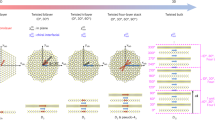
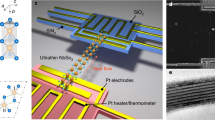
Abstract
Matter exhibits strikingly different structures and behaviours in strongly confined spaces compared with its bulk form. These differences become particularly pronounced when the confinement is reduced to the ångström scale, comparable with the characteristic size of atoms, ions and molecules. Advances in layered two-dimensional (2D) materials — including both van der Waals and non-van-der-Waals materials — have enabled the precise fabrication of ångström-scale 2D confinement systems, opening a new frontier for investigating matter properties and mass transport at this extreme scale. In this Review, we summarize the methods used for precisely fabricating such confined systems, the novel structural transformations of matter that arise and their associated physicochemical properties, and the unique molecular and ionic transport phenomena observed and their underlying mechanisms. We also critically assess the knowledge gaps, technological implications and untapped potential of this burgeoning field.

This is a preview of subscription content, access via your institution
Access options







Similar content being viewed by others
Change history
31 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41570-026-00823-w
References
Björneholm, O. et al. Water at interfaces. Chem. Rev. 116, 7698–7726 (2016).
Bocquet, L. & Charlaix, E. Nanofluidics, from bulk to interfaces. Chem. Soc. Rev. 39, 1073–1095 (2010).
Kavokine, N., Netz, R. R. & Bocquet, L. Fluids at the nanoscale: from continuum to subcontinuum transport. Annu. Rev. Fluid Mech. 53, 377–410 (2021).
Zheng, W. et al. Observation of liquid–solid transition of nanoconfined water at ambient temperature. Nat. Mater. https://doi.org/10.1038/s41563-025-02456-8 (2026).
Agre, P. Aquaporin water channels (Nobel Lecture). Angew. Chem. Int. Ed. 43, 4278–4290 (2004).
Kozono, D., Yasui, M., King, L. S. & Agre, P. Aquaporin water channels: atomic structure molecular dynamics meet clinical medicine. J. Clin. Investig. 109, 1395–1399 (2002).
Zhou, Y., Morais-Cabral, J. H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel–Fab complex at 2.0 Å resolution. Nature 414, 43–48 (2001).
MacKinnon, R. Potassium channels and the atomic basis of selective ion conduction (Nobel Lecture). Angew. Chem. Int. Ed. 43, 4265–4277 (2004).
Shen, J., Liu, G., Han, Y. & Jin, W. Artificial channels for confined mass transport at the sub-nanometre scale. Nat. Rev. Mater. 6, 294–312 (2021).
Emmerich, T. et al. Nanofluidics. Nat. Rev. Methods Primers 4, 69 (2024).
Bocquet, L. Nanofluidics coming of age. Nat. Mater. 19, 254–256 (2020).
Wang, L. et al. Fundamental transport mechanisms, fabrication and potential applications of nanoporous atomically thin membranes. Nat. Nanotechnol. 12, 509–522 (2017).
Aluru, N. R. et al. Fluids and electrolytes under confinement in single-digit nanopores. Chem. Rev. 123, 2737–2831 (2023).
Zhao, C. et al. Enhanced gating effects in responsive sub-nanofluidic ion channels. Acc. Mater. Res. 4, 786–797 (2023).
Geim, A. K. Exploring two-dimensional empty space. Nano Lett. 21, 6356–6358 (2021).
Kang, Y. et al. Functionalized 2D membranes for separations at the 1-nm scale. Chem. Soc. Rev. 53, 7939–7959 (2024).
Bhardwaj, A. et al. Fabrication of angstrom-scale two-dimensional channels for mass transport. Nat. Protoc. 19, 240–280 (2024).
Lei, D., Zhang, Z. & Jiang, L. Bioinspired 2D nanofluidic membranes for energy applications. Chem. Soc. Rev. 53, 2300–2325 (2024).
Hu, S. et al. Proton transport through one-atom-thick crystals. Nature 516, 227–230 (2014).
Mogg, L. et al. Atomically thin micas as proton-conducting membranes. Nat. Nanotechnol. 14, 962–966 (2019).
Ji, Y. et al. High proton conductivity through angstrom-porous titania. Nat. Commun. 15, 10546 (2024).
Cohen-Tanugi, D. & Grossman, J. C. Water desalination across nanoporous graphene. Nano Lett. 12, 3602–3608 (2012).
Jain, T. et al. Heterogeneous sub-continuum ionic transport in statistically isolated graphene nanopores. Nat. Nanotechnol. 10, 1053–1057 (2015).
Jiang, X. et al. Nonlinear electrohydrodynamic ion transport in graphene nanopores. Sci. Adv. 8, eabj2510 (2022).
Hoenig, E. et al. In situ generation of (sub) nanometer pores in MoS2 membranes for ion-selective transport. Nat. Commun. 15, 7911 (2024).
Feng, J. et al. Observation of ionic Coulomb blockade in nanopores. Nat. Mater. 15, 850–855 (2016).
Zhou, Z. et al. Gas permeation through graphdiyne-based nanoporous membranes. Nat. Commun. 13, 4031 (2022).
Graf, M. et al. Fabrication and practical applications of molybdenum disulfide nanopores. Nat. Protoc. 14, 1130–1168 (2019).
Novoselov, K. S. et al. Electric field effect in atomically thin carbon films. Science 306, 666–669 (2004).
Dreyer, D. R., Park, S., Bielawski, C. W. & Ruoff, R. S. The chemistry of graphene oxide. Chem. Soc. Rev. 39, 228–240 (2010).
Roy, S. et al. Structure, properties and applications of two-dimensional hexagonal boron nitride. Adv. Mater. 33, e2101589 (2021).
Chhowalla, M. et al. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 5, 263–275 (2013).
Wang, M. et al. Chemically engineered substrates for patternable growth of two-dimensional chalcogenide crystals. ACS Nano 10, 10317–10323 (2016).
Bandurin, D. A. et al. High electron mobility, quantum Hall effect and anomalous optical response in atomically thin InSe. Nat. Nanotechnol. 12, 223–227 (2017).
VahidMohammadi, A., Rosen, J. & Gogotsi, Y. The world of two-dimensional carbides and nitrides (MXenes). Science 372, eabf1581 (2021).
Carvalho, A. et al. Phosphorene: from theory to applications. Nat. Rev. Mater. 1, 16061 (2016).
Shao, J.-J., Raidongia, K., Koltonow, A. R. & Huang, J. Self-assembled two-dimensional nanofluidic proton channels with high thermal stability. Nat. Commun. 6, 7602 (2015).
Pan, X.-F. et al. Transforming ground mica into high-performance biomimetic polymeric mica film. Nat. Commun. 9, 2974 (2018).
Li, L., Ma, R., Ebina, Y., Iyi, N. & Sasaki, T. Positively charged nanosheets derived via total delamination of layered double hydroxides. Chem. Mater. 17, 4386–4391 (2005).
Wu, J. et al. High electron mobility and quantum oscillations in non-encapsulated ultrathin semiconducting Bi2O2Se. Nat. Nanotechnol. 12, 530–534 (2017).
Mao, L., Stoumpos, C. C. & Kanatzidis, M. G. Two-dimensional hybrid halide perovskites: principles and promises. J. Am. Chem. Soc. 141, 1171–1190 (2019).
Zhou, J. et al. A library of atomically thin metal chalcogenides. Nature 556, 355–359 (2018).
Hong, Y.-L. et al. Chemical vapor deposition of layered two-dimensional MoSi2N4 materials. Science 369, 670–674 (2020).
Qian, X. et al. CdPS3 nanosheets-based membrane with high proton conductivity enabled by Cd vacancies. Science 370, 596–600 (2020).
Wang, D. et al. Direct synthesis and chemical vapor deposition of 2D carbide and nitride MXenes. Science 379, 1242–1247 (2023).
Que, H. et al. Synthesis of two-dimensional transition metal phosphorous chalcogenides and their high-entropy alloys. Nat. Synth. 4, 582–591 (2025).
Dikin, D. A. et al. Preparation and characterization of graphene oxide paper. Nature 448, 457–460 (2007).
Coleman, J. N. et al. Two-dimensional nanosheets produced by liquid exfoliation of layered materials. Science 331, 568–571 (2011).
Zhang, L. et al. Metal telluride nanosheets by scalable solid lithiation and exfoliation. Nature 628, 313–319 (2024).
Zhu, X. et al. Scalable synthesis of bilayer graphene at ambient temperature. J. Am. Chem. Soc. 146, 6388–6396 (2024).
Yang, R. et al. High-yield production of mono- or few-layer transition metal dichalcogenide nanosheets by an electrochemical lithium ion intercalation-based exfoliation method. Nat. Protoc. 17, 358–377 (2022).
Radha, B. et al. Molecular transport through capillaries made with atomic-scale precision. Nature 538, 222–225 (2016).
Gopinadhan, K. et al. Complete steric exclusion of ions and proton transport through confined monolayer water. Science 363, 145–148 (2019).
Esfandiar, A. et al. Size effect in ion transport through angstrom-scale slits. Science 358, 511–513 (2017).
Raidongia, K. & Huang, J. Nanofluidic ion transport through reconstructed layered materials. J. Am. Chem. Soc. 134, 16528–16531 (2012).
Sapkota, B. et al. High permeability sub-nanometre sieve composite MoS2 membranes. Nat. Commun. 11, 2747 (2020).
Hoenig, E. et al. Controlling the structure of MoS2 membranes via covalent functionalization with molecular spacers. Nano Lett. 20, 7844–7851 (2020).
Ventura-Martinez, K., Zhu, Y., Booth, A. & Hatzell, K. B. Impact of asymmetric microstructure on ion transport in Ti3C2Tx membranes. Nano Lett. 24, 13551–13557 (2024).
Agmon, N. et al. Protons and hydroxide ions in aqueous systems. Chem. Rev. 116, 7642–7672 (2016).
Zhou, J. et al. Modular assembly of a library of hybrid superlattices and artificial quantum solids. Matter 7, 1131–1145 (2024).
Ries, L. et al. Enhanced sieving from exfoliated MoS2 membranes via covalent functionalization. Nat. Mater. 18, 1112–1117 (2019).
Ying, T. et al. Vacancy functionalized MoS2 nanolaminated membranes for efficient sieving in forward osmosis. Adv. Mater. 37, e04781 (2025).
Zhou, C. et al. Hybrid organic–inorganic two-dimensional metal carbide MXenes with amido- and imido-terminated surfaces. Nat. Chem. 15, 1722–1729 (2023).
Israelachvili, J. N. & Pashley, R. M. Molecular layering of water at surfaces and origin of repulsive hydration forces. Nature 306, 249–250 (1983).
Wang, M. et al. Tuning transport in graphene oxide membrane with single-site copper (II) cations. iScience 25, 104044 (2022).
Chen, L. et al. Ion sieving in graphene oxide membranes via cationic control of interlayer spacing. Nature 550, 380–383 (2017).
Ding, L. et al. Effective ion sieving with Ti3C2Tx MXene membranes for production of drinking water from seawater. Nat. Sustain. 3, 296–302 (2020).
Wang, M. et al. Anomalously enhanced ion transport and uptake in functionalized angstrom-scale two-dimensional channels. Proc. Natl Acad. Sci. USA 121, e2313616121 (2024).
Xia, Z. et al. Tunable ion transport with freestanding vermiculite membranes. ACS Nano 16, 18266–18273 (2022).
Hung, W.-S. et al. Cross-linking with diamine monomers to prepare composite graphene oxide-framework membranes with varying d-spacing. Chem. Mater. 26, 2983–2990 (2014).
Hwang, J. et al. Nano-confinement induced nucleation of ice-VII at room temperature. Preprint at https://doi.org/10.48550/arXiv.2411.12975 (2024).
Kapil, V. et al. The first-principles phase diagram of monolayer nanoconfined water. Nature 609, 512–516 (2022).
Lin, B., Jiang, J., Zeng, X. C. & Li, L. Temperature-pressure phase diagram of confined monolayer water/ice at first-principles accuracy with a machine-learning force field. Nat. Commun. 14, 4110 (2023).
Jiang, J. et al. Rich proton dynamics and phase behaviours of nanoconfined ices. Nat. Phys. 20, 456–464 (2024).
Jiang, J. et al. First-principles molecular dynamics simulations of the spontaneous freezing transition of 2D water in a nanoslit. J. Am. Chem. Soc. 143, 8177–8183 (2021).
Chin, H.-T. et al. Ferroelectric 2D ice under graphene confinement. Nat. Commun. 12, 6291 (2021).
Bramwell, S. T. Ferroelectric ice. Nature 397, 212–213 (1999).
Qiu, H. & Guo, W. Electromelting of confined monolayer ice. Phys. Rev. Lett. 110, 195701 (2013).
Algara-Siller, G. et al. Square ice in graphene nanocapillaries. Nature 519, 443–445 (2015).
Zhou, W. et al. The observation of square ice in graphene questioned. Nature 528, E1–E2 (2015).
Wu, D. et al. Probing structural superlubricity of two-dimensional water transport with atomic resolution. Science 384, 1254–1259 (2024).
Trushin, M., Andreeva, D. V., Peeters, F. M. & Novoselov, K. S. Structure and flow of low-dimensional water. Nat. Rev. Phys. 7, 502–513 (2025).
Smirnov, P., Yamaguchi, T., Kittaka, S., Takahara, S. & Kuroda, Y. X-ray diffraction study of water confined in mesoporous MCM-41 materials over a temperature range of 223−298 K. J. Phys. Chem. B 104, 5498–5504 (2000).
Ravindra, P., Advincula, X. R., Schran, C., Michaelides, A. & Kapil, V. Quasi-one-dimensional hydrogen bonding in nanoconfined ice. Nat. Commun. 15, 7301 (2024).
Wang, L. et al. Twisted bilayer Ice as a new class of hydrogen-bonding moiré materials. Nat. Commun. 16, 8762 (2025).
Fumagalli, L. et al. Anomalously low dielectric constant of confined water. Science 360, 1339–1342 (2018).
Wang, R. et al. In-plane dielectric constant and conductivity of confined water. Nature 646, 606–610 (2025).
Schlaich, A., Knapp, E. W. & Netz, R. R. Water dielectric effects in planar confinement. Phys. Rev. Lett. 117, 048001 (2016).
Zhang, C., Gygi, F. & Galli, G. Strongly anisotropic dielectric relaxation of water at the nanoscale. J. Phys. Chem. Lett. 4, 2477–2481 (2013).
Muñoz-Santiburcio, D. & Marx, D. Confinement-controlled aqueous chemistry within nanometric slit pores: focus review. Chem. Rev. 121, 6293–6320 (2021).
Nair, R. R., Wu, H. A., Jayaram, P. N., Grigorieva, I. V. & Geim, A. K. Unimpeded permeation of water through helium-leak–tight graphene-based membranes. Science 335, 442–444 (2012).
Raviv, U., Laurat, P. & Klein, J. Fluidity of water confined to subnanometre films. Nature 413, 51–54 (2001).
Marx, D. Proton transfer 200 years after von Grotthuss: insights from ab initio simulations. ChemPhysChem 7, 1848–1870 (2006).
Dasgupta, S., Saha, S. & Paesani, F. Sub-nanometer confinement suppresses autoionization of water. J. Am. Chem. Soc. 147, 25167–25173 (2025).
Ghosh, S., Sood, A. K. & Kumar, N. Carbon nanotube flow sensors. Science 299, 1042–1044 (2003).
Rabinowitz, J., Cohen, C. & Shepard, K. L. An electrically actuated, carbon-nanotube-based biomimetic ion pump. Nano Lett. 20, 1148–1153 (2020).
Keerthi, A. et al. Water friction in nanofluidic channels made from two-dimensional crystals. Nat. Commun. 12, 3092 (2021).
Secchi, E. et al. Massive radius-dependent flow slippage in carbon nanotubes. Nature 537, 210–213 (2016).
Xie, Q. et al. Fast water transport in graphene nanofluidic channels. Nat. Nanotechnol. 13, 238–245 (2018).
Devanathan, R., Chase-Woods, D., Shin, Y. & Gotthold, D. W. Molecular dynamics simulations reveal that water diffusion between graphene oxide layers is slow. Sci. Rep. 6, 29484 (2016).
Wei, N., Peng, X. & Xu, Z. Breakdown of fast water transport in graphene oxides. Phys. Rev. E 89, 012113 (2014).
Maali, A., Cohen-Bouhacina, T. & Kellay, H. Measurement of the slip length of water flow on graphite surface. Appl. Phys. Lett. 92, 053101 (2008).
Kavokine, N., Bocquet, M.-L. & Bocquet, L. Fluctuation-induced quantum friction in nanoscale water flows. Nature 602, 84–90 (2022).
Yu, X., Principi, A., Tielrooij, K.-J., Bonn, M. & Kavokine, N. Electron cooling in graphene enhanced by plasmon–hydron resonance. Nat. Nanotechnol. 18, 898–904 (2023).
Coquinot, B., Bocquet, L. & Kavokine, N. Quantum feedback at the solid-liquid interface: flow-induced electronic current and its negative contribution to friction. Phys. Rev. X 13, 011019 (2023).
Abraham, J. et al. Tunable sieving of ions using graphene oxide membranes. Nat. Nanotechnol. 12, 546–550 (2017).
Morelos-Gomez, A. et al. Effective NaCl and dye rejection of hybrid graphene oxide/graphene layered membranes. Nat. Nanotechnol. 12, 1083–1088 (2017).
Zhou, K.-G. et al. Electrically controlled water permeation through graphene oxide membranes. Nature 559, 236–240 (2018).
Zhao, F., Liang, Y., Cheng, H., Jiang, L. & Qu, L. Highly efficient moisture-enabled electricity generation from graphene oxide frameworks. Energy Environ. Sci. 9, 912–916 (2016).
Xia, H. et al. Electricity generated by upstream proton diffusion in two-dimensional nanochannels. Nat. Nanotechnol. 19, 1316–1322 (2024).
Nightingale, E. R. Phenomenological theory of ion solvation. Effective radii of hydrated ions. J. Phys. Chem. 63, 1381–1387 (1959).
Ohtaki, H. & Radnai, T. Structure and dynamics of hydrated ions. Chem. Rev. 93, 1157–1204 (1993).
Marcus, Y. The thermodynamics of solvation of ions. Part 2.—The enthalpy of hydration at 298.15 K. J. Chem. Soc. Faraday Trans. 1 83, 339–349 (1987).
Zhou, X. et al. Intrapore energy barriers govern ion transport and selectivity of desalination membranes. Sci. Adv. 6, eabd9045 (2020).
Yu, Y. et al. Dehydration impeding ionic conductance through two-dimensional angstrom-scale slits. Nanoscale 11, 8449–8457 (2019).
Zhang, Z., Wen, L. & Jiang, L. Nanofluidics for osmotic energy conversion. Nat. Rev. Mater. 6, 622–639 (2021).
Stein, D., Kruithof, M. & Dekker, C. Surface-charge-governed ion transport in nanofluidic channels. Phys. Rev. Lett. 93, 035901 (2004).
Yu, Y. et al. Charge asymmetry effect in ion transport through angstrom-scale channels. J. Phys. Chem. C 123, 1462–1469 (2019).
Zhou, K. & Xu, Z. Field-enhanced selectivity in nanoconfined ionic transport. Nanoscale 12, 6512–6521 (2020).
Wang, X. et al. Comediation of voltage gating and ion charge in MXene membrane for controllable and selective monovalent cation separation. Sci. Adv. 10, eado3998 (2024).
Cheng, C., Jiang, G., Simon, G. P., Liu, J. Z. & Li, D. Low-voltage electrostatic modulation of ion diffusion through layered graphene-based nanoporous membranes. Nat. Nanotechnol. 13, 685–690 (2018).
Xue, Y. et al. Atomic-scale ion transistor with ultrahigh diffusivity. Science 372, 501–503 (2021).
Yang, J. et al. Photo-induced ultrafast active ion transport through graphene oxide membranes. Nat. Commun. 10, 1171 (2019).
Wang, L. et al. Photo-switchable two-dimensional nanofluidic ionic diodes. Chem. Sci. 8, 4381–4386 (2017).
Xia, J. et al. Light-augmented multi-ion interaction in MXene membrane for simultaneous water treatment and osmotic power generation. ACS Nano 17, 25269–25278 (2023).
Lao, J. et al. Aqueous stable Ti3C2 MXene membrane with fast and photoswitchable nanofluidic transport. ACS Nano 12, 12464–12471 (2018).
Mouterde, T. et al. Molecular streaming and its voltage control in ångström-scale channels. Nature 567, 87–90 (2019).
Sun, P. et al. Electro- and magneto-modulated ion transport through graphene oxide membranes. Sci. Rep. 4, 6798 (2014).
Corry, B. & Thomas, M. Mechanism of ion permeation and selectivity in a voltage gated sodium channel. J. Am. Chem. Soc. 134, 1840–1846 (2012).
Hinds, B. J. Engineering small-ion transporter channels. Science 372, 459–460 (2021).
Bernèche, S. & Roux, B. Energetics of ion conduction through the K+ channel. Nature 414, 73–77 (2001).
Jin, Y. et al. Coupling effects in electromechanical ion transport in graphene nanochannels. Phys. Rev. E 102, 033112 (2020).
Ciarrocchi, A., Tagarelli, F., Avsar, A. & Kis, A. Excitonic devices with van der Waals heterostructures: valleytronics meets twistronics. Nat. Rev. Mater. 7, 449–464 (2022).
Zou, Y.-C. et al. Ion exchange in atomically thin clays and micas. Nat. Mater. 20, 1677–1682 (2021).
Tang, H. et al. On-chip multi-degree-of-freedom control of two-dimensional materials. Nature 632, 1038–1044 (2024).
Marcus, Y. & Hefter, G. Ion pairing. Chem. Rev. 106, 4585–4621 (2006).
van der Vegt, N. F. A. et al. Water-mediated ion pairing: occurrence and relevance. Chem. Rev. 116, 7626–7641 (2016).
Robin, P., Kavokine, N. & Bocquet, L. Modeling of emergent memory and voltage spiking in ionic transport through angstrom-scale slits. Science 373, 687–691 (2021).
Zhou, K. & Xu, Z. Nanoconfinement-enforced ion correlation and nanofluidic ion machinery. Nano Lett. 20, 8392–8398 (2020).
Zhao, W. et al. Two-dimensional monolayer salt nanostructures can spontaneously aggregate rather than dissolve in dilute aqueous solutions. Nat. Commun. 12, 5602 (2021).
Fong, K. D. et al. The interplay of solvation and polarization effects on ion pairing in nanoconfined electrolytes. Nano Lett. 24, 5024–5030 (2024).
Toquer, D., Bocquet, L. & Robin, P. Ionic association and Wien effect in 2D confined electrolytes. J. Chem. Phys. 162, 064703 (2025).
Zhao, X. et al. Anomalous phase behaviors of monolayer NaCl aqueous solutions induced by effective coulombic interactions within angstrom-scale slits. J. Phys. Chem. Lett. 13, 2704–2710 (2022).
Zhao, W. et al. Evidence of formation of monolayer hydrated salts in nanopores. J. Am. Chem. Soc. 144, 18976–18985 (2022).
Robin, P. et al. Long-term memory and synapse-like dynamics in two-dimensional nanofluidic channels. Science 379, 161–167 (2023).
Marcus, Y. Effect of ions on the structure of water: structure making and breaking. Chem. Rev. 109, 1346–1370 (2009).
Emmerich, T. et al. Nanofluidic logic with mechano–ionic memristive switches. Nat. Electron. 7, 271–278 (2024).
Wang, M. et al. Cooperative and inhibitory ion transport in functionalized angstrom-scale two-dimensional channels. Nat. Commun. 16, 5854 (2025).
Xu, R. et al. Regulate ion transport in subnanochannel membranes by ion-pairing. J. Am. Chem. Soc. 147, 17144–17151 (2025).
Lee, C.-H. & MacKinnon, R. Activation mechanism of a human SK-calmodulin channel complex elucidated by cryo-EM structures. Science 360, 508–513 (2018).
Rohaim, A. et al. Open and closed structures of a barium-blocked potassium channel. J. Mol. Biol. 432, 4783–4798 (2020).
Shi, G. et al. Two-dimensional Na–Cl crystals of unconventional stoichiometries on graphene surface from dilute solution at ambient conditions. Nat. Chem. 10, 776–779 (2018).
Zhang, W. et al. Unexpected stable stoichiometries of sodium chlorides. Science 342, 1502–1505 (2013).
Zhang, L. et al. Novel 2D CaCl crystals with metallicity, room-temperature ferromagnetism, heterojunction, piezoelectricity-like property and monovalent calcium ions. Natl Sci. Rev. 8, nwaa274 (2021).
Wang, L. et al. Microscopic kinetics pathway of salt crystallization in graphene nanocapillaries. Phys. Rev. Lett. 126, 136001 (2021).
Cui, Y. et al. Twisted epitaxy of gold nanodisks grown between twisted substrate layers of molybdenum disulfide. Science 383, 212–219 (2024).
Längle, M. et al. Two-dimensional few-atom noble gas clusters in a graphene sandwich. Nat. Mater. 23, 762–767 (2024).
Qian, J., Wu, H. & Wang, F. A generalized Knudsen theory for gas transport with specular and diffuse reflections. Nat. Commun. 14, 7386 (2023).
Arya, G., Chang, H.-C. & Maginn, E. J. Knudsen diffusivity of a hard sphere in a rough slit pore. Phys. Rev. Lett. 91, 026102 (2003).
Keerthi, A. et al. Ballistic molecular transport through two-dimensional channels. Nature 558, 420–424 (2018).
Qian, J., Li, Y., Wu, H. & Wang, F. Surface morphological effects on gas transport through nanochannels with atomically smooth walls. Carbon 180, 85–91 (2021).
Robeson, L. M. The upper bound revisited. J. Membr. Sci. 320, 390–400 (2008).
Li, H. et al. Ultrathin, molecular-sieving graphene oxide membranes for selective hydrogen separation. Science 342, 95–98 (2013).
Kim, H. W. et al. Selective gas transport through few-layered graphene and graphene oxide membranes. Science 342, 91–95 (2013).
Ding, L. et al. MXene molecular sieving membranes for highly efficient gas separation. Nat. Commun. 9, 155 (2018).
Zhang, P. et al. Strain-induced crumpling of graphene oxide lamellas to achieve fast and selective transport of H2 and CO2. Nat. Nanotechnol. 20, 1254–1261 (2025).
Kosinov, N., Gascon, J., Kapteijn, F. & Hensen, E. J. M. Recent developments in zeolite membranes for gas separation. J. Membr. Sci. 499, 65–79 (2016).
Knebel, A. & Caro, J. Metal–organic frameworks and covalent organic frameworks as disruptive membrane materials for energy-efficient gas separation. Nat. Nanotechnol. 17, 911–923 (2022).
Dave, S. H., Keller, B. D., Golmer, K. & Grossman, J. C. Six degrees of separation: connecting research with users and cost analysis. Joule 1, 410–415 (2017).
Zhou, D.-D. & Zhang, J.-P. On the role of flexibility for adsorptive separation. Acc. Chem. Res. 55, 2966–2977 (2022).
Wang, F. et al. Molecular transport under extreme confinement. Sci. China Phys. Mech. Astron. 65, 264601 (2022).
Faucher, S. et al. Critical knowledge gaps in mass transport through single-digit nanopores: a review and perspective. J. Phys. Chem. C 123, 21309–21326 (2019).
Yang, Q. et al. Ultrathin graphene-based membrane with precise molecular sieving and ultrafast solvent permeation. Nat. Mater. 16, 1198–1202 (2017).
Huang, L. et al. Reduced graphene oxide membranes for ultrafast organic solvent nanofiltration. Adv. Mater. 28, 8669–8674 (2016).
Chen, C. et al. Functionalized boron nitride membranes with ultrafast solvent transport performance for molecular separation. Nat. Commun. 9, 1902 (2018).
Guo, B.-Y. et al. MoS2 membranes for organic solvent nanofiltration: stability and structural control. J. Phys. Chem. Lett. 10, 4609–4617 (2019).
Wang, M. et al. Lanthanide transport in angstrom-scale MoS2-based two-dimensional channels. Sci. Adv. 10, eadh1330 (2024).
Dereka, B. et al. Crossover from hydrogen to chemical bonding. Science 371, 160–164 (2021).
Pagliero, D. et al. Slow water in engineered nanochannels revealed by color-center-enabled sensing. Nano Lett. 25, 9960–9966 (2025).
Li, Z. et al. Breakdown of the Nernst–Einstein relation in carbon nanotube porins. Nat. Nanotechnol. 18, 177–183 (2023).
Yang, Q. et al. Capillary condensation under atomic-scale confinement. Nature 588, 250–253 (2020).
Mi, B. Graphene oxide membranes for ionic and molecular sieving. Science 343, 740–742 (2014).
Li, F. et al. Highly efficient and selective extraction of gold by reduced graphene oxide. Nat. Commun. 13, 4472 (2022).
Wang, Z. et al. Understanding the aqueous stability and filtration capability of MoS2 membranes. Nano Lett. 17, 7289–7298 (2017).
Wang, Z. et al. Graphene oxide nanofiltration membranes for desalination under realistic conditions. Nat. Sustain. 4, 402–408 (2021).
Han, Q. et al. Highly efficient removal and sequestration of Cr(VI) in confined MoS2 interlayer nanochannels: performance and mechanism. Sep. Purif. Technol. 293, 121104 (2022).
Wang, Z. et al. Superselective removal of lead from water by two-dimensional MoS2 nanosheets and layer-stacked membranes. Environ. Sci. Technol. 54, 12602–12611 (2020).
Fu, Q. & Bao, X. Surface chemistry and catalysis confined under two-dimensional materials. Chem. Soc. Rev. 46, 1842–1874 (2017).
Li, H., Xiao, J., Fu, Q. & Bao, X. Confined catalysis under two-dimensional materials. Proc. Natl Acad. Sci. USA 114, 5930–5934 (2017).
Kim, S. et al. Neuromorphic van der Waals crystals for substantial energy generation. Nat. Commun. 12, 47 (2021).
Sugahara, A. et al. Negative dielectric constant of water confined in nanosheets. Nat. Commun. 10, 850 (2019).
Chen, W. et al. Two-dimensional quantum-sheet films with sub-1.2 nm channels for ultrahigh-rate electrochemical capacitance. Nat. Nanotechnol. 17, 153–158 (2022).
Yang, Y. et al. Ultrafast lithium-ion transport engineered by nanoconfinement effect. Adv. Mater. 37, e2416266 (2025).
Li, H. et al. Recent advances in zinc-ion dehydration strategies for optimized Zn–metal batteries. Chem. Soc. Rev. 53, 7742–7783 (2024).
Noy, A. & Darling, S. B. Nanofluidic computing makes a splash. Science 379, 143–144 (2023).
Suran, S. et al. Tunable release of ions from graphene oxide laminates for sustained antibacterial activity in a biomimetic environment. Small 21, 2304850 (2024).
Feynman, R. P. There’s plenty of room at the bottom. J. Microelectromechanical Syst. 1, 60–66 (1992).
Breck, D. W. in Zeolite Molecular Sieves: Structure, Chemistry, and Use 634–641 (Wiley, 1974).
Bondi, A. van der Waals volumes and radii. J. Phys. Chem. 68, 441–451 (1964).
Atkins, P. & De Paula, J. in Atkins’ Physical Chemistry 8th edn, 766 (W.H. Freeman, 2006).
Acknowledgements
X.C.Z. acknowledges support from the Hong Kong Global STEM Professorship Scheme and the Research Grants Council of Hong Kong (GRF Grant No. 11204123 and No. 11302225). W.Z. acknowledges the support from the Research Grants Council of Hong Kong (GRF CityU11310123 and CRF C1018-23G). M.W. acknowledges the support of the Global Research Assistant Professor Scheme of the CityUHK. J.J. acknowledges the funding support of the National Natural Science Foundation of China (Grant No. 22303072).
Author information
Authors and Affiliations
Contributions
M.W. and J.J. wrote the draft of the manuscript with input from all authors. C.H. contributed to the figure presentations. W.Z. and X.C.Z. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Chemistry thanks Limin Tong and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Jiang, J., Huang, C. et al. Matter in ångström-scale two-dimensional confinement. Nat Rev Chem (2026). https://doi.org/10.1038/s41570-026-00805-y
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41570-026-00805-y