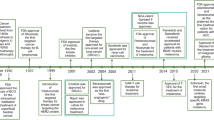
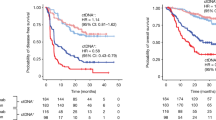
Abstract
The time required to conduct clinical trials limits the rate at which we can evaluate and deliver new treatment options to patients with cancer. New approaches to increase trial efficiency while maintaining rigor would benefit patients, especially in oncology, in which adjuvant trials hold promise for intercepting metastatic disease, but typically require large numbers of patients and many years to complete. We envision a standing platform — an infrastructure to support ongoing identification and trial enrolment of patients with cancer with early molecular evidence of disease (MED) after curative-intent therapy for early-stage cancer, based on the presence of circulating tumour DNA. MED strongly predicts subsequent recurrence, with the vast majority of patients showing radiographic evidence of disease within 18 months. Such a platform would allow efficient testing of many treatments, from small exploratory studies to larger pivotal trials. Trials enrolling patients with MED but without radiographic evidence of disease have the potential to advance drug evaluation because they can be smaller (given high probability of recurrence) and faster (given short time to recurrence) than conventional adjuvant trials. Circulating tumour DNA may also provide a valuable early biomarker of treatment effect, which would allow small signal-finding trials. In this Perspective, we discuss how such a platform could be established.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
IQVIA Institute for Human Data Science. Global oncology trends 2023: outlook to 2027. IQVIA Institute Reports https://www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/ global-oncology-trends-2023 (2023).
Moore, T. J., Zhang, H., Anderson, G. & Alexander, G. C. Estimated costs of pivotal trials for novel therapeutic agents approved by the US Food and Drug Administration, 2015–2016. JAMA Intern. Med. 178, 1451–1457 (2018).
Jenei, K., Haslam, A., Olivier, T., Miljkovic, M. & Prasad, V. What drives cancer clinical trial accrual? An empirical analysis of studies leading to FDA authorisation (2015–2020). BMJ Open 12, e064458 (2022).
McKinsey & Company. Accelerating clinical trials to improve biopharma R&D productivity. McKinsey & Company: Life Sciences. https://www.mckinsey.com/industries/life-sciences/our-insights/accelerating-clinical-trials-to-improve-biopharma-r-and-d-productivity (2024).
Getz, K., Smith, Z. & Kravet, M. Protocol design and performance benchmarks by phase and by oncology and rare disease subgroups. Ther. Innov. Regul. Sci. 57, 49–56 (2023).
U.S. Food & Drug Administration Center for Drug Evaluation and Research. Drug trials snapshots summary report 2022. FDA.gov. https://www.fda.gov/drugs/drug-approvals-and-databases/drug-trials-snapshots (2023).
Weber, J. S. et al. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): a randomised, phase 2b study. Lancet 403, 632–644 (2024).
Miller, K. D. et al. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 72, 409–436 (2022).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Arriagada, R. et al. Long-term results of the international adjuvant lung cancer trial evaluating adjuvant cisplatin-based chemotherapy in resected lung cancer. J. Clin. Oncol. 28, 35–42 (2010).
Spigel, D. R. et al. Five-year survival outcomes from the PACIFIC trial: durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. J. Clin. Oncol. 40, 1301–1311 (2022).
Taylor, M. D. et al. Tumor recurrence after complete resection for non-small cell lung cancer. Ann. Thorac. Surg. 93, 1813–1820 (2012).
Goldstraw, P. et al. The IASLC lung cancer staging project: proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J. Thorac. Oncol. 11, 39–51 (2016).
Andre, T. et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J. Clin. Oncol. 27, 3109–3116 (2009).
Grothey, A. et al. Duration of adjuvant chemotherapy for stage III colon cancer. N. Engl. J. Med. 378, 1177–1188 (2018).
Pan, H. et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N. Engl. J. Med. 377, 1836–1846 (2017).
Johnston, S. R. D. et al. Abemaciclib combined with endocrine therapy for the adjuvant treatment of HR+, HER2−, node-positive, high-risk, early breast cancer (monarchE). J. Clin. Oncol. 38, 3987–3998 (2020).
Johnston, S. R. D. et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 24, 77–90 (2023).
Albain, K. S. et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 11, 55–65 (2010).
Masuda, N. et al. Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N. Engl. J. Med. 376, 2147–2159 (2017).
Schmid, P. et al. Pembrolizumab for early triple-negative breast cancer. N. Engl. J. Med. 382, 810–821 (2020).
Shepherd, J. H. et al. CALGB 40603 (Alliance): long-term outcomes and genomic correlates of response and survival after neoadjuvant chemotherapy with or without carboplatin and bevacizumab in triple-negative breast cancer. J. Clin. Oncol. 40, 1323–1334 (2022).
von Minckwitz, G. et al. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N. Engl. J. Med. 380, 617–628 (2019).
Dieci, M. V. et al. Validation of the AJCC prognostic stage for HER2-positive breast cancer in the ShortHER trial. BMC Med. 17, 207 (2019).
Piccart, M. et al. Adjuvant pertuzumab and trastuzumab in early HER2-positive breast cancer in the APHINITY trial: 6 years’ follow-up. J. Clin. Oncol. 39, 1448–1457 (2021).
Garcia-Murillas, I. et al. Assessment of molecular relapse detection in early-stage breast cancer. JAMA Oncol. 5, 1473–1478 (2019).
Coombes, R. C. et al. Personalized detection of circulating tumor DNA antedates breast cancer metastatic recurrence. Clin. Cancer Res. 25, 4255–4263 (2019). This study demonstrates that ctDNA surveillance in patients with breast cancer can detect molecular evidence of disease up to two years before radiographic or clinical evidence of disease, providing a possible window for therapeutic intervention; evidence from this study was the primary basis on which Medicare decided to provide insurance coverage for adjuvant and recurrence monitoring in patients with stage IIb or higher breast cancer.
Garcia-Murillas, I. et al. Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Sci. Transl. Med. 7, 302ra133 (2015).
Olsson, E. et al. Serial monitoring of circulating tumor DNA in patients with primary breast cancer for detection of occult metastatic disease. EMBO Mol. Med. 7, 1034–1047 (2015).
Parikh, A. R. et al. Minimal residual disease detection using a plasma-only circulating tumor DNA assay in patients with colorectal cancer. Clin. Cancer Res. 27, 5586–5594 (2021).
Reinert, T. et al. Analysis of plasma cell-free DNA by ultradeep sequencing in patients with stages I to III colorectal cancer. JAMA Oncol. 5, 1124–1131 (2019). This study demonstrates that circulating tumour DNA surveillance in patients with colorectal cancer can potentially change postoperative management by enabling risk stratification, disease monitoring and identifying molecular evidence of disease up to 16 months earlier than radiographic evidence of disease; evidence from this study, among others, was the basis on which Medicare decided to provide insurance coverage for adjuvant and recurrence monitoring in patients with stage II–III colorectal cancer.
Schøler, L. V. et al. Clinical implications of monitoring circulating tumor DNA in patients with colorectal cancer. Clin. Cancer Res. 23, 5437–5445 (2017).
Henriksen, T. V. et al. Circulating tumor DNA in stage III colorectal cancer, beyond minimal residual disease detection, toward assessment of adjuvant therapy efficacy and clinical behavior of recurrences. Clin. Cancer Res. 28, 507–517 (2022).
Wang, Y. et al. Prognostic potential of circulating tumor DNA measurement in postoperative surveillance of nonmetastatic colorectal cancer. JAMA Oncol. 5, 1118–1123 (2019).
Tie, J. et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci. Transl. Med. 8, 346ra392 (2016).
Peng, M. et al. Circulating tumor DNA as a prognostic biomarker in localized non-small cell lung cancer. Front. Oncol. 10, 561598 (2020).
Abbosh, C. et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 545, 446–451 (2017). This is one of the first studies that demonstrates the potential of a personalized, tumour-informed circulating tumour DNA test to detect residual disease in early-stage lung cancer and identify molecular evidence of disease up to 11 months earlier than radiographic evidence of disease.
Chaudhuri, A. A. et al. Early detection of molecular residual disease in localized lung cancer by circulating tumor DNA profiling. Cancer Discov. 7, 1394–1403 (2017).
Qiu, B. et al. Dynamic recurrence risk and adjuvant chemotherapy benefit prediction by ctDNA in resected NSCLC. Nat. Commun. 12, 6770 (2021).
Zhang, J. T. et al. Longitudinal undetectable molecular residual disease defines potentially cured population in localized non-small cell lung cancer. Cancer Discov. 12, 1690–1701 (2022).
Lipsyc-Sharf, M. et al. Circulating tumor DNA and late recurrence in high-risk hormone receptor-positive, human epidermal growth factor receptor 2-negative breast cancer. J. Clin. Oncol. 40, 2408–2419 (2022).
Tie, J. et al. Circulating tumor DNA analyses as markers of recurrence risk and benefit of adjuvant therapy for stage III colon cancer. JAMA Oncol. 5, 1710–1717 (2019).
Moding, E. J. et al. Circulating tumor DNA dynamics predict benefit from consolidation immunotherapy in locally advanced non-small cell lung cancer. Nat. Cancer 1, 176–183 (2020).
Lebow, E. S. et al. ctDNA-based detection of molecular residual disease in stage I–III non-small cell lung cancer patients treated with definitive radiotherapy. Front. Oncol. 13, 1253629 (2023).
Gale, D. et al. Residual ctDNA after treatment predicts early relapse in patients with early-stage non-small cell lung cancer. Ann. Oncol. 33, 500–510 (2022).
Tarazona, N. et al. Targeted next-generation sequencing of circulating-tumor DNA for tracking minimal residual disease in localized colon cancer. Ann. Oncol. 30, 1804–1812 (2019).
Shaw, J. et al. Serial postoperative circulating tumor DNA assessment has strong prognostic value during long-term follow-up in patients with breast cancer. JCO Precis. Oncol. 8, e2300456 (2024).
Christensen, E. et al. Early detection of metastatic relapse and monitoring of therapeutic efficacy by ultra-deep sequencing of plasma cell-free DNA in patients with urothelial bladder carcinoma. J. Clin. Oncol. 37, 1547–1557 (2019).
Sausen, M. et al. Clinical implications of genomic alterations in the tumour and circulation of pancreatic cancer patients. Nat. Commun. 6, 7686 (2015).
Tan, L. et al. Prediction and monitoring of relapse in stage III melanoma using circulating tumor DNA. Ann. Oncol. 30, 804–814 (2019).
Natera, Inc. A personalized, tumor-informed approach to detect molecular residual disease with high sensitivity and specificity. Natera.com https://www.natera.com/wp-content/uploads/2020/11/Oncology-Clinical-A-personalized-tumor-informed-approach-to-detect-molecular-residual-disease-SGN_SR_WP.pdf (2020).
Zviran, A. et al. Genome-wide cell-free DNA mutational integration enables ultra-sensitive cancer monitoring. Nat. Med. 26, 1114–1124 (2020).
Gydush, G. et al. Massively parallel enrichment of low-frequency alleles enables duplex sequencing at low depth. Nat. Biomed. Eng. 6, 257–266 (2022).
Diehl, F. et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 14, 985–990 (2008).
Madsen, A. T., Hojbjerg, J. A., Sorensen, B. S. & Winther-Larsen, A. Day-to-day and within-day biological variation of cell-free DNA. eBioMedicine 49, 284–290 (2019).
Vittori, L. N., Tarozzi, A. & Latessa, P. M. Circulating cell-free DNA in physical activities. Methods Mol. Biol. 1909, 183–197 (2019).
Henriksen, T. V. et al. The effect of surgical trauma on circulating free DNA levels in cancer patients — implications for studies of circulating tumor DNA. Mol. Oncol. 14, 1670–1679 (2020).
Razavi, P. et al. High-intensity sequencing reveals the sources of plasma circulating cell-free DNA variants. Nat. Med. 25, 1928–1937 (2019).
Finn, O. The dawn of vaccines for cancer prevention. Nat. Rev. Immunol. 18, 183–194 (2018).
Martínez-Jiménez, F. et al. Pan-cancer whole-genome comparison of primary and metastatic solid tumors. Nature 618, 333–341 (2023).
Razavi, R. et al. The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 34, 427–438 (2018).
Kasi, P. M. et al. Impact of circulating tumor DNA-based detection of molecular residual disease on the conduct and design of clinical trials for solid tumors. JCO Precis. Oncol. 6, e2100181 (2022). This study comprehensively details the impact that circulating tumour DNA-based trial enrichment can have on the efficiency of molecular evidence of disease-based trials.
Magbanua, M. J. M. et al. Clinical significance and biology of circulating tumor DNA in high-risk early-stage HER2-negative breast cancer receiving neoadjuvant chemotherapy. Cancer Cell 41, 1091–1102 e1094 (2023).
Tie, J. et al. Circulating tumor DNA analysis guiding adjuvant therapy in stage II colon cancer. N. Engl. J. Med. 386, 2261–2272 (2022).
U.S. Food and Drug Administration Oncology Center of Excellence. Use of circulating tumor DNA for early-stage solid tumor drug development: draft guidance for industry. FDA.gov https://www.fda.gov/media/158072/download (2022). This FDA draft guidance for the use of circulating tumour DNA for early-stage solid tumour drug development validates the urgent need for molecular evidence of disease-based trials and helps biopharma companies consider how to plan them.
Manalac, T. FDA Adcomm backs new surrogate endpoint for accelerated approvals in blood cancer. Biospace.com https://www.biospace.com/article/fda-adcomm-backs-new-surrogate-endpoint-for-accelerated-approvals-in-blood-cancer/ (2024).
Morris, V. K. et al. Phase II results of circulating tumor DNA as a predictive biomarker in adjuvant chemotherapy in patients with stage II colon cancer: NRG-GI005 (COBRA) phase II/III study. J. Clin. Oncol. 42, 5–5 (2024).
Campbell, J. D. et al. Distinct patterns of somatic genome alterations in lung adenocarcinomas and squamous cell carcinomas. Nat. Genet. 48, 607–616 (2016).
Mishra-Kalyani, P. S. et al. External control arms in oncology: current use and future directions. Ann. Oncol. 33, 376–383 (2022).
CMS. Centers for Medicare & Medicaid Services. Billing and coding: MolDX: minimal residual disease testing for solid tumor cancers. https://www.cms.gov/medicare-coverage-database/view/article.aspx?articleId=58456 (2024).
Natera, Inc. Q4 and Full Year 2023 Earnings Call. https://s201.q4cdn.com/354493536/files/doc_financials/2023/q4/Q4-2023-Final-Slides.pdf (2024).
Martin-Alonso, C. et al. Priming agents transiently reduce the clearance of cell-free DNA to improve liquid biopsies. Science 383, eadf2341 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04567420 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT03285412 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04985266 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT05512364 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04915755 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT03145961 (2022).
Turner, N. C. et al. Results of the c-TRAK TN trial: a clinical trial utilising ctDNA mutation tracking to detect molecular residual disease and trigger intervention in patients with moderate- and high-risk early-stage triple-negative breast cancer. Ann. Oncol. 34, 200–211 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04434040 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT03803553 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04068103 (2024).
ANZCTR. The Australian and New Zealand Clinical Trial Registry. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=12615000381583 (2020).
ANZCTR. The Australian and New Zealand Clinical Trial Registry. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=12617001566325 (2020).
Tie, J. DYNAMIC-III: circulating tumour DNA analysis informing adjuvant chemotherapy in stage III colon cancer — a multicentre phase II/III randomised controlled study. In Australasian Gastro-Intestinal Trials Group Annual Scientific Meeting. https://asm.gicancer.org.au/agitg-abstracts/dynamic-iii/ (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04457297 (2022).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04050345 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT05174169 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04089631 (2020).
Folprecht, G. et al. The CIRCULATE trial: circulating tumor DNA based decision for adjuvant treatment in colon cancer stage II evaluation (AIO-KRK-0217). Clin. Colorectal Cancer 21, 170–174 (2022).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04120701 (2021).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04510285 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT03832569 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04660344 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT05987241 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04585477 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04367311 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04385368 (2023).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04642469 (2024).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/show/NCT04585490 (2024).
Amin, M. B. & Edge, S. B. AJCC Cancer Staging Manual (Springer, 2017).
Howlader, N., Cronin, K. A., Kurian, A. W. & Andridge, R. Differences in breast cancer survival by molecular subtypes in the United States. Cancer Epidemiol. Biomark. Prev. 27, 619–626 (2018).
Acknowledgements
The authors thank S. Kopetz, V. Adalsteinsson, T. Golub, B. Alexander and T. Hudson for providing reviews and comments.
Author information
Authors and Affiliations
Contributions
A.J.M., A.B.C. and A.R. researched data for the article. All authors contributed substantially to discussion of the content. A.J.M., A.B.C., A.R., E.S.L. and A.P. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
A.J.M. has served as an adviser/consultant for AstraZeneca, Guardant Health, Illumina, Myriad Genetics, Natera, SAGA Diagnostics and Science for America. A.J.M.’s research is supported by a National Institutes of Health K12 grant (K12CA087723). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. A.R. holds equity in Roche. N.W. is an employee of Genentech/Roche and has equity in Roche, Relay Therapeutics and Flare Therapeutics. L.G. is an employee of Genentech/Roche and has equity in Roche. A.P. has held Equity in C2i Genomics, XGenomes, Cadex, Vionix and Parithera. In the past 36 months, she has served as an adviser/consultant for Eli Lilly, Mirati, Pfizer, Inivata, Biofidelity, Checkmate Pharmaceuticals, FMI, Guardant, Abbvie, Bayer, Delcath, Taiho, CVS, Value Analytics Lab, Seagen, Saga, AZ, Scare Inc., Illumina, Taiho, Hookipa, Kahar Medical, Xilio Therapeutics, Sirtex, Takeda and Science for America. She receives fees from Up to Date. She has received travel fees from Karkinos Healthcare. She has been on the DSMC for a Roche study and on the Steering Committee for Exilixis. She has received research funding to the Institution from PureTech, PMV Pharmaceuticals, Plexxicon, Takeda, BMS, Mirati, Novartis, Erasca, Genentech, Daiichi Sankyo, Syndax, Revolution Medicine and Parthenon. A.P.’s research is supported by an NIH/NCI K08 grant (K08273688). A.B.C. and E.S.L. declare no competing interests.
Peer review
Peer review information
Nature Reviews Cancer thanks Cathy Eng, Stefan Fröhling and Jean-Charles Soria for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Baseline toxicity
-
Persistent side effects after cancer-directed therapy (for example, permanent neuropathy).
- Circulating tumour DNA
-
(ctDNA). Cell-free fragments of DNA released by tumour cells into the blood.
- Clinical evidence of disease
-
(CED). Clinical evidence of cancer detected on exam (for example, a palpable mass).
- Clinically high-risk disease
-
High-risk disease is determined by clinical factors (for example, tumour size and number of affected lymph nodes) and pathological factors (for example, high mitotic index or presence of residual disease after neoadjuvant therapy) that have been shown to be associated with a high risk of recurrence.
- Definitive treatment
-
Cancer-directed therapy, for example, surgery, radiation and chemotherapy, given with the intention of cure.
- Minimal residual disease
-
(MRD). Cancer in the blood (or bone marrow) that remains detectable after treatment.
- Molecular evidence of disease
-
(MED). Circulating tumour DNA (ctDNA) detectable via blood assay. In ‘early MED’, ctDNA is detected before radiographic findings or clinical evidence of cancer.
- Radiographic evidence of disease
-
(RED). Radiography images that visibly show the presence of cancer.
- Registrational trial
-
A trial that is used by regulatory authorities to determine whether a drug is safe and effective enough to be approved and administered to patients outside a clinical trial. Typically, registrational trials are large-scale, randomized controlled trials.
- Signal-finding trials
-
Clinical trials with the objective of identifying treatment (treatments) with an early indication of efficacy — usually measured using molecular biomarkers that reflect a relevant biological process — before advancing to larger, more definitive trials with robust clinical end points.
- Single-arm studies
-
A non-randomized clinical trial that uses only one experimental group of patients, without a control group.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Medford, A.J., Carmeli, A.B., Ritchie, A. et al. A standing platform for cancer drug development using ctDNA-based evidence of recurrence. Nat Rev Cancer 24, 810–821 (2024). https://doi.org/10.1038/s41568-024-00742-2
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41568-024-00742-2
This article is cited by
-
Advances in molecular pathology and therapy of non-small cell lung cancer
Signal Transduction and Targeted Therapy (2025)