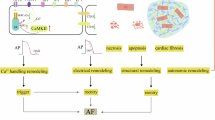
Abstract
Atrial fibrosis is one of the main manifestations of atrial cardiomyopathy, an array of electrical, mechanical and structural alterations associated with atrial fibrillation (AF), stroke and heart failure. Atrial fibrosis can be both a cause and a consequence of AF and, once present, it accelerates the progression of AF. The pathophysiological mechanisms leading to atrial fibrosis are diverse and include stretch-induced activation of fibroblasts, systemic inflammatory processes, activation of coagulation factors and fibrofatty infiltrations. Importantly, atrial fibrosis can occur in different forms, such as reactive and replacement fibrosis. The diversity of atrial fibrosis mechanisms and patterns depends on sex, age and comorbidity profile, hampering the development of therapeutic strategies. In addition, the presence and severity of comorbidities often change over time, potentially causing temporal changes in the mechanisms underlying atrial fibrosis development. This Review summarizes the latest knowledge on the molecular and cellular mechanisms of atrial fibrosis, its association with comorbidities and the sex-related differences. We describe how the various patterns of atrial fibrosis translate into electrophysiological mechanisms that promote AF, and critically appraise the clinical applicability and limitations of diagnostic tools to quantify atrial fibrosis. Finally, we provide an overview of the newest therapeutic interventions under development and discuss relevant knowledge gaps related to the association between clinical manifestations and pathological mechanisms of atrial fibrosis and to the translation of this knowledge to a clinical setting.
Key points
-
Atrial fibrosis occurs as reactive or reparative fibrosis, which differ in their underlying mechanisms; the epicardial layer of the atrium shows a higher degree of fibrosis than the endocardial bundle network.
-
Activated fibroblasts are the main cell type involved in collagen synthesis, but sensing of environmental factors that lead to atrial fibrosis involves other cell types, such as cardiomyocytes, adipocytes and immune cells.
-
Diffuse, reactive fibrosis on the millimetre scale results in discontinuous electrical conduction, longitudinal dissociation and endocardial–epicardial dissociation of electrical activity during atrial fibrillation; larger areas of patchy fibrosis (at the centimetre scale) are more likely to anchor macro re-entrant circuits.
-
Neither late gadolinium enhancement cardiac MRI nor low-voltage areas show a convincingly reliable and reproducible association with atrial fibrosis; therefore, the term ‘atrial fibrosis’ to refer to enhanced late gadolinium enhancement or low-voltage areas might lead to misinterpretation and should be avoided.
-
Angiotensin-converting enzyme inhibitors, angiotensin II receptor inhibitors and aldosterone receptor blockers are of limited use in preventing atrial collagen deposition, particularly if used independently from indications for cardiovascular diseases such as hypertension or heart failure.
-
MicroRNAs and mRNAs encapsulated in lipid nanoparticles are currently being explored as potential approaches for antifibrotic treatment; the advantage of these strategies is the high level of specificity for targeting molecular pathways.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Li, Z. et al. Atrial cardiomyopathy markers and new-onset atrial fibrillation risk in patients with acute myocardial infarction. Eur. J. Intern. Med. 102, 72–79 (2022).
Li, M. et al. Atrial cardiomyopathy: from cell to bedside. Esc. Heart Fail. 9, 3768–3784 (2022).
Miyauchi, S. et al. Relationship between fibrosis, endocardial endothelial damage, and thrombosis of left atrial appendage in atrial fibrillation. JACC Clin. Electrophysiol. 9, 1158–1168 (2023).
Nattel, S. Atrial fibrosis, endocardial damage, and thrombosis in atrial fibrillation: association with underlying conditions or causal? JACC Clin. Electrophysiol. 9, 1169–1171 (2023).
Kamel, H. et al. The atrial cardiopathy and antithrombotic drugs in prevention after cryptogenic stroke randomized trial: rationale and methods. Int. J. Stroke 14, 207–214 (2019).
Goette, A. et al. Atrial cardiomyopathy revisited – evolution of a concept. A clinical consensus statement of the European Heart Rhythm Association (EHRA) of the ESC, the Heart Rhythm Society (HRS), the Asian Pacific Heart Rhythm Association (APHRS), and the Latin American Heart Rhythm Society (LAHRS). Europace, https://doi.org/10.1093/europace/euae204 (2024).
Goette, A. et al. EHRA/HRS/APHRS/SOLAECE expert consensus on atrial cardiomyopathies: definition, characterization, and clinical implication. Europace 18, 1455–1490 (2016).
Schotten, U., Verheule, S., Kirchhof, P. & Goette, A. Pathophysiological mechanisms of atrial fibrillation: a translational appraisal. Physiol. Rev. 91, 265–325 (2011).
Nattel, S. Molecular and cellular mechanisms of atrial fibrosis in atrial fibrillation. JACC Clin. Electrophysiol. 3, 425–435 (2017).
Nattel, S. & Dobrev, D. Controversies about atrial fibrillation mechanisms: aiming for order in chaos and whether it matters. Circ. Res. 120, 1396–1398 (2017).
Heijman, J., Linz, D. & Schotten, U. Dynamics of atrial fibrillation mechanisms and comorbidities. Annu. Rev. Physiol. 83, 83–106 (2021).
Weber, K. T., Pick, R., Jalil, J. E., Janicki, J. S. & Carroll, E. P. Patterns of myocardial fibrosis. J. Mol. Cell Cardiol. 21, 121–131 (1989).
Verheule, S. & Schotten, U. Electrophysiological consequences of cardiac fibrosis. Cells 10, 3220 (2021).
Maesen, B. et al. Endomysial fibrosis, rather than overall connective tissue content, is the main determinant of conduction disturbances in human atrial fibrillation. Europace 24, 1015–1024 (2022).
Winters, J. et al. Heart failure, female sex, and atrial fibrillation are the main drivers of human atrial cardiomyopathy: results from the CATCH ME consortium. J. Am. Heart Assoc. 12, e031220 (2023).
Fabritz, L. et al. Dynamic risk assessment to improve quality of care in patients with atrial fibrillation: the 7th AFNET/EHRA consensus conference. Europace 23, 329–344 (2021).
Rohr, S. Arrhythmogenic implications of fibroblast–myocyte interactions. Circ. Arrhythm. Electrophysiol. 5, 442–452 (2012).
Weber, K. T., Sun, Y., Bhattacharya, S. K., Ahokas, R. A. & Gerling, I. C. Myofibroblast-mediated mechanisms of pathological remodelling of the heart. Nat. Rev. Cardiol. 10, 15–26 (2013).
Cardin, S. et al. Evolution of the atrial fibrillation substrate in experimental congestive heart failure: angiotensin-dependent and -independent pathways. Cardiovasc. Res. 60, 315–325 (2003).
Westerman, S. & Wenger, N. Gender differences in atrial fibrillation: a review of epidemiology, management, and outcomes. Curr. Cardiol. Rev. 15, 136–144 (2019).
Verheule, S. et al. Fibrillatory conduction in the atrial free walls of goats in persistent and permanent atrial fibrillation. Circ. Arrhythm. Electrophysiol. 3, 590–599 (2010).
Verheule, S. et al. Loss of continuity in the thin epicardial layer because of endomysial fibrosis increases the complexity of atrial fibrillatory conduction. Circ. Arrhythm. Electrophysiol. 6, 202–211 (2013).
Ravelli, F. et al. Quantitative assessment of transmural fibrosis profile in the human atrium: evidence for a three-dimensional arrhythmic substrate by slice-to-slice histology. Europace 25, 739–747 (2023).
Li, D., Fareh, S., Leung, T. K. & Nattel, S. Promotion of atrial fibrillation by heart failure in dogs: atrial remodeling of a different sort. Circulation 100, 87–95 (1999).
Platonov, P. G., Mitrofanova, L. B., Orshanskaya, V. & Ho, S. Y. Structural abnormalities in atrial walls are associated with presence and persistency of atrial fibrillation but not with age. J. Am. Coll. Cardiol. 58, 2225–2232 (2011).
van Brakel, T. J. et al. Fibrosis and electrophysiological characteristics of the atrial appendage in patients with atrial fibrillation and structural heart disease. J. Interv. Card. Electrophysiol. 38, 85–93 (2013).
Chen, J. et al. Extent and spatial distribution of left atrial arrhythmogenic sites, late gadolinium enhancement at magnetic resonance imaging, and low-voltage areas in patients with persistent atrial fibrillation: comparison of imaging vs. electrical parameters of fibrosis and arrhythmogenesis. Europace 21, 1484–1493 (2019).
Cochet, H. et al. Age, atrial fibrillation, and structural heart disease are the main determinants of left atrial fibrosis detected by delayed-enhanced magnetic resonance imaging in a general cardiology population. J. Cardiovasc. Electrophysiol. 26, 484–492 (2015).
Benito, E. M. et al. Preferential regional distribution of atrial fibrosis in posterior wall around left inferior pulmonary vein as identified by late gadolinium enhancement cardiac magnetic resonance in patients with atrial fibrillation. Europace 20, 1959–1965 (2018).
Nattel, S., Heijman, J., Zhou, L. & Dobrev, D. Molecular basis of atrial fibrillation pathophysiology and therapy: a translational perspective. Circ. Res. 127, 51–72 (2020).
Takawale, A., Aguilar, M., Bouchrit, Y. & Hiram, R. Mechanisms and management of thyroid disease and atrial fibrillation: impact of atrial electrical remodeling and cardiac fibrosis. Cells 11, 4047 (2022).
Harada, M. & Nattel, S. Implications of inflammation and fibrosis in atrial fibrillation pathophysiology. Card. Electrophysiol. Clin. 13, 25–35 (2021).
Gawalko, M. et al. Adiposity-associated atrial fibrillation: molecular determinants, mechanisms, and clinical significance. Cardiovasc. Res. 119, 614–630 (2023).
Kato, T. et al. Endothelial-mesenchymal transition in human atrial fibrillation. J. Cardiol. 69, 706–711 (2017).
Lai, Y. J. et al. miR-181b targets semaphorin 3A to mediate TGF-β-induced endothelial-mesenchymal transition related to atrial fibrillation. J. Clin. Invest. 132, e142548 (2022).
Simon, J. N., Ziberna, K. & Casadei, B. Compromised redox homeostasis, altered nitroso-redox balance, and therapeutic possibilities in atrial fibrillation. Cardiovasc. Res. 109, 510–518 (2016).
Yue, L., Xie, J. & Nattel, S. Molecular determinants of cardiac fibroblast electrical function and therapeutic implications for atrial fibrillation. Cardiovasc. Res. 89, 744–753 (2011).
Qiu, H. et al. Salvianolate reduces atrial fibrillation through suppressing atrial interstitial fibrosis by inhibiting TGF-β1/Smad2/3 and TXNIP/NLRP3 inflammasome signaling pathways in post-MI rats. Phytomedicine 51, 255–265 (2018).
Chen, Y. et al. JAK-STAT signalling and the atrial fibrillation promoting fibrotic substrate. Cardiovasc. Res. 113, 310–320 (2017).
He, X. et al. Atrial fibrillation induces myocardial fibrosis through angiotensin II type 1 receptor-specific Arkadia-mediated downregulation of Smad7. Circ. Res. 108, 164–175 (2011).
Serini, G. & Gabbiani, G. Mechanisms of myofibroblast activity and phenotypic modulation. Exp. Cell Res. 250, 273–283 (1999).
Al-U’datt, D., Allen, B. G. & Nattel, S. Role of the lysyl oxidase enzyme family in cardiac function and disease. Cardiovasc. Res. 115, 1820–1837 (2019).
Valiente-Alandi, I. et al. Inhibiting fibronectin attenuates fibrosis and improves cardiac function in a model of heart failure. Circulation 138, 1236–1252 (2018).
Hume, R. D. et al. Tropoelastin improves post-infarct cardiac function. Circ. Res. 132, 72–86 (2023).
Xia, P., Liu, Y. & Cheng, Z. Signaling pathways in cardiac myocyte apoptosis. Biomed. Res. Int. 2016, 9583268 (2016).
Mayyas, F. et al. Association of left atrial endothelin-1 with atrial rhythm, size, and fibrosis in patients with structural heart disease. Circ. Arrhythm. Electrophysiol. 3, 369–379 (2010).
Moreira, L. M. et al. Paracrine signalling by cardiac calcitonin controls atrial fibrogenesis and arrhythmia. Nature 587, 460–465 (2020).
Chen, G., Chelu, M. G., Dobrev, D. & Li, N. Cardiomyocyte inflammasome signaling in cardiomyopathies and atrial fibrillation: mechanisms and potential therapeutic implications. Front. Physiol. 9, 1115 (2018).
Yao, C. et al. Enhanced cardiomyocyte NLRP3 inflammasome signaling promotes atrial fibrillation. Circulation 138, 2227–2242 (2018).
Heijman, J. et al. Atrial myocyte NLRP3/CaMKII nexus forms a substrate for postoperative atrial fibrillation. Circ. Res. 127, 1036–1055 (2020).
Li, L. et al. Fibroblast-specific inflammasome activation predisposes to atrial fibrillation. Preprint at BioRxiv https://doi.org/10.1101/2023.05.18.541326 (2023).
Van Linthout, S., Miteva, K. & Tschope, C. Crosstalk between fibroblasts and inflammatory cells. Cardiovasc. Res. 102, 258–269 (2014).
Frangogiannis, N. G. The inflammatory response in myocardial injury, repair, and remodelling. Nat. Rev. Cardiol. 11, 255–265 (2014).
Hulsmans, M. et al. Recruited macrophages elicit atrial fibrillation. Science 381, 231–239 (2023).
Wang, Q. et al. Human epicardial adipose tissue activin a expression predicts occurrence of postoperative atrial fibrillation in patients receiving cardiac surgery. Heart Lung Circ. 28, 1697–1705 (2019).
Villarreal-Molina, M. T. & Antuna-Puente, B. Adiponectin: anti-inflammatory and cardioprotective effects. Biochimie 94, 2143–2149 (2012).
Matsuo, K. et al. Omentin functions to attenuate cardiac hypertrophic response. J. Mol. Cell Cardiol. 79, 195–202 (2015).
Antonopoulos, A. S. et al. Mutual regulation of epicardial adipose tissue and myocardial redox state by PPAR-γ/adiponectin signalling. Circ. Res. 118, 842–855 (2016).
Haemers, P. et al. Atrial fibrillation is associated with the fibrotic remodelling of adipose tissue in the subepicardium of human and sheep atria. Eur. Heart J. 38, 53–61 (2017).
Suffee, N. et al. Reactivation of the epicardium at the origin of myocardial fibro-fatty infiltration during the atrial cardiomyopathy. Circ. Res. 126, 1330–1342 (2020).
Chaumont, C. et al. Epicardial origin of cardiac arrhythmias: clinical evidences and pathophysiology. Cardiovasc. Res. 118, 1693–1702 (2022).
Ernault, A. C., Meijborg, V. M. F. & Coronel, R. Modulation of cardiac arrhythmogenesis by epicardial adipose tissue: JACC state-of-the-art review. J. Am. Coll. Cardiol. 78, 1730–1745 (2021).
Krishnan, A. et al. Are interactions between epicardial adipose tissue, cardiac fibroblasts and cardiac myocytes instrumental in atrial fibrosis and atrial fibrillation? Cells 10, 2501 (2021).
Kira, S. et al. Role of angiopoietin-like protein 2 in atrial fibrosis induced by human epicardial adipose tissue: analysis using an organo-culture system. Heart Rhythm. 17, 1591–1601 (2020).
D’Alessandro, E. et al. Coagulation factor Xa induces proinflammatory responses in cardiac fibroblasts via activation of protease-activated receptor-1. Cells 10, 2958 (2021).
Spronk, H. M. et al. Hypercoagulability causes atrial fibrosis and promotes atrial fibrillation. Eur. Heart J. 38, 38–50 (2017).
Spronk, H. M. et al. Pleiotropic effects of factor Xa and thrombin: what to expect from novel anticoagulants. Cardiovasc. Res. 101, 344–351 (2014).
Erlich, J. H. et al. Inhibition of the tissue factor-thrombin pathway limits infarct size after myocardial ischemia-reperfusion injury by reducing inflammation. Am. J. Pathol. 157, 1849–1862 (2000).
MacKenna, D. A., Dolfi, F., Vuori, K. & Ruoslahti, E. Extracellular signal-regulated kinase and c-Jun NH2-terminal kinase activation by mechanical stretch is integrin-dependent and matrix-specific in rat cardiac fibroblasts. J. Clin. Invest. 101, 301–310 (1998).
Hooper, C. L., Dash, P. R. & Boateng, S. Y. Lipoma preferred partner is a mechanosensitive protein regulated by nitric oxide in the heart. FEBS Open. Bio 2, 135–144 (2012).
Li, X., Garcia-Elias, A., Benito, B. & Nattel, S. The effects of cardiac stretch on atrial fibroblasts: analysis of the evidence and potential role in atrial fibrillation. Cardiovasc. Res. 118, 440–460 (2022).
Ruwhof, C., van Wamel, A. E., van der Valk, L. J., Schrier, P. I. & van der Laarse, A. Direct, autocrine and paracrine effects of cyclic stretch on growth of myocytes and fibroblasts isolated from neonatal rat ventricles. Arch. Physiol. Biochem. 109, 10–17 (2001).
Bomb, R. et al. Myofibroblast secretome and its auto-/paracrine signaling. Expert. Rev. Cardiovasc. Ther. 14, 591–598 (2016).
Li, Z. et al. Gender differences in fibrosis remodeling in patients with long-standing persistent atrial fibrillation. Oncotarget 8, 53714–53729 (2017).
Pappritz, K. et al. Sex- and age-related differences in the inflammatory properties of cardiac fibroblasts: impact on the cardiosplenic axis and cardiac fibrosis. Front. Cardiovasc. Med. 10, 1117419 (2023).
Bukowska, A. et al. Protective regulation of the ACE2/ACE gene expression by estrogen in human atrial tissue from elderly men. Exp. Biol. Med. 242, 1412–1423 (2017).
Akoum, N., Mahnkopf, C., Kholmovski, E. G., Brachmann, J. & Marrouche, N. F. Age and sex differences in atrial fibrosis among patients with atrial fibrillation. Europace 20, 1086–1092 (2018).
Masuda, M. et al. Gender differences in atrial fibrosis and cardiomyopathy assessed by left atrial low-voltage areas during catheter ablation of atrial fibrillation. Am. J. Cardiol. 203, 37–44 (2023).
Kuppe, C. et al. Spatial multi-omic map of human myocardial infarction. Nature 608, 766–777 (2022).
Heijman, J. et al. The value of basic research insights into atrial fibrillation mechanisms as a guide to therapeutic innovation: a critical analysis. Cardiovasc. Res. 109, 467–479 (2016).
Zlochiver, S. et al. Electrotonic myofibroblast-to-myocyte coupling increases propensity to reentrant arrhythmias in two-dimensional cardiac monolayers. Biophys. J. 95, 4469–4480 (2008).
Roney, C. H. et al. Modelling methodology of atrial fibrosis affects rotor dynamics and electrograms. Europace 18, iv146–iv155 (2016).
Vandersickel, N. et al. Dynamical anchoring of distant arrhythmia sources by fibrotic regions via restructuring of the activation pattern. PLoS Comput. Biol. 14, e1006637 (2018).
Jalife, J., Berenfeld, O. & Mansour, M. Mother rotors and fibrillatory conduction: a mechanism of atrial fibrillation. Cardiovasc. Res. 54, 204–216 (2002).
Tanaka, K. et al. Spatial distribution of fibrosis governs fibrillation wave dynamics in the posterior left atrium during heart failure. Circ. Res. 101, 839–847 (2007).
Koura, T. et al. Anisotropic conduction properties in canine atria analyzed by high-resolution optical mapping: preferential direction of conduction block changes from longitudinal to transverse with increasing age. Circulation 105, 2092–2098 (2002).
Xie, Y., Sato, D., Garfinkel, A., Qu, Z. & Weiss, J. N. So little source, so much sink: requirements for afterdepolarizations to propagate in tissue. Biophys. J. 99, 1408–1415 (2010).
Sato, D. et al. Synchronization of chaotic early afterdepolarizations in the genesis of cardiac arrhythmias. Proc. Natl Acad. Sci. USA 106, 2983–2988 (2009).
Colman, M. A., Sharma, R., Aslanidi, O. V. & Zhao, J. Patchy fibrosis promotes trigger-substrate interactions that both generate and maintain atrial fibrillation. Interface Focus. 13, 20230041 (2023).
Joyner, R. W. & van Capelle, F. J. Propagation through electrically coupled cells. How a small SA node drives a large atrium. Biophys. J. 50, 1157–1164 (1986).
Verheule, S. et al. Increased vulnerability to atrial fibrillation in transgenic mice with selective atrial fibrosis caused by overexpression of TGF-β1. Circ. Res. 94, 1458–1465 (2004).
Choi, E. K. et al. Triggered firing and atrial fibrillation in transgenic mice with selective atrial fibrosis induced by overexpression of TGF-β1. Circ. J. 76, 1354–1362 (2012).
Dhein, S. et al. Remodeling of cardiac passive electrical properties and susceptibility to ventricular and atrial arrhythmias. Front. Physiol. 5, 424 (2014).
Zhao, J., Schotten, U., Smaill, B. & Verheule, S. Loss of side-to-side connections affects the relative contributions of the sodium and calcium current to transverse propagation between strands of atrial myocytes. Front. Physiol. 9, 1212 (2018).
Spach, M. S. & Boineau, J. P. Microfibrosis produces electrical load variations due to loss of side-to-side cell connections: a major mechanism of structural heart disease arrhythmias. Pacing Clin. Electrophysiol. 20, 397–413 (1997).
Spach, M. S., Heidlage, J. F., Dolber, P. C. & Barr, R. C. Mechanism of origin of conduction disturbances in aging human atrial bundles: experimental and model study. Heart Rhythm. 4, 175–185 (2007).
Spach, M. S. & Dolber, P. C. Relating extracellular potentials and their derivatives to anisotropic propagation at a microscopic level in human cardiac muscle. Evidence for electrical uncoupling of side-to-side fiber connections with increasing age. Circ. Res. 58, 356–371 (1986).
Wesselink, R. et al. Does left atrial epicardial conduction time reflect atrial fibrosis and the risk of atrial fibrillation recurrence after thoracoscopic ablation? Post hoc analysis of the AFACT trial. BMJ Open. 12, e056829 (2022).
Berenfeld, O., Zaitsev, A. V., Mironov, S. F., Pertsov, A. M. & Jalife, J. Frequency-dependent breakdown of wave propagation into fibrillatory conduction across the pectinate muscle network in the isolated sheep right atrium. Circ. Res. 90, 1173–1180 (2002).
Ho, S. Y., Anderson, R. H. & Sanchez-Quintana, D. Atrial structure and fibres: morphologic bases of atrial conduction. Cardiovasc. Res. 54, 325–336 (2002).
Verheule, S. et al. Tissue structure and connexin expression of canine pulmonary veins. Cardiovasc. Res. 55, 727–738 (2002).
Hocini, M. et al. Electrical conduction in canine pulmonary veins: electrophysiological and anatomic correlation. Circulation 105, 2442–2448 (2002).
Li, D. et al. Effects of angiotensin-converting enzyme inhibition on the development of the atrial fibrillation substrate in dogs with ventricular tachypacing-induced congestive heart failure. Circulation 104, 2608–2614 (2001).
Hanna, N., Cardin, S., Leung, T. K. & Nattel, S. Differences in atrial versus ventricular remodeling in dogs with ventricular tachypacing-induced congestive heart failure. Cardiovasc. Res. 63, 236–244 (2004).
Li, D., Benardeau, A. & Nattel, S. Contrasting efficacy of dofetilide in differing experimental models of atrial fibrillation. Circulation 102, 104–112 (2000).
Shinagawa, K., Shi, Y.-F., Tardif, J.-C., Leung, T.-K. & Nattel, S. Dynamic nature of atrial fibrillation substrate during development and reversal of heart failure in dogs. Circulation 105, 2672–2678 (2002).
Schoonderwoerd, B. A. et al. Atrial ultrastructural changes during experimental atrial tachycardia depend on high ventricular rate. J. Cardiovasc. Electrophysiol. 15, 1167–1174 (2004).
Anne, W. et al. Self-terminating AF depends on electrical remodeling while persistent AF depends on additional structural changes in a rapid atrially paced sheep model. J. Mol. Cell Cardiol. 43, 148–158 (2007).
Guichard, J. B. et al. Role of atrial arrhythmia and ventricular response in atrial fibrillation induced atrial remodelling. Cardiovasc. Res. 117, 462–471 (2021).
Wijffels, M. C., Kirchhof, C. J., Dorland, R. & Allessie, M. A. Atrial fibrillation begets atrial fibrillation. A study in awake chronically instrumented goats. Circulation 92, 1954–1968 (1995).
Ausma, J. et al. Time course of atrial fibrillation-induced cellular structural remodeling in atria of the goat. J. Mol. Cell Cardiol. 33, 2083–2094 (2001).
Gharaviri, A. et al. Epicardial fibrosis explains increased endo-epicardial dissociation and epicardial breakthroughs in human atrial fibrillation. Front. Physiol. 11, 68 (2020).
Eckstein, J. et al. Time course and mechanisms of endo-epicardial electrical dissociation during atrial fibrillation in the goat. Cardiovasc. Res. 89, 816–824 (2011).
Verheule, S. et al. Role of endo-epicardial dissociation of electrical activity and transmural conduction in the development of persistent atrial fibrillation. Prog. Biophys. Mol. Biol. 115, 173–185 (2014).
Eckstein, J. et al. Transmural conduction is the predominant mechanism of breakthrough during atrial fibrillation: evidence from simultaneous endo-epicardial high-density activation mapping. Circ. Arrhythm. Electrophysiol. 6, 334–341 (2013).
Maesen, B. et al. Rearrangement of atrial bundle architecture and consequent changes in anisotropy of conduction constitute the 3-dimensional substrate for atrial fibrillation. Circ. Arrhythm. Electrophysiol. 6, 967–975 (2013).
Spach, M. S., Heidlage, J. F., Dolber, P. C. & Barr, R. C. Changes in anisotropic conduction caused by remodeling cell size and the cellular distribution of gap junctions and Na+ channels. J. Electrocardiol. 34, 69–76 (2001).
Abed, H. S. et al. Obesity results in progressive atrial structural and electrical remodeling: implications for atrial fibrillation. Heart Rhythm. 10, 90–100 (2013).
Polontchouk, L. et al. Effects of chronic atrial fibrillation on gap junction distribution in human and rat atria. J. Am. Coll. Cardiol. 38, 883–891 (2001).
Quinn, T. A. et al. Electrotonic coupling of excitable and nonexcitable cells in the heart revealed by optogenetics. Proc. Natl Acad. Sci. USA 113, 14852–14857 (2016).
Wang, Y. et al. Fibroblasts in heart scar tissue directly regulate cardiac excitability and arrhythmogenesis. Science 381, 1480–1487 (2023).
Elliott, A. D., Middeldorp, M. E., Van Gelder, I. C., Albert, C. M. & Sanders, P. Epidemiology and modifiable risk factors for atrial fibrillation. Nat. Rev. Cardiol. 20, 404–417 (2023).
Anné, W. et al. Matrix metalloproteinases and atrial remodeling in patients with mitral valve disease and atrial fibrillation. Cardiovasc. Res. 67, 655–666 (2005).
Ramos, K. et al. Degree of fibrosis in human atrial tissue is not the hallmark driving AF. Cells 11, 427 (2022).
Spach, M. S., Dolber, P. C. & Heidlage, J. F. Properties of discontinuous anisotropic propagation at a microscopic level. Ann. N. Y. Acad. Sci. 591, 62–74 (1990).
Verheule, S. et al. Alterations in atrial electrophysiology and tissue structure in a canine model of chronic atrial dilatation due to mitral regurgitation. Circulation 107, 2615–2622 (2003).
Krul, S. P. J. et al. Atrial fibrosis and conduction slowing in the left atrial appendage of patients undergoing thoracoscopic surgical pulmonary vein isolation for atrial fibrillation. Circ. Arrhythm. Electrophysiol. 8, 288–295 (2015).
Hansen, B. J. et al. Atrial fibrillation driven by micro-anatomic intramural re-entry revealed by simultaneous sub-epicardial and sub-endocardial optical mapping in explanted human hearts. Eur. Heart J. 36, 2390–2401 (2015).
Allessie, M. A. et al. Electropathological substrate of long-standing persistent atrial fibrillation in patients with structural heart disease: longitudinal dissociation. Circ. Arrhythm. Electrophysiol. 3, 606–615 (2010).
Lee, S. et al. Simultaneous biatrial high-density (510-512 electrodes) epicardial mapping of persistent and long-standing persistent atrial fibrillation in patients: new insights into the mechanism of its maintenance. Circulation 132, 2108–2117 (2015).
de Groot, N. M. et al. Electropathological substrate of longstanding persistent atrial fibrillation in patients with structural heart disease: epicardial breakthrough. Circulation 122, 1674–1682 (2010).
de Groot, N. et al. Direct proof of endo-epicardial asynchrony of the atrial wall during atrial fibrillation in humans. Circ. Arrhythm. Electrophysiol. 9, e003648 (2016).
Winters, J. et al. JavaCyte, a novel open-source tool for automated quantification of key hallmarks of cardiac structural remodeling. Sci. Rep. 10, 20074 (2020).
Ghosh, A. K., Rai, R., Flevaris, P. & Vaughan, D. E. Epigenetics in reactive and reparative cardiac fibrogenesis: the promise of epigenetic therapy. J. Cell Physiol. 232, 1941–1956 (2017).
Igarashi, T. et al. Connexin gene transfer preserves conduction velocity and prevents atrial fibrillation. Circulation 125, 216–225 (2012).
Zhao, J. et al. An image-based model of atrial muscular architecture: effects of structural anisotropy on electrical activation. Circ. Arrhythm. Electrophysiol. 5, 361–370 (2012).
Saliani, A., Irakoze, E. & Jacquemet, V. Simulation of diffuse and stringy fibrosis in a bilayer interconnected cable model of the left atrium. Europace 23, i169–i177 (2021).
Donal, E. et al. EACVI/EHRA expert consensus document on the role of multi-modality imaging for the evaluation of patients with atrial fibrillation. Eur. Heart J. Cardiovasc. Imaging 17, 355–383 (2016).
Lisi, M. et al. Left atrial strain by speckle tracking predicts atrial fibrosis in patients undergoing heart transplantation. Eur. Heart J. Cardiovasc. Imaging 23, 829–835 (2022).
Wessels, J. N. et al. Right atrial adaptation to precapillary pulmonary hypertension: pressure-volume, cardiomyocyte, and histological analysis. J. Am. Coll. Cardiol. 82, 704–717 (2023).
Eichenlaub, M. et al. Comparison of various late gadolinium enhancement magnetic resonance imaging methods with high-definition voltage and activation mapping for detection of atrial cardiomyopathy. Europace 24, 1102–1111 (2022).
Gunturiz-Beltran, C. et al. Quantification of right atrial fibrosis by cardiac magnetic resonance: verification of the method to standardize thresholds. Rev. Esp. Cardiol. 76, 173–182 (2023).
Farrag, N. A. et al. Assessment of left atrial fibrosis progression in canines following rapid ventricular pacing using 3D late gadolinium enhanced CMR images. PLoS ONE 17, e0269592 (2022).
Zhang, W. et al. Left atrial low voltage areas predicts recurrence of atrial fibrillation after catheter ablation: a meta-analysis. Heart Surg. Forum 27, E058–E067 (2024).
Masuda, M. et al. Prognostic impact of atrial cardiomyopathy: long-term follow-up of patients with and without low-voltage areas following atrial fibrillation ablation. Heart Rhythm. 21, 378–386 (2023).
Zahid, S. et al. Patient-derived models link re-entrant driver localization in atrial fibrillation to fibrosis spatial pattern. Cardiovasc. Res. 110, 443–454 (2016).
Goette, A. et al. Effect of atrial fibrillation on hematopoietic progenitor cells: a novel pathophysiological role of the atrial natriuretic peptide? Circulation 108, 2446–2449 (2003).
Lee, K. K. et al. Development and validation of a decision support tool for the diagnosis of acute heart failure: systematic review, meta-analysis, and modelling study. BMJ 377, e068424 (2022).
Nasab Mehrabi, E., Toupchi-Khosroshahi, V. & Athari, S. S. Relationship of atrial fibrillation and N terminal pro brain natriuretic peptide in heart failure patients. Esc. Heart Fail. 10, 3250–3257 (2023).
Mu, X. et al. N-terminal pro-B-type natriuretic peptide improves the predictive value of CHA2DS2-VASc risk score for long-term cardiovascular events in acute coronary syndrome patients with atrial fibrillation. Clin. Cardiol. 46, 810–817 (2023).
Sun, W. P., Du, X. & Chen, J. J. Biomarkers for predicting the occurrence and progression of atrial fibrillation: soluble suppression of tumorigenicity 2 protein and tissue inhibitor of matrix metalloproteinase-1. Int. J. Clin. Pract. 2022, 6926510 (2022).
Ruf, L., Bukowska, A., Gardemann, A. & Goette, A. Coagulation factor Xa has no effects on the expression of PAR1, PAR2, and PAR4 and no proinflammatory effects on HL-1 cells. Cells 12, 2849 (2023).
Bukowska, A. et al. Coagulation factor Xa induces an inflammatory signalling by activation of protease-activated receptors in human atrial tissue. Eur. J. Pharmacol. 718, 114–123 (2013).
Hennings, E. et al. Association of bone morphogenetic protein 10 and recurrent atrial fibrillation after catheter ablation. Europace 25, euad149 (2023).
Reyat, J. S. et al. Reduced left atrial cardiomyocyte PITX2 and elevated circulating BMP10 predict atrial fibrillation after ablation. JCI Insight 5, e139179 (2020).
Hijazi, Z. et al. Bone morphogenetic protein 10: a novel risk marker of ischaemic stroke in patients with atrial fibrillation. Eur. Heart J. 44, 208–218 (2023).
Hennings, E. et al. Bone morphogenetic protein 10 – a novel biomarker to predict adverse outcomes in patients with atrial fibrillation. J. Am. Heart Assoc. 12, e028255 (2023).
Winters, J. et al. Circulating BMP10 levels associate with late postoperative atrial fibrillation and left atrial endomysial fibrosis. JACC Clin. Electrophysiol. 10, 1326–1340 (2024).
McGann, C. et al. Atrial fibrillation ablation outcome is predicted by left atrial remodeling on MRI. Circ. Arrhythm. Electrophysiol. 7, 23–30 (2014).
Sohns, C. & Marrouche, N. F. Atrial fibrillation and cardiac fibrosis. Eur. Heart J. 41, 1123–1131 (2020).
Kapelios, C. J., Murrow, J. R., Nuhrenberg, T. G. & Montoro Lopez, M. N. Effect of mineralocorticoid receptor antagonists on cardiac function in patients with heart failure and preserved ejection fraction: a systematic review and meta-analysis of randomized controlled trials. Heart Fail. Rev. 24, 367–377 (2019).
Kumagai, K. et al. Effects of angiotensin II type 1 receptor antagonist on electrical and structural remodeling in atrial fibrillation. J. Am. Coll. Cardiol. 41, 2197–2204 (2003).
Li, Y. et al. Effects of cilazapril on atrial electrical, structural and functional remodeling in atrial fibrillation dogs. J. Electrocardiol. 40, 100.e1–100.e6 (2007).
Takemoto, Y. et al. Eplerenone reduces atrial fibrillation burden without preventing atrial electrical remodeling. J. Am. Coll. Cardiol. 70, 2893–2905 (2017).
Du, L. et al. Eplerenone prevents atrial fibrosis via the TGF-β signaling pathway. Cardiology 138, 55–62 (2017).
Goette, A. et al. Angiotensin II-antagonist in paroxysmal atrial fibrillation (ANTIPAF) trial. Circ. Arrhythm. Electrophysiol. 5, 43–51 (2012).
Goette, A. et al. Acute atrial tachyarrhythmia induces angiotensin II type 1 receptor-mediated oxidative stress and microvascular flow abnormalities in the ventricles. Eur. Heart J. 30, 1411–1420 (2009).
Wolke, C., Bukowska, A., Goette, A. & Lendeckel, U. Redox control of cardiac remodeling in atrial fibrillation. Biochim. Biophys. Acta 1850, 1555–1565 (2015).
Hai, Z., Wu, Y. & Ning, Z. Salidroside attenuates atrial fibrosis and atrial fibrillation vulnerability induced by angiotensin-II through inhibition of LOXL2-TGF-β1-Smad2/3 pathway. Heliyon 9, e21220 (2023).
Paw, M. et al. Hypoxia enhances anti-fibrotic properties of extracellular vesicles derived from hiPSCs via the miR302b-3p/TGFβ/SMAD2 axis. BMC Med. 21, 412 (2023).
Bukowska, A. et al. Anti-inflammatory effects of endothelin receptor blockade in left atrial tissue of spontaneously hypertensive rats. Int. J. Cardiol. Heart Vasc. 42, 101088 (2022).
Bukowska, A. et al. Activated clotting factor X mediates mitochondrial alterations and inflammatory responses via protease-activated receptor signaling in alveolar epithelial cells. Eur. J. Pharmacol. 869, 172875 (2020).
Liu, Y. et al. Differences in microRNA-29 and pro-fibrotic gene expression in mouse and human hypertrophic cardiomyopathy. Front. Cardiovasc. Med. 6, 170 (2019).
Rubis, P. et al. Twelve-month kinetics of circulating fibrosis-linked microRNAs (miR-21, miR-29, miR-30, and miR-133a) and the relationship with extracellular matrix fibrosis in dilated cardiomyopathy. Arch. Med. Sci. 18, 480–488 (2022).
Zhang, Y. et al. MiR-208b/miR-21 promotes the progression of cardiac fibrosis through the activation of the TGF-β1/Smad-3 signaling pathway: an in vitro and in vivo study. Front. Cardiovasc. Med. 9, 924629 (2022).
Shan, H. et al. Downregulation of miR-133 and miR-590 contributes to nicotine-induced atrial remodelling in canines. Cardiovasc. Res. 83, 465–472 (2009).
van den Berg, N. W. E. et al. MicroRNAs in atrial fibrillation target genes in structural remodelling. Cell Tissue Res. 394, 497–514 (2023).
Wang, J. et al. Integrated analysis of microRNA and mRNA expression profiles in the left atrium of patients with nonvalvular paroxysmal atrial fibrillation: role of miR-146b-5p in atrial fibrosis. Heart Rhythm. 12, 1018–1026 (2015).
Zhang, D., Hu, X., Henning, R. H. & Brundel, B. J. Keeping up the balance: role of HDACs in cardiac proteostasis and therapeutic implications for atrial fibrillation. Cardiovasc. Res. 109, 519–526 (2016).
Song, S. et al. EZH2 as a novel therapeutic target for atrial fibrosis and atrial fibrillation. J. Mol. Cell Cardiol. 135, 119–133 (2019).
van Ouwerkerk, A. F. et al. Epigenetic and transcriptional networks underlying atrial fibrillation. Circ. Res. 127, 34–50 (2020).
Guo, T. et al. Biomimetic nanoparticles loaded lutein functionalized by macrophage membrane for targeted amelioration pressure overload-induced cardiac fibrosis. Biomed. Pharmacother. 167, 115579 (2023).
Jung, H. N., Lee, S. Y., Lee, S., Youn, H. & Im, H. J. Lipid nanoparticles for delivery of RNA therapeutics: current status and the role of in vivo imaging. Theranostics 12, 7509–7531 (2022).
Jardin, B. & Epstein, J. A. Emerging mRNA therapies for cardiac fibrosis. Am. J. Physiol. Cell Physiol. 326, C107–C111 (2024).
Chen, Y. et al. Detailed characterization of microRNA changes in a canine heart failure model: relationship to arrhythmogenic structural remodeling. J. Mol. Cell Cardiol. 77, 113–124 (2014).
Walters, T. E. et al. Progression of atrial remodeling in patients with high-burden atrial fibrillation: implications for early ablative intervention. Heart Rhythm. 13, 331–339 (2016).
Marrouche, N. F. et al. Effect of MRI-guided fibrosis ablation vs conventional catheter ablation on atrial arrhythmia recurrence in patients with persistent atrial fibrillation: the DECAAF II randomized clinical trial. JAMA 327, 2296–2305 (2022).
Visone, R. et al. In vitro mechanical stimulation to reproduce the pathological hallmarks of human cardiac fibrosis on a beating chip and predict the efficacy of drugs and advanced therapies. Adv. Healthc. Mater. 13, e2301481 (2024).
Min, S. et al. Versatile human cardiac tissues engineered with perfusable heart extracellular microenvironment for biomedical applications. Nat. Commun. 15, 2564 (2024).
Dolber, P. C. & Spach, M. S. Structure of canine Bachmann’s bundle related to propagation of excitation. Am. J. Physiol. 257, H1446–H1457 (1989).
de Bakker, J. et al. Fractionated electrograms in dilated cardiomyopathy: origin and relation to abnormal conduction. J. Am. Coll. Cardiol. 27, 1071–1078 (1996).
Ausma, J. et al. Structural changes of atrial myocardium due to sustained atrial fibrillation in the goat. Circulation 96, 3157–3163 (1997).
Acknowledgements
The authors’ work was supported by the Netherlands Heart Foundation (CVON2014-09, RACE V (Reappraisal of Atrial Fibrillation: Interaction between hyperCoagulability, Electrical remodeling, and Vascular Destabilisation in the Progression of AF), and grant number 01-002-2022-0118, EmbRACE (Electro-Molecular Basis and the theRapeutic management of Atrial Cardiomyopathy, fibrillation and associated outcomEs)) and the European Union (ITN Network Personalize AF (Personalized Therapies for Atrial Fibrillation: a translational network), grant number 860974; CATCH ME (Characterizing Atrial fibrillation by Translating its Causes into Health Modifiers in the Elderly), grant number 633196; MAESTRIA (Machine Learning Artificial Intelligence Early Detection Stroke Atrial Fibrillation), grant number 965286; and REPAIR (Restoring cardiac mechanical function by polymeric artificial muscular tissue), grant number 952166).
Author information
Authors and Affiliations
Contributions
The authors contributed substantially to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
U.S. received consultancy fees or honoraria from Università della Svizzera Italiana (Switzerland), Roche Diagnostics, and YourRhythmics BV, a spin-off company of the Maastricht University (Netherlands); and is cofounder and shareholder of YourRhythmics BV. A.G. received speaker fees from Abbott, Astra Zeneca, Bayer Health Care, Berlin Chemie, Biotronik, Boehringer Ingelheim, BMS/Pfizer, Boston Scientific, Daiichi-Sankyo, Medtronic, Menarini, Omeicos, Sanofi-Aventis and Viofor. S.V. declares no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Na Li, David R. Van Wagoner and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Schotten, U., Goette, A. & Verheule, S. Translation of pathophysiological mechanisms of atrial fibrosis into new diagnostic and therapeutic approaches. Nat Rev Cardiol 22, 225–240 (2025). https://doi.org/10.1038/s41569-024-01088-w
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41569-024-01088-w
This article is cited by
-
Global drivers of heart failure attributable to atrial fibrillation/flutter: insights from Joinpoint regression, age-period-cohort analysis, and future projections from the GBD 2021 Study
BMC Medicine (2026)
-
Early differentiation between paroxysmal and persistent atrial fibrillation based on interpretable machine learning: a multicenter retrospective study
BioData Mining (2026)
-
Clinical and prognostic impact of left atrial strain in non-ischemic cardiomyopathies
The International Journal of Cardiovascular Imaging (2026)
-
Spectral photon-counting CT in first-pass myocardial perfusion imaging for very high-risk patients: a comparison with dual-energy CT
European Radiology Experimental (2025)
-
Exosomal miR-320c: A Novel Biomarker for Atrial Cardiomyopathy
Journal of Cardiovascular Translational Research (2025)