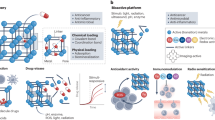
Abstract
In cancer diagnosis and treatment, ‘accurate diagnosis’ and ‘targeted therapy’ are the primary focus and key goals of many research groups. Metal–organic frameworks (MOFs) have attracted considerable attention in cancer diagnosis and treatment owing to their structural diversity and multifunctionality. Starting in 2010, MOFs with targeting capabilities have been gradually developed and applied in the field of biomedicine. This Review provides readers with a simple guide to help design bioenabled MOFs for specific bioapplications. First, construction strategies of MOFs with targeting functions are classified, including non-covalent interaction, covalent interaction and coordinative interaction strategies. Then, the applications of MOFs modified by antibodies, peptides, aptamers, carbohydrates, cell membranes and other targeting strategies are discussed. Finally, the future challenges and prospects for constructing MOFs with targeting functionalities are outlined to provide guidelines for the next generation of MOFs capable of achieving accurate cancer diagnosis and treatment and to accelerate the development of MOFs in clinical applications.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Kitagawa, S. Metal–organic frameworks (MOFs). Chem. Soc. Rev. 43, 5415–5418 (2014).
Li, B. et al. Emerging multifunctional metal–organic framework materials. Adv. Mater. 28, 8819–8860 (2016).
Tong, P.-H. et al. Metal–organic frameworks (MOFs) as host materials for the enhanced delivery of biomacromolecular therapeutics. Chem. Commun. 57, 12098–12110 (2021).
Wu, M. X. & Yang, Y. W. Metal–organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 29, 1606134 (2017).
Huang, X. et al. Advances in metal–organic framework-based nanozymes and their applications. Coord. Chem. Rev. 449, 214216 (2021).
Tong, P.-H. et al. Metal–organic frameworks (MOFs) for phototherapy and synergistic phototherapy of cancer. Coord. Chem. Rev. 526, 216381 (2025).
Kreno, L. E. et al. Metal–organic framework materials as chemical sensors. Chem. Rev. 112, 1105–1125 (2012).
Zhang, L. T., Zhou, Y. & Han, S. T. The role of metal–organic frameworks in electronic sensors. Angew. Chem. Int. Ed. 133, 15320–15340 (2021).
Luo, D. et al. Metal–organic frameworks (MOFs) as apt luminescent probes for the detection of biochemical analytes. J. Mater. Chem. B 11, 6802–6822 (2023).
Wang, A. et al. Biomedical metal–organic framework materials: perspectives and challenges. Adv. Funct. Mater. 34, 2308589 (2024).
Danhier, F. To exploit the tumor microenvironment: since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Rel. 244, 108–121 (2016).
Zhang, P. et al. Cancer nanomedicine toward clinical translation: obstacles, opportunities, and future prospects. Med 4, 147–167 (2023).
Wu, J. The enhanced permeability and retention (EPR) effect: the significance of the concept and methods to enhance its application. J. Pers. Med. 11, 771 (2021).
Byrne, J. D., Betancourt, T. & Brannon-Peppas, L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv. Drug Deliv. Rev. 60, 1615–1626 (2008).
Bazak, R., Houri, M., El Achy, S., Kamel, S. & Refaat, T. Cancer active targeting by nanoparticles: a comprehensive review of literature. J. Cancer Res. Clin. 141, 769–784 (2015).
Forgan, R. S. Reproducibility in research into metal–organic frameworks in nanomedicine. Commun. Mater. 5, 46 (2024).
Abánades Lázaro, I. et al. Metal–organic frameworks for biological applications. Nat. Rev. Methods Primers 4, 42 (2024).
Morales, M. A. & Halpern, J. M. Guide to selecting a biorecognition element for biosensors. Bioconjug. Chem. 29, 3231–3239 (2018).
Wang, Z. & Cohen, S. M. Postsynthetic modification of metal–organic frameworks. Chem. Soc. Rev. 38, 1315–1329 (2009).
Begum, S., Hassan, Z., Bräse, S., Wöll, C. & Tsotsalas, M. Metal–organic framework-templated biomaterials: recent progress in synthesis, functionalization, and applications. Acc. Chem. Res. 52, 1598–1610 (2019).
Mandal, S., Natarajan, S., Mani, P. & Pankajakshan, A. Post-synthetic modification of metal–organic frameworks toward applications. Adv. Funct. Mater. 31, 2006291 (2021).
Haque, A., Alenezi, K. M., Khan, M. S., Wong, W.-Y. & Raithby, P. R. Non-covalent interactions (NCIs) in π-conjugated functional materials: advances and perspectives. Chem. Soc. Rev. 52, 454–472 (2023).
Rest, C., Kandanelli, R. & Fernández, G. Strategies to create hierarchical self-assembled structures via cooperative non-covalent interactions. Chem. Soc. Rev. 44, 2543–2572 (2015).
Walkey, C. D. & Chan, W. C. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev. 41, 2780–2799 (2012).
Lee, J., Lee, J., Kim, J. Y. & Kim, M. Covalent connections between metal–organic frameworks and polymers including covalent organic frameworks. Chem. Soc. Rev. 52, 6379–6416 (2023).
Zhang, Z.-N. et al. Research and application of metal–organic framework in surface modification of biomaterials — a review. Metals 13, 1511 (2023).
Moses, J. E. & Moorhouse, A. D. The growing applications of click chemistry. Chem. Soc. Rev. 36, 1249–1262 (2007).
Kim, D., Kang, M., Ha, H., Hong, C. S. & Kim, M. Multiple functional groups in metal–organic frameworks and their positional regioisomerism. Coord. Chem. Rev. 438, 213892 (2021).
Paz, F. A. A. et al. Ligand design for functional metal–organic frameworks. Chem. Soc. Rev. 41, 1088–1110 (2012).
McGuire, C. V. & Forgan, R. S. The surface chemistry of metal–organic frameworks. Chem. Commun. 51, 5199–5217 (2015).
Kökçam-Demir, Ü et al. Coordinatively unsaturated metal sites (open metal sites) in metal–organic frameworks: design and applications. Chem. Soc. Rev. 49, 2751–2798 (2020).
Wu, A. M. & Senter, P. D. Arming antibodies: prospects and challenges for immunoconjugates. Nat. Biotechnol. 23, 1137–1146 (2005).
Schrama, D., Reisfeld, R. A. & Becker, J. C. Antibody targeted drugs as cancer therapeutics. Nat. Rev. Drug Discov. 5, 147–159 (2006).
Muyldermans, S., Cambillau, C. & Wyns, L. Recognition of antigens by single-domain antibody fragments: the superfluous luxury of paired domains. Trends Biochem. Sci. 26, 230–235 (2001).
Oh, J. Y. et al. Unveiling the power of cloaking metal–organic framework platforms via supramolecular antibody conjugation. ACS Nano 18, 15790–15801 (2024).
Hsieh, R. C.-E. et al. ATR-mediated CD47 and PD-L1 up-regulation restricts radiotherapy-induced immune priming and abscopal responses in colorectal cancer. Sci. Immunol. 7, eabl9330 (2022).
Ni, K. et al. Nanoscale metal–organic framework co-delivers TLR-7 agonists and anti-CD47 antibodies to modulate macrophages and orchestrate cancer immunotherapy. J. Am. Chem. Soc. 142, 12579–12584 (2020).
Tonigold, M. et al. Pre-adsorption of antibodies enables targeting of nanocarriers despite a biomolecular corona. Nat. Nanotechnol. 13, 862–869 (2018).
Garman, S. C., Wurzburg, B. A., Tarchevskaya, S. S., Kinet, J.-P. & Jardetzky, T. S. Structure of the Fc fragment of human IgE bound to its high-affinity receptor FcεRIα. Nature 406, 259–266 (2000).
Rader, C. Overview on concepts and applications of Fab antibody fragments. Curr. Protoc. Protein Sci. 55, 6–9 (2009).
Riley, R. S., June, C. H., Langer, R. & Mitchell, M. J. Delivery technologies for cancer immunotherapy. Nat. Rev. Drug Discov. 18, 175–196 (2019).
Alt, K. et al. Self-assembly of oriented antibody-decorated metal–organic framework nanocrystals for active-targeting applications. Adv. Mater. 34, 2106607 (2022).
Du, Y. et al. Potassium-based metal-organic framework with coordination-induced electrochemiluminescence for biosensing applications. Adv. Funct. Mater. 36, e13294 (2025).
David, A. Peptide ligand-modified nanomedicines for targeting cells at the tumor microenvironment. Adv. Drug Deliv. Rev. 119, 120–142 (2017).
Gray, B. P. & Brown, K. C. Combinatorial peptide libraries: mining for cell-binding peptides. Chem. Rev. 114, 1020–1081 (2014).
Wu, C.-H., Liu, I.-J., Lu, R.-M. & Wu, H.-C. Advancement and applications of peptide phage display technology in biomedical science. J. Biomed. Sci. 23, 1–14 (2016).
Merrifield, B. Solid phase synthesis. Science 232, 341–347 (1986).
Molek, P., Strukelj, B. & Bratkovic, T. Peptide phage display as a tool for drug discovery: targeting membrane receptors. Molecules 16, 857–887 (2011).
Segura, J. L., Mancheño, M. J. & Zamora, F. Covalent organic frameworks based on Schiff-base chemistry: synthesis, properties and potential applications. Chem. Soc. Rev. 45, 5635–5671 (2016).
Wu, S. et al. Peptide-functionalized metal–organic framework nanocomposite for ultrasensitive detection of secreted protein acidic and rich in cysteine with practical application. Biosens. Bioelectron. 169, 112613 (2020).
Tai, I. T. & Tang, M. J. SPARC in cancer biology: its role in cancer progression and potential for therapy. Drug Resist. Update 11, 231–246 (2008).
Inoue, M. et al. Identification of SPARC as a candidate target antigen for immunotherapy of various cancers. Int. J. Cancer 127, 1393–1403 (2010).
Zhang, L. et al. ZD2-engineered gold nanostar@metal-organic framework nanoprobes for T1-weighted magnetic resonance imaging and photothermal therapy specifically toward triple-negative breast cancer. Adv. Healthc. Mater. 7, 1801144 (2018).
Javid, H. et al. RGD peptide in cancer targeting: benefits, challenges, solutions, and possible integrin–RGD interactions. Cancer Med. 13, e6800 (2024).
Rowe, M. D., Thamm, D. H., Kraft, S. L. & Boyes, S. G. Polymer-modified gadolinium metal–organic framework nanoparticles used as multifunctional nanomedicines for the targeted imaging and treatment of cancer. Biomacromolecules 10, 983–993 (2009).
Wang, D. et al. In situ one-pot synthesis of MOF–polydopamine hybrid nanogels with enhanced photothermal effect for targeted cancer therapy. Adv. Sci. 5, 1800287 (2018).
He, Y. et al. Pulmonary targeting crosslinked cyclodextrin metal–organic frameworks for lung cancer therapy. Adv. Funct. Mater. 31, 2004550 (2021).
Cedrún-Morales, M. et al. Clickable polymer-based coatings for modulating the interaction of metal–organic framework nanocrystals with living cells. ACS Appl. Mater. Interfaces 17, 24994–25010 (2025).
Bao, J. et al. Coordination self-assembled AuTPyP-Cu metal–organic framework nanosheets with pH/ultrasound dual-responsiveness for synergistically triggering cuproptosis-augmented chemotherapy. ACS Nano 18, 9100–9113 (2024).
Del Pino, P. et al. Protein corona formation around nanoparticles — from the past to the future. Mater. Horiz. 1, 301–313 (2014).
Oh, J. Y. et al. Protein-precoated surface of metal–organic framework nanoparticles for targeted delivery. Small 19, 2300218 (2023).
Chen, Y. et al. Metal−organic frameworks nucleated by silk fibroin and modified with tumor-targeting peptides for targeted multimodal cancer therapy. Adv. Sci. 10, 2302700 (2023).
Huang, J. et al. Biomimetic metal–organic framework gated nanoplatform for sonodynamic therapy against extensively drug resistant bacterial lung infection. Adv. Sci. 11, 2402473 (2024).
Cai, M. et al. Enhanced lysosomal escape of cell penetrating peptide-functionalized metal–organic frameworks for co-delivery of survivin siRNA and oridonin. J. Colloid Interface Sci. 646, 370–380 (2023).
Chen, Y. L. et al. Fibrin-targeting metal–organic framework nanoagent for NIR-triggered photothermal thrombus ablation. Adv. Healthc. Mater. 10, e03394 (2025).
Song, Y. et al. A bimetallic metal–organic-framework-based biomimetic nanoplatform enhances anti-leukemia immunity via synchronizing DNA demethylation and RNA hypermethylation. Adv. Mater. 35, 2210895 (2023).
Lei, S. et al. ALPPL2-binding peptide facilitates targeted mRNA delivery for efficient hepatocellular carcinoma gene therapy. Adv. Funct. Mater. 32, 2204342 (2022).
Wang, Q. et al. Peptide-oligonucleotide nanohybrids designed for precise gene therapy of rheumatoid arthritis. Adv. Mater. 37, 2500883 (2025).
Cardle, I. I., Scherer, D. R., Jensen, M. C., Pun, S. H. & Sellers, D. L. In situ bioconjugation of synthetic peptides onto universal chimeric antigen receptor T cells for targeted cancer immunotherapies. ACS Nano 19, 5750–5768 (2025).
Zhou, J. & Rossi, J. Aptamers as targeted therapeutics: current potential and challenges. Nat. Rev. Drug Discov. 16, 181–202 (2017).
Zhou, P., Shi, R., Yao, J.-F., Sheng, C.-F. & Li, H. Supramolecular self-assembly of nucleotide–metal coordination complexes: from simple molecules to nanomaterials. Coord. Chem. Rev. 292, 107–143 (2015).
He, C., Lu, K., Liu, D. & Lin, W. Nanoscale metal–organic frameworks for the co-delivery of cisplatin and pooled siRNAs to enhance therapeutic efficacy in drug-resistant ovarian cancer cells. J. Am. Chem. Soc. 136, 5181–5184 (2014).
Ning, W. et al. Imparting designer biorecognition functionality to metal–organic frameworks by a DNA-mediated surface engineering strategy. Small 14, 1703812 (2018).
Wang, S. et al. DNA-functionalized metal–organic framework nanoparticles for intracellular delivery of proteins. J. Am. Chem. Soc. 141, 2215–2219 (2019).
Zhang, J. et al. PD-L1 aptamer-functionalized metal–organic framework nanoparticles for robust photo-immunotherapy against cancer with enhanced safety. Angew. Chem. Int. Ed. 62, e202214750 (2023).
Bobko, A. A. et al. Interstitial inorganic phosphate as a tumor microenvironment marker for tumor progression. Sci. Rep. 7, 41233 (2017).
Wei, Y. et al. Bioorthogonal activation of TLR7 agonists provokes innate immunity to reinforce aptamer-based checkpoint blockade. ACS Nano 17, 5808–5820 (2023).
Zhu, J. et al. Boosting endogenous copper (I) for biologically safe and efficient bioorthogonal catalysis via self-adaptive metal–organic frameworks. J. Am. Chem. Soc. 145, 1955–1963 (2023).
Saxon, E. & Bertozzi, C. R. Cell surface engineering by a modified Staudinger reaction. Science 287, 2007–2010 (2000).
Morris, W., Briley, W. E., Auyeung, E., Cabezas, M. D. & Mirkin, C. A. Nucleic acid–metal organic framework (MOF) nanoparticle conjugates. J. Am. Chem. Soc. 136, 7261–7264 (2014).
Jain, A. & Cheng, K. The principles and applications of avidin-based nanoparticles in drug delivery and diagnosis. J. Control. Rel. 245, 27–40 (2017).
Zhao, Q. et al. Target reprogramming lysosomes of CD8+ T cells by a mineralized metal–organic framework for cancer immunotherapy. Adv. Mater. 33, 2100616 (2021).
Marradi, M., Chiodo, F., García, I. & Penadés, S. Glyconanoparticles as multifunctional and multimodal carbohydrate systems. Chem. Soc. Rev. 42, 4728–4745 (2013).
Kang, B., Opatz, T., Landfester, K. & Wurm, F. R. Carbohydrate nanocarriers in biomedical applications: functionalization and construction. Chem. Soc. Rev. 44, 8301–8325 (2015).
Khan, H. et al. In glyco-nanoparticles: new drug delivery systems in cancer therapy. Semin. Cancer Biol. 69, 24–42 (2021).
Huang, K.-W. et al. Galactose derivative-modified nanoparticles for efficient siRNA delivery to hepatocellular carcinoma. Biomacromolecules 19, 2330–2339 (2018).
Xie, H.-N. et al. Targeted delivery of maytansine to liver cancer cells via galactose-modified supramolecular two-dimensional glycomaterial. Chem. Commun. 58, 5029–5032 (2022).
Ye, W. et al. Design, synthesis and biological evaluation of the positional isomers of the galactose conjugates able to target hepatocellular carcinoma cells via ASGPR-mediated cellular uptake and cytotoxicity. Eur. J. Med. Chem. 264, 115988 (2024).
Ye, Z. et al. An integrated therapeutic delivery system for enhanced treatment of hepatocellular carcinoma. Adv. Funct. Mater. 28, 1706600 (2018).
Hu, J. et al. Fabrication of glyco-metal-organic frameworks for targeted interventional photodynamic/chemotherapy for hepatocellular carcinoma through percutaneous transperitoneal puncture. Adv. Funct. Mater. 30, 1910084 (2020).
Alhussan, A. et al. Lipid-based nanoparticles external triggered release strategies in cancer nanomedicine. J. Nanobiotechnol. 23, 641 (2025).
Dou, Y., Hynynen, K. & Allen, C. To heat or not to heat: challenges with clinical translation of thermosensitive liposomes. J. Control. Rel. 249, 63–73 (2017).
Al-Nadaf, A. H., Dahabiyeh, L. A., Bardaweel, S., Mahmoud, N. N. & Jawarneh, S. Functionalized mesoporous silica nanoparticles by lactose and hydrophilic polymer as a hepatocellular carcinoma drug delivery system. J. Drug Deliv. Sci. Technol. 56, 101504 (2020).
Zhang, P. et al. Evoking and enhancing ferroptosis of cancer stem cells by a liver-targeted and metal–organic framework-based drug delivery system inhibits the growth and lung metastasis of hepatocellular carcinoma. Chem. Eng. J. 454, 140044 (2023).
Li, Y. et al. Lactate-responsive gene editing to synergistically enhance macrophage-mediated cancer immunotherapy. Small 19, 2301519 (2023).
Mastrotto, F. et al. Sulfation at glycopolymer side chains switches activity at the macrophage mannose receptor (CD206) in vitro and in vivo. J. Am. Chem. Soc. 144, 23134–23147 (2022).
Jaynes, J. M. et al. Mannose receptor (CD206) activation in tumor-associated macrophages enhances adaptive and innate antitumor immune responses. Sci. Transl. Med. 12, eaax6337 (2020).
Demir Duman, F., Monaco, A., Foulkes, R., Becer, C. R. & Forgan, R. S. Glycopolymer-functionalized MOF-808 nanoparticles as a cancer-targeted dual drug delivery system for carboplatin and floxuridine. ACS Appl. Nano Mater. 5, 13862–13873 (2022).
Sun, Q. et al. Hyaluronic acid-targeted and pH-responsive drug delivery system based on metal–organic frameworks for efficient antitumor therapy. Biomaterials 223, 119473 (2019).
Peach, R. J., Hollenbaugh, D., Stamenkovic, I. & Aruffo, A. Identification of hyaluronic acid binding sites in the extracellular domain of CD44. J. Cell Biol. 122, 257–264 (1993).
Mattheolabakis, G., Milane, L., Singh, A. & Amiji, M. M. Hyaluronic acid targeting of CD44 for cancer therapy: from receptor biology to nanomedicine. J. Drug Target. 23, 605–618 (2015).
Pan, M. M. et al. Bimetallic ions functionalized metal–organic-framework nanozyme for tumor microenvironment regulating and enhanced photodynamic therapy for hypoxic tumor. Adv. Healthc. Mater. 12, 2300821 (2023).
Liu, W. et al. Recent advances in cell membrane coated metal–organic frameworks (MOFs) for tumor therapy. J. Mater. Chem. B 9, 4459–4474 (2021).
Yan, H. et al. Engineering cell membrane-based nanotherapeutics to target inflammation. Adv. Sci. 6, 1900605 (2019).
Fang, R. H., Gao, W. & Zhang, L. Targeting drugs to tumours using cell membrane-coated nanoparticles. Nat. Rev. Clin. Oncol. 20, 33–48 (2023).
Dash, P., Piras, A. M. & Dash, M. Cell membrane coated nanocarriers — an efficient biomimetic platform for targeted therapy. J. Control. Rel. 327, 546–570 (2020).
Nguyen, P. H. D., Jayasinghe, M. K., Le, A. H., Peng, B. & Le, M. T. Advances in drug delivery systems based on red blood cells and their membrane-derived nanoparticles. ACS Nano 17, 5187–5210 (2023).
Wang, S. et al. Drug targeting via platelet membrane-coated nanoparticles. Small Struct. 1, 2000018 (2020).
Oroojalian, F., Beygi, M., Baradaran, B., Mokhtarzadeh, A. & Shahbazi, M. A. Immune cell membrane-coated biomimetic nanoparticles for targeted cancer therapy. Small 17, 2006484 (2021).
Wang, H. et al. Cell membrane biomimetic nanoparticles for inflammation and cancer targeting in drug delivery. Biomater. Sci. 8, 552–568 (2020).
Zhuang, J. et al. Targeted gene silencing in vivo by platelet membrane-coated metal–organic framework nanoparticles. Sci. Adv. 6, eaaz6108 (2020).
Wei, X. et al. Nanoparticle functionalization with platelet membrane enables multifactored biological targeting and detection of atherosclerosis. ACS Nano 12, 109–116 (2018).
Hamzeh-Cognasse, H. et al. Platelets and infections — complex interactions with bacteria. Front. Immunol. 6, 82 (2015).
Zhang, L. et al. Erythrocyte membrane cloaked metal–organic framework nanoparticle as biomimetic nanoreactor for starvation-activated colon cancer therapy. ACS Nano 12, 10201–10211 (2018).
Wang, S. et al. Membrane cholesterol depletion enhances enzymatic activity of cell-membrane-coated metal-organic-framework nanoparticles. Angew. Chem. Int. Ed. 134, e202203115 (2022).
Pan, H. et al. At the crossroad of nanotechnology and cancer cell membrane coating: expanding horizons with engineered nanoplatforms for advanced cancer therapy harnessing homologous tumor targeting. Coord. Chem. Rev. 506, 215712 (2024).
Alsaiari, S. K. et al. Sustained and targeted delivery of checkpoint inhibitors by metal–organic frameworks for cancer immunotherapy. Sci. Adv. 7, eabe7174 (2021).
Liu, Z. et al. A nature-inspired metal–organic framework discriminator for differential diagnosis of cancer cell subtypes. Angew. Chem. Int. Ed. 133, 15564–15572 (2021).
Low, P. S., Henne, W. A. & Doorneweerd, D. D. Discovery and development of folic-acid-based receptor targeting for imaging and therapy of cancer and inflammatory diseases. Acc. Chem. Res. 41, 120–129 (2008).
Zhao, X., Li, H. & Lee, R. J. Targeted drug delivery via folate receptors. Exp. Opin. Drug Deliv. 5, 309–319 (2008).
Wei, Y. et al. MOFs modulate copper trafficking in tumor cells for bioorthogonal therapy. Nano Lett. 24, 1341–1350 (2024).
Liu, J., Liu, T., Du, P., Zhang, L. & Lei, J. Metal–organic framework (MOF) hybrid as a tandem catalyst for enhanced therapy against hypoxic tumor cells. Angew. Chem. Int. Ed. 131, 7890–7894 (2019).
Zielonka, J. et al. Mitochondria-targeted triphenylphosphonium-based compounds: syntheses, mechanisms of action, and therapeutic and diagnostic applications. Chem. Rev. 117, 10043–10120 (2017).
Cheng, X. et al. Application prospects of triphenylphosphine-based mitochondria-targeted cancer therapy. Cancers 15, 666 (2023).
Wang, F. et al. A biocompatible heterogeneous MOF–Cu catalyst for in vivo drug synthesis in targeted subcellular organelles. Angew. Chem. Int. Ed. 58, 6987–6992 (2019).
Peng, X. et al. Autonomous metal–organic framework nanorobots for active mitochondria-targeted cancer therapy. Sci. Adv. 9, eadh1736 (2023).
Liu, L., Ma, X., Chang, Y., Guo, H. & Wang, W. Biosensors with boronic acid-based materials as the recognition elements and signal labels. Biosensors 13, 785 (2023).
Liang, J., Liu, J., Lord, M. S., Wang, Y. & Liang, K. De Novo engineering of metal–organic framework-printed in vitro diagnostic devices for specific capture and release of tumor cells. Small 17, 2103590 (2021).
Zhang, S. et al. Boronic acid-rich lanthanide metal–organic frameworks enable deep proteomics with ultratrace biological samples. Adv. Mater. 36, 2401559 (2024).
Fytory, M. et al. Dual-ligated metal–organic framework as novel multifunctional nanovehicle for targeted drug delivery for hepatic cancer treatment. Sci. Rep. 11, 19808 (2021).
Björgvinsdóttir, U. J., Larsen, J. B., Bak, M., Andresen, T. L. & Münter, R. Targeting antibodies dissociate from drug delivery liposomes during blood circulation. J. Control. Rel. 379, 982–992 (2025).
Ioannidis, J. P., Kim, B. Y. & Trounson, A. How to design preclinical studies in nanomedicine and cell therapy to maximize the prospects of clinical translation. Nat. Biomed. Eng. 2, 797–809 (2018).
Stater, E. P., Sonay, A. Y., Hart, C. & Grimm, J. The ancillary effects of nanoparticles and their implications for nanomedicine. Nat. Nanotechnol. 16, 1180–1194 (2021).
Ettlinger, R. et al. Toxicity of metal–organic framework nanoparticles: from essential analyses to potential applications. Chem. Soc. Rev. 51, 464–484 (2022).
Acknowledgements
M.L. thanks the support of the National Natural Science Foundation of China (grant no. 2230822), Shenzhen University Third-Phase Project of Constructing High-Level University (grant no. 000001032104), the Research Team Cultivation Program of Shenzhen University (grant no. 2023QNT005) and Guangdong Province Key Areas Special Project for Regular Colleges and Universities (grant no. 2024ZDZX2018). The authors also thank the support from the Shenzhen University 2035 Program for Excellent Research (nos. 00000208 and 00000225) and the Shenzhen Science and Technology Program (grant no. RCBS20231211090515015). X.C. thanks the support of the Guangdong Basic and Applied Basic Research Foundation (2023B1515120001). P.T. thanks the support of the China Postdoctoral Science Foundation (grant no. 2025M771167). T.D.J. thanks the University of Bath and the Open Research Fund of the School of Chemistry and Chemical Engineering, Henan Normal University (2020ZD01) for their support.
Author information
Authors and Affiliations
Contributions
P.T. and M.L. wrote and researched data for the manuscript. All authors contributed substantially to the discussion of the content and research data for the article. X.C., M.L., T.D.J. and X.P. edited and revised the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Chemistry thanks David Fairen-Jimenez, who co-reviewed with Nathalie Tarabay; Ja-Hyoung Ryu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tong, P., Wang, R., Zhou, Y. et al. Enhancing the targeting ability of metal–organic frameworks in cancer therapy and diagnosis. Nat Rev Chem 10, 258–276 (2026). https://doi.org/10.1038/s41570-026-00807-w
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41570-026-00807-w