Abstract
Hypertension is the leading cause of death globally, primarily due to its strong association with cardiovascular disease. The global prevalence of hypertension has surged over the past three decades, driven by rising rates of diabetes mellitus and obesity. Despite current antihypertensive therapies, only a small proportion of patients with hypertension achieve adequate blood pressure control, necessitating novel therapeutic strategies. In this Review we explore the challenges and emerging opportunities in hypertension management. Aprocitentan, a dual endothelin receptor antagonist, is the first agent from a novel class of antihypertensive drug to be licensed since 2007 and exemplifies innovative treatments on the horizon. Here we also address the complex factors contributing to poor hypertension control, including genetic influences, lifestyle factors, therapeutic inertia and poor patient adherence. We discuss the limitations of existing therapies and highlight promising new pharmacological approaches to hypertension management. Integrating these novel treatments alongside current pharmaceuticals combined with improved diagnostic and management strategies could substantially reduce the global burden of hypertension and associated cardiovascular disease.
Key points
-
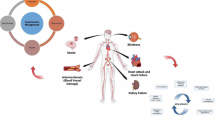
Hypertension, or high blood pressure (BP), is the leading cause of morbidity and mortality worldwide, mostly because of its well-established association with a range of cardiovascular diseases, including coronary heart disease, heart failure and stroke.
-
Although effective treatment of hypertension reduces morbidity and mortality, rates of hypertension control are low, and this deficit is likely to become an even greater issue as BP treatment targets are lowered.
-
Almost half of all patients with hypertension have not had it diagnosed, only around half of those with a diagnosis are treated and only around half of treated individuals achieve adequate BP control; therefore, improvements are needed in both the diagnosis and management of hypertension.
-
A strong evidence base supports starting therapy with two antihypertensive medicines and shows that combining different antihypertensive classes into a single-pill combination reduces BP more effectively than doubling the dose of a drug in any one class.
-
The dual endothelin receptor antagonist aprocitentan, approved for the indication of uncontrolled hypertension, was the first new class of drug to be added to the hypertension armamentarium for almost two decades and the first to target a new pathway in almost four decades.
-
New antihypertensive approaches in development include synthetic small interfering RNAs targeting angiotensinogen synthesis, aldosterone synthase inhibitors and non-steroidal mineralocorticoid inhibitors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Murray, C. J. L. et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1223–1249 (2020).
Frohlich, E. D. et al. The heart in hypertension. N. Engl. J. Med. 327, 998–1008 (1992).
Ho, J. E. et al. Predicting heart failure with preserved and reduced ejection fraction: the International Collaboration on heart failure subtypes. Circ. Heart Fail. 9, 1–9 (2016).
Feigin, V. L. et al. Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 20, 795–820 (2021).
Klag, M. J. et al. Blood pressure and end-stage renal disease in men. N. Engl. J. Med. 334, 13–18 (1996).
Iadecola, C. Hypertension and dementia. Hypertension 64, 3–5 (2014).
Non-Communicable Disease Risk Factor Collaboration. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet 398, 957–980 (2021).
Zhou, B., Perel, P., Mensah, G. A. & Ezzati, M. Global epidemiology, health burden and effective interventions for elevated blood pressure and hypertension. Nat. Rev. Cardiol. 18, 785–802 (2021).
Sun, Z. Aging, arterial stiffness, and hypertension. Hypertension 65, 252–256 (2015).
Foreman, K. J. et al. Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 392, 2052–2090 (2018).
Wierzejska, E. et al. A global perspective on the costs of hypertension: a systematic review. Arch. Med. Sci. 16, 1078–1091 (2020).
Rahimi, K. et al. Pharmacological blood pressure lowering for primary and secondary prevention of cardiovascular disease across different levels of blood pressure: an individual participant-level data meta-analysis. Lancet 397, 1625–1636 (2021).
Wright, J. T. Jr. et al. A randomized trial of intensive versus standard blood-pressure control. N. Engl. J. Med. 373, 2103–2116 (2015).
Zhang, W. et al. Trial of intensive blood-pressure control in older patients with hypertension. N. Engl. J. Med. 385, 1268–1279 (2021).
Guo, X. et al. Multifaceted intensive blood pressure control model in older and younger individuals with hypertension: a randomized clinical trial. JAMA Cardiol. 9, 781–790 (2024).
Liu, J. et al. Lowering systolic blood pressure to less than 120 mm Hg versus less than 140 mm Hg in patients with high cardiovascular risk with and without diabetes or previous stroke: an open-label, blinded-outcome, randomised trial. Lancet 404, 245–255 (2024).
Brown, M. J. Aliskiren. Circulation 118, 773–784 (2008).
Richardson, L. C., Vaughan, A. S., Wright, J. S. & Coronado, F. Examining the hypertension control cascade in adults with uncontrolled hypertension in the US. JAMA Netw. Open. 7, e2431997 (2024).
Scheltens, T., Bots, M. L., Numans, M. E., Grobbee, D. E. & Hoes, A. W. Awareness, treatment and control of hypertension: the ‘rule of halves’ in an era of risk-based treatment of hypertension. J. Hum. Hypertens. 21, 99–106 (2007).
Dale, C. E. et al. The impact of the COVID-19 pandemic on cardiovascular disease prevention and management. Nat. Med. 29, 219–225 (2023).
Stergiou, G. S. et al. 2021 European Society of Hypertension practice guidelines for office and out-of-office blood pressure measurement. J. Hypertens. 39, 1293–1302 (2021).
Validated blood pressure monitoring. Stride BP https://www.stridebp.org/bp-monitors/ (2025).
Lu, Y. et al. Quantifying blood pressure visit-to-visit variability in the real-world setting: a retrospective cohort study. Circ. Cardiovasc. Qual. 16, e009258 (2023).
Huang, Q. F. et al. Ambulatory blood pressure monitoring to diagnose and manage hypertension. Hypertension 77, 254–264 (2021).
Ali, D. H. et al. Therapeutic inertia in the management of hypertension in primary care. J. Hypertens. 39, 1238–1245 (2021).
Niiranen, T. J., Mäki, J., Puukka, P., Karanko, H. & Jula, A. M. Office, home, and ambulatory blood pressures as predictors of cardiovascular risk. Hypertension 64, 281–286 (2014).
Hayek, M. A., Giannouchos, T., Lawley, M. & Kum, H. C. Economic evaluation of blood pressure monitoring techniques in patients with hypertension: a systematic review. JAMA Netw. Open. 6, e2344372 (2023).
Roerecke, M., Kaczorowski, J. & Myers, M. G. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension: a systematic review and meta-analysis. JAMA Intern. Med. 179, 351–362 (2019).
Muntner, P. et al. Measurement of blood pressure in humans: a scientific statement from the American Heart Association. Hypertension 73, e35–e66 (2019).
Whelton, P. K., O’Connell, S., Mills, K. T. & He, J. Optimal antihypertensive systolic blood pressure: a systematic review and meta-analysis. Hypertension 81, 2329–2339 (2024).
Mach, F. et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: the task force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Eur. Heart J. 41, 111–188 (2019).
Schutte, A. E. & Jennings, G. L. R. To harmonize or to hinder… do we need two sets of European guidelines in 2024? Hypertension 82, 8–10 (2025).
McCarthy, C. P., Jackson, R., McEvoy, J. W. & Rahimi, K. From treating hypertension to lowering cardiovascular disease risk. Hypertension 81, 1655–1658 (2024).
Oparil, S. et al. Hypertension. Nat. Rev. Dis. Primers 4, 18014 (2018).
Ellison, D. H. & Welling, P. Insights into salt handling and blood pressure. N. Engl. J. Med. 385, 1981–1993 (2021).
Padmanabhan, S. & Dominiczak, A. F. Genomics of hypertension: the road to precision medicine. Nat. Rev. Cardiol. 18, 235–250 (2021).
McCallum, L. et al. UMOD genotype-blinded trial of ambulatory blood pressure response to torasemide. Hypertension 81, 2049–2059 (2024).
Padmanabhan, S., Caulfield, M. & Dominiczak, A. F. Genetic and molecular aspects of hypertension. Circ. Res. 116, 937–959 (2015).
Brown, M. J. Renin: friend or foe? Heart 93, 1026–1033 (2007).
Hypertension in adults: diagnosis and management (NG136) (National Institute for Health and Care Excellence, 2023); https://www.nice.org.uk/guidance/ng136.
Rayner, B. L. & Spence, J. D. Physiological treatment of hypertension in black patients: time for action. Circulation 143, 2367–2369 (2021).
Mancia, G. et al. 2023 ESH guidelines for the management of arterial hypertension The Task Force for the Management of Arterial hypertension of the European Society of Hypertension: endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA). J. Hypertens. 41, 1874–2071 (2023).
Whelton, P. K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Hypertension 71, 1269–1324 (2018).
Cheung, A. K. et al. KDIGO 2021 clinical practice guideline for the management of blood pressure in chronic kidney disease. Kidney Int. 99, S1–S87 (2021).
McEvoy, J. W. et al. 2024 ESC guidelines for the management of elevated blood pressure and hypertension: developed by the task force on the management of elevated blood pressure and hypertension of the European Society of Cardiology (ESC) and endorsed by the European Society of Endocrinology (ESE) and the European Stroke Organisation (ESO). Eur. Heart J. 45, 3912–4018 (2024).
Visseren, F. L. J. et al. 2021 ESC guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart. J. 42, 3227–3337 (2021).
Bailey, M. A. & Dhaun, N. Salt sensitivity: causes, consequences, and recent advances. Hypertension 81, 476–489 (2024).
Chan, R. J., Parikh, N., Ahmed, S., Ruzicka, M. & Hiremath, S. Blood pressure control should focus on more potassium: controversies in hypertension. Hypertension 81, 501–509 (2024).
Blumenthal, J. A. et al. Effects of lifestyle modification on patients with resistant hypertension: results of the TRIUMPH randomized clinical trial. Circulation 144, 1212–1226 (2021).
Okonofua, E. C. et al. Therapeutic inertia is an impediment to achieving the healthy people 2010 blood pressure control goals. Hypertension 47, 345–351 (2006).
An, D. W., Li, Y. & Staessen, J. A. Action points for implementation of the lowest well-tolerated blood pressure. Eur. J. Intern. Med. 123, 55–57 (2024).
Agarwal, R., Bills, J. E., Hecht, T. J. W. & Light, R. P. Role of home blood pressure monitoring in overcoming therapeutic inertia and improving hypertension control. Hypertension 57, 29–38 (2011).
Serumaga, B. et al. Effect of pay for performance on the management and outcomes of hypertension in the United Kingdom: interrupted time series study. Br. Med. J. 342, d108 (2011).
Josiah Willock, R. et al. Therapeutic inertia and treatment intensification. Curr. Hypertens. Rep. 20, 4 (2018).
Burnier, M. Medication adherence and persistence as the cornerstone of effective antihypertensive therapy. Am. J. Hypertens. 19, 1190–1196 (2006).
Carey, R. M., Sakhuja, S., Calhoun, D. A., Whelton, P. K. & Muntner, P. Prevalence of apparent treatment-resistant hypertension in the United States. Hypertension 73, 424–431 (2019).
Fischer, M. A. et al. Trouble getting started: predictors of primary medication nonadherence. Am. J. Med. 124, 9–22 (2011).
Hameed, M. A., Tebbit, L., Jacques, N., Thomas, M. & Dasgupta, I. Non-adherence to antihypertensive medication is very common among resistant hypertensives: results of a directly observed therapy clinic. J. Hum. Hypertens. 30, 83–89 (2016).
Mennini, F. S. et al. Cost of poor adherence to anti-hypertensive therapy in five European countries. Eur. J. Health. Econ. 16, 65–72 (2015).
Hamilton, G. A. Measuring adherence in a hypertension clinical trial. Eur. J. Cardiovasc. Nurs. 2, 219–228 (2003).
Burnier, M. & Egan, B. M. Adherence in hypertension. Circ. Res. 124, 1124–1140 (2019).
Lane, D. et al. Nonadherence in hypertension: how to develop and implement chemical adherence testing. Hypertension 79, 12–23 (2022).
Goldman, D. P., Joyce, G. F. & Zheng, Y. Prescription drug cost sharing: associations with medication and medical utilization and spending and health. JAMA 298, 61–69 (2007).
Roumie, C. L. et al. Patient centered primary care is associated with patient hypertension medication adherence. J. Behav. Med. 34, 244–253 (2011).
Choudhry, N. K. et al. Medication adherence and blood pressure control: a scientific statement from the American Heart Association. Hypertension 79, e1–e14 (2022).
MacDonald, T. M. et al. Combination therapy is superior to sequential monotherapy for the initial treatment of hypertension: a double-blind randomized controlled trial. J. Am. Heart Assoc. 6, e006986 (2017).
Alshehri, A. A. et al. Impact of the pharmacist-led intervention on the control of medical cardiovascular risk factors for the primary prevention of cardiovascular disease in general practice: a systematic review and meta-analysis of randomised controlled trials. Br. J. Clin. Pharmacol. 86, 29–38 (2020).
Kario, K., Harada, N. & Okura, A. Digital therapeutics in hypertension: evidence and perspectives. Hypertension 79, 2148–2158 (2022).
Johnson, A. G., Nguyen, T. V. & Day, R. O. Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann. Intern. Med. 121, 289–300 (1994).
Parati, G., Kjeldsen, S., Coca, A., Cushman, W. C. & Wang, J. Adherence to single-pill versus free-equivalent combination therapy in hypertension: a systematic review and meta-analysis. Hypertension 77, 692–705 (2021).
Wald, D. S., Law, M., Morris, J. K., Bestwick, J. P. & Wald, N. J. Combination therapy versus monotherapy in reducing blood pressure: meta-analysis on 11,000 participants from 42 trials. Am. J. Med. 122, 290–300 (2009).
Egan, B. M. et al. Initial monotherapy and combination therapy and hypertension control the first year. Hypertension 59, 1124–1131 (2012).
Gradman, A. H. et al. Initial combination therapy reduces the risk of cardiovascular events in hypertensive patients. Hypertension 61, 309–318 (2013).
Schmieder, R. E. et al. Improved persistence to medication, decreased cardiovascular events and reduced all-cause mortality in hypertensive patients with use of single-pill combinations: results from the START-Study. Hypertension 80, 1127–1135 (2023).
Machnicki, G., Ong, S. H., Chen, W., Wei, Z. J. & Kahler, K. H. Comparison of amlodipine/valsartan/hydrochlorothiazide single pill combination and free combination: adherence, persistence, healthcare utilization and costs. Curr. Med. Res. Opin. 31, 2287–2296 (2015).
Webster, R. et al. Fixed low-dose triple combination antihypertensive medication vs usual care for blood pressure control in patients with mild to moderate hypertension in Sri Lanka: a randomized clinical trial. JAMA 320, 566–579 (2018).
Ojji, D. B. et al. Low-dose triple-pill vs standard-care protocols for hypertension treatment in Nigeria: a randomized clinical trial. JAMA 332, 1070–1079 (2024).
Tsioufis, K., Kreutz, R., Sykara, G., van Vugt, J. & Hassan, T. Impact of single-pill combination therapy on adherence, blood pressure control, and clinical outcomes: a rapid evidence assessment of recent literature. J. Hypertens. 38, 1016–1028 (2020).
Rodgers, A. et al. Efficacy and safety of a novel low-dose triple single-pill combination compared with placebo for initial treatment of hypertension. J. Am. Coll. Cardiol. 84, 2393–2403 (2024).
Rodgers, A. et al. Efficacy and safety of a novel low-dose triple single-pill combination of telmisartan, amlodipine and indapamide, compared with dual combinations for treatment of hypertension: a randomised, double-blind, active-controlled, international clinical trial. Lancet 404, 1536–1546 (2024).
Antonopoulou, M. & Chliveros, K. A novel low-dose triple single-pill combination for hypertension. Lancet 404, 1496–1498 (2024).
Mahmud, A. & Feely, J. Low-dose quadruple antihypertensive combination. Hypertension 49, 272–275 (2007).
Chow, C. K. et al. Initial treatment with a single pill containing quadruple combination of quarter doses of blood pressure medicines versus standard dose monotherapy in patients with hypertension (QUARTET): a phase 3, randomised, double-blind, active-controlled trial. Lancet 398, 1043–1052 (2021).
Huffman, M. D. et al. Efficacy and safety of a four-drug, quarter-dose treatment for hypertension: the QUARTET USA randomized trial. Hypertens. Res. 47, 1668–1677 (2024).
Nolde, J. M. et al. Effect of initial treatment with a single pill containing quadruple combination of quarter doses of blood pressure medicines versus standard dose monotherapy in patients with hypertension on ambulatory blood pressure indices: results from the QUARTET study. Circulation 148, 375–377 (2023).
Ranasinghe, P., Addison, M. L., Dear, J. W. & Webb, D. J. Small interfering RNA: discovery, pharmacology and clinical development-an introductory review. Br. J. Pharmacol. 180, 2697–2720 (2023).
Addison, M. L., Ranasinghe, P. & Webb, D. J. Emerging insights and future prospects for therapeutic application of siRNA targeting angiotensinogen in hypertension. Expert Rev. Clin. Pharmacol. 16, 1025–1033 (2023).
Ribo announces the latest research news for three siRNA therapeutic drugs at the European Society of Cardiology Congress 2023 (Ribocure Pharmaceuticals, 2023); https://ribocure.com/ribo-announces-the-latest-research-news-for-three-sirna-therapeutic-drugs-at-the-european-society-of-cardiology-congress-2023.
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06501586 (2024).
Desai, A. S. et al. Zilebesiran, an RNA interference therapeutic agent for hypertension. N. Engl. J. Med. 389, 228–238 (2023).
Carvalho, T. RNA interference treatment targeting angiotensinogen lowers blood pressure. Nat. Med. 29, 2962–2963 (2023).
Bakris, G. L. et al. RNA interference with zilebesiran for mild to moderate hypertension: the KARDIA-1 randomized clinical trial. JAMA 331, 740–749 (2024).
KARDIA-2: novel BP-lowering drug reduces SBP with just one injection (American College of Cardiology, 2024); https://www.acc.org/Latest-in-Cardiology/Articles/2024/04/02/17/02/sun-8am-phase2-kardia2-acc-2024.
MacGregor, G. A. et al. Captopril in essential hypertension; contrasting effects of adding hydrochlorothiazide or propranolol. Br. Med. J. 284, 693–696 (1982).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06272487 (2024).
Ranasinghe, P., Addison, M. L. & Webb, D. J. Small interfering RNA therapeutics in hypertension: a viewpoint on vasopressor and vasopressor-sparing strategies for counteracting blood pressure lowering by angiotensinogen-targeting small interfering RNA. J. Am. Heart Assoc. 11, e027694 (2022).
Uijl, E. et al. Conventional vasopressor and vasopressor-sparing strategies to counteract the blood pressure-lowering effect of small interfering RNA targeting angiotensinogen. J. Am. Heart Assoc. 11, e026426 (2022).
Ye, D. et al. Counteracting angiotensinogen small-interfering RNA-mediated antihypertensive effects with REVERSIR. Hypertension 81, 1491–1499 (2024).
Morgan, E. S. et al. Antisense inhibition of angiotensinogen with IONIS-AGT-L(Rx): results of phase 1 and phase 2 studies. JACC Basic Transl. Sci. 6, 485–496 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04714320 (2023).
Uijl, E. et al. Strong and sustained antihypertensive effect of small interfering RNA targeting liver angiotensinogen. Hypertension 73, 1249–1257 (2019).
Staessen, J., Lijnen, P., Fagard, R., Verschueren, L. J. & Amery, A. Rise in plasma concentration of aldosterone during long-term angiotensin II suppression. J. Endocrinol. 91, 457–465 (1981).
Calhoun, D. A. et al. Effects of a novel aldosterone synthase inhibitor for treatment of primary hypertension: results of a randomized, double-blind, placebo- and active-controlled phase 2 trial. Circulation 124, 1945–1955 (2011).
Brixius-Anderko, S. & Scott, E. E. Aldosterone synthase structure with cushing disease drug LCI699 highlights avenues for selective CYP11B drug design. Hypertension 78, 751–759 (2021).
Freeman, M. W. et al. Phase 2 trial of baxdrostat for treatment-resistant hypertension. N. Engl. J. Med. 388, 395–405 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06034743 (2025).
Laffin, L. J. et al. Aldosterone synthase inhibition with lorundrostat for uncontrolled hypertension: the target-HTN randomized clinical trial. JAMA 330, 1140–1150 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05769608 (2024).
Mineralys Therapeutics completes target enrollment in pivotal advance-HTN Trial of lorundrostat for the treatment of hypertension (Mineralys, 2024); https://ir.mineralystx.com/news-events/press-releases/detail/49/mineralys-therapeutics-completes-target-enrollment-in.
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06153693 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05968430 (2025).
Romagnani, P. et al. Chronic kidney disease. Nat. Rev. Dis. Primers 3, 17088 (2017).
Burnier, M. & Damianaki, A. Hypertension as cardiovascular risk factor in chronic kidney disease. Circ. Res. 132, 1050–1063 (2023).
Peralta, C. A. et al. Control of hypertension in adults with chronic kidney disease in the United States. Hypertension 45, 1119–1124 (2005).
Tuttle, K. R. et al. Efficacy and safety of aldosterone synthase inhibition with and without empagliflozin for chronic kidney disease: a randomised, controlled, phase 2 trial. Lancet 403, 379–390 (2024).
Tsai, W. C. et al. Cardiovascular and renal efficacy and safety of sodium-glucose cotransporter-2 inhibitors in patients without diabetes: a systematic review and meta-analysis of randomised placebo-controlled trials. BMJ Open 12, e060655 (2022).
Good, D. W. Nongenomic actions of aldosterone on the renal tubule. Hypertension 49, 728–739 (2007).
Heidenreich, P. A. et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation 145, e895–e1032 (2022).
Williams, B. et al. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet 386, 2059–2068 (2015).
Williams, B. et al. Endocrine and haemodynamic changes in resistant hypertension, and blood pressure responses to spironolactone or amiloride: the PATHWAY-2 mechanisms substudies. Lancet Diabetes Endocrinol. 6, 464–475 (2018).
Hobbs, F. D. R. et al. Low-dose spironolactone and cardiovascular outcomes in moderate stage chronic kidney disease: a randomized controlled trial. Nat. Med. 30, 3634–3645 (2024).
Kolkhof, P. & Bärfacker, L. 30 Years of the mineralocorticoid receptor: mineralocorticoid receptor antagonists: 60 years of research and development. J. Endocrinol. 234, 125–140 (2017).
Azizi, M. Decreasing the effects of aldosterone in resistant hypertension — a success story. N. Engl. J. Med. 388, 461–463 (2023).
Brown, N. J. Eplerenone. Circulation 107, 2512–2518 (2003).
Parthasarathy, H. K. et al. A double-blind, randomized study comparing the antihypertensive effect of eplerenone and spironolactone in patients with hypertension and evidence of primary aldosteronism. J. Hypertens. 29, 980–990 (2011).
Blazek, O. & Bakris, G. L. A review of novel endothelin antagonists and overview of non-steroidal mineralocorticoid antagonists for treating resistant hypertension: an update. Eur. J. Pharmacol. 979, 176752 (2024).
Jhund, P. S. et al. Mineralocorticoid receptor antagonists in heart failure: an individual patient level meta-analysis. Lancet 404, 1119–1131 (2024).
Ito, S. et al. Double-blind randomized phase 3 study comparing esaxerenone (CS-3150) and eplerenone in patients with essential hypertension (ESAX-HTN Study). Hypertension 75, 51–58 (2020).
Rakugi, H., Ito, S., Itoh, H., Okuda, Y. & Yamakawa, S. Long-term phase 3 study of esaxerenone as mono or combination therapy with other antihypertensive drugs in patients with essential hypertension. Hypertens. Res. 42, 1932–1941 (2019).
Pitt, B. et al. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N. Engl. J. Med. 385, 2252–2263 (2021).
Bakris, G. L. et al. Effect of finerenone on chronic fidney disease outcomes in type 2 diabetes. N. Engl. J. Med. 383, 2219–2229 (2020).
Bakris, G. L. et al. Effect of finerenone on albuminuria in patients with diabetic nephropathy: a randomized clinical trial. JAMA 314, 884–894 (2015).
Kintscher, U., Bakris, G. L. & Kolkhof, P. Novel non-steroidal mineralocorticoid receptor antagonists in cardiorenal disease. Br. J. Pharmacol. 179, 3220–3234 (2022).
Bakris, G. et al. Effect of KBP-5074 on blood pressure in advanced chronic kidney disease: results of the BLOCK-CKD Study. Hypertension 78, 74–81 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04968184 (2024).
Novo Nordisk stops the ocedurenone CLARION-CKD trial and recognises impairment loss (Novo Nordisk, 2024); https://www.novonordisk.com/news-and-media/news-and-ir-materials/news-details.html?id=168529.
Kario, K. & Williams, B. Angiotensin receptor–neprilysin inhibitors for hypertension — hemodynamic effects and relevance to hypertensive heart disease. Hypertens. Res. 45, 1097–1110 (2022).
Gu, J. et al. Pharmacokinetics and pharmacodynamics of LCZ696, a novel dual-acting angiotensin receptor-neprilysin inhibitor (ARNi). J. Clin. Pharmacol. 50, 401–414 (2010).
Ferro, C. J., Spratt, J. C., Haynes, W. G. & Webb, D. J. Inhibition of neutral endopeptidase causes vasoconstriction of human resistance vessels in vivo. Circulation 97, 2323–2330 (1998).
Kostis, J. B. et al. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial. Am. J. Hypertens. 17, 103–111 (2004).
Chen, J. et al. Effect of sacubitril/valsartan or valsartan on ventricular remodeling and myocardial fibrosis in perimenopausal women with hypertension. J. Hypertens. 41, 1077–1083 (2023).
Rakugi, H. et al. Efficacy of sacubitril/valsartan versus olmesartan in Japanese patients with essential hypertension: a randomized, double-blind, multicenter study. Hypertens. Res. 45, 824–833 (2022).
McMurray, J. J. et al. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 371, 993–1004 (2014).
Böhm, M. et al. Systolic blood pressure, cardiovascular outcomes and efficacy and safety of sacubitril/valsartan (LCZ696) in patients with chronic heart failure and reduced ejection fraction: results from PARADIGM-HF. Eur. Heart J. 38, 1132–1143 (2017).
Kario, K. et al. Twenty‐four‐hour blood pressure‐lowering efficacy of sacubitril/valsartan versus olmesartan in Japanese patients with essential hypertension based on nocturnal blood pressure dipping status: a post hoc analysis of data from a randomized, double‐blind multicenter study. J. Am. Heart Assoc. 12, e027612 (2023).
Kario, K. et al. Efficacy and safety of LCZ696, a first-in-class angiotensin receptor neprilysin inhibitor, in Asian patients with hypertension. Hypertension 63, 698–705 (2014).
Yamamoto, K. & Rakugi, H. Angiotensin receptor-neprilysin inhibitors: comprehensive review and implications in hypertension treatment. Hypertens. Res. 44, 1239–1250 (2021).
Lin, D. S. et al. Angiotensin receptor neprilysin inhibitor as a novel antihypertensive drug: evidence from Asia and around the globe. J. Clin. Hypertens. 23, 556–567 (2021).
Kario, K. et al. Seven-action approaches for the management of hypertension in Asia — the HOPE Asia network. J. Clin. Hypertens. 24, 213–223 (2022).
Wang, T. D. et al. 2022 Guidelines of the Taiwan society of cardiology and the Taiwan hypertension society for the management of hypertension. Acta Cardiol. Sin. 38, 225–325 (2022).
Goetze, J. P. et al. Cardiac natriuretic peptides. Nat. Rev. Cardiol. 17, 698–717 (2020).
Dunn, M. E. et al. Agonist antibody to guanylate cyclase receptor NPR1 regulates vascular tone. Nature 633, 654–661 (2024).
Yanagisawa, M. et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332, 411–415 (1988).
Dhaun, N. & Webb, D. J. Endothelins in cardiovascular biology and therapeutics. Nat. Rev. Cardiol. 16, 491–502 (2019).
Krum, H., Viskoper, R. J., Lacourciere, Y., Budde, M. & Charlon, V. The effect of an endothelin-receptor antagonist, bosentan, on blood pressure in patients with essential hypertension. N. Engl. J. Med. 338, 784–791 (1998).
Roumen, N., Egon, P. & Siegfried, E. Darusentan: an effective endothelin A receptor antagonist for treatment of hypertension. Am. J. Hypertens. 15, 583–589 (2002).
Weber, M. A. et al. A selective endothelin-receptor antagonist to reduce blood pressure in patients with treatment-resistant hypertension: a randomised, double-blind, placebo-controlled trial. Lancet 374, 1423–1431 (2009).
Bakris, G. L. et al. Divergent results using clinic and ambulatory blood pressures: report of a darusentan-resistant hypertension trial. Hypertension 56, 824–830 (2010).
Webb, D. J. DORADO: opportunity postponed: lessons from studies of endothelin receptor antagonists in treatment-resistant hypertension. Hypertension 56, 806–807 (2010).
US Food and Drug Administration. Highlights of prescribing information tryvio (aprocitentan) (FDA, 2024); https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/217686s000lbl.pdf.
Schlaich, M. P. et al. Dual endothelin antagonist aprocitentan for resistant hypertension (PRECISION): a multicentre, blinded, randomised, parallel-group, phase 3 trial. Lancet 400, 1927–1937 (2022).
Heerspink, H. J. L. et al. Atrasentan and renal events in patients with type 2 diabetes and chronic kidney disease (SONAR): a double-blind, randomised, placebo-controlled trial. Lancet 393, 1937–1947 (2019).
Heerspink, H. J. L., Kohan, D. E. & de Zeeuw, D. New insights from SONAR indicate adding sodium glucose co-transporter 2 inhibitors to an endothelin receptor antagonist mitigates fluid retention and enhances albuminuria reduction. Kidney Int. 99, 346–349 (2021).
Dupuis, J. & Hoeper, M. M. Endothelin receptor antagonists in pulmonary arterial hypertension. Eur. Respir. J. 31, 407–415 (2008).
Wei, A. et al. Clinical adverse effects of endothelin receptor antagonists: insights from the meta-analysis of 4894 patients from 24 randomized double-blind placebo-controlled clinical trials. J. Am. Heart Assoc. 5, e003896 (2016).
Oliver, J. J., Melville, V. P. & Webb, D. J. Effect of regular phosphodiesterase type 5 inhibition in hypertension. Hypertension 48, 622–627 (2006).
Wolk, R. et al. Blood pressure lowering effects of a new long-acting inhibitor of phosphodiesterase 5 in patients with mild to moderate hypertension. Hypertension 53, 1091–1097 (2009).
Kloner, R. A., Brown, M., Prisant, L. M. & Collins, M. Effect of sildenafil in patients with erectile dysfunction taking antihypertensive therapy. Sildenafil Study Group. Am. J. Hypertens. 14, 70–73 (2001).
Kloner, R. A., Mitchell, M. & Emmick, J. T. Cardiovascular effects of tadalafil in patients on common antihypertensive therapies. Am. J. Cardiol. 92, 47–57 (2003).
Pickering, T. G. et al. Sildenafil citrate for erectile dysfunction in men receiving multiple antihypertensive agents: a randomized controlled trial. Am. J. Hypertens. 17, 1135–1142 (2004).
Nehra, A. Erectile dysfunction and cardiovascular disease: efficacy and safety of phosphodiesterase type 5 inhibitors in men with both conditions. Mayo Clin. Proc. 84, 139–148 (2009).
Schwartz, B. G. & Kloner, R. A. Drug interactions with phosphodiesterase-5 inhibitors used for the treatment of erectile dysfunction or pulmonary hypertension. Circulation 122, 88–95 (2010).
Oliver, J. J., Dear, J. W. & Webb, D. J. Clinical potential of combined organic nitrate and phosphodiesterase type 5 inhibitor in treatment-resistant hypertension. Hypertension 56, 62–67 (2010).
Ali, Q., Patel, S. & Hussain, T. Angiotensin AT2 receptor agonist prevents salt-sensitive hypertension in obese Zucker rats. Am. J. Physiol. Ren. Physiol. 308, 1379–1385 (2015).
Hanrahan, J. P. et al. A randomized, placebo-controlled, multiple-ascending-dose study to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of the soluble guanylate cyclase stimulator praliciguat in healthy subjects. Clin. Pharmacol. Drug Dev. 8, 564–575 (2019).
Hanrahan, J. P. et al. An exploratory, randomised, placebo-controlled, 14 day trial of the soluble guanylate cyclase stimulator praliciguat in participants with type 2 diabetes and hypertension. Diabetologia 63, 733–743 (2020).
Ferdinand, K. C. et al. Efficacy and safety of firibastat, a first-in-class brain aminopeptidase A inhibitor, in hypertensive overweight patients of multiple ethnic origins. Circulation 140, 138–146 (2019).
First-in-class aminopeptidase-a inhibitor fails to reduce treatment-resistant hypertension (American Heart Association, 2022); https://sessions.hub.heart.org/aha-22/article/22539144/firstinclass-aminopeptidasea-inhibitor-fails-to-reduce-treatmentresistant-hypertension.
Schaffer, R. Firibistat fails to improve BP in resistant hypertension: FRESH (Healio, 2022); https://www.healio.com/news/cardiology/20221111/firibastat-fails-to-improve-bp-in-resistant-hypertension-fresh.
Lewington, S., Clarke, R., Qizilbash, N., Peto, R. & Collins, R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 360, 1903–1913 (2002).
Rovin, B. H. et al. Efficacy and safety of sparsentan versus irbesartan in patients with IgA nephropathy (PROTECT): 2-year results from a randomised, active-controlled, phase 3 trial. Lancet 402, 2077–2090 (2023).
Bakris, G. L. et al. Divergent results using clinic and ambulatory blood pressures. Hypertension 56, 824–830 (2010).
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811 (1998).
Bartel, D. P. Metazoan microRNAs. Cell 173, 20–51 (2018).
Svoboda, P. Key mechanistic principles and considerations concerning RNA interference. Front. Plant. Sci. 11, 1237 (2020).
Touyz, R. M. Silencing angiotensinogen in hypertension. N. Engl. J. Med. 389, 278–281 (2023).
Bogman, K. et al. Preclinical and early clinical profile of a highly selective and potent oral inhibitor of aldosterone synthase (CYP11B2). Hypertension 69, 189–196 (2017).
Lenzini, L., Zanotti, G., Bonchio, M. & Rossi, G. P. Aldosterone synthase inhibitors for cardiovascular diseases: a comprehensive review of preclinical, clinical and in silico data. Pharmacol. Res. 163, 105332 (2021).
Lundberg, J. O., Gladwin, M. T. & Weitzberg, E. Strategies to increase nitric oxide signalling in cardiovascular disease. Nat. Rev. Drug Discov. 14, 623–641 (2015).
Sandner, P., Becker-Pelster, E. M. & Stasch, J. P. Discovery and development of sGC stimulators for the treatment of pulmonary hypertension and rare diseases. Nitric Oxide 77, 88–95 (2018).
Weber, M. A. et al. Clinical practice guidelines for the management of hypertension in the community: a statement by the American Society of Hypertension and the International Society of Hypertension. J. Clin. Hypertens. 16, 14–26 (2014).
Whelton, P. K. et al. Harmonization of the American College of Cardiology/American Heart Association and European Society of Cardiology/European Society of Hypertension blood pressure/hypertension guidelines: comparisons, reflections, and recommendations. Circulation 146, 868–877 (2022).
Carey, R. M. et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension 72, e53–e90 (2018).
Sim, J. J. et al. Comparative risk of renal, cardiovascular, and mortality outcomes in controlled, uncontrolled resistant, and nonresistant hypertension. Kidney Int. 88, 622–632 (2015).
Daugherty, S. L. et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation 125, 1635–1642 (2012).
Acelajado, M. C., Hughes, Z. H., Oparil, S. & Calhoun, D. A. Treatment of resistant and refractory hypertension. Circ. Res. 124, 1061–1070 (2019).
Cohen, J. B. et al. Cardiovascular events and mortality in white coat hypertension: a systematic review and meta-analysis. Ann. Intern. Med. 170, 853–862 (2019).
Huang, J.-F. et al. Efficacy of antihypertensive treatment for target organ protection in patients with masked hypertension (ANTI-MASK): a multicentre, double-blind, placebo-controlled trial. eClinicalMedicine 74, 102736 (2024).
Kario, K., Thijs, L. & Staessen, J. A. Blood pressure measurement and treatment decisions. Circ. Res. 124, 990–1008 (2019).
Author information
Authors and Affiliations
Contributions
The authors contributed substantially to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
D.J.W. has been involved in the development of ERAs over many years and was involved in the pivotal licensing study for aprocitentan as well as the first-in-human studies with zilebesiran, but did not receive personal funding for this work. D.J.W. has provided consultancy in the field of hypertension research for Johnson & Johnson, Nugerontix and Silence Therapeutics. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Josep Redon, Jan Staessen and Ji-Guang Wang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
We used accumulated knowledge and references gathered over more than 65 years of combined experience in the field of hypertension research and clinical practice. References were also identified through PubMed searches and from relevant articles, guidelines and conference news announcements. PubMed search terms included ‘hypertension’, ‘treatment resistant hypertension’ and ‘uncontrolled hypertension’, combined with the individual drug class names. Articles published in English between 1981 and September 2024 were considered and the most relevant articles were included in the manuscript.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sayer, M., Webb, D.J. & Dhaun, N. Novel pharmacological approaches to lowering blood pressure and managing hypertension. Nat Rev Cardiol 22, 649–663 (2025). https://doi.org/10.1038/s41569-025-01131-4
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41569-025-01131-4
This article is cited by
-
An online machine learning model for predicting medication adherence in hypertensive patients: data from the China health and retirement longitudinal study (CHARLS)
International Journal of Clinical Pharmacy (2026)
-
Hypertension in 2025: are we ready for bold precision public health approaches worldwide?
Nature Reviews Cardiology (2025)