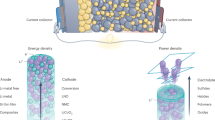
Abstract
The potential increase in cost of lithium-ion batteries owing to the limited supply of lithium has prompted investigations into alternative and complementary rechargeable batteries that use post-lithium charge carriers with higher elemental abundance. However, achieving highly reversible post-lithium metal anodes with sufficient kinetics remains challenging. The addition of co-solvents to conventional electrolytes is emerging as an important strategy to resolve these issues. In this Perspective, we discuss the progress of the co-solvent strategy for sodium, potassium, magnesium, calcium, zinc and aluminium post-lithium metal batteries. The coordination ability of co-solvents with post-lithium charge carriers is presented as a useful guide for selecting co-solvents for the respective battery electrolytes, owing to its correlation with several influential factors that affect the electrochemical performance of the metal anodes, such as solvation structure, de-solvation process and solid electrolyte interphase formation. Additionally, a discussion is provided on the importance of unravelling the effects beyond the solvation sheath of cationic charge carriers and for the development of sustainable electrolytes.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tian, Y. et al. Promises and challenges of next-generation ‘beyond Li-ion’ batteries for electric vehicles and grid decarbonization. Chem. Rev. 121, 1623–1669 (2021).
Vaalma, C., Buchholz, D., Weil, M. & Passerini, S. A cost and resource analysis of sodium-ion batteries. Nat. Rev. Mater. 3, 18013 (2018).
Ji, H., Wang, J., Ma, J., Cheng, H. M. & Zhou, G. Fundamentals, status and challenges of direct recycling technologies for lithium ion batteries. Chem. Soc. Rev. 52, 8194–8244 (2023).
Canepa, P. et al. Odyssey of multivalent cathode materials: open questions and future challenges. Chem. Rev. 117, 4287–4341 (2017).
Gao, Y., Yu, Q., Yang, H., Zhang, J. & Wang, W. The enormous potential of sodium/potassium-ion batteries as the mainstream energy storage technology for large-scale commercial applications. Adv. Mater. 36, e2405989 (2024).
Gautam, G. S. et al. First-principles evaluation of multi-valent cation insertion into orthorhombic V2O5. Chem. Commun. 51, 13619–13622 (2015).
Innocenti, A., Beringer, S. & Passerini, S. Cost and performance analysis as a valuable tool for battery material research. Nat. Rev. Mater. 9, 347–357 (2024).
Wang, X. et al. Building stable anodes for high-rate Na-metal batteries. Adv. Mater. 36, 2311256 (2024).
Lei, Y. J. et al. Progress and prospects of emerging potassium–sulfur batteries. Adv. Energy Mater. 12, 2202523 (2022).
Li, Y. et al. Interfacial engineering to achieve an energy density of over 200 Wh kg−1 in sodium batteries. Nat. Energy 7, 511–519 (2022).
Hobold, G. M. et al. Moving beyond 99.9% Coulombic efficiency for lithium anodes in liquid electrolytes. Nat. Energy 6, 951–960 (2021).
Li, M. et al. Design strategies for nonaqueous multivalent-ion and monovalent-ion battery anodes. Nat. Rev. Mater. 5, 276–294 (2020).
Wu, W., Luo, W. & Huang, Y. Less is more: a perspective on thinning lithium metal towards high-energy-density rechargeable lithium batteries. Chem. Soc. Rev. 52, 2553–2572 (2023).
Yamada, Y. & Yamada, A. Review — superconcentrated electrolytes for lithium batteries. J. Electrochem. Soc. 162, A2406–A2423 (2015).
Le, P. M. L. et al. Excellent cycling stability of sodium anode enabled by a stable solid electrolyte interphase formed in ether-based electrolytes. Adv. Funct. Mater. 30, 2001151 (2020).
Ha, S. Y. et al. Magnesium(II) bis(trifluoromethane sulfonyl) imide-based electrolytes with wide electrochemical windows for rechargeable magnesium batteries. ACS Appl. Mater. Interfaces 6, 4063–4073 (2014).
Zhang, N. et al. Cation-deficient spinel ZnMn2O4 cathode in Zn(CF3SO3)2 electrolyte for rechargeable aqueous Zn-ion battery. J. Am. Chem. Soc. 138, 12894–12901 (2016).
Gao, X. et al. Alkoxy-functionalized ionic liquid electrolytes: understanding ionic coordination of calcium ion speciation for the rational design of calcium electrolytes. Energy Environ. Sci. 13, 2559–2569 (2020).
Lee, J. et al. Ultraconcentrated sodium bis(fluorosulfonyl)imide-based electrolytes for high-performance sodium metal batteries. ACS Appl. Mater. Interfaces 9, 3723–3732 (2017).
Elia, G. A., Hoeppner, K. & Hahn, R. Comparison of chloroaluminate melts for aluminium graphite dual-ion battery application. Batter. Supercaps 4, 368–373 (2021).
Rajput, N. N., Qu, X., Sa, N., Burrell, A. K. & Persson, K. A. The coupling between stability and ion pair formation in magnesium electrolytes from first-principles quantum mechanics and classical molecular dynamics. J. Am. Chem. Soc. 137, 3411–3420 (2015).
Miao, L. et al. Aqueous electrolytes with hydrophobic organic cosolvents for stabilizing zinc metal anodes. ACS Nano 16, 9667–9678 (2022). This literature reports the use of a hydrophobic diethyl carbonate (DEC) with a low dielectric constant as a co-solvent for 2 M Zn(SO3CF3) aqueous electrolyte. The preferential adsorption of DEC molecules onto the zinc metal anode surface to create a water-poor electrical double layer is demonstrated, which contributes to the suppressed hydrogen evolution.
Li, Z., Fuhr, O., Fichtner, M. & Zhao-Karger, Z. Towards stable and efficient electrolytes for room-temperature rechargeable calcium batteries. Energy Environ. Sci. 12, 3496–3501 (2019).
Li, S. et al. Customized electrolyte and host structures enabling high-energy-density anode-free potassium-metal batteries. ACS Energy Lett. 8, 3467–3475 (2023).
Zheng, J. et al. Extremely stable sodium metal batteries enabled by localized high-concentration electrolytes. ACS Energy Lett. 3, 315–321 (2018). This literature reports an early example of the use of low-viscosity, non-solvating hydrofluoroether co-solvents in the construction of localized high-concentration electrolytes for sodium metal batteries.
Hu, L. et al. Restructuring electrolyte solvation by a versatile diluent toward beyond 99.9% Coulombic efficiency of sodium plating/stripping at ultralow temperatures. Adv. Mater. 36, 2312161 (2024). A cyclic ether co-solvent with weaker solvating ability than the original linear ether solvent in the low-concentration NaPF6 electrolyte is demonstrated, enhancing the coordination between Na+ and anion, which leads to preferentially anion-derived solid electrolyte interphase and promotes reversibility of sodium plating and stripping.
Xu, C., Diemant, T., Liu, X. & Passerini, S. Locally concentrated deep eutectic liquids electrolytes for low-polarization aluminum metal batteries. Adv. Mater. 36, 2400263 (2024).
You, C. et al. An inexpensive electrolyte with double-site hydrogen bonding and a regulated Zn2+ solvation structure for aqueous Zn-ion batteries capable of high-rate and ultra-long low-temperature operation. Energy Environ. Sci. 16, 5096–5107 (2023). Formamide with a high dielectric constant is adopted as a co-solvent for low-cost zinc acetate aqueous electrolytes, which coordinate with Zn2+ reducing water in the metal ion solvation sheath. It also reorganizes the hydrogen-bonded network and modifies the morphology of zinc metal anode through surface adsorption.
Hou, S. et al. Solvation sheath reorganization enables divalent metal batteries with fast interfacial charge transfer kinetics. Science 374, 172–178 (2021). A family of methoxyethyl-amine co-solvents exhibiting stronger coordination ability than the original 1,2-dimethoxyethane (DME) solvent towards Mg2+ is reported as co-solvents for non-aqueous magnesium metal batteries. The co-solvent replaces DME in the solvation sheath of Mg2+, leading to reduced polarization and considerably promoting magnesium stripping and plating reversibility.
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4417 (2004).
Fong, R., von Sacken, U. & Dahn, J. R. Studies of lithium intercalation into carbons using nonaqueous electrochemical cells. J. Electrochem. Soc. 137, 2009–2013 (1990).
Guyomard, D. & Tarascon, J. M. Rechargeable Li1+xMn2O4/carbon cells with a new electrolyte composition: potentiostatic studies and application to practical cells. J. Electrochem. Soc. 140, 3071–3081 (1993).
Geysens, P. et al. Solvation structure of sodium bis(fluorosulfonyl)imide-glyme solvate ionic liquids and its influence on cycling of Na-MNC cathodes. J. Phys. Chem. B 122, 275–289 (2018).
Cao, R. et al. Enabling room temperature sodium metal batteries. Nano Energy 30, 825–830 (2016).
Schafzahl, L., Hanzu, I., Wilkening, M. & Freunberger, S. A. An electrolyte for reversible cycling of sodium metal and intercalation compounds. ChemSusChem 10, 401–408 (2017).
Wang, Y. et al. Enhanced sodium metal/electrolyte interface by a localized high-concentration electrolyte for sodium metal batteries: first-principles calculations and experimental studies. ACS Appl. Energy Mater. 4, 7376–7384 (2021).
He, J., Bhargav, A., Shin, W. & Manthiram, A. Stable dendrite-free sodium-sulfur batteries enabled by a localized high-concentration electrolyte. J. Am. Chem. Soc. 143, 20241–20248 (2021).
Guo, D., Wang, J., Lai, T., Henkelman, G. & Manthiram, A. Electrolytes with solvating inner sheath engineering for practical Na–S batteries. Adv. Mater. 35, 2300841 (2023).
Chen, S. et al. High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30, 1706102 (2018).
Cao, X., Jia, H., Xu, W. & Zhang, J.-G. Review — localized high-concentration electrolytes for lithium batteries. J. Electrochem. Soc. 168, 010522 (2021).
Zhou, X. et al. Anion-reinforced solvation for a gradient inorganic-rich interphase enables high-rate and stable sodium batteries. Angew. Chem. Int. Ed. Engl. 61, e202205045 (2022). The addition of non-solvating co-solvents to high-concentration electrolytes is demonstrated, facilitating more anions to enter the solvation sheath of Na+, which leads to an inorganic-rich solid electrolyte interphase and the improved performance.
Yi, Q. et al. Fluorinated ether based electrolyte enabling sodium-metal batteries with exceptional cycling stability. ACS Appl. Mater. Interfaces 11, 46965–46972 (2019).
Zheng, X. et al. Bridging the immiscibility of an all-fluoride fire extinguishant with highly-fluorinated electrolytes toward safe sodium metal batteries. Energy Environ. Sci. 13, 1788–1798 (2020).
Seh, Z. W., Sun, J., Sun, Y. & Cui, Y. A highly reversible room-temperature sodium metal anode. ACS Cent. Sci. 1, 449–455 (2015).
Wang, S. et al. Stable sodium metal batteries via manipulation of electrolyte solvation structure. Small Methods 4, 1900856 (2020).
Thenuwara, A. C. et al. Enabling highly reversible sodium metal cycling across a wide temperature range with dual-salt electrolytes. J. Mater. Chem. A 9, 10992–11000 (2021).
Tomich, A. W. et al. A carboranyl electrolyte enabling highly reversible sodium metal anodes via a ‘fluorine-free’ SEI. Angew. Chem. Int. Ed. Engl. 61, e202208158 (2022).
Doi, K. et al. Reversible sodium metal electrodes: is fluorine an essential interphasial component? Angew. Chem. Int. Ed. Engl. 58, 8024–8028 (2019).
Li, Y. et al. Ultralow-concentration electrolyte for Na-ion batteries. ACS Energy Lett. 5, 1156–1158 (2020).
Yamada, Y., Wang, J., Ko, S., Watanabe, E. & Yamada, A. Advances and issues in developing salt-concentrated battery electrolytes. Nat. Energy 4, 269–280 (2019).
Zhou, J. et al. Low-temperature and high-rate sodium metal batteries enabled by electrolyte chemistry. Energy Storage Mater. 50, 47–54 (2022).
Wang, C. et al. Extending the low-temperature operation of sodium metal batteries combining linear and cyclic ether-based electrolyte solutions. Nat. Commun. 13, 4934 (2022).
Wang, Z. et al. Promoting fast Na ion transport at low temperatures for sodium metal batteries. ACS Appl. Mater. Interfaces 14, 40985–40991 (2022).
Holoubek, J. et al. Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Nat. Energy 6, 303–313 (2021).
Guo, D. et al. Low‐temperature sodium–sulfur batteries enabled by ionic liquid in localized high concentration electrolytes. Adv. Funct. Mater. 34, 2409494 (2024).
Xiao, N., Gourdin, G. & Wu, Y. Simultaneous stabilization of potassium metal and superoxide in K–O2 batteries on the basis of electrolyte reactivity. Angew. Chem. Int. Ed. Engl. 57, 10864–10867 (2018).
Liu, Q. et al. An ultra-low concentration electrolyte with fluorine-free bulky anions for stable potassium metal batteries. Nano Res. 16, 8290–8296 (2023).
Zhang, F. et al. Weakly solvated electrolyte driven anion interface chemistry for potassium batteries/hybrid capacitors. ACS Energy Lett. 8, 4895–4902 (2023).
Yu, Z. et al. Designing electrolytes with steric hindrance and film-forming booster for high-voltage potassium metal batteries. Adv. Funct. Mater. 34, 2315446 (2024).
Xiao, N., McCulloch, W. D. & Wu, Y. Reversible dendrite-free potassium plating and stripping electrochemistry for potassium secondary batteries. J. Am. Chem. Soc. 139, 9475–9478 (2017).
Liu, S. et al. An intrinsically non-flammable electrolyte for high-performance potassium batteries. Angew. Chem. Int. Ed. Engl. 59, 3638–3644 (2020).
Chen, J. et al. Low-temperature high-areal-capacity rechargeable potassium-metal batteries. Adv. Mater. 34, 2205678 (2022).
Chen, C. et al. Regulating the solvation structure of potassium ions using a multidentate ether in potassium metal batteries. ACS Appl. Energy Mater. 5, 10366–10374 (2022).
Yi, X. et al. Safe electrolyte for long-cycling alkali-ion batteries. Nat. Sustain. 7, 326–337 (2024).
Chen, X. et al. Phase transfer-mediated degradation of ether-based localized high-concentration electrolytes in alkali metal batteries. Angew. Chem. Int. Ed. Engl. 61, e202207018 (2022). The chemical degradation occurring between the ether-based localized high-concentration electrolytes and alkali metals is demonstrated, which is accelerated going from lithium to sodium, and particularly potassium, owing to their increasing trend to generate metal anions in a dynamic equilibrium with solvated electrons.
Chen, X., Meng, Y., Xiao, D., Wu, Y. & Qin, L. Tunning solvation structure in non-flammable, localized high-concentration electrolytes with enhanced stability towards all aluminum substrate-based K batteries. Energy Storage Mater. 61, 102923 (2023).
Gao, P. et al. Ultrastable dendrite-free potassium metal batteries enabled by weakly-solvated electrolyte. ACS Nano 17, 20325–20333 (2023).
Elia, G. A. et al. An overview and future perspectives of aluminum batteries. Adv. Mater. 28, 7564–7579 (2016).
Meng, J. et al. Rapid-charging aluminium–sulfur batteries operated at 85 °C with a quaternary molten salt electrolyte. Nat. Commun. 15, 596 (2024).
Pang, Q. et al. Fast-charging aluminium–chalcogen batteries resistant to dendritic shorting. Nature 608, 704–711 (2022).
Lin, M. C. et al. An ultrafast rechargeable aluminium-ion battery. Nature 520, 325–328 (2015).
Jiao, H., Wang, C., Tu, J., Tian, D. & Jiao, S. A rechargeable Al-ion battery: Al/molten AlCl3-urea/graphite. Chem. Commun. 53, 2331–2334 (2017).
Angell, M. et al. High Coulombic efficiency aluminum-ion battery using an AlCl3–urea ionic liquid analog electrolyte. Proc. Natl Acad. Sci. USA 114, 834–839 (2017).
Abood, H. M. A., Abbott, A. P., Ballantyne, A. D. & Ryder, K. S. Do all ionic liquids need organic cations? Characterisation of [AlCl2·nAmide]+ AlCl4− and comparison with imidazolium based systems. Chem. Commun. 47, 3523–3525 (2011).
Xu, C. et al. Locally concentrated ionic liquid electrolytes for wide-temperature-range aluminum–sulfur batteries. Angew. Chem. Int. Ed. Engl. 63, e202318204 (2024). The addition of non-solvating co-solvent can effectively promote the fluidity and ionic conductivity without affecting the AlCl4−/Al2Cl7− equilibrium of the electrolyte, which promotes the kinetics of aluminium stripping and plating.
Liebenow, C. Reversibility of electrochemical magnesium deposition from Grignard solutions. J. Appl. Electrochem. 27, 221–225 (1997).
Aurbach, D. et al. Prototype systems for rechargeable magnesium batteries. Nature 407, 724–727 (2000).
Mizrahi, O. et al. Electrolyte solutions with a wide electrochemical window for rechargeable magnesium batteries. J. Electrochem. Soc. 155, A103 (2008).
Zhao, W. et al. Chloride-free electrolytes for high-voltage magnesium metal batteries: challenges, strategies, and perspectives. Chem. Eur. J. 29, e202203334 (2023).
Ding, M. S., Diemant, T., Behm, R. J., Passerini, S. & Giffin, G. A. Dendrite growth in Mg metal cells containing Mg(TFSI)2/glyme electrolytes. J. Electrochem. Soc. 165, A1983–A1990 (2018).
Zhao, W. et al. Tailoring coordination in conventional ether-based electrolytes for reversible magnesium-metal anodes. Angew. Chem. Int. Ed. Engl. 61, e202205187 (2022). Trimethyl phosphate, partially replacing 1,2-dimethoxyethane (DME) molecules in the solvation sheath of Mg2+, is adopted as co-solvent for the Ca(TFSI)2–DME electrolyte, which promotes the de-solvation kinetics and modifies solid electrolyte interphase for magnesium metal anodes with reduced polarization.
Wang, F. et al. Solvent molecule design enables excellent charge transfer kinetics for a magnesium metal anode. ACS Energy Lett. 8, 780–789 (2023).
Bakulin, I. K. & Orekhov, M. A. Effect of long range interactions on the reduction of divalent ions in N,O-chelating solvents. Phys. Chem. Chem. Phys. 25, 20686–20692 (2023).
Du, Y. et al. Strong solvent coordination effect inducing gradient solid-electrolyte-interphase formation for highly efficient Mg plating/stripping. Energy Storage Mater. 62, 102939 (2023). This literature replaces the previously adopted Mg(TFSI)2 salt with a cheaper one, such as Mg(SO3CF3), and clarifies the relationship between the solvation sheath in the electrolyte and the solid electrolyte interphase structure on magnesium metal anodes.
Zhang, D. et al. Constructing efficient Mg(CF3SO3)2 electrolyte via tailoring solvation and interface chemistry for high-performance rechargeable magnesium batteries. Adv. Energy Mater. 13, 2301795 (2023).
Wang, M. et al. Synergy between the coordination and trace ionization of co-solvents enables reversible magnesium electroplating/stripping behavior. Energy Environ. Sci. 17, 630–641 (2023). A trace ionization of amine and ether co-solvents is proposed, which leads to the formation of protonated molecules. These species promote the formation of MgH2 in the solid electrolyte interphase and chemically associate with the TFSI− to neutrally charged ion pairs, which mitigate the migration and decomposition of TFSI− anion on magnesium metal anode and, therefore, promote the reversibility.
Fan, S. et al. A simple halogen-free magnesium electrolyte for reversible magnesium deposition through cosolvent assistance. ACS Appl. Mater. Interfaces 12, 10252–10260 (2020).
Aurbach, D., Skaletsky, R. & Gofer, Y. The electrochemical behavior of calcium electrodes in a few organic electrolytes. J. Electrochem. Soc. 138, 3536–3545 (1991).
Ponrouch, A., Frontera, C., Bardé, F. & Palacín, M. R. Towards a calcium-based rechargeable battery. Nat. Mater. 15, 169–172 (2016).
Wang, D. et al. Plating and stripping calcium in an organic electrolyte. Nat. Mater. 17, 16–20 (2018).
Kisu, K. et al. Monocarborane cluster as a stable fluorine-free calcium battery electrolyte. Sci. Rep. 11, 7563 (2021).
Shyamsunder, A., Blanc, L. E., Assoud, A. & Nazar, L. F. Reversible calcium plating and stripping at room temperature using a borate salt. ACS Energy Lett. 4, 2271–2276 (2019).
Hou, Z. et al. Correlation between electrolyte chemistry and solid electrolyte interphase for reversible Ca metal anodes. Angew. Chem. Int. Ed. Engl. 61, e202214796 (2022).
Ye, L. et al. A rechargeable calcium–oxygen battery that operates at room temperature. Nature 626, 313–318 (2024). Dimethyl sulfoxide as a solvating co-solvent for an ionic liquid electrolyte using Ca(TFSI)2 as salt and EmimBF4 ionic liquid as solvent, which facilitates both the kinetics and reversibility of calcium metal anodes.
Dong, Y. et al. Dissolution, solvation and diffusion in low-temperature zinc electrolyte design. Nat. Rev. Chem. 9, 102–117 (2025).
Luo, J. Y. & Xia, Y. Y. Aqueous lithium-ion battery LiTi2(PO4)3/LiMn2O4 with high power and energy densities as well as superior cycling stability. Adv. Funct. Mater. 17, 3877–3884 (2007).
Pang, G. et al. Enhanced performance of aqueous sodium-ion batteries using electrodes based on the NaTi2(PO4)3/MWNTs-Na0.44MnO2 System. Energy Technol. 2, 705–712 (2014).
Ming, J., Guo, J., Xia, C., Wang, W. & Alshareef, H. N. Zinc-ion batteries: materials, mechanisms, and applications. Mater. Sci. Eng. R. 135, 58–84 (2019).
Dong, Y. et al. Non-concentrated aqueous electrolytes with organic solvent additives for stable zinc batteries. Chem. Sci. 12, 5843–5852 (2021).
Ming, F. et al. Co-solvent electrolyte engineering for stable anode-free zinc metal batteries. J. Am. Chem. Soc. 144, 7160–7170 (2022).
Li, Z. et al. A co-solvent in aqueous electrolyte towards ultralong-life rechargeable zinc-ion batteries. Energy Storage Mater. 56, 174–182 (2023).
Dong, Y. et al. Cell-nucleus structured electrolyte for low-temperature aqueous zinc batteries. J. Energy Chem. 83, 324–332 (2023). When the content of 1,2-dimethoxyethane is not higher than 30%, it does not replace water in the solvation sheath of Zn2+ but pushes more anions to enter the Zn2+ solvation, which results in the formation of inorganic-rich solid electrolyte interphase on zinc metal anodes.
Guan, K. et al. A dual salt/dual solvent electrolyte enables ultrahigh utilization of zinc metal anode for aqueous batteries. Adv. Mater. 36, 2405889 (2024).
Cao, L. et al. Solvation structure design for aqueous Zn metal batteries. J. Am. Chem. Soc. 142, 21404–21409 (2020).
Hao, J. et al. Boosting zinc electrode reversibility in aqueous electrolytes by using low-cost antisolvents. Angew. Chem. Int. Ed. Engl. 60, 7366–7375 (2021).
Wang, Y. et al. Sulfolane-containing aqueous electrolyte solutions for producing efficient ampere-hour-level zinc metal battery pouch cells. Nat. Commun. 14, 1828 (2023). Sulfolane is adopted as an organic co-solvent for the 3 M Zn(SO3CF3)2 aqueous electrolyte. The addition of sulfolane leads to the formation of a micelle structure, in which sulfolane molecules constrain water in nanodomains to hinder proton reduction on zinc metal anodes.
Feng, D. et al. Immunizing aqueous Zn batteries against dendrite formation and side reactions at various temperatures via electrolyte additives. Small 17, 2103195 (2021).
Meng, C. et al. Multifunctional water–organic hybrid electrolyte for rechargeable zinc ions batteries. Chem. Eng. J. 450, 138265 (2022).
Shi, X. et al. Metallic zinc anode working at 50 and 50 mAh cm−2 with high depth of discharge via electrical double layer reconstruction. Adv. Funct. Mater. 33, 2211917 (2023).
Deng, W., Xu, Z. & Wang, X. High-donor electrolyte additive enabling stable aqueous zinc-ion batteries. Energy Storage Mater. 52, 52–60 (2022).
Hao, Y. et al. Hybrid electrolyte engineering enables reversible Zn metal anodes at ultralow current densities. J. Power Sources 584, 233631 (2023).
Zhang, T. et al. A solubility-limited, non-protonic polar small molecule co-solvent reveals additive selection in inorganic zinc salts. Energy Storage Mater. 65, 103085 (2024).
Liu, S. et al. Monolithic phosphate interphase for highly reversible and stable Zn metal anode. Angew. Chem. Int. Ed. Engl. 62, e202215600 (2023). This literature demonstrates the positive effect of the solid electrolyte interphase generated in the presence of a dimethyl methylphosphonate co-solvent on the cyclability of zinc metal anodes in aqueous electrolytes.
Ma, Q. et al. Regulation of outer solvation shell toward superior low-temperature aqueous zinc-ion batteries. Adv. Mater. 34, 2207344 (2022).
Han, J. et al. A thin and uniform fluoride-based artificial interphase for the zinc metal anode enabling reversible Zn/MnO2 batteries. ACS Energy Lett. 6, 3063–3071 (2021).
Li, T. C. et al. A universal additive strategy to reshape electrolyte solvation structure toward reversible Zn storage. Adv. Energy Mater. 12, 2103231 (2022).
Jiang, W. et al. Breaking the trade-off between capacity and stability in vanadium-based zinc-ion batteries. Chem. Sci. 15, 2601–2611 (2024).
Han, K. S. et al. Factors influencing preferential anion interactions during solvation of multivalent cations in ethereal solvents. J. Phys. Chem. C 125, 6005–6012 (2021).
Sun, Q. et al. Dipole–dipole interaction induced electrolyte interfacial model to stabilize antimony anode for high-safety lithium-ion batteries. ACS Energy Lett. 7, 3545–3556 (2022).
Liu, X. et al. Difluorobenzene-based locally concentrated ionic liquid electrolyte enabling stable cycling of lithium metal batteries with nickel-rich cathode. Adv. Energy Mater. 12, 2200862 (2022).
Liu, X. et al. Locally concentrated ionic liquid electrolyte with partially solvating diluent for lithium/sulfurized polyacrylonitrile batteries. Adv. Mater. 34, 2207155 (2022).
Liu, X. et al. PFAS-free locally concentrated ionic liquid electrolytes for lithium metal batteries. ACS Energy Lett. 9, 3049–3057 (2024).
Liu, X. et al. Development of PFAS-free locally concentrated ionic liquid electrolytes for high-energy lithium and aluminium metal batteries. Acc. Chem. Res. 58, 354–365 (2025).
Acknowledgements
Financial support from the Helmholtz Association, National Key R & D Program (2022YFB2404600), Natural Science Foundation of China (Key Project of 52131306), Project on Carbon Emission Peak and Neutrality of Jiangsu Province (BE2022031-4), the Start-up Research Fund of Southeast University (4003002418) and the Big Data Computing Center of Southeast University are acknowledged.
Author information
Authors and Affiliations
Contributions
X.L. and S.P. conceived the idea. X.L. and X.D. read and summarized the relevant literature. X.L. and X.D. wrote the original draft. H.A. revised the original draft. Y.W. revised the original draft. S.P. revised the original draft, supervised the project and acquired the funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Chemistry thanks Oleg Borodin, Weiyang Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
European Chemical Agency receiving the PFASs restriction proposal from five national authorities: https://echa.europa.eu/-/echa-receives-pfass-restriction-proposal-from-five-national-authorities
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Dong, X., Adenusi, H. et al. Co-solvent strategy for rechargeable post-lithium metal batteries. Nat Rev Chem 9, 415–426 (2025). https://doi.org/10.1038/s41570-025-00714-6
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41570-025-00714-6