, O. & Rappich, J. Unraveling the light-induced degradation mechanisms of CH3NH3PbI3 perovskite films. Adv. Electron. Mater. 3, 1700158 (2017).
Kerner, R. A., Xu, Z., Larson, B. W. & Rand, B. P. The role of halide oxidation in perovskite halide phase separation. Joule 5, 2273–2295 (2021).
Chen, S. et al. Atomic-scale imaging of CH3NH3PbI3 structure and its decomposition pathway. Nat. Commun. 12, 5516 (2021).
Mathew, P. S., Samu, G. F., Janáky, C. & Kamat, P. V. Iodine (I) expulsion at photoirradiated mixed halide perovskite interface. Should I stay or should I go? ACS Energy Lett. 5, 1872–1880 (2020).
Samu, G. F. et al. Electrochemical hole injection selectively expels iodide from mixed halide perovskite films. J. Am. Chem. Soc. 141, 10812–10820 (2019).
McGovern, L., Futscher, M. H., Muscarella, L. A. & Ehrler, B. Understanding the stability of MAPbBr3 versus MAPbI3: suppression of methylammonium migration and reduction of halide migration. J. Phys. Chem. Lett. 11, 7127–7132 (2020).
Minns, J., Zajdel, P., Chernyshov, D., Van Beek, W. & Green, M. Structure and interstitial iodide migration in hybrid perovskite methylammonium lead iodide. Nat. Commun. 8, 15152 (2017).
Liu, Y. et al. Direct observation of photoinduced ion migration in lead halide perovskites. Adv. Funct. Mater. 31, 2008777 (2021).
Nguyen, L. L. et al. Multitechnique detection of lead iodide hybrid perovskite degradation pathways under varying electric fields. ACS Nano 19, 21927–21941 (2025).
Lee, M., Vigil, J. A., Jiang, Z. & Karunadasa, H. I. Evidence for I2 loss from the perovskite-gas interface upon light-induced halide segregation. Chem. Sci. 16, 9662–9668 (2025).
Li, W., Hao, M., Baktash, A., Wang, L. & Etheridge, J. The role of ion migration, octahedral tilt, and the A-site cation on the instability of Cs1 − xFAxPbI3. Nat. Commun. 14, 8523 (2023).
Emelianov, N. A. et al. Electric-field-induced aging dynamics of triple-cation lead iodide perovskite at nanoscale. Sol. Energy Mater. Sol. Cells 282, 113305 (2025).
Kerner, R. A. et al. Low threshold voltages electrochemically drive gold migration in halide perovskite devices. ACS Energy Lett. 5, 3352–3356 (2020).
Kerner, R. A. et al. Amine additive reactions induced by the soft Lewis acidity of Pb2+ in halide perovskites. Part II: impacts of amido Pb impurities in methylammonium lead triiodide thin films. J. Mater. Chem. C 7, 5244–5250 (2019).
Hu, J., Kerner, R. A., Pelczer, I., Rand, B. P. & Schwartz, J. Organoammonium-ion-based perovskites can degrade to Pb0 via amine-Pb(II) coordination. ACS Energy Lett. 6, 2262–2267 (2021).
Wang, S., Jiang, Y., Juarez-Perez, E. J., Ono, L. K. & Qi, Y. Accelerated degradation of methylammonium lead iodide perovskites induced by exposure to iodine vapour. Nat. Energy 2, 16195 (2016).
Ren, X. et al. Mobile iodides capture for highly photolysis- and reverse-bias-stable perovskite solar cells. Nat. Mater. 23, 810–817 (2024).
Ishihara, K. & Muniz, K. Iodine Catalysis in Organic Synthesis (Wiley, 2022).
Ye, J. et al. Extending shelf-life of two-step method precursor solutions through targeted regulation for highly efficient and reproducible perovskite solar cells. Angew. Chem. Int. Ed. 63, e202411708 (2024).
Dong, Q. et al. Critical role of organoamines in the irreversible degradation of a metal halide perovskite precursor colloid: mechanism and inhibiting strategy. ACS Energy Lett. 7, 481–489 (2021).
Liang, Z. et al. Unraveling the irreversible transformation by nucleophilic substitution: a hint for fully transparent perovskite. EcoMat 4, e12211 (2022).
Wang, X. et al. Perovskite solution aging: what happened and how to inhibit? Chem 6, 1369–1378 (2020).
Wang, M. et al. Ammonium cations with high pKa in perovskite solar cells for improved high-temperature photostability. Nat. Energy 8, 1229–1239 (2023).
Zhou, S. et al. Reactive passivation of wide-bandgap organic-inorganic perovskites with benzylamine. J. Am. Chem. Soc. 146, 27405–27416 (2024).
Tay, D. J. J. et al. Understanding the mechanisms of methylammonium-induced thermal instability in mixed-FAMA perovskites. Small 21, 2403389 (2025).
Akbulatov, A. F. et al. Temperature dynamics of MAPbI3 and PbI2 photolysis: revealing the interplay between light and heat, two enemies of perovskite photovoltaics. J. Phys. Chem. Lett. 12, 4362–4367 (2021).
Li, G. et al. Highly efficient p-i-n perovskite solar cells that endure temperature variations. Science 379, 399–403 (2023).
Jiang, Q. et al. Towards linking lab and field lifetimes of perovskite solar cells. Nature 623, 313–318 (2023).
Wang, L. et al. A Eu3+-Eu2+ ion redox shuttle imparts operational durability to Pb-I perovskite solar cells. Science 363, 265–270 (2019).
Meng, H. et al. Inhibition of halide oxidation and deprotonation of organic cations with dimethylammonium formate for air-processed p-i-n perovskite solar cells. Nat. Energy 9, 536–547 (2024).
Zhang, Y. et al. Uncovering aging chemistry of perovskite precursor solutions and anti-aging mechanism of additives. Angew. Chem. Int. Ed. 135, e202215799 (2023).
Wang, L. et al. Over 19% efficiency perovskite solar modules by simultaneously suppressing cation deprotonation and iodide oxidation. ACS Appl. Mater. Interfaces 16, 4751–4762 (2024).
Gou, Y. et al. Suppressing deprotonation to extend the shelf life of perovskite precursor solutions and enhance the stability and efficiency of perovskite solar cells. Adv. Funct. Mater. 35, 2505965 (2025).
Wu, S. et al. Redox mediator-stabilized wide-bandgap perovskites for monolithic perovskite-organic tandem solar cells. Nat. Energy 9, 411–421 (2024).
Yang, Y. et al. Amidination of ligands for chemical and field-effect passivation stabilizes perovskite solar cells. Science 386, 898–902 (2024).
Peng, Z. et al. Revealing degradation mechanisms in 3D/2D perovskite solar cells under photothermal accelerated ageing. Energy Environ. Sci. 17, 8313–8324 (2024).
Teale, S., Degani, M., Chen, B., Sargent, E. H. & Grancini, G. Molecular cation and low-dimensional perovskite surface passivation in perovskite solar cells. Nat. Energy 9, 779–792 (2024).
Zhong, C. et al. Instability of two-dimensional hybrid perovskites underpinned by organic molecule loss under light illumination. Mater. Chem. Front 8, 2836–2844 (2024).
Mathew, P. S., Szabó, G., Kuno, M. & Kamat, P. V. Phase segregation and sequential expulsion of iodide and bromide in photoirradiated Ruddlesden-Popper 2D perovskite films. ACS Energy Lett. 7, 3982–3988 (2022).
Acknowledgements
This work was supported by RGC CRF project 7018−20G, RGC GRF project 17311422, HRZZ project PZS-2019-02-2068 and Russian Science Foundation project no. 19-73-30020P.
Author information
Authors and Affiliations
Contributions
A.B.D. wrote the first draft of the manuscript. Images and boxes were prepared by Z.R., S.D., Z.Y. and V.K. The manuscript was written with contributions from all authors. All authors have given approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ren, Z., Dolić, S., Kojić, V. et al. Irreversible organic cations chemistry limits organic–inorganic halide perovskite stability under illumination or bias. Nat Energy (2026). https://doi.org/10.1038/s41560-026-01965-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41560-026-01965-3