Abstract
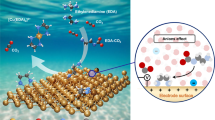
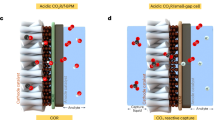
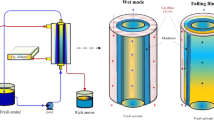
Operating anion-exchange membrane fuel cells (AEMFCs) with CO2-containing ambient-air feed is conventionally seen as a difficult challenge detrimental to cell performance, because CO2 reacts with hydroxide ions generated at the cathode, forming (bi)carbonate ions that reduce AEM ionic conductivity. In this Perspective, we discuss the effect of CO2 in operando AEMFCs, which involves a complex interplay of multi-anion transport, concentration polarization, back-diffusion, changes in water distribution, cation stability and variations in local pH. We argue that the negative effects of CO2 may be managed to minimize them, and even exploited to advantage. We introduce two concepts: CO2 management and the positive stabilizing effect of CO2. The first, analogous to the water-management effect, relates to phenomena and strategies to balance, transport and utilize CO2-related species to enhance AEMFC performance. The second, although counterintuitive, relates to the potentially positive effects that CO2 may impart to ambient-air cell operation, in particular towards improving long-term performance stability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yang, Y. et al. Electrocatalysis in alkaline media and alkaline membrane-based energy technologies. Chem. Rev. 122, 6117–6321 (2022).
Yang, Y. et al. Anion-exchange membrane water electrolyzers and fuel cells. Chem. Soc. Rev. 51, 9620–9693 (2022).
Adabi, H. et al. High-performing commercial Fe–N–C cathode electrocatalyst for anion-exchange membrane fuel cells. Nat. Energy 6, 834–843 (2021).
Hossen, M. d. M. et al. State-of-the-art and developmental trends in platinum group metal-free cathode catalyst for anion exchange membrane fuel cell (AEMFC). Appl. Catal. B 325, 121733 (2023).
Ni, W. et al. An efficient nickel hydrogen oxidation catalyst for hydroxide exchange membrane fuel cells. Nat. Mater. 21, 804–810 (2022).
Ren, J.-T., Chen, L., Wang, H.-Y., Feng, Y. & Yuan, Z.-Y. Hydrogen oxidation electrocatalysts for anion-exchange membrane fuel cells: activity descriptors, stability regulation, and perspectives. Energy Environ. Sci. 17, 3960–4009 (2024).
Liu, X. et al. Magnetic-field-oriented mixed-valence-stabilized ferrocenium anion-exchange membranes for fuel cells. Nat. Energy 7, 329–339 (2022).
Fan, J. et al. Poly(bis-arylimidazoliums) possessing high hydroxide ion exchange capacity and high alkaline stability. Nat. Commun. 10, 2306 (2019).
Wang, J. et al. Poly(aryl piperidinium) membranes and ionomers for hydroxide exchange membrane fuel cells. Nat. Energy 4, 392–398 (2019).
Chen, N. et al. Poly(fluorenyl aryl piperidinium) membranes and ionomers for anion exchange membrane fuel cells. Nat. Commun. 12, 2367 (2021).
Abbasi, R., Setzler, B. P. & Yan, Y. Material and system development needs for widespread deployment of hydroxide exchange membrane fuel cells in light-duty vehicles. Energy Environ. Sci. 16, 4404–4422 (2023).
Hyun, J. & Kim, H.-T. Powering the hydrogen future: current status and challenges of anion exchange membrane fuel cells. Energy Environ. Sci. 16, 5633–5662 (2023).
Douglin, J. C. et al. High-performance ionomerless cathode anion-exchange membrane fuel cells with ultra-low-loading Ag–Pd alloy electrocatalysts. Nat. Energy 8, 1262–1272 (2023).
Ul Hassan, N. et al. Achieving high-performance and 2000 h stability in anion exchange membrane fuel cells by manipulating ionomer properties and electrode optimization. Adv. Energy Mater. 10, 2001986 (2020).
Ziv, N., Mustain, W. E. & Dekel, D. R. The effect of ambient carbon dioxide on anion-exchange membrane fuel cells. ChemSusChem 11, 1136–1150 (2018).
Peng, J., Roy, A. L., Greenbaum, S. G. & Zawodzinski, T. A. Effect of CO2 absorption on ion and water mobility in an anion exchange membrane. J. Power Sources 380, 64–75 (2018).
Shi, L. et al. A shorted membrane electrochemical cell powered by hydrogen to remove CO2 from the air feed of hydroxide exchange membrane fuel cells. Nat. Energy 7, 238–247 (2022).
Liang, Z. et al. Designing the feasible membrane systems for CO2 removal from air-fed anion-exchange membrane fuel cells. Sep. Purif. Technol. 289, 120713 (2022).
Dekel, D. R. Review of cell performance in anion exchange membrane fuel cells. J. Power Sources 375, 158–169 (2018).
Piana, M. et al. H2/air alkaline membrane fuel cell performance and durability, using novel ionomer and non-platinum group metal cathode catalyst. J. Power Sources 195, 5875–5881 (2010).
Li, G. et al. Carbonation effects on the performance of alkaline polymer electrolyte fuel cells. Int. J. Hydrogen Energy 40, 6655–6660 (2015).
Grew, K. N., Ren, X. & Chu, D. Effects of temperature and carbon dioxide on anion exchange membrane conductivity. Electrochem. Solid State Lett. 14, B127–B131 (2011).
Krewer, U., Weinzierl, C., Ziv, N. & Dekel, D. R. Impact of carbonation processes in anion exchange membrane fuel cells. Electrochim. Acta 263, 433–446 (2018).
Zhegur-Khais, A., Kubannek, F., Krewer, U. & Dekel, D. R. Measuring the true hydroxide conductivity of anion exchange membranes. J. Membr. Sci. 612, 118461 (2020).
Gerhardt, M. R., Pant, L. M. & Weber, A. Z. Along-the-channel impacts of water management and carbon-dioxide contamination in hydroxide-exchange-membrane fuel cells: a modeling study. J. Electrochem. Soc. 166, F3180–F3192 (2019).
Zheng, Y. et al. Quantifying and elucidating the effect of CO2 on the thermodynamics, kinetics and charge transport of AEMFCs. Energy Environ. Sci. 12, 2806–2819 (2019).
Li, Q. et al. Origin of performance decline in carbonated anion exchange membrane fuel cells. J. Am. Chem. Soc. 146, 33587–33594 (2024).
Xue, J. et al. High-temperature anion-exchange membrane fuel cells with balanced water management and enhanced stability. Joule 8, 1457–1477 (2024).
Diesendruck, C. E. & Dekel, D. R. Water—a key parameter in the stability of anion exchange membrane fuel cells. Curr. Opin. Electrochem. 9, 173–178 (2018).
Wrubel, J. A. et al. Anion exchange membrane ionic conductivity in the presence of carbon dioxide under fuel cell operating conditions. J. Electrochem. Soc. 164, F1063–F1073 (2017).
Lang, C. M., Kim, K. & Kohl, P. A. High-energy density, room-temperature carbonate fuel cell. Electrochem. Solid State Lett. 9, A545–A548 (2006).
Wain, A. J. & Smith, G. Monitoring carbonate and bicarbonate within an anion exchange membrane during self-purge using infrared spectroelectrochemistry. ACS Electrochem. 1, 1794–1802 (2025).
Zheng, Y. et al. Effect of reacting gas flowrates and hydration on the carbonation of anion exchange membrane fuel cells in the presence of CO2. J. Power Sources 467, 228350 (2020).
Divekar, A. G. et al. In-depth understanding of the CO2 limitation of air fed anion exchange membrane fuel cells. Sustain. Energy Fuels 4, 1801–1811 (2020).
Katayama, Y. et al. Anion-exchange membrane fuel cells with improved CO2 tolerance: impact of chemically induced bicarbonate ion consumption. ACS Appl. Mater. Interfaces 9, 28650–28658 (2017).
Page, M., Dekel, D. R., Gottesfeld, Z. & Gottesfeld, S. Use of ammonia as source of hydrogen fuel and as a getter for air–CO2 in alkaline membrane fuel cells. European patent 2,735,046 (2017).
Chatenet, M. et al. Water electrolysis: from textbook knowledge to the latest scientific strategies and industrial developments. Chem. Soc. Rev. 51, 4583–4762 (2022).
Tuckerman, M. E., Marx, D. & Parrinello, M. The nature and transport mechanism of hydrated hydroxide ions in aqueous solution. Nature 417, 925–929 (2002).
Leung, K., Nielsen, I. M. B. & Kurtz, I. Ab Initio molecular dynamics study of carbon dioxide and bicarbonate hydration and the nucleophilic attack of hydroxide on CO2. J. Phys. Chem. B 111, 4453–4459 (2007).
Srebnik, S., Pusara, S. & Dekel, D. R. Effect of carbonate anions on quaternary ammonium–hydroxide interaction. J. Phys. Chem. C 123, 15956–15962 (2019).
Zelovich, T., Simari, C., Nicotera, I., Dekel, D. R. & Tuckerman, M. E. The impact of carbonation on hydroxide diffusion in nano-confined anion exchange membranes. J. Mater. Chem. A 10, 11137–11149 (2022).
Willdorf-Cohen, S., Li, S., Srebnik, S., Diesendruck, C. E. & Dekel, D. R. Does the presence of CO2 affect the alkaline stability of anion-exchange membranes? J. Mater. Chem. A 13, 35316–35328 (2025).
Weiss, R., Schwab, O. & Hampel, F. Ion-pair strain as the driving force for hypervalent adduct formation between iodide ions and substituted iodobenzenes: structural alternatives to Meisenheimer complexes. Chem. Eur. J. 5, 968–974 (1999).
Zahan, S. M. et al. Can anion-exchange membrane fuel cells operate with ambient air? ACS Energy Lett. 11, 1688–1695 (2026).
Zou, X. et al. Unsupervised learning-guided accelerated discovery of alkaline anion exchange membranes for fuel cells. Angew. Chem. Int. Ed. 62, e202300388 (2023).
Zhang, Q. et al. Machine learning-aided design of highly conductive anion exchange membranes for fuel cells and water electrolyzers. Adv. Mater. 36, 2404981 (2024).
Acknowledgements
This work was partially funded by the Nancy & Stephen Grand Technion Energy Program (GTEP), by the Israeli Science Foundation (ISF), grant 169/22, by the Israeli Council for Higher Education, Flagship Initiatives and Applied Research Strategy and International Affairs Division on Sustainability and the Climate Crisis, grant 2072133, and with the support of the Israel National Institute for Energy Storage (INIES). M.D.G. acknowledges the support of the State Key Laboratory of Engines, Tianjin University, China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Gaohong He and Lorenz Gubler for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yassin, K., Willdorf-Cohen, S., Guiver, M.D. et al. Addressing the challenge of carbon dioxide in anion-exchange membrane fuel cells. Nat Energy (2026). https://doi.org/10.1038/s41560-026-01999-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41560-026-01999-7