Abstract
Chimeric antigen receptor (CAR) T cell therapy is a ‘living drug’ in which the T cells of patients are genetically engineered with an artificial receptor that directs them to attack diseased cells. CAR T cell therapies have had remarkable impact, curing subsets of patients with previously untreatable, late-stage cancers. However, limitations persist, including severe toxicities, limited survival of engineered cells, and therapeutic resistance. Genetically encoded small-molecule control systems have been developed to address these limitations. They can halt toxicities by eliminating CAR T cells or switching off their function. Furthermore, they can enhance therapy by directly targeting antigens or broadening cell killing ability through cytotoxic pro-drug activation. Small-molecule controllers include protease inhibitors, protein dimerizers, protein degraders, bi-specific adaptors and conditionally activated chemotherapeutics. Here, we outline small-molecule-based control approaches, categorizing them by function and detailing their molecular mechanisms. We emphasize systems in the clinic and highlight emerging applications and unmet areas.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Luster, A. D., Alon, R. & von Andrian, U. H. Immune cell migration in inflammation: present and future therapeutic targets. Nat. Immunol. 6, 1182–1190 (2005).
Kochenderfer, J. N. et al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood 116, 4099–4102 (2010).
June, C. H. & Sadelain, M. Chimeric antigen receptor therapy. N. Engl. J. Med. 379, 64–73 (2018).
Srivastava, S. & Riddell, S. R. Engineering CAR-T cells: design concepts. Trends Immunol. 36, 494–502 (2015).
Jayaraman, J. et al. CAR-T design: elements and their synergistic function. EBioMedicine 58, 102931 (2020).
Park, J. H. et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 378, 449–459 (2018).
Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Melenhorst, J. J. et al. Decade-long leukaemia remissions with persistence of CD4+ CAR T cells. Nature 602, 503–509 (2022).
Mackensen, A. et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat. Med. 28, 2124–2132 (2022).
Xu, X., Qiu, J. & Sun, Y. The basics of CAR T design and challenges in immunotherapy of solid tumors — ovarian cancer as a model. Hum. Vaccin. Immunother. 13, 1548–1555 (2017).
Zmievskaya, E. et al. Application of CAR-T cell therapy beyond oncology: autoimmune diseases and viral infections. Biomedicines https://doi.org/10.3390/biomedicines9010059 (2021).
Rurik, J. G. et al. CAR T cells produced in vivo to treat cardiac injury. Science 375, 91–96 (2022).
Porter, D., Frey, N., Wood, P. A., Weng, Y. & Grupp, S. A. Grading of cytokine release syndrome associated with the CAR T cell therapy tisagenlecleucel. J. Hematol. Oncol. 11, 35 (2018).
Morgan, R. A. et al. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol. Ther. 18, 843–851 (2010).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
Mei, H. et al. Neurological toxicities and coagulation disorders in the cytokine release syndrome during CAR-T therapy. Br. J. Haematol. 181, 689–692 (2018).
Khurana, A. et al. Chimeric antigen receptor T-cell therapy associated hemophagocytic lymphohistiocytosis syndrome: clinical presentation, outcomes, and management. Blood Cancer J. 14, 136 (2024).
Fugere, T. et al. Immune effector cell-associated HLH-like syndrome: a review of the literature of an increasingly recognized entity. Cancers https://doi.org/10.3390/cancers15215149 (2023).
Sharma, N., Reagan, P. M. & Liesveld, J. L. Cytopenia after CAR-T cell therapy — a brief review of a complex problem. Cancers https://doi.org/10.3390/cancers14061501 (2022).
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023). This review covers the long-term outcomes from cancer patients treated with approved CAR T cell therapies including durability of response and toxicities.
Le, R. Q. et al. FDA approval summary: tocilizumab for treatment of chimeric antigen receptor T cell-induced severe or life-threatening cytokine release syndrome. Oncologist 23, 943–947 (2018).
Bonifant, C. L., Jackson, H. J., Brentjens, R. J. & Curran, K. J. Toxicity and management in CAR T-cell therapy. Mol. Ther. Oncolytics 3, 16011 (2016).
Kochenderfer, J. N. et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119, 2709–2720 (2012).
Casucci, M., Hawkins, R. E., Dotti, G. & Bondanza, A. Overcoming the toxicity hurdles of genetically targeted T cells. Cancer Immunol. Immunother. 64, 123–130 (2015).
Lamers, C. H. et al. Treatment of metastatic renal cell carcinoma with CAIX CAR-engineered T cells: clinical evaluation and management of on-target toxicity. Mol. Ther. 21, 904–912 (2013).
Parkhurst, M. R. et al. T cells targeting carcinoembryonic antigen can mediate regression of metastatic colorectal cancer but induce severe transient colitis. Mol. Ther. 19, 620–626 (2011).
Zhai, X., Mao, L., Wu, M., Liu, J. & Yu, S. Challenges of anti-mesothelin CAR-T-cell therapy. Cancers https://doi.org/10.3390/cancers15051357 (2023).
Elsallab, M. et al. Second primary malignancies after commercial CAR T-cell therapy: analysis of the FDA adverse events reporting system. Blood 143, 2099–2105 (2024).
Suran, M. FDA adds boxed warning to CAR T-cell therapies, but says benefits outweigh risks of secondary cancers. JAMA 331, 818–820 (2024).
Ghilardi, G. et al. T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy. Nat. Med. 30, 984–989 (2024).
Verdun, N. & Marks, P. Secondary cancers after chimeric antigen receptor T-cell therapy. N. Engl. J. Med. 390, 584–586 (2024).
Tix, T. et al. Second primary malignancies after CAR T-cell therapy: a systematic review and meta-analysis of 5,517 lymphoma and myeloma patients. Clin. Cancer Res. 30, 4690–4700 (2024).
Schambach, A., Morgan, M. & Fehse, B. Two cases of T cell lymphoma following PiggyBac-mediated CAR T cell therapy. Mol. Ther. 29, 2631–2633 (2021).
Rotte, A. et al. Dose-response correlation for CAR-T cells: a systematic review of clinical studies. J. Immunother. Cancer https://doi.org/10.1136/jitc-2022-005678 (2022).
Huan, T. et al. Activation-induced cell death in CAR-T cell therapy. Hum. Cell 35, 441–447 (2022).
Poorebrahim, M. et al. Counteracting CAR T cell dysfunction. Oncogene 40, 421–435 (2021).
Long, A. H. et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat. Med. 21, 581–590 (2015).
Calderon, H., Mamonkin, M. & Guedan, S. Analysis of CAR-mediated tonic signaling. Methods Mol. Biol. 2086, 223–236 (2020).
Liu, Z. et al. Immunosuppression in tumor immune microenvironment and its optimization from CAR-T cell therapy. Theranostics 12, 6273–6290 (2022).
Majzner, R. G. & Mackall, C. L. Tumor antigen escape from CAR T-cell therapy. Cancer Discov. 8, 1219–1226 (2018).
Chen, N., Li, X., Chintala, N. K., Tano, Z. E. & Adusumilli, P. S. Driving CARs on the uneven road of antigen heterogeneity in solid tumors. Curr. Opin. Immunol. 51, 103–110 (2018).
Kirouac, D. C., Zmurchok, C. & Morris, D. Making drugs from T cells: the quantitative pharmacology of engineered T cell therapeutics. NPJ Syst. Biol. Appl. 10, 31 (2024). This paper provides a mathematical modelling perspective on the pharmacology of CAR T cells.
Milone, M. C. & Bhoj, V. G. The pharmacology of T cell therapies. Mol. Ther. Methods Clin. Dev. 8, 210–221 (2018).
Lu, L., Xie, M., Yang, B., Zhao, W. B. & Cao, J. Enhancing the safety of CAR-T cell therapy: synthetic genetic switch for spatiotemporal control. Sci. Adv. 10, eadj6251 (2024).
Pan, Y. et al. Mechanogenetics for the remote and noninvasive control of cancer immunotherapy. Proc. Natl Acad. Sci. USA 115, 992–997 (2018).
Seo, D. et al. A mechanogenetic toolkit for interrogating cell signaling in space and time. Cell 165, 1507–1518 (2016).
Long, X., Ye, J., Zhao, D. & Zhang, S. J. Magnetogenetics: remote non-invasive magnetic activation of neuronal activity with a magnetoreceptor. Sci. Bull. 60, 2107–2119 (2015).
Bai, P. et al. A fully human transgene switch to regulate therapeutic protein production by cooling sensation. Nat. Med. 25, 1266–1273 (2019).
Miller, I. C., Gamboa Castro, M., Maenza, J., Weis, J. P. & Kwong, G. A. Remote control of mammalian cells with heat-triggered gene switches and photothermal pulse trains. ACS Synth. Biol. 7, 1167–1173 (2018).
Piraner, D. I., Abedi, M. H., Moser, B. A., Lee-Gosselin, A. & Shapiro, M. G. Tunable thermal bioswitches for in vivo control of microbial therapeutics. Nat. Chem. Biol. 13, 75–80 (2017).
Stanley, S. A. et al. Radio-wave heating of iron oxide nanoparticles can regulate plasma glucose in mice. Science 336, 604–608 (2012).
Kortmann, J. & Narberhaus, F. Bacterial RNA thermometers: molecular zippers and switches. Nat. Rev. Microbiol. 10, 255–265 (2012).
Zhao, B. et al. An optogenetic controllable T cell system for hepatocellular carcinoma immunotherapy. Theranostics 9, 1837–1850 (2019).
Oide, M. et al. Blue light-excited light-oxygen-voltage-sensing domain 2 (LOV2) triggers a rearrangement of the kinase domain to induce phosphorylation activity in Arabidopsis phototropin1. J. Biol. Chem. 291, 19975–19984 (2016).
Polstein, L. R. & Gersbach, C. A. A light-inducible CRISPR-Cas9 system for control of endogenous gene activation. Nat. Chem. Biol. 11, 198–200 (2015).
Huse, M. Photochemical approaches to T-cell activation. Immunology 130, 151–157 (2010).
Celichowski, P. et al. Tuning CARs: recent advances in modulating chimeric antigen receptor (CAR) T cell activity for improved safety, efficacy, and flexibility. J. Transl. Med. 21, 197 (2023).
Foster, M. C. et al. Utility of a safety switch to abrogate CD19.CAR T-cell-associated neurotoxicity. Blood 137, 3306–3309 (2021). This letter reports a case study in which the iCasp9 suicide switch was successfully deployed in a patient to resolve CAR T cell-induced neurotoxicity.
Del Bufalo, F. et al. GD2-CART01 for relapsed or refractory high-risk neuroblastoma. N. Engl. J. Med. 388, 1284–1295 (2023).
Stein, M. N. et al. PSCA-targeted BPX-601 CAR T cells with pharmacological activation by rimiducid in metastatic pancreatic and prostate cancer: a phase 1 dose escalation trial. Nat. Commun. 15, 10743 (2024).
Bonini, C. et al. HSV-TK gene transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science 276, 1719–1724 (1997).
Moolten, F. L. Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: paradigm for a prospective cancer control strategy. Cancer Res. 46, 5276–5281 (1986).
Fan, L., Freeman, K. W., Khan, T., Pham, E. & Spencer, D. M. Improved artificial death switches based on caspases and FADD. Hum. Gene Ther. 10, 2273–2285 (1999).
Casucci, M. et al. Extracellular NGFR spacers allow efficient tracking and enrichment of fully functional CAR-T cells co-expressing a suicide gene. Front. Immunol. 9, 507 (2018).
Sasaki, T., Sakoda, Y., Adachi, K., Tokunaga, Y. & Tamada, K. Therapeutic effects of anti-GM2 CAR-T cells expressing IL-7 and CCL19 for GM2-positive solid cancer in xenograft model. Cancer Med. 12, 12569–12580 (2023).
Ciceri, F. et al. Infusion of suicide-gene-engineered donor lymphocytes after family haploidentical haemopoietic stem-cell transplantation for leukaemia (the TK007 trial): a non-randomised phase I-II study. Lancet Oncol. 10, 489–500 (2009).
Tiberghien, P. et al. Administration of herpes simplex-thymidine kinase-expressing donor T cells with a T-cell-depleted allogeneic marrow graft. Blood 97, 63–72 (2001).
Di Stasi, A. et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N. Engl. J. Med. 365, 1673–1683 (2011).
Berger, C., Flowers, M. E., Warren, E. H. & Riddell, S. R. Analysis of transgene-specific immune responses that limit the in vivo persistence of adoptively transferred HSV-TK-modified donor T cells after allogeneic hematopoietic cell transplantation. Blood 107, 2294–2302 (2006).
Keu, K. V. et al. Reporter gene imaging of targeted T cell immunotherapy in recurrent glioma. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aag2196 (2017).
Straathof, K. C. et al. An inducible caspase 9 safety switch for T-cell therapy. Blood 105, 4247–4254 (2005).
Hoyos, V. et al. Engineering CD19-specific T lymphocytes with interleukin-15 and a suicide gene to enhance their anti-lymphoma/leukemia effects and safety. Leukemia 24, 1160–1170 (2010).
Iuliucci, J. D. et al. Intravenous safety and pharmacokinetics of a novel dimerizer drug, AP1903, in healthy volunteers. J. Clin. Pharmacol. 41, 870–879 (2001).
Renatus, M., Stennicke, H. R., Scott, F. L., Liddington, R. C. & Salvesen, G. S. Dimer formation drives the activation of the cell death protease caspase 9. Proc. Natl Acad. Sci. USA 98, 14250–14255 (2001).
Clackson, T. et al. Redesigning an FKBP-ligand interface to generate chemical dimerizers with novel specificity. Proc. Natl Acad. Sci. USA 95, 10437–10442 (1998).
Diaconu, I. et al. Inducible caspase-9 selectively modulates the toxicities of CD19-specific chimeric antigen receptor-modified T cells. Mol. Ther. 25, 580–592 (2017).
Budde, L. E. et al. Combining a CD20 chimeric antigen receptor and an inducible caspase 9 suicide switch to improve the efficacy and safety of T cell adoptive immunotherapy for lymphoma. PLoS ONE 8, e82742 (2013).
Minagawa, K., Al-Obaidi, M. & Di Stasi, A. Generation of suicide gene-modified chimeric antigen receptor-redirected T-cells for cancer immunotherapy. Methods Mol. Biol. 1895, 57–73 (2019).
Stavrou, M. et al. A rapamycin-activated caspase 9-based suicide gene. Mol. Ther. 26, 1266–1276 (2018). This manuscript reports the development and pre-clinical characterization of the RapaCasp9 suicide switch system to deplete CAR T cells.
Zhou, X. et al. Inducible caspase-9 suicide gene controls adverse effects from alloreplete T cells after haploidentical stem cell transplantation. Blood 125, 4103–4113 (2015).
Ghosn, M. et al. Image-guided interventional radiological delivery of chimeric antigen receptor (CAR) T cells for pleural malignancies in a phase I/II clinical trial. Lung Cancer 165, 1–9 (2022).
Heczey, A. et al. CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol. Ther. 25, 2214–2224 (2017).
Majzner, R. G. et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature 603, 934–941 (2022).
Tey, S. K., Dotti, G., Rooney, C. M., Heslop, H. E. & Brenner, M. K. Inducible caspase 9 suicide gene to improve the safety of allodepleted T cells after haploidentical stem cell transplantation. Biol. Blood Marrow Transpl. 13, 913–924 (2007).
Donnelly, M. L. L. et al. Analysis of the aphthovirus 2A/2B polyprotein ‘cleavage’ mechanism indicates not a proteolytic reaction, but a novel translational effect: a putative ribosomal ‘skip’. J. Gen. Virol. 82, 1013–1025 (2001).
MacCorkle, R. A., Freeman, K. W. & Spencer, D. M. Synthetic activation of caspases: artificial death switches. Proc. Natl Acad. Sci. USA 95, 3655–3660 (1998).
Zajc, C. U., Sylvander, E., Lehner, M. & Traxlmayr, M. W. Small molecule-regulated switches to provide functional control of CAR T cells within the patient. Expert Opin. Biol. Ther. 24, 425–432 (2024).
Bouquet, L. et al. RapaCaspase-9-based suicide gene applied to the safety of IL-1RAP CAR-T cells. Gene Ther. 30, 706–713 (2023).
Lee, S. M. et al. A chemical switch system to modulate chimeric antigen receptor T cell activity through proteolysis-targeting chimaera technology. ACS Synth. Biol. 9, 987–992 (2020).
Juillerat, A. et al. Modulation of chimeric antigen receptor surface expression by a small molecule switch. BMC Biotechnol. 19, 44 (2019).
Richman, S. A. et al. Ligand-induced degradation of a CAR permits reversible remote control of CAR T cell activity in vitro and in vivo. Mol. Ther. 28, 1600–1613 (2020).
Jan, M. et al. Reversible ON- and OFF-switch chimeric antigen receptors controlled by lenalidomide. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.abb6295 (2021). This paper describes the creation and characterization of several ON switch and OFF switch CARs regulated by the molecular glue lenalidomide.
Carbonneau, S. et al. An IMiD-inducible degron provides reversible regulation for chimeric antigen receptor expression and activity. Cell Chem. Biol. 28, 802–812.e6 (2021).
Lee, I. K. et al. A genetically encoded protein tag for control and quantitative imaging of CAR T cell therapy. Mol. Ther. 31, 3564–3578 (2023).
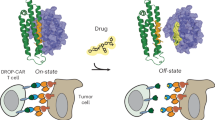
Giordano-Attianese, G. et al. A computationally designed chimeric antigen receptor provides a small-molecule safety switch for T-cell therapy. Nat. Biotechnol. 38, 426–432 (2020). This manuscript reports the development of a PPI-based CAR OFF switch that uses a clever computational design to develop the small-molecule disruptable protein heterodimerization interface.
Wu, C. Y., Roybal, K. T., Puchner, E. M., Onuffer, J. & Lim, W. A. Remote control of therapeutic T cells through a small molecule-gated chimeric receptor. Science 350, aab4077 (2015). This paper describes the development of the first CAR ON switch using small-molecule dimerizers to assemble CAR fragments.
Zheng, Y. et al. Generation of regulable EGFRvIII targeted chimeric antigen receptor T cells for adoptive cell therapy of glioblastoma. Biochem. Biophys. Res. Commun. 507, 59–66 (2018).
Leung, W. H. et al. Sensitive and adaptable pharmacological control of CAR T cells through extracellular receptor dimerization. JCI Insight https://doi.org/10.1172/jci.insight.124430 (2019).
Juillerat, A. et al. Design of chimeric antigen receptors with integrated controllable transient functions. Sci. Rep. 6, 18950 (2016).
Mata, M. et al. Inducible activation of MyD88 and CD40 in CAR T cells results in controllable and potent antitumor activity in preclinical solid tumor models. Cancer Discov. 7, 1306–1319 (2017). This paper develops a MyD88/CD40 signalling switch for rimiducid-regulated augmentation of CAR T cell expansion and antitumour function.
Kim, M. S. et al. Redirection of genetically engineered CAR-T cells using bifunctional small molecules. J. Am. Chem. Soc. 137, 2832–2835 (2015).
Lee, Y. G. et al. Regulation of CAR T cell-mediated cytokine release syndrome-like toxicity using low molecular weight adapters. Nat. Commun. 10, 2681 (2019).
Labanieh, L. et al. Enhanced safety and efficacy of protease-regulated CAR-T cell receptors. Cell 185, 1745–1763.e22 (2022). This manuscript describes the design and characterization of CARs regulated by viral protease inhibitors, showing robust control of activity and augmentation through T cell ‘rest’.
Li, H. S. et al. High-performance multiplex drug-gated CAR circuits. Cancer Cell 40, 1294–1305.e4 (2022).
Hotblack, A. et al. Tunable control of CAR T cell activity through tetracycline mediated disruption of protein-protein interaction. Sci. Rep. 11, 21902 (2021).
Jha, R. et al. Designer small-molecule control system based on minocycline-induced disruption of protein-protein interaction. ACS Chem. Biol. 19, 308–324 (2024). This paper reports the development of a novel PPI disruption system triggered by minocycline and its use for a variety of applications including building a CAR OFF switch, activation and antigen targeting.
Gu, X., He, D., Li, C., Wang, H. & Yang, G. Development of inducible CD19-CAR T cells with a Tet-On system for controlled activity and enhanced clinical safety. Int. J. Mol. Sci. https://doi.org/10.3390/ijms19113455 (2018).
Sakemura, R. et al. A Tet-On inducible system for controlling CD19-chimeric antigen receptor expression upon drug administration. Cancer Immunol. Res. 4, 658–668 (2016).
Drent, E. et al. Feasibility of controlling CD38-CAR T cell activity with a Tet-on inducible CAR design. PLoS ONE 13, e0197349 (2018).
Kotter, B. et al. Titratable pharmacological regulation of CAR T cells using zinc finger-based transcription factors. Cancers https://doi.org/10.3390/cancers13194741 (2021).
Chakravarti, D., Caraballo, L. D., Weinberg, B. H. & Wong, W. W. Inducible gene switches with memory in human T cells for cellular immunotherapy. ACS Synth. Biol. 8, 1744–1754 (2019). This manuscript describes the development of recombinase-based ON switch and OFF switch for stable regulation of CAR expression by a single dose of tamoxifen.
Zhang, R. Y. et al. Doxycycline inducible chimeric antigen receptor T cells targeting CD147 for hepatocellular carcinoma therapy. Front. Cell Dev. Biol. 7, 233 (2019).
Ali Hosseini Rad, S. M., Poudel, A., Tan, G. M. Y. & McLellan, A. D. Optimisation of Tet-On inducible systems for sleeping beauty-based chimeric antigen receptor (CAR) applications. Sci. Rep. 10, 13125 (2020).
Sahillioglu, A. C., Toebes, M., Apriamashvili, G., Gomez, R. & Schumacher, T. N. CRASH-IT switch enables reversible and dose-dependent control of TCR and CAR T-cell function. Cancer Immunol. Res. 9, 999–1007 (2021).
Li, H. S. et al. Multidimensional control of therapeutic human cell function with synthetic gene circuits. Science 378, 1227–1234 (2022). This paper details the development of zinc finger transcription factors controlled by several small-molecule drugs and their application to transcriptional control of CARs and other therapeutic payloads.
Weber, E. W. et al. Pharmacologic control of CAR-T cell function using dasatinib. Blood Adv. 3, 711–717 (2019).
Weber, E. W. et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science https://doi.org/10.1126/science.aba1786 (2021). This paper introduces the concept of resting CAR T cells with small-molecule ON switches to improve their function.
Fink, E. C. & Ebert, B. L. The novel mechanism of lenalidomide activity. Blood 126, 2366–2369 (2015).
Bekes, M., Langley, D. R. & Crews, C. M. PROTAC targeted protein degraders: the past is prologue. Nat. Rev. Drug Discov. 21, 181–200 (2022).
Bondeson, D. P. et al. Catalytic in vivo protein knockdown by small-molecule PROTACs. Nat. Chem. Biol. 11, 611–617 (2015).
Sakamoto, K. M. et al. Protacs: chimeric molecules that target proteins to the Skp1-Cullin-F box complex for ubiquitination and degradation. Proc. Natl Acad. Sci. USA 98, 8554–8559 (2001).
Harper, S. et al. Discovery of MK-5172, a macrocyclic hepatitis C virus NS3/4a protease inhibitor. ACS Med. Chem. Lett. 3, 332–336 (2012).
Jiang, Y. et al. Discovery of danoprevir (ITMN-191/R7227), a highly selective and potent inhibitor of hepatitis C virus (HCV) NS3/4A protease. J. Med. Chem. 57, 1753–1769 (2014).
Scola, P. M. et al. The discovery of asunaprevir (BMS-650032), an orally efficacious NS3 protease inhibitor for the treatment of hepatitis C virus infection. J. Med. Chem. 57, 1730–1752 (2014).
Salzer, B. et al. Engineering AvidCARs for combinatorial antigen recognition and reversible control of CAR function. Nat. Commun. 11, 4166 (2020).
Auslander, S. & Fussenegger, M. Engineering gene circuits for mammalian cell-based applications. Cold Spring Harb. Perspect. Biol. https://doi.org/10.1101/cshperspect.a023895 (2016).
Cronin, C. A., Gluba, W. & Scrable, H. The lac operator-repressor system is functional in the mouse. Genes Dev. 15, 1506–1517 (2001).
Gossen, M. & Bujard, H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc. Natl Acad. Sci. USA 89, 5547–5551 (1992).
Cabrera, A. et al. The sound of silence: transgene silencing in mammalian cell engineering. Cell Syst. 13, 950–973 (2022).
Yang, G., Nowsheen, S., Aziz, K. & Georgakilas, A. G. Toxicity and adverse effects of tamoxifen and other anti-estrogen drugs. Pharmacol. Ther. 139, 392–404 (2013).
Branda, C. S. & Dymecki, S. M. Talking about a revolution: the impact of site-specific recombinases on genetic analyses in mice. Dev. Cell 6, 7–28 (2004).
Mestermann, K. et al. The tyrosine kinase inhibitor dasatinib acts as a pharmacologic on/off switch for CAR T cells. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aau5907 (2019).
Tamada, K. et al. Redirecting gene-modified T cells toward various cancer types using tagged antibodies. Clin. Cancer Res. 18, 6436–6445 (2012).
Urbanska, K. et al. A universal strategy for adoptive immunotherapy of cancer through use of a novel T-cell antigen receptor. Cancer Res. 72, 1844–1852 (2012).
Rodgers, D. T. et al. Switch-mediated activation and retargeting of CAR-T cells for B-cell malignancies. Proc. Natl Acad. Sci. USA 113, E459–E468 (2016).
Cho, J. H., Collins, J. J. & Wong, W. W. Universal chimeric antigen receptors for multiplexed and logical control of T cell responses. Cell 173, 1426–1438.e11 (2018).
Rodriguez-Garcia, A. et al. CAR-T cell-mediated depletion of immunosuppressive tumor-associated macrophages promotes endogenous antitumor immunity and augments adoptive immunotherapy. Nat. Commun. 12, 877 (2021).
Cresswell, G. M. et al. Folate receptor beta designates immunosuppressive tumor-associated myeloid cells that can be reprogrammed with folate-targeted drugs. Cancer Res. 81, 671–684 (2021).
Tummers, Q. R. et al. Intraoperative imaging of folate receptor alpha positive ovarian and breast cancer using the tumor specific agent EC17. Oncotarget 7, 32144–32155 (2016).
Lu, Y. J. et al. Preclinical evaluation of bispecific adaptor molecule controlled folate receptor CAR-T cell therapy with special focus on pediatric malignancies. Front. Oncol. 9, 151 (2019).
Lee, Y. G. et al. Use of a single CAR T cell and several bispecific adapters facilitates eradication of multiple antigenically different solid tumors. Cancer Res. 79, 387–396 (2019). This manuscript describes the development of several heterobifunctional adaptors that target universal CAR T cells to recognize a variety of antigens.
Lu, Y. et al. Preclinical pharmacokinetics, tissue distribution, and antitumor activity of a folate-hapten conjugate-targeted immunotherapy in hapten-immunized mice. Mol. Cancer Ther. 5, 3258–3267 (2006).
Zajc, C. U. et al. A conformation-specific ON-switch for controlling CAR T cells with an orally available drug. Proc. Natl Acad. Sci. USA 117, 14926–14935 (2020).
Kvorjak, M. et al. Conditional control of universal CAR T cells by cleavable OFF-switch adaptors. ACS Synth. Biol. 12, 2996–3007 (2023).
Carlson, J. C. T., Mikula, H. & Weissleder, R. Unraveling tetrazine-triggered bioorthogonal elimination enables chemical tools for ultrafast release and universal cleavage. J. Am. Chem. Soc. 140, 3603–3612 (2018).
Ruffo, E. et al. Post-translational covalent assembly of CAR and synNotch receptors for programmable antigen targeting. Nat. Commun. 14, 2463 (2023).
Minutolo, N. G. et al. Quantitative control of gene-engineered T-cell activity through the covalent attachment of targeting ligands to a universal immune receptor. J. Am. Chem. Soc. 142, 6554–6568 (2020).
Stepanov, A. V. et al. Control of the antitumour activity and specificity of CAR T cells via organic adapters covalently tethering the CAR to tumour cells. Nat. Biomed. Eng. 8, 529–543 (2024). This paper reports the development of covalent small-molecule adaptors for targeting universal CAR T cells and use of a DNA-barcoded library to identify a new small-molecule ligand that recognizes the HER2 antigen.
Serniuck, N. J. et al. Electrophilic proximity-inducing synthetic adapters enhance universal T cell function by covalently enforcing immune receptor signaling. Mol. Ther. Oncol. 32, 200842 (2024).
Bulcha, J. T., Wang, Y., Ma, H., Tai, P. W. L. & Gao, G. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target. Ther. 6, 53 (2021).
Narayanan, P. et al. A composite MyD88/CD40 switch synergistically activates mouse and human dendritic cells for enhanced antitumor efficacy. J. Clin. Invest. 121, 1524–1534 (2011).
Foster, A. E. et al. Regulated expansion and survival of chimeric antigen receptor-modified T cells using small molecule-dependent inducible MyD88/CD40. Mol. Ther. 25, 2176–2188 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT02744287 (2023).
Cook, P. J. et al. A chemically inducible IL-2 receptor signaling complex allows for effective in vitro and in vivo selection of engineered CD4+ T cells. Mol. Ther. 31, 2472–2488 (2023).
Sudarsanam, H., Buhmann, R. & Henschler, R. Influence of culture conditions on ex vivo expansion of T lymphocytes and their function for therapy: current insights and open questions. Front. Bioeng. Biotechnol. 10, 886637 (2022).
Baluna, R. & Vitetta, E. S. Vascular leak syndrome: a side effect of immunotherapy. Immunopharmacology 37, 117–132 (1997).
Inniss, M. C. et al. Carbonic anhydrase 2-derived drug-responsive domain regulates membrane-bound cytokine expression and function in engineered T cells. Commun. Biol. 8, 28 (2025).
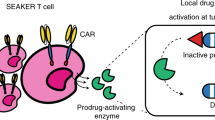
Gardner, T. J. et al. Engineering CAR-T cells to activate small-molecule drugs in situ. Nat. Chem. Biol. 18, 216–225 (2022). This paper describes the development of CAR T cells that can activate cytotoxic pro-drugs to enhance their antitumour activity.
Zhang, J., Kale, V. & Chen, M. Gene-directed enzyme prodrug therapy. AAPS J. 17, 102–110 (2015).
Sharma, S. K. & Bagshawe, K. D. Antibody directed enzyme prodrug therapy (ADEPT): trials and tribulations. Adv. Drug Deliv. Rev. 118, 2–7 (2017).
Wolfsberg, E. et al. Machine-guided dual-objective protein engineering for deimmunization and therapeutic functions. Cell Syst. https://doi.org/10.1016/j.cels.2025.101299 (2025).
Chen, Y. Y., Jensen, M. C. & Smolke, C. D. Genetic control of mammalian T-cell proliferation with synthetic RNA regulatory systems. Proc. Natl Acad. Sci. USA 107, 8531–8536 (2010).
Kapcan, E. et al. Mimicry of molecular glues using dual covalent chimeras. Nat. Commun. 16, 2855 (2025).
Peterson, A. A. & Liu, D. R. Small-molecule discovery through DNA-encoded libraries. Nat. Rev. Drug Discov. 22, 699–722 (2023).
Hasselgren, C. & Oprea, T. I. Artificial intelligence for drug discovery: are we there yet? Annu. Rev. Pharmacol. Toxicol. 64, 527–550 (2024).
Sanchez-Paulete, A. R. et al. Targeting macrophages with CAR T cells delays solid tumor progression and enhances antitumor immunity. Cancer Immunol. Res. 10, 1354–1369 (2022).
Huang, B. et al. Use of a universal targeting CAR T cell to simultaneously kill cancer cells and cancer-associated fibroblasts. Front. Immunol. 16, 1539265 (2025).
Millul, J. et al. An ultra-high-affinity small organic ligand of fibroblast activation protein for tumor-targeting applications. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.2101852118 (2021).
Maher, J. & Davies, D. M. CAR based immunotherapy of solid tumours — a clinically based review of target antigens. Biology https://doi.org/10.3390/biology12020287 (2023).
Sharma, A. et al. Hypoxia-targeted drug delivery. Chem. Soc. Rev. 48, 771–813 (2019).
Boedtkjer, E. & Pedersen, S. F. The acidic tumor microenvironment as a driver of cancer. Annu. Rev. Physiol. 82, 103–126 (2020).
Gobin, E. et al. A pan-cancer perspective of matrix metalloproteases (MMP) gene expression profile and their diagnostic/prognostic potential. BMC Cancer 19, 581 (2019).
Wang, H. et al. Selective in vivo metabolic cell-labeling-mediated cancer targeting. Nat. Chem. Biol. 13, 415–424 (2017).
Nguyen, H. T. X. et al. Engineering TME-gated inducible CAR-T cell therapy for solid tumors. Mol. Ther. https://doi.org/10.1016/j.ymthe.2025.04.037 (2025).
Muller, F. et al. CD19 CAR T-cell therapy in autoimmune disease — a case series with follow-up. N. Engl. J. Med. 390, 687–700 (2024).
Mougiakakos, D. et al. CD19-targeted CAR T cells in refractory systemic lupus erythematosus. N. Engl. J. Med. 385, 567–569 (2021).
Guffroy, A., Jacquel, L., Guffroy, B. & Martin, T. CAR-T cells for treating systemic lupus erythematosus: a promising emerging therapy. Jt. Bone Spine 91, 105702 (2024).
Short, L., Holt, R. A., Cullis, P. R. & Evgin, L. Direct in vivo CAR T cell engineering. Trends Pharmacol. Sci. 45, 406–418 (2024).
Bui, T. A., Mei, H., Sang, R., Ortega, D. G. & Deng, W. Advancements and challenges in developing in vivo CAR T cell therapies for cancer treatment. EBioMedicine 106, 105266 (2024).
Guzman, G., Reed, M. R., Bielamowicz, K., Koss, B. & Rodriguez, A. CAR-T therapies in solid tumors: opportunities and challenges. Curr. Oncol. Rep. 25, 479–489 (2023).
Albelda, S. M. CAR T cell therapy for patients with solid tumours: key lessons to learn and unlearn. Nat. Rev. Clin. Oncol. 21, 47–66 (2024).
Daei Sorkhabi, A. et al. The current landscape of CAR T-cell therapy for solid tumors: mechanisms, research progress, challenges, and counterstrategies. Front. Immunol. 14, 1113882 (2023).
Garcia, J. et al. Naturally occurring T cell mutations enhance engineered T cell therapies. Nature 626, 626–634 (2024).
Farber, D. L., Yudanin, N. A. & Restifo, N. P. Human memory T cells: generation, compartmentalization and homeostasis. Nat. Rev. Immunol. 14, 24–35 (2014).
Hudecek, M. et al. The nonsignaling extracellular spacer domain of chimeric antigen receptors is decisive for in vivo antitumor activity. Cancer Immunol. Res. 3, 125–135 (2015).
Sadelain, M., Brentjens, R. & Riviere, I. The basic principles of chimeric antigen receptor design. Cancer Discov. 3, 388–398 (2013).
Guedan, S., Calderon, H., Posey, A. D. Jr. & Maus, M. V. Engineering and design of chimeric antigen receptors. Mol. Ther. Methods Clin. Dev. 12, 145–156 (2019).
Imai, C. et al. Chimeric receptors with 4-1BB signaling capacity provoke potent cytotoxicity against acute lymphoblastic leukemia. Leukemia 18, 676–684 (2004).
Maher, J., Brentjens, R. J., Gunset, G., Riviere, I. & Sadelain, M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRζ/CD28 receptor. Nat. Biotechnol. 20, 70–75 (2002).
Qiu, Y. et al. Mechanical force determines chimeric antigen receptor microclustering and signaling. Mol. Ther. 32, 1016–1032 (2024).
Wu, L., Wei, Q., Brzostek, J. & Gascoigne, N. R. J. Signaling from T cell receptors (TCRs) and chimeric antigen receptors (CARs) on T cells. Cell Mol. Immunol. 17, 600–612 (2020).
Hennecke, J. & Wiley, D. C. T cell receptor-MHC interactions up close. Cell 104, 1–4 (2001).
Rosenberg, S. A., Restifo, N. P., Yang, J. C., Morgan, R. A. & Dudley, M. E. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat. Rev. Cancer 8, 299–308 (2008).
Du, S., Yan, J., Xue, Y., Zhong, Y. & Dong, Y. Adoptive cell therapy for cancer treatment. Exploration 3, 20210058 (2023).
Levine, B. L., Miskin, J., Wonnacott, K. & Keir, C. Global manufacturing of CAR T cell therapy. Mol. Ther. Methods Clin. Dev. 4, 92–101 (2017).
Wang, X. & Riviere, I. Clinical manufacturing of CAR T cells: foundation of a promising therapy. Mol. Ther. Oncolytics 3, 16015 (2016).
Goyco Vera, D., Waghela, H., Nuh, M., Pan, J. & Lulla, P. Approved CAR-T therapies have reproducible efficacy and safety in clinical practice. Hum. Vaccin. Immunother. 20, 2378543 (2024).
Labanieh, L. & Mackall, C. L. CAR immune cells: design principles, resistance and the next generation. Nature 614, 635–648 (2023). This review covers key design principles of CARs, current clinical data, and next-generation designs for augmenting activity.
Acknowledgements
This work was supported by NIH grants R01 GM142007 (to J.L. and A.D.), R01 CA290866 (to J.L. and A.D.) and T32 EB001026 (to V.S.).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article and contributed substantially to discussion of the content. E.L.A., A.C.M., V.S., A.D. and J.L. wrote the article. A.C.M., V.S., A.D. and J.L. reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
J.L. and A.D. have personal financial interests in the form of stocks in the privately traded company Monarch Therapeutics, LLC. They are also inventors on patent applications filed by the University of Pittsburgh (J.L.: US11117936B2, awarded, mSA2-CAR technology; J.L. and A.D.: WO2020072764A, pending, SNAP-CAR technology; J.L. and A.D.: WO2022099065A, pending, conditional adaptor switches; J.L. and A.D.: 63/541,954, pending, small-molecule adaptors). Aspects of WO2020072764A are licensed by Coeptis Therapeutics, and aspects of WO2020072764A, US11117936B2, WO2022099065A and 63/541,954 are licensed by Monarch Therapeutics.
Peer review
Peer review information
Nature Reviews Chemistry thanks Kui Cheng, Michael Traxlmayr, Elise Sylvander and Dhruv K. Sethi for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Adams, E.L., McGovern, A.C., So, V. et al. Small-molecule control of CAR T cells. Nat Rev Chem 9, 809–825 (2025). https://doi.org/10.1038/s41570-025-00768-6
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41570-025-00768-6