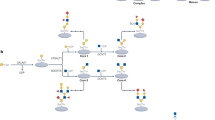
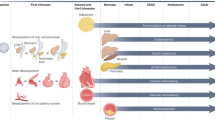
Abstract
Glycogen storage diseases are rare conditions affecting both sexes that are caused by inherited deficiencies of enzymes involved either in glycogen synthesis or breakdown, or in glycolysis. The liver and skeletal muscle are usually the most affected tissues. However, because glycogen has an important role in cardiac development and function, several glycogen storage diseases are associated, at least indirectly, with cardiac disorders, some of which have severe consequences from the first months of life. Early identification of these conditions is, therefore, an important issue, and implementation of strategies to prevent fatal outcomes due to cardiovascular disease is vital. In this Review, we discuss the pathophysiological mechanisms and the preclinical, clinical and epidemiological evidence for cardiovascular involvement in various glycogen storage diseases. We also describe interventions that can help preserve heart function, including changes in nutrition and exercise, as well as the few available molecular therapies to address the underlying metabolic anomalies.
Key points
-
Glycogen is needed for normal cardiac development and metabolism, but the precise expression and role of the many enzymes involved in glycogen metabolism remain to be elucidated.
-
Glycogen storage diseases (GSDs) are rare genetic conditions affecting both sexes and are caused by deficiencies of enzymes involved either in glycogen synthesis or breakdown, or in glycolysis.
-
Many GSDs are directly or indirectly associated with an increased risk of cardiovascular disease, with a broad spectrum of clinical presentations and outcomes.
-
Some GSDs manifest early in life with stereotypical features, whereas others present later and can be confused with other disorders; cardiologists should be vigilant for ‘red flags’ suggesting undiagnosed GSDs.
-
Supportive cardiovascular care, ranging from routine medication to heart transplantation, is required in patients with GSDI; enzyme replacement therapy (which is available only for GSDII) can improve cardiac outcomes.
-
Novel therapies are needed to improve cardiac outcomes in GSDs; gene therapies addressing underlying genetic abnormalities have shown promise in early-phase clinical trials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gumus, E. & Ozen, H. Glycogen storage diseases: an update. World J. Gastroenterol. 29, 3932–3963 (2023).
Pederson, B. A. et al. Abnormal cardiac development in the absence of heart glycogen. Mol. Cell Biol. 24, 7179–7187 (2004).
ESC Working Group on Myocardial and Pericardial Diseases. Diagnosis and management of cardiac manifestations in Anderson Fabry disease and glycogen storage diseases. European Society of Cardiology https://www.escardio.org/static-file/Escardio/Subspecialty/Working%20Groups/Myocardial%20and%20Pericardial%20Diseases/y.%20Documents/Booklet-WG-Diseases-Fabry&Glycogen.pdf (2025).
Sukigara, S. et al. Muscle glycogen storage disease 0 presenting recurrent syncope with weakness and myalgia. Neuromuscul. Disord. 22, 162–165 (2012).
Kollberg, G. et al. Cardiomyopathy and exercise intolerance in muscle glycogen storage disease 0. N. Engl. J. Med. 357, 1507–1514 (2007).
Cameron, J. M. et al. Identification of a novel mutation in GYS1 (muscle-specific glycogen synthase) resulting in sudden cardiac death, that is diagnosable from skin fibroblasts. Mol. Genet. Metab. 98, 378–382 (2009).
Musumeci, O. et al. A new phenotype of muscle glycogen synthase deficiency (GSD0B) characterized by an adult onset myopathy without cardiomyopathy. Neuromuscul. Disord. 32, 582–589 (2022).
Kishnani, P. S. et al. Diagnosis and management of glycogen storage disease type I: a practice guideline of the American College of Medical Genetics and Genomics. Genet. Med. 16, e1 (2014).
Bali D. S. et al. Glycogen Storage Disease Type I (eds Adam M. P. et al.) www.ncbi.nlm.nih.gov/books/NBK1312/ (GeneReviews, 2021).
Yiu, W. H. et al. Angiotensin mediates renal fibrosis in the nephropathy of glycogen storage disease type Ia. Kidney Int. 73, 716–723 (2008).
Jonas, A. J., Verani, R. R., Howell, R. R. & Conley, S. B. Hypertension in a child with type IA glycogen storage disease. Am. J. Kidney Dis. 11, 264–266 (1988).
Bhowmik, E., Ghosh, M., Sabui, T. K. & Mondal, R. Glycogen storage disease type I presenting with hypertension during infancy. Indian J. Pediatr. 82, 767 (2015).
Chen, Y. T., Coleman, R. A., Scheinman, J. I., Kolbeck, P. C. & Sidbury, J. B. Renal disease in type I glycogen storage disease. N. Engl. J. Med. 318, 7–11 (1988).
Rake, J. P. et al. Glycogen storage disease type I: diagnosis, management, clinical course and outcome. results of the European Study on Glycogen Storage Disease type I (ESGSD I). Eur. J. Pediatr. 161, S20–S34 (2002).
Hamaoka, K., Nakagawa, M., Furukawa, N. & Sawada, T. Pulmonary hypertension in type I glycogen storage disease. Pediatr. Cardiol. 11, 54–56 (1990).
Humbert, M. et al. Pulmonary arterial hypertension and type-I glycogen-storage disease: the serotonin hypothesis. Eur. Respir. J. 20, 59–65 (2002).
Pizzo, C. J. Type I glycogen storage disease with focal nodular hyperplasia of the liver and vasoconstrictive pulmonary hypertension. Pediatrics 65, 341–343 (1980).
Furukawa, N. et al. Type I glycogen storage disease with vasoconstrictive pulmonary hypertension. J. Inherit. Metab. Dis. 13, 102–107 (1990).
Ohura, T. et al. Progressive pulmonary hypertension: a fatal complication of type I glycogen storage disease. J. Inherit. Metab. Dis. 18, 361–362 (1995).
Bolz, D., Stocker, F. & Zimmermann, A. Pulmonary vascular disease in a child with atrial septal defect of the secundum type and type I glycogen storage disease. Pediatr. Cardiol. 17, 265–267 (1996).
Kishnani, P., Bengur, A. R. & Chen, Y. T. Pulmonary hypertension in glycogen storage disease type I. J. Inherit. Metab. Dis. 19, 213–216 (1996).
Kim, Y. M. et al. Predominance of the c.648G>T G6PC gene mutation and late complications in Korean patients with glycogen storage disease type Ia. Orphanet J. Rare Dis. 15, 45 (2020).
Ueno, M., Murakami, T., Takeda, A. & Kubota, M. Efficacy of oral sildenafil in a beraprost-treated patient with severe pulmonary hypertension secondary to type I glycogen storage disease. Circ. J. 73, 1965–1968 (2009).
Torok, R. D. et al. Pulmonary arterial hypertension in glycogen storage disease type I. J. Inborn Errors Metab. Screen. 5, 2326409817707773 (2017).
Tavil, B., Cetin, M. & Gumruk, F. Sea-blue histiocytes in the bone marrow of a boy with severe congenital neutropenia associated with G6PC3 mutation. Br. J. Haematol. 165, 426 (2014).
Weinstein, D. A. et al. Safety and efficacy of DTX401, an AAV8-mediated liver-directed gene therapy, in adults with glycogen storage disease type I a (GSDIa). J. Inherit. Metab. Dis. 48, e70014 (2025).
Dornelles, A. D. et al. Efficacy and safety of enzyme replacement therapy with alglucosidase alfa for the treatment of patients with infantile-onset Pompe disease: a systematic review and metanalysis. Front. Pediatr. 12, 1310317 (2024).
Levine, J. C., Kishnani, P. S., Chen, Y. T., Herlong, J. R. & Li, J. S. Cardiac remodeling after enzyme replacement therapy with acid α-glucosidase for infants with Pompe disease. Pediatr. Cardiol. 29, 1033–1042 (2008).
Geel, T. M., McLaughlin, P. M., de Leij, L. F., Ruiters, M. H. & Niezen-Koning, K. E. Pompe disease: current state of treatment modalities and animal models. Mol. Genet. Metab. 92, 299–307 (2007).
Desai, A. K. et al. An updated management approach of Pompe disease patients with high-sustained anti-rhGAA IgG antibody titers: experience with bortezomib-based immunomodulation. Front. Immunol. 15, 1360369 (2024).
Reuser, A. J. J. et al. GAA variants and phenotypes among 1,079 patients with Pompe disease: data from the Pompe Registry. Hum. Mutat. 40, 2146–2164 (2019).
Colburn, R. & Lapidus, D. An analysis of Pompe newborn screening data: a new prevalence at birth, insight and discussion. Front. Pediatr. 11, 1221140 (2023).
Ceron-Rodriguez, M. et al. Classic infantile-onset Pompe disease with histopathological neurologic findings linked to a novel GAA gene 4 bp deletion: a case study. Mol. Genet. Genom. Med. 10, e1957 (2022).
Hamdan, M. A., El-Zoabi, B. A., Begam, M. A., Mirghani, H. M. & Almalik, M. H. Antenatal diagnosis of Pompe disease by fetal echocardiography: impact on outcome after early initiation of enzyme replacement therapy. J. Inherit. Metab. Dis. 33, S333–S339 (2010).
Cohen, J. L. et al. In utero enzyme-replacement therapy for infantile-onset Pompe’s disease. N. Engl. J. Med. 387, 2150–2158 (2022).
Saccconi, J. Y. et al. CRISPR-Cas9 generated Pompe knock-in murine model exhibits early-onset hypertrophic cardiomyopathy and skeletal muscle weakness. Sci. Rep. 10, 10321 (2020).
Belfiore, M. P. et al. Aortopathies in mouse models of Pompe, Fabry and mucopolysaccharidosis IIIB lysosomal storage diseases. PLoS ONE 15, e0233050 (2020).
Huang, W. et al. Mitochondrial dysfunction is associated with hypertrophic cardiomyopathy in Pompe disease-specific induced pluripotent stem cell-derived cardiomyocytes. Cell Prolif. 57, e13573 (2024).
Lim, J. A., Li, L., Kakhlon, O., Myerowitz, R. & Raben, N. Defects in calcium homeostasis and mitochondria can be reversed in Pompe disease. Autophagy 11, 385–402 (2015).
van den Hout, H. M. et al. The natural course of infantile Pompe’s disease: 20 original cases compared with 133 cases from the literature. Pediatrics 112, 332–340 (2003).
Bharati, S. et al. The conduction system in Pompe’s disease. Pediatr. Cardiol. 2, 25–32 (1982).
Hagemans, M. L. et al. Clinical manifestation and natural course of late-onset Pompe’s disease in 54 Dutch patients. Brain 128, 671–677 (2005).
Mori, M. et al. Severe cardiomyopathy as the isolated presenting feature in an adult with late-onset Pompe disease: a case report. JIMD Rep. 31, 79–83 (2017).
Soliman, O. I. et al. Cardiac involvement in adults with Pompe disease. J. Intern. Med. 264, 333–339 (2008).
Boentert, M., Florian, A., Drager, B., Young, P. & Yilmaz, A. Pattern and prognostic value of cardiac involvement in patients with late-onset Pompe disease: a comprehensive cardiovascular magnetic resonance approach. J. Cardiovasc. Magn. Reson. 18, 91 (2016).
Sacconi, S. et al. Atrio-ventricular block requiring pacemaker in patients with late onset Pompe disease. Neuromuscul. Disord. 24, 648–650 (2014).
Morris, D. A. et al. Structural and functional cardiac analyses using modern and sensitive myocardial techniques in adult Pompe disease. Int. J. Cardiovasc. Imaging 31, 947–956 (2015).
Fayssoil, A., Nardi, O., Annane, D. & Orlikowski, D. Right ventricular function in late-onset Pompe disease. J. Clin. Monit. Comput. 28, 419–421 (2014).
van Kooten, H. A. et al. Cardiovascular disease in non-classic Pompe disease: a systematic review. Neuromuscul. Disord. 31, 79–90 (2021).
Hobson-Webb, L. D. et al. Autopsy findings in late-onset Pompe disease: a case report and systematic review of the literature. Mol. Genet. Metab. 106, 462–469 (2012).
van der Walt, J. D., Swash, M., Leake, J. & Cox, E. L. The pattern of involvement of adult-onset acid maltase deficiency at autopsy. Muscle Nerve 10, 272–281 (1987).
Hensel, O. et al. Morphology and function of cerebral arteries in adults with pompe disease. JIMD Rep. 20, 27–33 (2015).
Goeber, V., Banz, Y., Kaeberich, A. & Carrel, T. Huge aneurysm of the ascending aorta in a patient with adult-type Pompe’s disease: histological findings mimicking fibrillinopathy. Eur. J. Cardiothorac. Surg. 43, 193–195 (2013).
El-Gharbawy, A. H. et al. Expanding the clinical spectrum of late-onset Pompe disease: dilated arteriopathy involving the thoracic aorta, a novel vascular phenotype uncovered. Mol. Genet. Metab. 103, 362–366 (2011).
Li, H. P. et al. Pulmonary hypertension in glycogen storage disease type II. Chin. Med. J. 131, 1375–1376 (2018).
Kobayashi, H. et al. Prognostic factors for the late onset Pompe disease with enzyme replacement therapy: from our experience of 4 cases including an autopsy case. Mol. Genet. Metab. 100, 14–19 (2010).
Yang, C. F. et al. Late-onset Pompe disease with left-sided bronchomalacia. Respir. Care 60, e26–e29 (2015).
Kishnani, P. S. et al. Pompe disease diagnosis and management guideline. Genet. Med. 8, 267–288 (2006).
Tarnopolsky, M. et al. Pompe disease: diagnosis and management. Evidence-based guidelines from a Canadian expert panel. Can. J. Neurol. Sci. 43, 472–485 (2016).
van der Ploeg, A. T. et al. European consensus for starting and stopping enzyme replacement therapy in adult patients with Pompe disease: a 10-year experience. Eur. J. Neurol. 24, 768–e31 (2017).
Parenti, G. et al. The European Reference Network for Metabolic Diseases (MetabERN) clinical pathway recommendations for Pompe disease (acid maltase deficiency, glycogen storage disease type II). Orphanet J. Rare Dis. 19, 408 (2024).
Schoser, B. et al. Start, switch and stop (triple-S) criteria for enzyme replacement therapy of late-onset Pompe disease: European Pompe Consortium recommendation update 2024. Eur. J. Neurol. 31, e16383 (2024).
Angelini, C. Exercise, nutrition and enzyme replacement therapy are efficacious in adult Pompe patients: report from EPOC consortium. Eur. J. Transl. Myol. 31, 9798 (2021).
Arbelo, E. et al. 2023 ESC Guidelines for the management of cardiomyopathies. Eur. Heart J. 44, 3503–3626 (2023).
Schoser, B. et al. Safety and efficacy of cipaglucosidase alfa plus miglustat versus alglucosidase alfa plus placebo in late-onset Pompe disease (PROPEL): an international, randomised, double-blind, parallel-group, phase 3 trial. Lancet Neurol. 20, 1027–1037 (2021).
Fiege, L., Duran, I. & Marquardt, T. Improved enzyme replacement therapy with cipaglucosidase alfa/miglustat in infantile Pompe disease. Pharmaceuticals 16, 1199 (2023).
Blair, H. A. Cipaglucosidase alfa: first approval. Drugs 83, 739–745 (2023).
Kishnani, P. S. et al. Safety and efficacy of avalglucosidase alfa in individuals with infantile-onset Pompe disease enrolled in the phase 2, open-label Mini-COMET study: The 6-month primary analysis report. Genet. Med. 25, 100328 (2023).
Zhu, D. et al. A multi-centre prospective study of the efficacy and safety of alglucosidase alfa in Chinese patients with infantile-onset Pompe disease. Front. Pharmacol. 13, 903488 (2022).
Mozaffar, T. et al. Efficacy of avalglucosidase alfa on forced vital capacity percent predicted in treatment-naive patients with late-onset Pompe disease: a pooled analysis of clinical trials. Mol. Genet. Metab. Rep. 40, 101109 (2024).
Dimachkie, M. M. et al. Long-term safety and efficacy of avalglucosidase alfa in patients with late-onset Pompe disease. Neurology 99, e536–e548 (2022).
Xu, S. et al. Improved efficacy of a next-generation ERT in murine Pompe disease. JCI Insight 4, e125358 (2019).
Schoser, B., Raben, N., Varfaj, F., Walzer, M. & Toscano, A. Acid α-glucosidase (GAA) activity and glycogen content in muscle biopsy specimens of patients with Pompe disease: a systematic review. Mol. Genet. Metab. Rep. 39, 101085 (2024).
McDowell, R. et al. Arrhythmias in patients receiving enzyme replacement therapy for infantile Pompe disease. Genet. Med. 10, 758–762 (2008).
Kishnani, P. S. et al. Immune response to enzyme replacement therapies in lysosomal storage diseases and the role of immune tolerance induction. Mol. Genet. Metab. 117, 66–83 (2016).
Kishnani, P. S. et al. Cross-reactive immunologic material status affects treatment outcomes in Pompe disease infants. Mol. Genet. Metab. 99, 26–33 (2010).
Desai, A. K., Baloh, C. H., Sleasman, J. W., Rosenberg, A. S. & Kishnani, P. S. Benefits of prophylactic short-course immune tolerance induction in patients with infantile Pompe disease: demonstration of long-term safety and efficacy in an expanded cohort. Front. Immunol. 11, 1727 (2020).
Kazi, Z. B. et al. Sustained immune tolerance induction in enzyme replacement therapy-treated CRIM-negative patients with infantile Pompe disease. JCI Insight 2, e94328 (2017).
Li, C. et al. Transforming the clinical outcome in CRIM-negative infantile Pompe disease identified via newborn screening: the benefits of early treatment with enzyme replacement therapy and immune tolerance induction. Genet. Med. 23, 845–855 (2021).
Banugaria, S. G. et al. Bortezomib in the rapid reduction of high sustained antibody titers in disorders treated with therapeutic protein: lessons learned from Pompe disease. Genet. Med. 15, 123–131 (2013).
Douillard-Guilloux, G. et al. Modulation of glycogen synthesis by RNA interference: towards a new therapeutic approach for glycogenosis type II. Hum. Mol. Genet. 17, 3876–3886 (2008).
Douillard-Guilloux, G. et al. Restoration of muscle functionality by genetic suppression of glycogen synthesis in a murine model of Pompe disease. Hum. Mol. Genet. 19, 684–696 (2010).
Clayton, N. P. et al. Antisense oligonucleotide-mediated suppression of muscle glycogen synthase 1 synthesis as an approach for substrate reduction therapy of Pompe disease. Mol. Ther. Nucleic Acids 3, e206 (2014).
Ullman, J. C. et al. Small-molecule inhibition of glycogen synthase 1 for the treatment of Pompe disease and other glycogen storage disorders. Sci. Transl. Med. 16, eadf1691 (2024).
Ullman, J. C. et al. First-in-human evaluation of safety, pharmacokinetics and muscle glycogen lowering of a novel glycogen synthase 1 inhibitor for the treatment of Pompe disease. Clin. Pharmacol. Ther. 116, 1580–1592 (2024).
Unnisa, Z., Yoon, J. K., Schindler, J. W., Mason, C. & van Til, N. P. Gene therapy developments for Pompe disease. Biomedicines 10, 302 (2022).
Bordoli, C., Murphy, E., Varley, I., Sharpe, G. & Hennis, P. A systematic review investigating the effectiveness of exercise training in glycogen storage diseases. Ther. Adv. Rare Dis. 3, 26330040221076497 (2022).
Fletcher, G. F. et al. Promoting physical activity and exercise: JACC Health Promotion Series. J. Am. Coll. Cardiol. 72, 1622–1639 (2018).
Lavie, C. J., Ozemek, C., Carbone, S., Katzmarzyk, P. T. & Blair, S. N. Sedentary behavior, exercise, and cardiovascular health. Circ. Res. 124, 799–815 (2019).
Myers, J. et al. Exercise capacity and mortality among men referred for exercise testing. N. Engl. J. Med. 346, 793–801 (2002).
Kokkinos, P. et al. Exercise capacity and mortality in black and white men. Circulation 117, 614–622 (2008).
Kavanagh, T. et al. Peak oxygen intake and cardiac mortality in women referred for cardiac rehabilitation. J. Am. Coll. Cardiol. 42, 2139–2143 (2003).
Ross, R. et al. Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a Scientific Statement from the American Heart Association. Circulation 134, e653–e699 (2016).
Evins, A. et al. Glycogen storage disease type III: a mixed-methods study to assess the burden of disease. Ther. Adv. Endocrinol. Metab. 15, 20420188231224233 (2024).
Hijazi, G. et al. A retrospective longitudinal study and comprehensive review of adult patients with glycogen storage disease type III. Mol. Genet. Metab. Rep. 29, 100821 (2021).
Berling, E. et al. Narrative review of glycogen storage disorder type III with a focus on neuromuscular, cardiac and therapeutic aspects. J. Inherit. Metab. Dis. 44, 521–533 (2021).
Sentner, C. P. et al. Glycogen storage disease type III: diagnosis, genotype, management, clinical course and outcome. J. Inherit. Metab. Dis. 39, 697–704 (2016).
Vertilus, S. M. et al. Echocardiographic manifestations of glycogen storage disease III: increase in wall thickness and left ventricular mass over time. Genet. Med. 12, 413–423 (2010).
Mogahed, E. A. et al. Skeletal and cardiac muscle involvement in children with glycogen storage disease type III. Eur. J. Pediatr. 174, 1545–1548 (2015).
Carvalho, J. S., Matthews, E. E., Leonard, J. V. & Deanfield, J. Cardiomyopathy of glycogen storage disease type III. Heart Vessels 8, 155–159 (1993).
Austin, S. L. et al. Cardiac pathology in glycogen storage disease type III. JIMD Rep. 6, 65–72 (2012).
Tada, H. et al. Glycogen storage disease type III associated with ventricular tachycardia. Am. Heart J. 130, 911–912 (1995).
Shen, J., Bao, Y. & Chen, Y. T. A nonsense mutation due to a single base insertion in the 3′-coding region of glycogen debranching enzyme gene associated with a severe phenotype in a patient with glycogen storage disease type IIIa. Hum. Mutat. 9, 37–40 (1997).
Miller, C. G., Alleyne, G. A. & Brooks, S. E. Gross cardiac involvement in glycogen storage disease type 3. Br. Heart J. 34, 862–864 (1972).
Akazawa, H. et al. Specific heart muscle disease associated with glycogen storage disease type III: clinical similarity to the dilated phase of hypertrophic cardiomyopathy. Eur. Heart J. 18, 532–533 (1997).
Lee, T. M., Berman-Rosenzweig, E. S., Slonim, A. E. & Chung, W. K. Two cases of pulmonary hypertension associated with type III glycogen storage disease. JIMD Rep. 1, 79–82 (2011).
Wicker, C. et al. French recommendations for the management of glycogen storage disease type III. Eur. J. Med. Res. 28, 253 (2023).
Kishnani, P. S. et al. Glycogen storage disease type III diagnosis and management guidelines. Genet. Med. 12, 446–463 (2010).
Mayorandan, S., Meyer, U., Hartmann, H. & Das, A. M. Glycogen storage disease type III: modified Atkins diet improves myopathy. Orphanet J. Rare Dis. 9, 196 (2014).
Kumru Akin, B., Ozturk Hismi, B. & Daly, A. Improvement in hypertrophic cardiomyopathy after using a high-fat, high-protein and low-carbohydrate diet in a non-adherent child with glycogen storage disease type IIIa. Mol. Genet. Metab. Rep. 32, 100904 (2022).
Marusic, T. et al. Data highlighting effects of ketogenic diet on cardiomyopathy and hepatopathy in glycogen storage disease type IIIA. Data Brief. 32, 106205 (2020).
Dagli, A. I. et al. Reversal of glycogen storage disease type IIIa-related cardiomyopathy with modification of diet. J. Inherit. Metab. Dis. 32, S103–S106 (2009).
Marusic, T. et al. Normalization of obstructive cardiomyopathy and improvement of hepatopathy on ketogenic diet in patient with glycogen storage disease (GSD) type IIIa. Mol. Genet. Metab. Rep. 24, 100628 (2020).
Brambilla, A. et al. Improvement of cardiomyopathy after high-fat diet in two siblings with glycogen storage disease type III. JIMD Rep. 17, 91–95 (2014).
Valayannopoulos, V. et al. Successful treatment of severe cardiomyopathy in glycogen storage disease type III with D,L-3-hydroxybutyrate, ketogenic and high-protein diet. Pediatr. Res. 70, 638–641 (2011).
Francini-Pesenti, F., Tresso, S. & Vitturi, N. Modified Atkins ketogenic diet improves heart and skeletal muscle function in glycogen storage disease type III. Acta Myol. 38, 17–20 (2019).
Sentner, C. P., Caliskan, K., Vletter, W. B. & Smit, G. P. Heart failure due to severe hypertrophic cardiomyopathy reversed by low calorie, high protein dietary adjustments in a glycogen storage disease type IIIa patient. JIMD Rep. 5, 13–16 (2012).
Lim, J. A., Choi, S. J., Gao, F., Kishnani, P. S. & Sun, B. A novel gene therapy approach for GSD III using an AAV vector encoding a bacterial glycogen debranching enzyme. Mol. Ther. Methods Clin. Dev. 18, 240–249 (2020).
Gardin, A. et al. A functional mini-GDE transgene corrects impairment in models of glycogen storage disease type III. J. Clin. Invest. 134, e172018 (2024).
Yi, H. et al. Correction of glycogen storage disease type III with rapamycin in a canine model. J. Mol. Med. 92, 641–650 (2014).
Pursell, N. et al. Inhibition of glycogen synthase II with RNAi prevents liver injury in mouse models of glycogen storage diseases. Mol. Ther. 26, 1771–1782 (2018).
Magoulas, P. L. & El-Hattab, A. W. Glycogen Storage Disease Type IV (eds Adam, M. P. et al.) https://www.ncbi.nlm.nih.gov/books/NBK115333/ (GeneReviews, 2019).
Wilke, M. et al. A broad characterization of glycogen storage disease IV patients: a clinical, genetic, and histopathological study. Biomedicines 11, 363 (2023).
Kiely, B. T. et al. A novel approach to characterize phenotypic variation in GSD IV: reconceptualizing the clinical continuum. Front. Genet. 13, 992406 (2022).
Lee, Y. C., Chang, C. J., Bali, D., Chen, Y. T. & Yan, Y. T. Glycogen-branching enzyme deficiency leads to abnormal cardiac development: novel insights into glycogen storage disease IV. Hum. Mol. Genet. 20, 455–465 (2011).
Ndugga-Kabuye, M. K., Maleszewski, J., Chanprasert, S. & Smith, K. D. Glycogen storage disease type IV: dilated cardiomyopathy as the isolated initial presentation in an adult patient. BMJ Case Rep. 12, e230068 (2019).
Mochel, F. et al. Adult polyglucosan body disease: natural history and key magnetic resonance imaging findings. Ann. Neurol. 72, 433–441 (2012).
Nase, S. et al. A new variant of type IV glycogenosis with primary cardiac manifestation and complete branching enzyme deficiency. In vivo detection by heart muscle biopsy. Eur. Heart J. 16, 1698–1704 (1995).
Schroder, J. M., May, R., Shin, Y. S., Sigmund, M. & Nase-Huppmeier, S. Juvenile hereditary polyglucosan body disease with complete branching enzyme deficiency (type IV glycogenosis). Acta Neuropathol. 85, 419–430 (1993).
Bao, Y., Kishnani, P., Wu, J. Y. & Chen, Y. T. Hepatic and neuromuscular forms of glycogen storage disease type IV caused by mutations in the same glycogen-branching enzyme gene. J. Clin. Invest. 97, 941–948 (1996).
Tang, T. T. et al. Neonatal hypotonia and cardiomyopathy secondary to type IV glycogenosis. Acta Neuropathol. 87, 531–536 (1994).
Servidei, S. et al. Severe cardiopathy in branching enzyme deficiency. J. Pediatr. 111, 51–56 (1987).
Eminoglu, T. F. et al. Multisystem involvement in a patient due to accumulation of amylopectin-like material with diminished branching enzyme activity. J. Inherit. Metab. Dis. 31, S255–S259 (2008).
Aksu, T., Colak, A. & Tufekcioglu, O. Cardiac involvement in glycogen storage disease type IV: two cases and the two ends of a spectrum. Case Rep. Med. 2012, 764286 (2012).
Szymanska, E. et al. Variable clinical presentation of glycogen storage disease type IV: from severe hepatosplenomegaly to cardiac insufficiency. Some discrepancies in genetic and biochemical abnormalities. Arch. Med. Sci. 14, 237–247 (2018).
Aquaro, G. D. et al. Diagnostic and prognostic role of late gadolinium enhancement in cardiomyopathies. Eur. Heart J. Suppl. 25, C130–C136 (2023).
Lyo, S., Miles, J., Meisner, J. & Guelfguat, M. Case report: adult-onset manifesting heterozygous glycogen storage disease type IV with dilated cardiomyopathy and absent late gadolinium enhancement on cardiac magnetic resonance imaging. Eur. Heart J. Case Rep. 4, 1–6 (2020).
Sokal, E. M. et al. Progressive cardiac failure following orthotopic liver transplantation for type IV glycogenosis. Eur. J. Pediatr. 151, 200–203 (1992).
Willot, S., Marchand, V., Rasquin, A., Alvarez, F. & Martin, S. R. Systemic progression of type IV glycogen storage disease after liver transplantation. J. Pediatr. Gastroenterol. Nutr. 51, 661–664 (2010).
Koch, R. L. et al. Diagnosis and management of glycogen storage disease type IV, including adult polyglucosan body disease: a clinical practice resource. Mol. Genet. Metab. 138, 107525 (2023).
Santalla, A. et al. Genotypic and phenotypic features of all Spanish patients with McArdle disease: a 2016 update. BMC Genom. 18, 819 (2017).
Nogales-Gadea, G. et al. McArdle disease: update of reported mutations and polymorphisms in the PYGM gene. Hum. Mutat. 36, 669–678 (2015).
Lucia, A. et al. Clinical practice guidelines for glycogen storage disease V & VII (McArdle disease and Tarui disease) from an international study group. Neuromuscul. Disord. 31, 1296–1310 (2021).
Soria-Navarro, R. et al. Cardiac arrest as a manifestation of unknown type V glycogenosis: a case report. Esc. Heart Fail. 9, 3625–3629 (2022).
Lucia, A. et al. Genotypic and phenotypic features of McArdle disease: insights from the Spanish National Registry. J. Neurol. Neurosurg. Psychiatry 83, 322–328 (2012).
Lucia, A. et al. McArdle disease: what do neurologists need to know? Nat. Clin. Pract. Neurol. 4, 568–577 (2008).
Santos-Lozano, A. et al. Exercise intolerance in McArdle disease: a role for cardiac impairment? A preliminary study in humans and mice. Med. Sci. Sports Exerc. 56, 2241–2255 (2024).
Villarreal-Salazar, M. et al. Low aerobic capacity in McArdle disease: a role for mitochondrial network impairment? Mol. Metab. 66, 101648 (2022).
Nicholls, D. P., Campbell, N. P., Stevenson, H. P. & Patterson, V. H. Angina in McArdle’s disease. Heart 76, 372–373 (1996).
Hoxhaj, D. et al. Cardiac comorbidities in McArdle disease: case report and systematic review. Neurol. Sci. 45, 4757–4765 (2024).
Ratinov, G., Baker, W. P. & Swaiman, K. F. Mcardle’s syndrome with previously unreported electrocardiographic and serum enzyme abnormalities. Ann. Intern. Med. 62, 328–334 (1965).
Moustafa, S., Patton, D. J. & Connelly, M. S. Unforeseen cardiac involvement in McArdle’s disease. Heart Lung Circ. 22, 769–771 (2013).
Jones, D. M., Lopes, L., Quinlivan, R., Elliott, P. M. & Khanji, M. Y. Cardiac manifestations of McArdle disease. Eur. Heart J. 40, 397–398 (2019).
Vissing, J. & Haller, R. G. A diagnostic cycle test for McArdle’s disease. Ann. Neurol. 54, 539–542 (2003).
Salazar-Martinez, E. et al. The second wind in McArdle patients: fitness matters. Front. Physiol. 12, 744632 (2021).
Valenzuela, P. L. et al. Dose-response effect of pre-exercise carbohydrates under muscle glycogen unavailability: insights from McArdle disease. J. Sport. Health Sci. 13, 398–408 (2024).
Vissing, J. & Haller, R. G. The effect of oral sucrose on exercise tolerance in patients with McArdle’s disease. N. Engl. J. Med. 349, 2503–2509 (2003).
Haller, R. G., Wyrick, P., Taivassalo, T. & Vissing, J. Aerobic conditioning: an effective therapy in McArdle’s disease. Ann. Neurol. 59, 922–928 (2006).
Porcelli, S., Marzorati, M., Morandi, L. & Grassi, B. Home-based aerobic exercise training improves skeletal muscle oxidative metabolism in patients with metabolic myopathies. J. Appl. Physiol. 121, 699–708 (2016).
Andersen, S. T., Haller, R. G. & Vissing, J. Effect of oral sucrose shortly before exercise on work capacity in McArdle disease. Arch. Neurol. 65, 786–789 (2008).
Garcia, M. et al. Phosphofructo-1-kinase deficiency leads to a severe cardiac and hematological disorder in addition to skeletal muscle glycogenosis. PLoS Genet. 5, e1000615 (2009).
Toscano, A. & Musumeci, O. Tarui disease and distal glycogenoses: clinical and genetic update. Acta Myol. 26, 105–107 (2007).
Finsterer, J. & Stollberger, C. Progressive mitral valve thickening and progressive muscle cramps as manifestations of glycogenosis VII (Tarui’s disease). Cardiology 110, 238–240 (2008).
Amit, R. et al. Fatal familial infantile glycogen storage disease: multisystem phosphofructokinase deficiency. Muscle Nerve 15, 455–458 (1992).
Lefeuvre, C. et al. Glycogenin-1 deficiency mimicking limb-girdle muscular dystrophy. Mol. Genet. Metab. Rep. 24, 100597 (2020).
Malfatti, E. et al. A new muscle glycogen storage disease associated with glycogenin-1 deficiency. Ann. Neurol. 76, 891–898 (2014).
Moslemi, A. R. et al. Glycogenin-1 deficiency and inactivated priming of glycogen synthesis. N. Engl. J. Med. 362, 1203–1210 (2010).
Hedberg-Oldfors, C. et al. Cardiomyopathy as presenting sign of glycogenin-1 deficiency-report of three cases and review of the literature. J. Inherit. Metab. Dis. 40, 139–149 (2017).
Mancheno, N. et al. Cardiac phenotype in glycogen storage disease type XV: a rare cardiomyopathy to bear in mind. Rev. Esp. Cardiol. 74, 99–101 (2021).
Testoni, G. et al. Lack of glycogenin causes glycogen accumulation and muscle function impairment. Cell Metab. 26, 256–266.e4 (2017).
Gollob, M. H., Green, M. S., Tang, A. S. & Roberts, R. PRKAG2 cardiac syndrome: familial ventricular preexcitation, conduction system disease, and cardiac hypertrophy. Curr. Opin. Cardiol. 17, 229–234 (2002).
MacRae, C. A. et al. Familial hypertrophic cardiomyopathy with Wolff–Parkinson–White syndrome maps to a locus on chromosome 7q3. J. Clin. Invest. 96, 1216–1220 (1995).
Gollob, M. H. et al. Identification of a gene responsible for familial Wolff–Parkinson–White syndrome. N. Engl. J. Med. 344, 1823–1831 (2001).
Arad, M. et al. Constitutively active AMP kinase mutations cause glycogen storage disease mimicking hypertrophic cardiomyopathy. J. Clin. Invest. 109, 357–362 (2002).
Murphy, R. T. et al. Adenosine monophosphate-activated protein kinase disease mimicks hypertrophic cardiomyopathy and Wolff–Parkinson–White syndrome: natural history. J. Am. Coll. Cardiol. 45, 922–930 (2005).
Arad, M. et al. Glycogen storage diseases presenting as hypertrophic cardiomyopathy. N. Engl. J. Med. 352, 362–372 (2005).
Gruner, C. et al. Sarcomere protein gene mutations in patients with apical hypertrophic cardiomyopathy. Circ. Cardiovasc. Genet. 4, 288–295 (2011).
Sternick, E. B. et al. Clinical, electrocardiographic, and electrophysiologic characteristics of patients with a fasciculoventricular pathway: the role of PRKAG2 mutation. Heart Rhythm. 8, 58–64 (2011).
Zhang, L. P., Hui, B. & Gao, B. R. High risk of sudden death associated with a PRKAG2-related familial Wolff–Parkinson–White syndrome. J. Electrocardiol. 44, 483–486 (2011).
Pöyhönen, P. et al. Cardiovascular magnetic resonance findings in patients with PRKAG2 gene mutations. J. Cardiovasc. Magn. Reson. 17, 89 (2015).
Banankhah, P., Fishbein, G. A., Dota, A. & Ardehali, R. Cardiac manifestations of PRKAG2 mutation. BMC Med. Genet. 19, 1 (2018).
Thevenon, J. et al. High prevalence of arrhythmic and myocardial complications in patients with cardiac glycogenosis due to PRKAG2 mutations. Europace 19, 651–659 (2017).
Lopez-Sainz, A. et al. Clinical features and natural history of PRKAG2 variant cardiac glycogenosis. J. Am. Coll. Cardiol. 76, 186–197 (2020).
Ahamed, H. et al. Phenotypic expression and clinical outcomes in a South Asian PRKAG2 cardiomyopathy cohort. Sci. Rep. 10, 20610 (2020).
Hu, D. et al. Identification, clinical manifestation and structural mechanisms of mutations in AMPK associated cardiac glycogen storage disease. EBioMedicine 54, 102723 (2020).
van der Steld, L. P. et al. PRKAG2 syndrome, a rare hypertrophic cardiomyopathy: a Brazilian long-term follow-up with extracardiac disorders. Einstein 22, eAO0549 (2024).
Porto, A. G. et al. Clinical spectrum of PRKAG2 syndrome. Circ. Arrhythm. Electrophysiol. 9, e003121 (2016).
Xu, J. et al. Danon disease: a case report and literature review. Diagn. Pathol. 16, 39 (2021).
D’Souza, R. S. et al. Danon disease: clinical features, evaluation, and management. Circ. Heart Fail. 7, 843–849 (2014).
Danon, M. J. et al. Lysosomal glycogen storage disease with normal acid maltase. Neurology 31, 51–57 (1981).
Nishino, I. et al. Primary LAMP-2 deficiency causes X-linked vacuolar cardiomyopathy and myopathy (Danon disease). Nature 406, 906–910 (2000).
Rowland, T. J., Sweet, M. E., Mestroni, L. & Taylor, M. R. Danon disease – dysregulation of autophagy in a multisystem disorder with cardiomyopathy. J. Cell Sci. 129, 2135–2143 (2016).
Nascimbeni, A. C., Fanin, M., Angelini, C. & Sandri, M. Autophagy dysregulation in Danon disease. Cell Death Dis. 8, e2565 (2017).
Maron, B. J. et al. Clinical outcome and phenotypic expression in LAMP2 cardiomyopathy. JAMA 301, 1253–1259 (2009).
Boucek, D., Jirikowic, J. & Taylor, M. Natural history of Danon disease. Genet. Med. 13, 563–568 (2011).
Lacoste-Collin, L. et al. Danon’s disease (X-linked vacuolar cardiomyopathy and myopathy): a case with a novel Lamp-2 gene mutation. Neuromuscul. Disord. 12, 882–885 (2002).
Echaniz-Laguna, A. et al. Novel Lamp-2 gene mutation and successful treatment with heart transplantation in a large family with Danon disease. Muscle Nerve 33, 393–397 (2006).
Lobrinus, J. A. et al. Morphological, clinical and genetic aspects in a family with a novel LAMP-2 gene mutation (Danon disease). Neuromuscul. Disord. 15, 293–298 (2005).
Yang, Z. et al. Danon disease as an underrecognized cause of hypertrophic cardiomyopathy in children. Circulation 112, 1612–1617 (2005).
Charron, P. et al. Danon’s disease as a cause of hypertrophic cardiomyopathy: a systematic survey. Heart 90, 842–846 (2004).
Balmer, C. et al. Familial X-linked cardiomyopathy (Danon disease): diagnostic confirmation by mutation analysis of the LAMP2 gene. Eur. J. Pediatr. 164, 509–514 (2005).
Regelsberger, G. et al. Danon disease: case report and detection of new mutation. J. Inherit. Metab. Dis. 32, S115–S122 (2009).
Dougu, N. et al. Novel LAMP-2 mutation in a family with Danon disease presenting with hypertrophic cardiomyopathy. Circ. J. 73, 376–380 (2009).
Sabourdy, F. et al. Danon disease: further clinical and molecular heterogeneity. Muscle Nerve 39, 837–844 (2009).
Nadeau, A., Therrien, C., Karpati, G. & Sinnreich, M. Danon disease due to a novel splice mutation in the LAMP2 gene. Muscle Nerve 37, 338–342 (2008).
Musumeci, O. et al. Asymptomatic hyperCKemia in a case of Danon disease due to a missense mutation in Lamp-2 gene. Neuromuscul. Disord. 15, 409–411 (2005).
Di Blasi, C., Jarre, L., Blasevich, F., Dassi, P. & Mora, M. Danon disease: a novel LAMP2 mutation affecting the pre-mRNA splicing and causing aberrant transcripts and partial protein expression. Neuromuscul. Disord. 18, 962–966 (2008).
Sugie, K. et al. Clinicopathological features of genetically confirmed Danon disease. Neurology 58, 1773–1778 (2002).
Brambatti, M. et al. Danon disease: gender differences in presentation and outcomes. Int. J. Cardiol. 286, 92–98 (2019).
Elliott, P. M. et al. 2014 ESC guidelines on diagnosis and management of hypertrophic cardiomyopathy: the Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC). Eur. Heart J. 35, 2733–2779 (2014).
Ommen, S. R. et al. 2020 AHA/ACC guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 76, 3022–3055 (2020).
Rapezzi, C. et al. Diagnostic work-up in cardiomyopathies: bridging the gap between clinical phenotypes and final diagnosis. A position statement from the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 34, 1448–1458 (2013).
Hong, K. N. et al. International consensus on differential diagnosis and management of patients with Danon disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 82, 1628–1647 (2023).
Limongelli, G. et al. Diagnosis and management of rare cardiomyopathies in adult and paediatric patients. A position paper of the Italian Society of Cardiology (SIC) and Italian Society of Paediatric Cardiology (SICP). Int. J. Cardiol. 357, 55–71 (2022).
Hong, K. N. et al. Cardiac transplantation in Danon disease. J. Card. Fail. 28, 664–669 (2022).
Koeberl, D. D. et al. Gene therapy for glycogen storage diseases. J. Inherit. Metab. Dis. 47, 93–118 (2024).
Manso, A. M. et al. Systemic AAV9.LAMP2B injection reverses metabolic and physiologic multiorgan dysfunction in a murine model of Danon disease. Sci. Transl. Med. 12, eaax1744 (2020).
Greenberg, B. et al. Phase 1 study of AAV9.LAMP2B gene therapy in Danon disease. N. Engl. J. Med. 392, 972–983 (2025).
Scalco, R. S. et al. Data from the European registry for patients with McArdle disease and other muscle glycogenoses (EUROMAC). Orphanet J. Rare Dis. 15, 330 (2020).
Hannah, W. B. et al. Glycogen storage diseases. Nat. Rev. Dis. Prim. 9, 46 (2023).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Young, F. G. Claude Bernard and the discovery of glycogen; a century of retrospect. Br. Med. J. 1, 1431–1437 (1957).
Kornberg, A. Remembering our teachers. J. Biol. Chem. 276, 3–11 (2001).
Von Gierke, E. Hepato-nephro-megalia glycogenica (Glykogenspeicher-krankheit der Leber und Nieren). Beitr. Pathol. Anat. 82, 497–513 (1929).
Schönheimer, R. Über eine eigenartige Störung des Kohlehydrat-Stoffwechsels [German]. Hoppe Seylers Z. Physiol. Chem. 182, 148–150 (1929).
Cori, G. T. & Cori, C. F. Glucose-6-phosphatase of the liver in glycogen storage disease. J. Biol. Chem. 199, 661–667 (1952).
Cori, C. F. & Cori, G. T. Mechanism of formation of hexosemonophosphate in muscle and isolation of a new phosphate ester. Proc. Soc. Exp. Biol. Med. 34, 702–705 (1936).
Lim, J. A., Li, L. & Raben, N. Pompe disease: from pathophysiology to therapy and back again. Front. Aging Neurosci. 6, 177 (2014).
Bischoff, G. Zum klinischen Bild der Glykogen-Speicherungskrankheit (Glykogenose) [German]. Z. Kinderheilkd. 52, 722–726 (1932).
Putschar, W. Glykogenspeicherkrankheit des Herzens: “Thesaurismosis glycogenica” (v. Gierke) [German]. Beitr. Pathol. Anat. 90, 222–232 (1932).
Cori, G. T. Enzymes and glycogen structure in glycogenosis. Osterr Z. Kinderheilkd. Kinderfuersorge 10, 38–42 (1954).
Hers, H. G. α-Glucosidase deficiency in generalized glycogen-storage disease (Pompe’s disease). Biochem. J. 86, 11–16 (1963).
Almodovar-Paya, A. et al. Preclinical research in glycogen storage diseases: a comprehensive review of current animal models. Int. J. Mol. Sci. 21, 9621 (2020).
Bijvoet, A. G. et al. Generalized glycogen storage and cardiomegaly in a knockout mouse model of Pompe disease. Hum. Mol. Genet. 7, 53–62 (1998).
O’Sullivan, B. M. et al. Generalised glycogenosis in Brahman cattle. Aust. Vet. J. 57, 227–229 (1981).
Murakami, H., Takagi, A., Nanaka, S., Ishiura, S. & Sugita, H. Glycogenosis II in Japanese quails. Jikken Dobutsu 29, 475–478 (1980).
Allard, M. F. et al. Glycogen metabolism in the aerobic hypertrophied rat heart. Circulation 96, 676–682 (1997).
Goodwin, G. W., Ahmad, F., Doenst, T. & Taegtmeyer, H. Energy provision from glycogen, glucose, and fatty acids on adrenergic stimulation of isolated working rat hearts. Am. J. Physiol. 274, H1239–H1247 (1998).
Goodwin, G. W., Taylor, C. S. & Taegtmeyer, H. Regulation of energy metabolism of the heart during acute increase in heart work. J. Biol. Chem. 273, 29530–29539 (1998).
Henning, S. L., Wambolt, R. B., Schonekess, B. O., Lopaschuk, G. D. & Allard, M. F. Contribution of glycogen to aerobic myocardial glucose utilization. Circulation 93, 1549–1555 (1996).
Nielsen, J., Johnsen, J., Pryds, K., Ortenblad, N. & Botker, H. E. Myocardial subcellular glycogen distribution and sarcoplasmic reticulum Ca2+ handling: effects of ischaemia, reperfusion and ischaemic preconditioning. J. Muscle Res. Cell Motil. 42, 17–31 (2021).
Depre, C., Vanoverschelde, J. L. & Taegtmeyer, H. Glucose for the heart. Circulation 99, 578–588 (1999).
Botker, H. E., Helligso, P., Kimose, H. H., Thomassen, A. R. & Nielsen, T. T. Determination of high energy phosphates and glycogen in cardiac and skeletal muscle biopsies, with special reference to influence of biopsy technique and delayed freezing. Cardiovasc. Res. 28, 524–527 (1994).
Shelley, H. J. Glycogen reserves and their changes at birth and in anoxia. Br. Med. Bull. 17, 137–143 (1961).
Reichelt, M. E., Mellor, K. M., Curl, C. L., Stapleton, D. & Delbridge, L. M. Myocardial glycophagy – a specific glycogen handling response to metabolic stress is accentuated in the female heart. J. Mol. Cell Cardiol. 65, 67–75 (2013).
Goldfarb, A. H., Bruno, J. F. & Buckenmeyer, P. J. Intensity and duration effects of exercise on heart cAMP, phosphorylase, and glycogen. J. Appl. Physiol. 60, 1268–1273 (1986).
Schneider, C. A., Nguyen, V. T. & Taegtmeyer, H. Feeding and fasting determine postischemic glucose utilization in isolated working rat hearts. Am. J. Physiol. 260, H542–H548 (1991).
Segel, L. D., Chung, A., Mason, D. T. & Amsterdam, E. A. Cardiac glycogen in long-evans rats: diurnal pattern and response to exercise. Am. J. Physiol. 229, 398–401 (1975).
Kerem, D., Hammond, D. D. & Elsner, R. Tissue glycogen levels in the Weddell seal, Leptonychotes weddelli: a possible adaptation to asphyxial hypoxia. Comp. Biochem. Physiol. A Comp. Physiol. 45, 731–736 (1973).
Mellor, K. M. et al. Myocardial glycophagy flux dysregulation and glycogen accumulation characterize diabetic cardiomyopathy. J. Mol. Cell Cardiol. 189, 83–89 (2024).
Bhavnani, B. R. Ontogeny of some enzymes of glycogen metabolism in rabbit fetal heart, lungs, and liver. Can. J. Biochem. Cell Biol. 61, 191–197 (1983).
Gutierrez-Correa, J., Hod, M., Passoneau, J. V. & Freinkel, N. Glycogen and enzymes of glycogen metabolism in rat embryos and fetal organs. Biol. Neonate 59, 294–302 (1991).
Roach, P. J., Depaoli-Roach, A. A., Hurley, T. D. & Tagliabracci, V. S. Glycogen and its metabolism: some new developments and old themes. Biochem. J. 441, 763–787 (2012).
Delbridge, L. M. D., Mellor, K. M., Taylor, D. J. & Gottlieb, R. A. Myocardial stress and autophagy: mechanisms and potential therapies. Nat. Rev. Cardiol. 14, 412–425 (2017).
Delbridge, L. M., Mellor, K. M., Taylor, D. J. & Gottlieb, R. A. Myocardial autophagic energy stress responses – macroautophagy, mitophagy, and glycophagy. Am. J. Physiol. Heart Circ. Physiol. 308, H1194–H1204 (2015).
Zhao, H., Tang, M., Liu, M. & Chen, L. Glycophagy: an emerging target in pathology. Clin. Chim. Acta 484, 298–303 (2018).
Caulfield, J. & Klionsky, B. Myocardial ischemia and early infarction: an electron microscopic study. Am. J. Pathol. 35, 489–523 (1959).
Nielsen, J., Dubillot, P., Stausholm, M. H. & Ortenblad, N. Specific ATPases drive compartmentalized glycogen utilization in rat skeletal muscle. J. Gen. Physiol. 154, e202113071 (2022).
Bers, D. M. Excitation-contraction coupling and cardiac contractile force 2nd edn (Springer, 2001) [Series Developments in Cardiovascular Medicine Vol. 237].
Dizon, J. et al. Metabolic inhibition in the perfused rat heart: evidence for glycolytic requirement for normal sodium homeostasis. Am. J. Physiol. 274, H1082–H1089 (1998).
Geisbuhler, T. et al. Adenine nucleotide metabolism and compartmentalization in isolated adult rat heart cells. Circ. Res. 54, 536–546 (1984).
Bricknell, O. L. & Opie, L. H. Glycolytic ATP and its production during ischemia in isolated Langendorff-perfused rat hearts. Recent. Adv. Stud. Card. Struct. Metab. 11, 509–519 (1976).
Mercer, R. W. & Dunham, P. B. Membrane-bound ATP fuels the Na/K pump. Studies on membrane-bound glycolytic enzymes on inside-out vesicles from human red cell membranes. J. Gen. Physiol. 78, 547–568 (1981).
Campbell, J. D. & Paul, R. J. The nature of fuel provision for the Na+,K+-ATPase in porcine vascular smooth muscle. J. Physiol. 447, 67–82 (1992).
Fossel, E. T. & Solomon, A. K. Relation between red cell membrane (Na+ + K+)-ATPase and band 3 protein. Biochim. Biophys. Acta 649, 557–571 (1981).
Singh, P., Salih, M., Leddy, J. J. & Tuana, B. S. The muscle-specific calmodulin-dependent protein kinase assembles with the glycolytic enzyme complex at the sarcoplasmic reticulum and modulates the activity of glyceraldehyde-3-phosphate dehydrogenase in a Ca2+/calmodulin-dependent manner. J. Biol. Chem. 279, 35176–35182 (2004).
Boehm, E., Ventura-Clapier, R., Mateo, P., Lechene, P. & Veksler, V. Glycolysis supports calcium uptake by the sarcoplasmic reticulum in skinned ventricular fibres of mice deficient in mitochondrial and cytosolic creatine kinase. J. Mol. Cell Cardiol. 32, 891–902 (2000).
Takeuchi, K. et al. Improving glucose metabolism and/or sarcoplasmic reticulum Ca2+-ATPase function is warranted for immature pressure overload hypertrophied myocardium. Jpn. Circ. J. 65, 1064–1070 (2001).
Zima, A. V., Kockskamper, J. & Blatter, L. A. Cytosolic energy reserves determine the effect of glycolytic sugar phosphates on sarcoplasmic reticulum Ca2+ release in cat ventricular myocytes. J. Physiol. 577, 281–293 (2006).
Acknowledgements
Research by T.P., C.F.-L. and M.A.M. is funded by the Spanish Ministry of Economy and Competitiveness and Fondos Feder (grants PI22/00201, PI21/00381 and FORT23/00023, respectively). R.M.C. is supported in part by the National Institute for Health and Care Research (NIHR) Leeds Biomedical Research Centre (BRC) (NIHR203331). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care. Research by C.F.-L. and A.L. is funded by Wereld Kanker Onderzoek Fonds (WKOF) as part of the World Cancer Research Fund International grant programme (IIG_FULL_2021_007).
Author information
Authors and Affiliations
Contributions
T.P., R.M.C., C.F.-L., A.S.-L., J.N., N.Ø. and A.L. researched data for the article. T.P., R.M.C., A.S., C.F.-L., A.S.-L., J.N., N.Ø. and A.L. contributed to discussions of content. T.P., J.N., N.Ø. and A.L. wrote the manuscript. T.P., R.M.C., A.S., C.F.-L., M.A.M., J.A., J.N., N.Ø. and A.L. reviewed/edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
R.M.C. has received speaker’s fees from Janssen Oncology. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Priya Kishnani, who co-reviewed with Rebecca Koch and Monika Williams; Giuseppe Limongelli, who co-reviewed with Emanuele Monda; and Ros Quinlivan for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Amylopectin-like polyglucosan bodies
-
Aberrant glucose polymers that differ in structure and appearance from glycogen and are less soluble. They accumulate in tissues and are a common feature of some GSDs (GSDIV and GSDXV), neurodegenerative diseases and physiological ageing.
- Fasciculoventricular pathways
-
Uncommon pre‐excitation variants characterized by accessory connections between the bundle of His and the ventricles.
- Limit dextrins
-
When a branched polysaccharide (such as glycogen) is hydrolysed enzymatically, glucose units are removed one at a time until a branch point (that is, a limit dextrin) is reached. Further hydrolysis requires a different enzyme, glycogen phosphorylase.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pinós, T., Cubbon, R.M., Santalla, A. et al. Cardiovascular involvement in glycogen storage diseases. Nat Rev Cardiol 23, 39–59 (2026). https://doi.org/10.1038/s41569-025-01171-w
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41569-025-01171-w