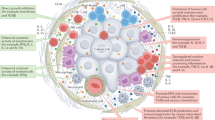
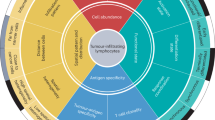
Abstract
Interleukins and associated cytokines serve as the means of communication for innate and adaptive immune cells as well as non-immune cells and tissues. Thus, interleukins have a critical role in cancer development, progression and control. Interleukins can nurture an environment enabling and favouring cancer growth while simultaneously being essential for a productive tumour-directed immune response. These properties of interleukins can be exploited to improve immunotherapies to promote effectiveness as well as to limit side effects. This Review aims to unravel some of these complex interactions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dunn, G. P., Old, L. J. & Schreiber, R. D. The three Es of cancer immunoediting. Annu. Rev. Immunol. 22, 329–360 (2004).
Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J. & Schreiber, R. D. Cancer immunoediting: from immunosurveillance to tumor escape. Nat. Immunol. 3, 991–998 (2002). Dunn and colleagues formulate a theory of individual cancer evolution under immunological pressure.
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Bruni, D., Angell, H. K. & Galon, J. The immune contexture and Immunoscore in cancer prognosis and therapeutic efficacy. Nat. Rev. Cancer https://doi.org/10.1038/s41568-020-0285-7 (2020).
Pages, F. et al. International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391, 2128–2139 (2018). This research validates the prognostic value of colon cancer classification according to the immune status of the tumour.
Mascaux, C. et al. Immune evasion before tumour invasion in early lung squamous carcinogenesis. Nature 571, 570–575 (2019).
Yuzhalin, A. E. & Kutikhin, A. G. Interleukins in Cancer Biology: their Heterogeneous Role (Academic Press, 2014).
Waldmann, T. A. Cytokines in cancer immunotherapy. Cold Spring Harb. Perspect. Biol. https://doi.org/10.1101/cshperspect.a028472 (2018).
Dinarello, C. A. The IL-1 family of cytokines and receptors in rheumatic diseases. Nat. Rev. Rheumatol. 15, 612–632 (2019).
Dinarello, C. A. Why not treat human cancer with interleukin-1 blockade? Cancer Metastasis Rev. 29, 317–329 (2010). Here IL-1 blockade is proposed for the first time as the means to tame chronic inflammation and halting tumor progression.
Elinav, E. et al. Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat. Rev. Cancer 13, 759–771 (2013).
Mantovani, A., Dinarello, C. A., Molgora, M. & Garlanda, C. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity 50, 778–795 (2019).
Lamkanfi, M. & Dixit, V. M. Mechanisms and functions of inflammasomes. Cell 157, 1013–1022 (2014).
Zheng, D., Liwinski, T. & Elinav, E. Inflammasome activation and regulation: toward a better understanding of complex mechanisms. Cell Discov. 6, 36 (2020).
Taniguchi, S. et al. Tumor-initiating cells establish an IL-33-TGF-beta niche signaling loop to promote cancer progression. Science https://doi.org/10.1126/science.aay1813 (2020). A mechanism by which tumour-initiating cells create a favourable environment that fosters tumour growth is demonstrated here.
Andersson, P. et al. Molecular mechanisms of IL-33-mediated stromal interactions in cancer metastasis. JCI Insight https://doi.org/10.1172/jci.insight.122375 (2018).
Tu, S. et al. Overexpression of interleukin-1 beta induces gastric inflammation and cancer and mobilizes myeloid-derived suppressor cells in mice. Cancer Cell 14, 408–419 (2008).
Whitley, S. K. et al. IL-1R signaling promotes STAT3 and NF-kappaB factor recruitment to distal cis-regulatory elements that regulate Il17a/f transcription. J. Biol. Chem. 293, 15790–15800 (2018).
Dmitrieva-Posocco, O. et al. Cell-type-specific responses to interleukin-1 control microbial invasion and tumor-elicited inflammation in colorectal cancer. Immunity 50, 166–180 e167 (2019). Dmitrieva-Posocco and colleagues demonstrate differential cell type-specific immune responses to IL-1 in chronic inflammation and colorectal cancer.
Grivennikov, S. I. et al. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17-mediated tumour growth. Nature 491, 254–258 (2012).
Jin, C. et al. Commensal microbiota promote lung cancer development via gammadelta T cells. Cell 176, 998–1013 e1016 (2019). This publication delineates a mechanism by which the commensal microbiota may trigger a cascade of protumorigenic interleukin-mediated responses in the lung.
Voigt, C. et al. Cancer cells induce interleukin-22 production from memory CD4+ T cells via interleukin-1 to promote tumor growth. Proc. Natl Acad. Sci. USA 114, 12994–12999 (2017).
Dudakov, J. A., Hanash, A. M. & van den Brink, M. R. Interleukin-22: immunobiology and pathology. Annu. Rev. Immunol. 33, 747–785 (2015).
Ngo, V. L. et al. A cytokine network involving IL-36gamma, IL-23, and IL-22 promotes antimicrobial defense and recovery from intestinal barrier damage. Proc. Natl Acad. Sci. USA 115, E5076–E5085 (2018).
Gronke, K. et al. Interleukin-22 protects intestinal stem cells against genotoxic stress. Nature 566, 249–253 (2019). Gronke and colleagues define a mechanism by which IL-22 prevents the malignant transformation of the intestinal epithelium.
Huber, S. et al. IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature 491, 259–263 (2012).
Perez, L. G. et al. TGF-beta signaling in Th17 cells promotes IL-22 production and colitis-associated colon cancer. Nat. Commun. 11, 2608 (2020).
Kobold, S. et al. Interleukin-22 is frequently expressed in small- and large-cell lung cancer and promotes growth in chemotherapy-resistant cancer cells. J. Thorac. Oncol. 8, 1032–1042 (2013).
Kryczek, I. et al. IL-22+CD4+ T cells promote colorectal cancer stemness via STAT3 transcription factor activation and induction of the methyltransferase DOT1L. Immunity 40, 772–784 (2014).
Katara, G. K. et al. Interleukin-22 promotes development of malignant lesions in a mouse model of spontaneous breast cancer. Mol. Oncol. 14, 211–224 (2020).
Khosravi, N. et al. IL-22 promotes Kras-mutant lung cancer by induction of a protumor immune response and protection of stemness properties. Cancer Immunol. Res. 6, 788–797 (2018).
Perusina Lanfranca, M. et al. Interleukin 22 signaling regulates acinar cell plasticity to promote pancreatic tumor development in mice. Gastroenterology 158, 1417–1432 e1411 (2020).
Lu, S. W. et al. IL-20 antagonist suppresses PD-L1 expression and prolongs survival in pancreatic cancer models. Nat. Commun. 11, 4611 (2020).
Huynh, J., Chand, A., Gough, D. & Ernst, M. Therapeutically exploiting STAT3 activity in cancer - using tissue repair as a road map. Nat. Rev. Cancer 19, 82–96 (2019).
Heinrich, P. C. et al. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem. J. 374, 1–20 (2003).
Jones, S. A. & Jenkins, B. J. Recent insights into targeting the IL-6 cytokine family in inflammatory diseases and cancer. Nat. Rev. Immunol. 18, 773–789 (2018).
Hirano, T. IL-6 in inflammation, autoimmunity and cancer. Int. Immunol. 33, 127–148 (2021).
Putoczki, T. L. et al. Interleukin-11 is the dominant IL-6 family cytokine during gastrointestinal tumorigenesis and can be targeted therapeutically. Cancer Cell 24, 257–271 (2013). This work defines a dominant role of IL-11 as a driver of procarcinogenic chronic inflammation in the intestine.
Grivennikov, S. I. IL-11: a prominent pro-tumorigenic member of the IL-6 family. Cancer Cell 24, 145–147 (2013).
Lokau, J. et al. Proteolytic cleavage governs interleukin-11 trans-signaling. Cell Rep. 14, 1761–1773 (2016).
Dong, C., Davis, R. J. & Flavell, R. A. MAP kinases in the immune response. Annu. Rev. Immunol. 20, 55–72 (2002).
Atsumi, T. et al. Inflammation amplifier, a new paradigm in cancer biology. Cancer Res. 74, 8–14 (2014).
Hunter, C. A. & Jones, S. A. IL-6 as a keystone cytokine in health and disease. Nat. Immunol. 16, 448–457 (2015).
Rose-John, S. Interleukin-6 family cytokines. Cold Spring Harb. Perspect Biol. https://doi.org/10.1101/cshperspect.a028415 (2018).
Ernst, M. et al. STAT3 and STAT1 mediate IL-11-dependent and inflammation-associated gastric tumorigenesis in gp130 receptor mutant mice. J. Clin. Invest. 118, 1727–1738 (2008).
Hu, B. et al. Microbiota-induced activation of epithelial IL-6 signaling links inflammasome-driven inflammation with transmissible cancer. Proc. Natl Acad. Sci. USA 110, 9862–9867 (2013).
Bollrath, J. et al. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15, 91–102 (2009).
Phesse, T. J. et al. Partial inhibition of gp130-Jak-Stat3 signaling prevents Wnt-beta-catenin-mediated intestinal tumor growth and regeneration. Sci. Signal. 7, ra92 (2014).
Wu, Y. S. et al. Paracrine IL-6 signaling mediates the effects of pancreatic stellate cells on epithelial-mesenchymal transition via Stat3/Nrf2 pathway in pancreatic cancer cells. Biochim. Biophys. Acta Gen. Subj. 1861, 296–306 (2017).
van Duijneveldt, G., Griffin, M. D. W. & Putoczki, T. L. Emerging roles for the IL-6 family of cytokines in pancreatic cancer. Clin. Sci. 134, 2091–2115 (2020).
Caetano, M. S. et al. IL-6 blockade reprograms the lung tumor microenvironment to limit the development and progression of Kras-mutant lung cancer. Cancer Res. 76, 3189–3199 (2016).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1beta through HIF-1alpha. Nature 496, 238–242 (2013).
Biswas, S. K. Metabolic reprogramming of immune cells in cancer progression. Immunity 43, 435–449 (2015).
Knochelmann, H. M. et al. IL-6 fuels durable memory for Th17 cell-mediated responses to tumors. Cancer Res. 80, 3920–3932 (2020).
Liu, Y. et al. NCR− group 3 innate lymphoid cells orchestrate IL-23/IL-17 axis to promote hepatocellular carcinoma development. EBioMedicine 41, 333–344 (2019).
Teng, M. W. et al. IL-12 and IL-23 cytokines: from discovery to targeted therapies for immune-mediated inflammatory diseases. Nat. Med. 21, 719–729 (2015).
Langowski, J. L. et al. IL-23 promotes tumour incidence and growth. Nature 442, 461–465 (2006).
Guo, L. et al. IL-1 family members and STAT activators induce cytokine production by Th2, Th17, and Th1 cells. Proc. Natl Acad. Sci. USA 106, 13463–13468 (2009).
Wu, L. et al. A novel IL-17 signaling pathway controlling keratinocyte proliferation and tumorigenesis via the TRAF4-ERK5 axis. J. Exp. Med. 212, 1571–1587 (2015).
Zhao, J., Chen, X., Herjan, T. & Li, X. The role of interleukin-17 in tumor development and progression. J. Exp. Med. https://doi.org/10.1084/jem.20190297 (2020).
Zepp, J. A. et al. IL-17A-induced PLET1 expression contributes to tissue repair and colon tumorigenesis. J. Immunol. 199, 3849–3857 (2017).
Chen, X. et al. IL-17R-EGFR axis links wound healing to tumorigenesis in Lrig1+ stem cells. J. Exp. Med. 216, 195–214 (2019).
Merrouche, Y. et al. IL-17E synergizes with EGF and confers in vitro resistance to EGFR-targeted therapies in TNBC cells. Oncotarget 7, 53350–53361 (2016).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Kitamura, T., Qian, B. Z. & Pollard, J. W. Immune cell promotion of metastasis. Nat. Rev. Immunol. 15, 73–86 (2015).
MacCarthy-Morrogh, L. & Martin, P. The hallmarks of cancer are also the hallmarks of wound healing. Sci. Signal. https://doi.org/10.1126/scisignal.aay8690 (2020).
Voronov, E. et al. IL-1 is required for tumor invasiveness and angiogenesis. Proc. Natl Acad. Sci. USA 100, 2645–2650 (2003).
Moossavi, M., Parsamanesh, N., Bahrami, A., Atkin, S. L. & Sahebkar, A. Role of the NLRP3 inflammasome in cancer. Mol. Cancer 17, 158 (2018).
Lee, H. E. et al. Inhibition of NLRP3 inflammasome in tumor microenvironment leads to suppression of metastatic potential of cancer cells. Sci. Rep. 9, 12277 (2019).
Deswaerte, V. et al. Inflammasome adaptor ASC suppresses apoptosis of gastric cancer cells by an IL-18-mediated inflammation-independent mechanism. Cancer Res. 78, 1293–1307 (2018).
Park, S., Cheon, S. & Cho, D. The dual effects of interleukin-18 in tumor progression. Cell Mol. Immunol. 4, 329–335 (2007).
Terme, M. et al. IL-18 induces PD-1-dependent immunosuppression in cancer. Cancer Res. 71, 5393–5399 (2011).
Guo, X. et al. Blocking NF-kappaB is essential for the immunotherapeutic effect of recombinant IL-18 in pancreatic cancer. Clin. Cancer Res. 22, 5939–5950 (2016).
Kumari, N., Dwarakanath, B. S., Das, A. & Bhatt, A. N. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol. 37, 11553–11572 (2016).
De Simone, V. et al. Th17-type cytokines, IL-6 and TNF-alpha synergistically activate STAT3 and NF-kB to promote colorectal cancer cell growth. Oncogene 34, 3493–3503 (2015).
Lim, C. & Savan, R. The role of the IL-22/IL-22R1 axis in cancer. Cytokine Growth Factor. Rev. 25, 257–271 (2014).
Markota, A., Endres, S. & Kobold, S. Targeting interleukin-22 for cancer therapy. Hum. Vaccin. Immunother. 14, 2012–2015 (2018).
Brabletz, T., Kalluri, R., Nieto, M. A. & Weinberg, R. A. EMT in cancer. Nat. Rev. Cancer 18, 128–134 (2018).
Liu, X. et al. Epithelial-type systemic breast carcinoma cells with a restricted mesenchymal transition are a major source of metastasis. Sci. Adv. 5, eaav4275 (2019).
Dongre, A. & Weinberg, R. A. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 20, 69–84 (2019).
Li, R. et al. Chronic IL-1beta-induced inflammation regulates epithelial-to-mesenchymal transition memory phenotypes via epigenetic modifications in non-small cell lung cancer. Sci. Rep. 10, 377 (2020).
Castano, Z. et al. IL-1beta inflammatory response driven by primary breast cancer prevents metastasis-initiating cell colonization. Nat. Cell Biol. 20, 1084–1097 (2018).
Lee, C. C. et al. Macrophage-secreted interleukin-35 regulates cancer cell plasticity to facilitate metastatic colonization. Nat. Commun. 9, 3763 (2018).
Cao, H. et al. IL-13/STAT6 signaling plays a critical role in the epithelial-mesenchymal transition of colorectal cancer cells. Oncotarget 7, 61183–61198 (2016).
Al-Ismaeel, Q. et al. ZEB1 and IL-6/11-STAT3 signalling cooperate to define invasive potential of pancreatic cancer cells via differential regulation of the expression of S100 proteins. Br. J. Cancer 121, 65–75 (2019).
Chen, D. et al. Interleukin-23 promotes the epithelial-mesenchymal transition of oesophageal carcinoma cells via the Wnt/beta-catenin pathway. Sci. Rep. 5, 8604 (2015).
Gu, K. et al. Interleukin-17-induced EMT promotes lung cancer cell migration and invasion via NF-kappaB/ZEB1 signal pathway. Am. J. Cancer Res. 5, 1169–1179 (2015).
Long, X. et al. IL-8, a novel messenger to cross-link inflammation and tumor EMT via autocrine and paracrine pathways (Review). Int. J. Oncol. 48, 5–12 (2016).
Shankaran, V. et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410, 1107–1111 (2001).
Wang, Y. et al. The IL-15-AKT-XBP1s signaling pathway contributes to effector functions and survival in human NK cells. Nat. Immunol. 20, 10–17 (2019).
Wu, Y., Tian, Z. & Wei, H. Developmental and functional control of natural killer cells by cytokines. Front. Immunol. 8, 930 (2017).
Lazarevic, V., Glimcher, L. H. & Lord, G. M. T-bet: a bridge between innate and adaptive immunity. Nat. Rev. Immunol. 13, 777–789 (2013).
Zhou, X. et al. The deubiquitinase Otub1 controls the activation of CD8+ T cells and NK cells by regulating IL-15-mediated priming. Nat. Immunol. 20, 879–889 (2019).
Vesely, M. D., Kershaw, M. H., Schreiber, R. D. & Smyth, M. J. Natural innate and adaptive immunity to cancer. Annu. Rev. Immunol. 29, 235–271 (2011).
Fehniger, T. A. et al. Differential cytokine and chemokine gene expression by human NK cells following activation with IL-18 or IL-15 in combination with IL-12: implications for the innate immune response. J. Immunol. 162, 4511–4520 (1999).
Gerosa, F. et al. Reciprocal activating interaction between natural killer cells and dendritic cells. J. Exp. Med. 195, 327–333 (2002).
Zhou, T. et al. IL-18BP is a secreted immune checkpoint and barrier to IL-18 immunotherapy. Nature 583, 609–614 (2020). This publication suggests a decoy-resistant cytokine variant as a way to enable effective IL-18-based cancer immunotherapy.
Dixon, K. O. & Kuchroo, V. K. IL-18: throwing off the shackles to boost anti-tumor immunity. Cell Res. 30, 831–832 (2020).
Molgora, M. et al. IL-1R8 is a checkpoint in NK cells regulating anti-tumour and anti-viral activity. Nature 551, 110–114 (2017).
Hollande, C. et al. Inhibition of the dipeptidyl peptidase DPP4 (CD26) reveals IL-33-dependent eosinophil-mediated control of tumor growth. Nat. Immunol. 20, 257–264 (2019).
Grisaru-Tal, S., Itan, M., Klion, A. D. & Munitz, A. A new dawn for eosinophils in the tumour microenvironment. Nat. Rev. Cancer 20, 594–607 (2020).
Sheppard, P. et al. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat. Immunol. 4, 63–68 (2003).
Lazear, H. M., Schoggins, J. W. & Diamond, M. S. Shared and distinct functions of type i and type III interferons. Immunity 50, 907–923 (2019).
Zhu, J., Yamane, H. & Paul, W. E. Differentiation of effector CD4 T cell populations. Annu. Rev. Immunol. 28, 445–489 (2010).
Testa, U., Pelosi, E. & Castelli, G. CD123 as a therapeutic target in the treatment of hematological malignancies. Cancers https://doi.org/10.3390/cancers11091358 (2019).
Dolgin, E. First CD123-targeted drug approved after wowing in rare cancer. Nat. Biotechnol. 37, 202–203 (2019).
Tugues, S. et al. New insights into IL-12-mediated tumor suppression. Cell Death Differ. 22, 237–246 (2015).
Tait Wojno, E. D., Hunter, C. A. & Stumhofer, J. S. The immunobiology of the interleukin-12 family: room for discovery. Immunity 50, 851–870 (2019).
Garris, C. S. et al. Successful anti-PD-1 cancer immunotherapy requires T cell-dendritic cell crosstalk involving the cytokines IFN-gamma and IL-12. Immunity 49, 1148–1161 e1147 (2018).
Santana Carrero, R. M. et al. IL-15 is a component of the inflammatory milieu in the tumor microenvironment promoting antitumor responses. Proc. Natl Acad. Sci. USA 116, 599–608 (2019).
Mumm, J. B. et al. IL-10 elicits IFNgamma-dependent tumor immune surveillance. Cancer Cell 20, 781–796 (2011).
DuPage, M. & Bluestone, J. A. Harnessing the plasticity of CD4(+) T cells to treat immune-mediated disease. Nat. Rev. Immunol. 16, 149–163 (2016).
Lu, Y. et al. Th9 cells represent a unique subset of CD4+ T cells endowed with the ability to eradicate advanced tumors. Cancer Cell 33, 1048–1060 e1047 (2018). This publication highlights the potential of TH9 polarized cells for the improvement of immunotherapy for solid tumours.
Angkasekwinai, P. & Dong, C. IL-9-producing T cells: potential players in allergy and cancer. Nat. Rev. Immunol. https://doi.org/10.1038/s41577-020-0396-0 (2020).
Kim, I. K. et al. Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells. Nat. Med. 21, 1010–1017 (2015).
Vegran, F. et al. The transcription factor IRF1 dictates the IL-21-dependent anticancer functions of TH9 cells. Nat. Immunol. 15, 758–766 (2014).
Xue, G., Jin, G., Fang, J. & Lu, Y. IL-4 together with IL-1beta induces antitumor Th9 cell differentiation in the absence of TGF-beta signaling. Nat. Commun. 10, 1376 (2019).
Spolski, R. & Leonard, W. J. Interleukin-21: a double-edged sword with therapeutic potential. Nat. Rev. Drug Discov. 13, 379–395 (2014).
Zander, R. et al. CD4+ T cell help is required for the formation of a cytolytic CD8+ T cell subset that protects against chronic infection and cancer. Immunity 51, 1028–1042 e1024 (2019).
Kourko, O., Seaver, K., Odoardi, N., Basta, S. & Gee, K. IL-27, IL-30, and IL-35: a cytokine triumvirate in cancer. Front. Oncol. 9, 969 (2019).
Chihara, N. et al. Induction and transcriptional regulation of the co-inhibitory gene module in T cells. Nature 558, 454–459 (2018).
Li, S. et al. Cancer immunotherapy via targeted TGF-beta signalling blockade in Th cells. Nature https://doi.org/10.1038/s41586-020-2850-3 (2020).
Zitvogel, L., Tesniere, A. & Kroemer, G. Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat. Rev. Immunol. 6, 715–727 (2006).
Hatzioannou, A. et al. An intrinsic role of IL-33 in Treg cell-mediated tumor immunoevasion. Nat. Immunol. 21, 75–85 (2020).
Schiering, C. et al. The alarmin IL-33 promotes regulatory T-cell function in the intestine. Nature 513, 564–568 (2014).
Sawant, D. V. et al. Adaptive plasticity of IL-10+ and IL-35+ Treg cells cooperatively promotes tumor T cell exhaustion. Nat. Immunol. 20, 724–735 (2019). This work demonstrates a pathway by which Treg cell-derived IL-10 and IL-35 cooperatively exhaust CD8+ T cells in tumours.
Smith, L. K. et al. Interleukin-10 directly inhibits CD8+ T cell function by enhancing N-glycan branching to decrease antigen sensitivity. Immunity 48, 299–312 e295 (2018).
Sledzinska, A. et al. Regulatory T cells restrain interleukin-2- and Blimp-1-dependent acquisition of cytotoxic function by CD4+ T cells. Immunity 52, 151–166 e156 (2020).
Vitiello, G. A. & Miller, G. Targeting the interleukin-17 immune axis for cancer immunotherapy. J. Exp. Med. https://doi.org/10.1084/jem.20190456 (2020).
Coffelt, S. B. et al. IL-17-producing gammadelta T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
Koh, J. et al. IL23-producing human lung cancer cells promote tumor growth via conversion of innate lymphoid cell 1 (ILC1) into ILC3. Clin. Cancer Res. 25, 4026–4037 (2019).
Bakouny, Z. & Choueiri, T. K. IL-8 and cancer prognosis on immunotherapy. Nat. Med. 26, 650–651 (2020).
Yuen, K. C. et al. High systemic and tumor-associated IL-8 correlates with reduced clinical benefit of PD-L1 blockade. Nat. Med. 26, 693–698 (2020).
Nakamura, K. et al. Dysregulated IL-18 is a key driver of immunosuppression and a possible therapeutic target in the multiple myeloma microenvironment. Cancer Cell 33, 634–648 e635 (2018).
Shani, O. et al. Fibroblast-derived IL-33 facilitates breast cancer metastasis by modifying the immune microenvironment and driving type 2 immunity. Cancer Res. 80, 5317–5329 (2020).
Coussens, L. M., Zitvogel, L. & Palucka, A. K. Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science 339, 286–291 (2013).
Schuijs, M. J. et al. ILC2-driven innate immune checkpoint mechanism antagonizes NK cell antimetastatic function in the lung. Nat. Immunol. 21, 998–1009 (2020).
Gu, M. et al. NF-kappaB-inducing kinase maintains T cell metabolic fitness in antitumor immunity. Nat. Immunol. 22, 193–204 (2021).
Kesh, K. et al. Stroma secreted IL-6 selects for “stem-like” population and alters pancreatic tumor microenvironment by reprogramming metabolic pathways. Cell Death Dis. 11, 967 (2020).
Fousek, K., Horn, L. A. & Palena, C. Interleukin-8: A chemokine at the intersection of cancer plasticity, angiogenesis, and immune suppression. Pharmacol. Therap. https://doi.org/10.1016/j.pharmthera.2020.107692 (2020).
Ngiow, S. F., Teng, M. W. L. & Smyth, M. J. A balance of interleukin-12 and -23 in cancer. Trends Immunol. 34, 548–555 (2013).
Feng, L. et al. Serum levels of IL-6, IL-8, and IL-10 are indicators of prognosis in pancreatic cancer. J. Int. Med. Res. 46, 5228–5236 (2018).
Lippitz, B. E. Cytokine patterns in patients with cancer: a systematic review. Lancet Oncol. 14, e218–e228 (2013).
Ridker, P. M. et al. Effect of interleukin-1beta; inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet 390, 1833–1842 (2017).
Libby, P. Interleukin-1 beta as a target for atherosclerosis therapy: biological basis of CANTOS and beyond. J. Am. Coll. Cardiol. 70, 2278–2289 (2017).
Wong, C. C. et al. Inhibition of IL-1β by canakinumab may be effective against diverse molecular subtypes of lung cancer: an exploratory analysis of the CANTOS trial. Cancer Res. https://doi.org/10.1158/0008-5472.Can-19-3176 (2020). This work impressively demonstrates how patients with cancer could benefit from IL-1β neutralization.
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03447769 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04239157 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03631199 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03626545 (2018).
Lust, J. A. et al. Induction of a chronic disease state in patients with smoldering or indolent multiple myeloma by targeting interleukin 1beta-induced interleukin 6 production and the myeloma proliferative component. Mayo Clin. Proc. 84, 114–122 (2009).
Lust, J. A. et al. Reduction in C-reactive protein indicates successful targeting of the IL-1/IL-6 axis resulting in improved survival in early stage multiple myeloma. Am. J. Hematol. 91, 571–574 (2016).
Nguyen, M. L. T. et al. Targeting interleukin 6 signaling by monoclonal antibody siltuximab on cholangiocarcinoma. J. Gastroenterol. Hepatol. https://doi.org/10.1111/jgh.15307 (2020).
Brighton, T. A. et al. Randomized, double-blind, placebo-controlled, multicenter study of siltuximab in high-risk smoldering multiple myeloma. Clin. Cancer Res. 25, 3772–3775 (2019).
Orlowski, R. Z. et al. A phase 2, randomized, double-blind, placebo-controlled study of siltuximab (anti-IL-6 mAb) and bortezomib versus bortezomib alone in patients with relapsed or refractory multiple myeloma. Am. J. Hematol. 90, 42–49 (2015).
San-Miguel, J. et al. Phase 2 randomized study of bortezomib-melphalan-prednisone with or without siltuximab (anti-IL-6) in multiple myeloma. Blood 123, 4136–4142 (2014).
Dorff, T. B. et al. Clinical and correlative results of SWOG S0354: a phase II trial of CNTO328 (siltuximab), a monoclonal antibody against interleukin-6, in chemotherapy-pretreated patients with castration-resistant prostate cancer. Clin. Cancer Res. 16, 3028–3034 (2010).
Rossi, J. F. et al. A phase I/II study of siltuximab (CNTO 328), an anti-interleukin-6 monoclonal antibody, in metastatic renal cell cancer. Br. J. Cancer 103, 1154–1162 (2010).
Angevin, E. et al. A phase I/II, multiple-dose, dose-escalation study of siltuximab, an anti-interleukin-6 monoclonal antibody, in patients with advanced solid tumors. Clin. Cancer Res. 20, 2192–2204 (2014).
Dijkgraaf, E. M. et al. A phase I trial combining carboplatin/doxorubicin with tocilizumab, an anti-IL-6R monoclonal antibody, and interferon-alpha2b in patients with recurrent epithelial ovarian cancer. Ann. Oncol. 26, 2141–2149 (2015).
Johnson, D. E., O’Keefe, R. A. & Grandis, J. R. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat. Rev. Clin. Oncol. 15, 234–248 (2018).
Bergmann, J. et al. IL-6 trans-signaling is essential for the development of hepatocellular carcinoma in mice. Hepatology 65, 89–103 (2017).
Schmidt, S. et al. ADAM17 is required for EGF-R-induced intestinal tumors via IL-6 trans-signaling. J. Exp. Med. 215, 1205–1225 (2018).
Jostock, T. et al. Soluble gp130 is the natural inhibitor of soluble interleukin-6 receptor transsignaling responses. Eur. J. Biochem. 268, 160–167 (2001).
Bilusic, M. et al. Phase I trial of HuMax-IL8 (BMS-986253), an anti-IL-8 monoclonal antibody, in patients with metastatic or unresectable solid tumors. J. Immunother. Cancer 7, 240–240 (2019).
Schalper, K. A. et al. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nat. Med. 26, 688–692 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03400332 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04123379 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04050462 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03689699 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04572451 (2020).
Nagaoka, K. et al. Deep immunophenotyping at the single-cell level identifies a combination of anti-IL-17 and checkpoint blockade as an effective treatment in a preclinical model of data-guided personalized immunotherapy. J. Immunother. Cancer 8, e001358 (2020).
Zhang, Q. et al. Targeting Th17-IL-17 pathway in prevention of micro-invasive prostate cancer in a mouse model. Prostate 77, 888–899 (2017).
Calcinotto, A. et al. Microbiota-driven interleukin-17-producing cells and eosinophils synergize to accelerate multiple myeloma progression. Nat. Commun. 9, 4832–4832 (2018).
Prabhala, R. H. et al. Targeting IL-17A in multiple myeloma: a potential novel therapeutic approach in myeloma. Leukemia 30, 379–389 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03111992 (2017).
Calcinotto, A. et al. IL-23 secreted by myeloid cells drives castration-resistant prostate cancer. Nature 559, 363–369 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04458311 (2020).
Kudo-Saito, C. et al. IL-33 is a key driver of treatment resistance of cancer. Cancer Res. 80, 1981–1990 (2020).
Rosenberg, S. A. IL-2: the first effective immunotherapy for human cancer. J. Immunol. 192, 5451–5458 (2014). This work provides an overview of the treatment of patients with cancer with recombinant IL-2, the first interleukin approved for cancer therapy.
Charych, D. H. et al. NKTR-214, an engineered cytokine with biased IL-2 receptor binding, increased tumor exposure, and marked efficacy in mouse tumor models. Clin. Cancer Res. 22, 680–690 (2016).
Bentebibel, S.-E. et al. A first-in-human study and biomarker analysis of NKTR-214, a novel IL2Rβγ-biased cytokine, in patients with advanced or metastatic solid tumors. Cancer Discov. 9, 711–721 (2019).
Diab, A. et al. Bempegaldesleukin (NKTR-214) plus nivolumab in patients with advanced solid tumors: phase I dose-escalation study of safety, efficacy, and immune activation (PIVOT-02). Cancer Discov. 10, 1158–1173 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04209114 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03729245 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03635983 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04009681 (2019).
Sahin, D. et al. An IL-2-grafted antibody immunotherapy with potent efficacy against metastatic cancer. Nat. Commun. 11, 6440–6440 (2020).
Silva, D.-A. et al. De novo design of potent and selective mimics of IL-2 and IL-15. Nature 565, 186–191 (2019).
Mortara, L. et al. Anti-cancer therapies employing IL-2 cytokine tumor targeting: contribution of innate, adaptive and immunosuppressive cells in the anti-tumor efficacy. Front. Immunol. https://doi.org/10.3389/fimmu.2018.02905 (2018).
Naing, A. et al. Safety, antitumor activity, and immune activation of pegylated recombinant human interleukin-10 (AM0010) in patients with advanced solid tumors. J. Clin. Oncol. 34, 3562–3569 (2016).
Naing, A. et al. PEGylated IL-10 (pegilodecakin) induces systemic immune activation, CD8(+) T cell invigoration and polyclonal T cell expansion in cancer patients. Cancer Cell 34, 775–791.e773 (2018).
Naing, A. et al. Pegilodecakin combined with pembrolizumab or nivolumab for patients with advanced solid tumours (IVY): a multicentre, multicohort, open-label, phase 1b trial. Lancet Oncol. 20, 1544–1555 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03382912 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03382899 (2017).
Qiao, J. et al. Targeting tumors with IL-10 prevents dendritic cell-mediated CD8(+) T Cell Apoptosis. Cancer Cell 35, 901–915.e904 (2019).
Nguyen, K. G. et al. Localized interleukin-12 for cancer immunotherapy. Front. Immunol. https://doi.org/10.3389/fimmu.2020.575597 (2020).
Algazi, A. et al. Intratumoral delivery of tavokinogene telseplasmid yields systemic immune responses in metastatic melanoma patients. Ann. Oncol. 31, 532–540 (2020).
Algazi, A. P. et al. Phase II trial of IL-12 plasmid transfection and PD-1 blockade in immunologically quiescent melanoma. Clin. Cancer Res. 26, 2827–2837 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03567720 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03823131 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04526730 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03132675 (2017).
Mansurov, A. et al. Collagen-binding IL-12 enhances tumour inflammation and drives the complete remission of established immunologically cold mouse tumours. Nat. Biomed. Eng. 4, 531–543 (2020).
Li, Y. et al. Multifunctional oncolytic nanoparticles deliver self-replicating IL-12 RNA to eliminate established tumors and prime systemic immunity. Nat. Cancer 1, 882–893 (2020).
Waldmann, T. A., Dubois, S., Miljkovic, M. D. & Conlon, K. C. IL-15 in the combination immunotherapy of cancer. Front. Immunol. 11, 868–868 (2020).
Margolin, K. et al. Phase I Trial of ALT-803, A novel recombinant IL-15 complex, in patients with advanced solid tumors. Clin. Cancer Res. 24, 5552–5561 (2018).
Romee, R. et al. First-in-human phase 1 clinical study of the IL-15 superagonist complex ALT-803 to treat relapse after transplantation. Blood 131, 2515–2527 (2018).
Rosser, C. J., Nix, J., Ferguson, L., Hernandez, L. & Wong, H. C. Phase Ib trial of ALT-803, an IL-15 superagonist, plus BCG for the treatment of BCG-naïve patients with non-muscle-invasive bladder cancer. J. Clin. Oncol. 36, 510–510 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02138734 (2014).
Waldmann, T. A., Miljkovic, M. D. & Conlon, K. C. Interleukin-15 (dys)regulation of lymphoid homeostasis: Implications for therapy of autoimmunity and cancer. J. Exp. Med. 217, e20191062 (2020).
Hewitt, S. L. et al. Durable anticancer immunity from intratumoral administration of IL-23, IL-36gamma, and OX40L mRNAs. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aat9143 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03739931 (2018).
Nguyen, L. T. et al. Phase II clinical trial of adoptive cell therapy for patients with metastatic melanoma with autologous tumor-infiltrating lymphocytes and low-dose interleukin-2. Cancer Immunol. Immunother. 68, 773–785 (2019).
Junghans, R. P. et al. Phase I trial of anti-PSMA designer CAR-T cells in prostate cancer: possible role for interacting interleukin-2-T cell pharmacodynamics as a determinant of clinical response. Prostate 76, 1257–1270 (2016).
Sockolosky, J. T. et al. Selective targeting of engineered T cells using orthogonal IL-2 cytokine-receptor complexes. Science 359, 1037–1042 (2018).
Jin, L. et al. CXCR1- or CXCR2-modified CAR T cells co-opt IL-8 for maximal antitumor efficacy in solid tumors. Nat Commun 10, 4016 (2019).
Chmielewski, M., Kopecky, C., Hombach, A. A. & Abken, H. IL-12 release by engineered T cells expressing chimeric antigen receptors can effectively muster an antigen-independent macrophage response on tumor cells that have shut down tumor antigen expression. Cancer Res. 71, 5697–5706 (2011). This study provides a proof of concept for the expression of interleukins by CAR T cells to recruit innate immune cells and mount an antigen-independent immune response.
Zhang, L. et al. Tumor-infiltrating lymphocytes genetically engineered with an inducible gene encoding interleukin-12 for the immunotherapy of metastatic melanoma. Clin. Cancer Res. 21, 2278–2288 (2015). This is the first report of interleukin-expressing tumour-infiltrating lymphocytes.
Etxeberria, I. et al. Intratumor adoptive transfer of IL-12 mRNA transiently engineered antitumor CD8+ T cells. Cancer Cell 36, 613–629.e617 (2019).
Subbiah, V. et al. Cytokines produced by dendritic cells administered intratumorally correlate with clinical outcome in patients with diverse cancers. Clin. Cancer Res. 24, 3845–3856 (2018).
Adachi, K. et al. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat. Biotechnol. 36, 346–351 (2018).
Xu, X. et al. NKT cells coexpressing a GD2-specific chimeric antigen receptor and IL-15 show enhanced in vivo persistence and antitumor activity against neuroblastoma. Clin. Cancer Res. 25, 7126–7138 (2019).
Hurton, L. V. et al. Tethered IL-15 augments antitumor activity and promotes a stem-cell memory subset in tumor-specific T cells. Proc. Natl. Acad. Sci. USA 113, E7788–E7797 (2016).
Heczey, A. et al. Anti-GD2 CAR-NKT cells in patients with relapsed or refractory neuroblastoma: an interim analysis. Nat. Med. 26, 1686–1690 (2020).
Tang, L. et al. Enhancing T cell therapy through TCR-signaling-responsive nanoparticle drug delivery. Nat. Biotechnol. 36, 707–716 (2018).
Ma, X. et al. Interleukin-23 engineering improves CAR T cell function in solid tumors. Nat. biotechnol. 38, 448–459 (2020).
Espinosa-Cotton, M. et al. Interleukin-1 alpha increases anti-tumor efficacy of cetuximab in head and neck squamous cell carcinoma. J. Immunother. Cancer 7, 79 (2019).
Gottschlich, A., Endres, S. & Kobold, S. Therapeutic strategies for targeting IL-1 in cancer. Cancers 13, 477 (2021).
Li, S. et al. Role for nuclear interleukin-37 in the suppression of innate immunity. Proc. Natl Acad. Sci. USA 116, 4456–4461 (2019).
Weinstein, A. M. et al. Association of IL-36gamma with tertiary lymphoid structures and inflammatory immune infiltrates in human colorectal cancer. Cancer Immunol. Immunother. 68, 109–120 (2019).
van de Veerdonk, F. L., de Graaf, D. M., Joosten, L. A. & Dinarello, C. A. Biology of IL-38 and its role in disease. Immunol. Rev. 281, 191–196 (2018).
Kinoshita, F. et al. Interleukin-38 promotes tumor growth through regulation of CD8+tumor-infiltrating lymphocytes in lung cancer tumor microenvironment. Cancer Immunol. Immunother. 70, 123–135 (2021).
Damoiseaux, J. The IL-2 - IL-2 receptor pathway in health and disease: The role of the soluble IL-2 receptor. Clin. Immunol. 218, 108515 (2020).
Suzuki, A., Leland, P., Joshi, B. H. & Puri, R. K. Targeting of IL-4 and IL-13 receptors for cancer therapy. Cytokine 75, 79–88 (2015).
Lin, J. et al. The role of IL-7 in immunity and cancer. Anticancer Res. 37, 963–967 (2017).
Ferretti, E., Corcione, A. & Pistoia, V. The IL-31/IL-31 receptor axis: general features and role in tumor microenvironment. J. Leukoc. Biol. 102, 711–717 (2017).
Ouyang, W. & O’Garra, A. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity 50, 871–891 (2019).
Chen, Y. Y., Li, C. F., Yeh, C. H., Chang, M. S. & Hsing, C. H. Interleukin-19 in breast cancer. Clin. Dev. Immunol. 2013, 294320 (2013).
Hsu, Y. H. et al. Anti-IL-20 monoclonal antibody suppresses breast cancer progression and bone osteolysis in murine models. J. Immunol. 188, 1981–1991 (2012).
Menezes, M. E. et al. MDA-7/IL-24: multifunctional cancer killing cytokine. Adv. Exp. Med. Biol. 818, 127–153 (2014).
Hu, H. J. et al. The armed oncolytic adenovirus ZD55-IL-24 eradicates melanoma by turning the tumor cells from the self-state into the nonself-state besides direct killing. Cell Death Dis. 11, 1022 (2020).
Trotter, T. N. et al. IL-26, a noncanonical mediator of DNA inflammatory stimulation, promotes TNBC engraftment and progression in association with neutrophils. Cancer Res 80, 3088–3100 (2020).
Larochette, V. et al. IL-26, a cytokine with roles in extracellular DNA-induced inflammation and microbial defense. Front. Immunol. 10, 204 (2019).
Yan, J., Smyth, M. J. & Teng, M. W. L. Interleukin (IL)-12 and IL-23 and their conflicting roles in cancer. Cold Spring Harb. Perspect Biol. https://doi.org/10.1101/cshperspect.a028530 (2018).
Park, Y. J. et al. IL-27 confers a protumorigenic activity of regulatory T cells via CD39. Proc. Natl Acad. Sci. USA 116, 3106–3111 (2019).
Figueiredo, M. L. et al. Ligand-mediated targeting of cytokine interleukin-27 enhances its bioactivity in vivo. Mol. Ther. Methods Clin. Dev. 17, 739–751 (2020).
Bie, Q., Jin, C., Zhang, B. & Dong, H. IL-17B: A new area of study in the IL-17 family. Mol. Immunol. 90, 50–56 (2017).
Jungnickel, C. et al. IL-17C mediates the recruitment of tumor-associated neutrophils and lung tumor growth. Oncogene 36, 4182–4190 (2017).
Liu, X., Sun, S. & Liu, D. IL-17D: a less studied cytokine of IL-17 family. Int. Arch. Allergy Immunol. 181, 618–623 (2020).
Hasegawa, K. et al. Anti-tumor immunity elicited by direct intratumoral administration of a recombinant adenovirus expressing either IL-28A/IFN-lambda2 or IL-29/IFN-lambda1. Cancer Gene Ther. 23, 266–277 (2016).
Ford, R. et al. Identification of B-cell growth factors (interleukin-14; high molecular weight-B-cell growth factors) in effusion fluids from patients with aggressive B-cell lymphomas. Blood 86, 283–293 (1995).
Richmond, J., Tuzova, M., Cruikshank, W. & Center, D. Regulation of cellular processes by interleukin-16 in homeostasis and cancer. J. Cell Physiol. 229, 139–147 (2014).
Sloot, Y. J. E., Smit, J. W., Joosten, L. A. B. & Netea-Maier, R. T. Insights into the role of IL-32 in cancer. Semin. Immunol. 38, 24–32 (2018).
Baghdadi, M. et al. Interleukin-34, a comprehensive review. J. Leukoc. Biol. 104, 931–951 (2018).
Franzè, E., Stolfi, C., Troncone, E., Scarozza, P. & Monteleone, G. Role of interleukin-34 in cancer. Cancers 12, 252 (2020).
Grupp, S. A. et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N. Engl. J. Med. 368, 1509–1518 (2013).
Davila, M. L. et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci. Transl. Med. 6, 224ra225 (2014).
Norelli, M. et al. Monocyte-derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nat. Med. 24, 739–748 (2018).
Giavridis, T. et al. CAR T cell-induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade. Nat. Med. 24, 731–738 (2018).
Ando, K. et al. Tocilizumab, a proposed therapy for the cachexia of Interleukin-6-expressing lung cancer. PloS ONE 9, e102436 (2014).
Rigas, J. R. et al. Efect of ALD518, a humanized anti-IL-6 antibody, on lean body mass loss and symptoms in patients with advanced non-small cell lung cancer (NSCLC): results of a phase II randomized, double-blind safety and efficacy trial. J. Clin. Oncol. 28, 7622–7622 (2010).
Flint, T. R. et al. Tumor-induced IL-6 reprograms host metabolism to suppress anti-tumor immunity. Cell Metab. 24, 672–684 (2016).
Pettersen, K. et al. Cancer cachexia associates with a systemic autophagy-inducing activity mimicked by cancer cell-derived IL-6 trans-signaling. Sci. Rep. 7, 2046 (2017).
McDonald, J. J., McMillan, D. C. & Laird, B. J. A. Targeting IL-1alpha in cancer cachexia: a narrative review. Curr. Opin. Support Palliat. Care 12, 453–459 (2018).
Hickish, T. et al. MABp1 as a novel antibody treatment for advanced colorectal cancer: a randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 18, 192–201 (2017).
Hong, D. S. et al. Xilonix, a novel true human antibody targeting the inflammatory cytokine interleukin-1 alpha, in non-small cell lung cancer. Invest. New Drugs 33, 621–631 (2015).
Callaway, C. S. et al. IL-8 released from human pancreatic cancer and tumor-associated stromal cells signals through a CXCR2-ERK1/2 axis to induce muscle atrophy. Cancers 11, 1863 (2019).
Costamagna, D. et al. Interleukin-4 administration improves muscle function, adult myogenesis, and lifespan of colon carcinoma-bearing mice. J. Cachexia Sarcopenia Muscle 11, 783–801 (2020).
Acknowledgements
This study was supported by the Marie Skłodowska-Curie Programme Training Network for the Immunotherapy of Cancer and Training Network for Optimizing Adoptive T Cell Therapy of Cancer funded by the Horizon 2020 programme of the European Union (grant 641549 to S.E. and S.K. and grant 955575 to S.K), the Hector Foundation, the international doctoral programme i Target: Immunotargeting of Cancer funded by the Elite Network of Bavaria (S.K. and S.E.), Melanoma Research Alliance grant 409510 (to S.K.), the Else Kröner-Fresenius-Stiftung (S.K.), the German Cancer Aid (S.K.), the Ernst-Jung-Stiftung (S.K.), LMU Munich’s institutional strategy LMUexcellent within the framework of the German Excellence Initiative (S.E. and S.K.), the Bundesministerium für Bildung und Forschung project Oncoattract (S.E. and S.K.), European Research Council grant 756017, ARMOR-T (to S.K.), the German Research Foundation (S.K.), the Fritz Bender Foundation (S.K.), the Bavarian Ministry of Economical Affairs (m4 award to S.K. and S.E.) and the José Carreras Foundation (to S.K.). The authors apologize to any colleagues whose important work could not be cited or commented on due to space restrictions.
Author information
Authors and Affiliations
Contributions
S.K., D.B. and J.D. contributed equally to all aspects of the article. S.E., P.L. and C.A.D. contributed to researching literature for the article and discussion of the content, and reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.D. has received remuneration from Novartis for work unrelated to this Review. P.L. is an unpaid consultant to, or involved in clinical trials for, Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, Medimmune, Merck, Norvo Nordisk, Novartis, Pfizer and Sanofi-Regeneron. P.L. is a member of scientific advisory board for Amgen, Caristo, Cartesian, Corvidia Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint, Kowa Pharmaceuticals, Olatec Therapeutics, Medimmune, Novartis, PlaqueTec and XBiotech, Inc. P.L.’s laboratory has received research funding in the past 2 years from Novartis. P.L. is on the Board of Directors of XBiotech, and has a financial interest in Xbiotech, a company developing therapeutic human antibodies. P.L.’s interests were reviewed and are managed by Brigham and Women’s Hospital and Partners HealthCare in accordance with their conflict-of-interest policies. S.K. and S.E. are inventors named on several patents in the field of immuno-oncology unrelated to the present work. S.K. and S.E. received research support from TCR2 Inc. and Arcus Bioscience for work unrelated to this Review. S.K. and S.E. have licensed intellectual property to TCR2 Inc. S.K. has received honoraria from GlaxoSmithKline and Novartis. D.B. and C.A.D. declare no competing interests.
Additional information
Peer review information
Nature Reviews Cancer thanks L. Apetoh, who co-reviewed with F. Vegran; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Danger-associated molecular patterns
-
(DAMPs). Danger-associated molecular patterns and alarmins are endogenous, constitutively expressed, chemotactic and immune-activating proteins and peptides that are released upon degranulation, cell injury or death or in response to immune induction.
- Pathogen-associated molecular patterns
-
(PAMPs). Exogenous conserved microbial structures recognized by the innate immune receptors.
- Tumour-associated macrophages
-
Macrophage subtype that is highly abundant in tumours and associated with immunosuppression, tumour progression and metastasis.
- γδ T cells
-
A subset of T cells differing from conventional T cells by expressing a distinct γδ T cell receptor in contrast to the usual αβ T cell receptor. They do not depend on histocompatibility complex presentation and possess properties of both innate and adaptive immunity
- Group 3 innate lymphoid cells
-
(ILC3s). Cells with characteristics of lymphoid cells but without rearranged antigen-specific surface receptors that are crucial for maintaining homeostasis in the gut microbiota.
- Regulatory T cell
-
(Treg cell). A subtype of T cells maintaining self-tolerance and preventing excessive immune responses, mainly by the inhibition of proliferation and activation of cytotoxic T cells.
- Adoptive cell transfer
-
(ACT). Treatment of patients with ex vivo expanded or modified immune cells usually derived from the patient.
- Natural killer (NK) cells
-
Cytotoxic lymphocytes that can eliminate virus-infected cells or tumour cells by the recognition of non-self antigens presented on major histocompatibility complex class I molecules of the target cell.
- Natural killer T cells
-
(NKT cells). A subset of T cells exhibiting antigen-specific immunity via a restricted T cell receptor recognizing CD1-presented lipid antigens, but also sharing natural killer cell functions in terms of their independence from major histocompatibility complex.
- Antigen-presenting cells
-
(APCs). Dendritic cells, macrophages and B cells that present non-self peptide antigens to T cells via major histocompatibility complex class II molecules.
- M1 macrophage
-
A macrophage subtype associated with inflammation that is characterized by the secretion of the proinflammatory cytokines IL-1, IL-6 and TNF.
- Immunotherapy
-
Treatment of patients with factors shaping an immune response to enable the patient’s immune system to react to the disease.
- TH1-type responses
-
Proinflammatory responses inducing killing of intracellular pathogens and tumour cells mediated mainly by interferon-γ.
- M2 macrophages
-
An inhibitory macrophage subtype associated with tissue remodelling and repair that secrete anti-inflammatory cytokines such as IL-10 and transforming growth factor-β (TGFβ).
- Myeloid-derived suppressor cells
-
(MDSCs). A heterogeneous group of myeloid cells often recruited by tumour cells that lead to pronounced immunosuppression by inhibiting a wide range of inflammatory immune cells.
- Group 2 innate lymphoid cells
-
(ILC2). Cells with characteristics of lymphoid cells but without rearranged antigen-specific surface receptors. Similarly to CD4+ T cells, they produce type 2 cytokines and are also referred to as natural or innate helper cells.
- TH2-type responses
-
Immune responses against mainly extracellular antigens mediated by IL-4, IL-5 and IL-13.
- Immune checkpoint inhibitors
-
(ICIs). Drugs used for therapeutic neutralization of signalling pathways that activate immune checkpoints, which normally lead to the inhibition of immune cell function to keep immune responses under control.
- Tumour-infiltrating lymphocytes
-
Lymphocytes that migrated into the tumour and can be isolated, expanded and re-infused into the patient as therapy.
Rights and permissions
About this article
Cite this article
Briukhovetska, D., Dörr, J., Endres, S. et al. Interleukins in cancer: from biology to therapy. Nat Rev Cancer 21, 481–499 (2021). https://doi.org/10.1038/s41568-021-00363-z
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41568-021-00363-z
This article is cited by
-
Inflame and restrain — the paradoxical roles of IL-12 and IL-23 in immunity
Nature Reviews Immunology (2026)
-
Contextualizing TH17 cells in cancer
Nature Reviews Immunology (2026)
-
Epithelial-mesenchymal transition orchestrates tumor microenvironment: current perceptions and challenges
Journal of Translational Medicine (2025)
-
Intratumoral microbiome: a crucial regulating factor in development and progression of colorectal cancer
Molecular Biomedicine (2025)
-
Extracellular matrix: unlocking new avenues in cancer treatment
Biomarker Research (2025)