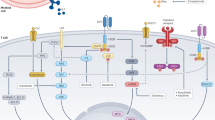
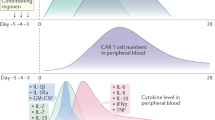
Abstract
As clinical advances with chimeric antigen receptor (CAR) T cells are increasingly described and the potential for extending their therapeutic benefit grows, optimizing the implementation of this therapeutic modality is imperative. The recognition and management of cytokine release syndrome (CRS) marked a milestone in this field; however, beyond the understanding gained in treating CRS, a host of additional toxicities and/or potential late effects of CAR T cell therapy warrant further investigation. A multicentre initiative involving experts in paediatric cell therapy, supportive care and/or study of late effects from cancer and haematopoietic stem cell transplantation was convened to facilitate the comprehensive study of extended CAR T cell-mediated toxicities and establish a framework for new systematic investigations of CAR T cell-related adverse events. Together, this group identified six key focus areas: extended monitoring of neurotoxicity and neurocognitive function, psychosocial considerations, infection and immune reconstitution, other end organ toxicities, evaluation of subsequent neoplasms, and strategies to optimize remission durability. Herein, we present the current understanding, gaps in knowledge and future directions of research addressing these CAR T cell-related outcomes. This systematic framework to study extended toxicities and optimization strategies will facilitate the translation of acquired experience and knowledge for optimal application of CAR T cell therapies.
Key points
-
A host of extended subacute toxicities and/or potential late effects of CAR T cell therapy and cytokine release syndrome (CRS) in children warrant further investigation with a systematic approach and with prospective studies.
-
Herein, we provide a contextual framework for evaluating toxicities of novel CAR T cell constructs, as well as an overview of strategies currently used to optimize responses and assess other long-term outcomes of CAR T cell therapy.
-
Longitudinal neurocognitive evaluations, established time points for and interpretation of neuroimaging, and development of biomarkers for assessment of neurotoxicity risk and quantification of neurological injury across CAR T cell trials are needed to further optimize neurological outcomes.
-
Comprehensive care models that include the patient, family, and referring and primary teams should be embedded into all CAR T cell therapy programmes to provide comfort, address expectations, and understand psychosocial risk factors and educational needs.
-
CAR T cell recipients are at high risk of infection owing to a host of factors and developing optimal guidelines for infection prevention during both acute CRS and longitudinally is necessary to optimize outcomes. Monitoring immune reconstitution after treatment will help identify future risks of infection and how vaccination strategies might be effective in this population.
-
Extending the durability of remission following CAR T cell treatment remains a primary goal, for which the study of late effects remains imperative. The results of ongoing studies of a host of strategies will inform the role of remission consolidation and risk stratification in identifying patients at highest risk of relapse.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Grupp, S. A. et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N. Engl. J. Med. 368, 1509–1518 (2013).
Maude, S. L. et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl. J. Med. 371, 1507–1517 (2014).
Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
Schuster, S. J. et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N. Engl. J. Med. 380, 45–56 (2019).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
US Food and Drug Administration. FDA approves brexucabtagene autoleucel for relapsed or refractory mantle cell lymphoma. fda.gov https://www.fda.gov/drugs/fda-approves-brexucabtagene-autoleucel-relapsed-or-refractory-mantle-cell-lymphoma (2020).
Rosenbaum, L. Tragedy, perseverance, and chance – the story of CAR-T therapy. N. Engl. J. Med. 377, 1313–1315 (2017).
Lee, D. W. et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 124, 188–195 (2014).
Lee, D. W. et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol. Blood Marrow Transplant. 25, 625–638 (2019).
Brudno, J. N. & Kochenderfer, J. N. Recent advances in CAR T-cell toxicity: mechanisms, manifestations and management. Blood Rev. 34, 45–55 (2019).
Maus, M. V. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune effector cell-related adverse events. J. Immunother. Cancer 8 e001511 (2020).
Strati, P. et al. Clinical efficacy of anakinra to mitigate CAR T-cell therapy-associated toxicity in large B-cell lymphoma. Blood Adv. 4, 3123–3127 (2020).
Shah, N. N. et al. CD4/CD8 T-cell selection affects chimeric antigen receptor (CAR) T-cell potency and toxicity: updated results from a phase I anti-CD22 CAR T-cell trial. J. Clin. Oncol. 38, 1938–1950 (2020).
Gardner, R. A. et al. Preemptive mitigation of CD19 CAR T-cell cytokine release syndrome without attenuation of antileukemic efficacy. Blood. 134, 2149–2158 (2019).
Perales, M. A., Kebriaei, P., Kean, L. S. & Sadelain, M. Building a safer and faster CAR: seatbelts, airbags, and CRISPR. Biol. Blood Marrow Transplant. 24, 27–31 (2018).
Weber, E. W. et al. Pharmacologic control of CAR-T cell function using dasatinib. Blood Adv. 3, 711–717 (2019).
Teachey, D. T. et al. Identification of predictive biomarkers for cytokine release syndrome after chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Cancer Discov. 6, 664–679 (2016).
Lee D. W. & Shah N. N. Chimeric Antigen Receptor T-Cell Therapies for Cancer: A Practical Guide, 1st edn (Elsevier, 2019).
Gust, J., Taraseviciute, A. & Turtle, C. J. Neurotoxicity associated with CD19-targeted CAR-T cell therapies. CNS Drugs 32, 1091–1101 (2018).
Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 385, 517–528 (2015).
Gardner, R. A. et al. Intent-to-treat leukemia remission by CD19 CAR T cells of defined formulation and dose in children and young adults. Blood. 129, 3322–3331 (2017).
Fry, T. J. et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. 24, 20–28 (2018).
Gust, J. et al. Endothelial activation and blood-brain barrier disruption in neurotoxicity after adoptive immunotherapy with CD19 CAR-T cells. Cancer Discov. 7, 1404–1419 (2017).
Schuster, S. J. et al. Chimeric antigen receptor T cells in refractory B-cell lymphomas. N. Engl. J. Med. 377, 2545–2554 (2017).
Torre, M. et al. Neuropathology of a case with fatal CAR T-cell-associated cerebral edema. J. Neuropathol. Exp. Neurol. 77, 877–882 (2018).
Santomasso, B. D. et al. Clinical and biological correlates of neurotoxicity associated with CAR T-cell therapy in patients with B-cell acute lymphoblastic leukemia. Cancer Discov. 8, 958–971 (2018).
Gofshteyn, J. S. et al. Neurotoxicity after CTL019 in a pediatric and young adult cohort. Ann. Neurol. 84, 537–546 (2018).
Newman, H. et al. CD19-targeted chimeric antigen receptor (CAR) T cells in CNS relapsed acute lymphoblastic leukemia (ALL) [abstract]. J. Clin. Oncol. 38 (Suppl. 15), 10511 (2020).
Teachey, D. T., Bishop, M. R., Maloney, D. G. & Grupp, S. A. Toxicity management after chimeric antigen receptor T cell therapy: one size does not fit ‘ALL’. Nat. Rev. Clin. Oncol. 15, 218 (2018).
Gust, J. et al. Glial injury in neurotoxicity after pediatric CD19-directed chimeric antigen receptor T cell therapy. Ann. Neurol. 86, 42–54. (2019).
Shalabi, H. et al. Systematic evaluation of neurotoxicity in children and young adults undergoing CD22 chimeric antigen receptor T-cell therapy. J. Immunother. 41, 350–358 (2018).
Ruark, J. et al. Patient-reported neuropsychiatric outcomes of long-term survivors after chimeric antigen receptor T cell therapy. Biol. Blood Marrow Transplant. 26, 34–43 (2020).
Cheung, Y. T. et al. Leukoencephalopathy and long-term neurobehavioural, neurocognitive, and brain imaging outcomes in survivors of childhood acute lymphoblastic leukaemia treated with chemotherapy: a longitudinal analysis. Lancet Haematol. 3, e456–e466 (2016).
Cheung, Y. T. & Krull, K. R. Neurocognitive outcomes in long-term survivors of childhood acute lymphoblastic leukemia treated on contemporary treatment protocols: a systematic review. Neurosci. Biobehav. Rev. 53, 108–120 (2015).
Mulhern, R. K. & Butler, R. W. Neurocognitive sequelae of childhood cancers and their treatment. Pediatr. Rehabil. 7, 1–14 discussion 15-16 (2004).
Mahadeo, K. M. et al. Management guidelines for paediatric patients receiving chimeric antigen receptor T cell therapy. Nat. Rev. Clin. Oncol. 16, 45–63 (2019).
Hay, K. A. et al. Kinetics and biomarkers of severe cytokine release syndrome after CD19 chimeric antigen receptor-modified T-cell therapy. Blood. 130, 2295–2306 (2017).
Khalil, M. et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 14, 577–589 (2018).
Steineck, A. et al. Psychosocial care for children receiving chimeric antigen receptor (CAR) T-cell therapy. Pediatr. Blood Cancer 67, e28249 (2020).
Sun, W. et al. Outcome of children with multiply relapsed B-cell acute lymphoblastic leukemia: a therapeutic advances in childhood leukemia & lymphoma study. Leukemia. 32, 2316–2325 (2018).
Callahan, C. et al. Pediatric survivorship: considerations following CAR T-cell therapy. Clin. J. Oncol. Nurs. 23, 35–41 (2019).
Leahy, A. B., Feudtner, C. & Basch, E. Symptom monitoring in pediatric oncology using patient-reported outcomes: why, how, and where next. Patient 11, 147–153 (2018).
Kluetz, P. G., O’Connor, D. J. & Soltys, K. Incorporating the patient experience into regulatory decision making in the USA, Europe, and Canada. Lancet Oncol. 19, e267–e274 (2018).
Kirch, R. et al. Advancing a comprehensive cancer care agenda for children and their families: Institute of Medicine Workshop highlights and next steps. CA Cancer J. Clin. 66, 398–407 (2016).
Kazak, A. E. et al. Psychosocial assessment as a standard of care in pediatric cancer. Pediatr. Blood Cancer 62, S426–S459 (2015).
Kim, J. et al. Use of PRO measures to inform tolerability in oncology trials: implications for clinical review, IND safety reporting, and clinical site inspections. Clin. Cancer Res. 24, 1780–1784 (2018).
Schepers, S. A., Haverman, L., Zadeh, S., Grootenhuis, M. A. & Wiener, L. Healthcare professionals’ preferences and perceived barriers for routine assessment of patient-reported outcomes in pediatric oncology practice: moving toward international processes of change. Pediatr. Blood Cancer 63, 2181–2188 (2016).
Yen, H. J. et al. Patient-reported outcomes in survivors of childhood hematologic malignancies with hematopoietic stem cell transplant. Blood. 135, 1847–1858 (2020).
Maziarz, R. T. et al. Patient-reported long-term quality of life after tisagenlecleucel in relapsed/refractory diffuse large B-cell lymphoma. Blood Adv. 4, 629–637 (2020).
Sidana, S. et al. Patient experience of chimeric antigen receptor (CAR)-T cell therapy vs. stem cell transplant: longitudinal patient reported adverse events, cognition and quality of life [abstract]. Blood. 134 (Suppl. 1), 794 (2019).
Laetsch, T. W. et al. Patient-reported quality of life after tisagenlecleucel infusion in children and young adults with relapsed or refractory B-cell acute lymphoblastic leukaemia: a global, single-arm, phase 2 trial. Lancet Oncol. 20, 1710–1718 (2019).
Chakraborty, R. et al. Patient-reported outcomes with chimeric antigen receptor T cell therapy: challenges and opportunities. Biol. Blood Marrow Transplant. 25, e155–e162 (2019).
Centers for Medicare & Medicaid Services. MEDCAC Meeting 8/22/2018 - Chimeric Antigen Receptor (CAR) T-Cell Therapy and Patient Reported Outcomes. 2018.
Pinheiro, L. C. et al. Child and adolescent self-report symptom measurement in pediatric oncology research: a systematic literature review. Qual. Life Res. 27, 291–319 (2018).
Vora, S. B. et al. Infectious complications following CD19 chimeric antigen receptor T-cell therapy for children, adolescents, and young adults. Open. Forum Infect. Dis. 7, ofaa121 (2020).
Hill, J. A. et al. Infectious complications of CD19-targeted chimeric antigen receptor-modified T-cell immunotherapy. Blood. 131, 121–130 (2018).
Park, J. H. et al. Cytokine release syndrome grade as a predictive marker for infections in patients with relapsed or refractory B-cell acute lymphoblastic leukemia treated with chimeric antigen receptor T cells. Clin. Infect. Dis. 67, 533–540 (2018).
Mahmoudjafari, Z. et al. American Society for Blood and Marrow Transplantation Pharmacy Special Interest Group survey on chimeric antigen receptor T cell therapy administrative, logistic, and toxicity management practices in the United States. Biol. Blood Marrow Transplant. 25, 26–33 (2019).
Logue, J. M. et al. Immune reconstitution and associated infections following axicabtagene ciloleucel in relapsed or refractory large B-cell lymphoma. Haematologica https://doi.org/10.3324/haematol.2019.238634 (2020).
Cordeiro, A. et al. Late events after treatment with CD19-targeted chimeric antigen receptor modified T cells. Biol. Blood Marrow Transplant. 26, 26–33 (2020).
Fried, S. et al. Early and late hematologic toxicity following CD19 CAR-T cells. Bone Marrow Transplant. 54, 1643–1650 (2019).
Locke, F. L. et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 20, 31–42 (2019).
Kochenderfer, J. N. et al. Long-duration complete remissions of diffuse large B cell lymphoma after anti-CD19 chimeric antigen receptor T cell therapy. Mol. Ther. 25, 2245–2253 (2017).
Bhoj, V. G. et al. Persistence of long-lived plasma cells and humoral immunity in individuals responding to CD19-directed CAR T-cell therapy. Blood. 128, 360–370 (2016).
Hill, J. A. et al. Durable preservation of antiviral antibodies after CD19-directed chimeric antigen receptor T-cell immunotherapy. Blood Adv. 3, 3590–3601 (2019).
Deya-Martinez, A. et al. Kinetics of humoral deficiency in CART19-treated children and young adults with acute lymphoblastic leukaemia. Bone Marrow Transplant. https://doi.org/10.1038/s41409-020-01027-6 (2020).
Arnold, D. E. et al. Subcutaneous immunoglobulin replacement following CD19-specific chimeric antigen receptor T-cell therapy for B-cell acute lymphoblastic leukemia in pediatric patients. Pediatr. Blood Cancer 67, e28092 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04410900 (2020).
Hill, J. A. & Seo, S. How we prevent infections in patients receiving CD19-targeted chimeric antigen receptor T-cells for B-cell malignancies. Blood. 136, 925–935 (2020).
Sandler, R. D. et al. Diagnosis and management of secondary HLH/MAS following HSCT and CAR-T cell therapy in adults; a review of the literature and a survey of practice within EBMT centres on behalf of the Autoimmune Diseases Working Party (ADWP) and Transplant Complications Working Party (TCWP). Front. Immunol. 11, 524 (2020).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy – assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
Hashmi, H. et al. Haemophagocytic lymphohistiocytosis has variable time to onset following CD19 chimeric antigen receptor T cell therapy. Br. J. Haematol. 187, e35–e38. (2019).
Jain, T. et al. Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies. Blood Adv. 4, 3776–3787 (2020).
Strati, P. et al. Hematopoietic recovery and immune reconstitution after axicabtagene ciloleucel in patients with large B-cell lymphoma. Haematologica https://doi.org/10.3324/haematol.2020.254045 (2020).
Feng, X. et al. Cytokine signature profiles in acquired aplastic anemia and myelodysplastic syndromes. Haematologica. 96, 602–606 (2011).
Morales-Mantilla, D. E. & King, K. Y. The role of interferon-gamma in hematopoietic stem cell development, homeostasis, and disease. Curr. Stem Cell Rep. 4, 264–271 (2018).
Wang, S., Degar, B. A., Zieske, A., Shafi, N. Q. & Rose, M. G. Hemophagocytosis exacerbated by G-CSF/GM-CSF treatment in a patient with myelodysplasia. Am. J. Hematol. 77, 391–396 (2004).
Kuter, D. J. Thrombopoietin and thrombopoietin mimetics in the treatment of thrombocytopenia. Annu. Rev. Med. 60, 193–206 (2009).
Sterner, R. M. et al. GM-CSF inhibition reduces cytokine release syndrome and neuroinflammation but enhances CAR-T cell function in xenografts. Blood. 133, 697–709 (2019).
Haen, S. P. et al. Poor graft function can be durably and safely improved by CD34+-selected stem cell boosts after allogeneic unrelated matched or mismatched hematopoietic cell transplantation. J. Cancer Res. Clin. Oncol. 141, 2241–2251 (2015).
Mainardi, C. et al. CD34(+) selected stem cell boosts can improve poor graft function after paediatric allogeneic stem cell transplantation. Br. J. Haematol. 180, 90–99 (2018).
Guha, A. et al. Cardiovascular events associated with chimeric antigen receptor T cell therapy: cross-sectional FDA Adverse Events Reporting System analysis. Biol Blood Marrow Transplant. 26, 2211–2216 (2020).
Burstein, D. S. et al. Cardiac profile of chimeric antigen receptor T cell therapy in children: a single-institution experience. Biol. Blood Marrow Transplant. 24, 1590–1595 (2018).
Ganatra, S. et al. Chimeric antigen receptor T-cell therapy for cancer and heart: JACC Council Perspectives. J. Am. Coll. Cardiol. 74, 3153–3163 (2019).
Alvi, R. M. et al. Cardiovascular events among adults treated with chimeric antigen receptor T-cells (CAR-T). J. Am. Coll. Cardiol. 74, 3099–3108 (2019).
Shalabi, H. et al. Impact of cytokine release syndrome on cardiac function following CD19 CAR-T cell therapy in children and young adults with hematological malignancies. J. Immunother. Cancer 8, e001159 (2020).
Yates, B. et al. Efficacy and kinetics of CAR-T cell therapy in children and young adults with extramedullary acute lymphoblastic leukemia (ALL) and non-Hodgkin lymphoma (NHL) [abstract 65]. Biol. Blood Marrow Transplant. 24 (Suppl. 3), S67–S68 (2018).
Gutierrez, C. et al. The chimeric antigen receptor-intensive care unit (CAR-ICU) initiative: surveying intensive care unit practices in the management of CAR T-cell associated toxicities. J. Crit. Care 58, 58–64 (2020).
Mumtaz, A. A. & Li, A. Ocular toxicity and mangement of chimeric antigen receptor T-cell therapy [abstract]. Investig. Ophthalmol. Vis. Sci. 61, 5178 (2020).
Sharma, T., Grewal, J., Gupta, S. & Murray, P. I. Ophthalmic manifestations of acute leukaemias: the ophthalmologist’s role. Eye 18, 663–672 (2004).
Denton, C. C. et al. Bilateral retinal detachment after chimeric antigen receptor T-cell therapy. Blood Adv. 4, 2158–2162 (2020).
Fischer, A. & Li, A. CAR T-cell therapy associated ocular adverse events reported to the Food and Drug Administration [abstract]. Investig. Ophthalmol. Vis. Sci. 61, 3636 (2020).
Jhaveri, K. D. & Rosner, M. H. Chimeric antigen receptor T cell therapy and the kidney: what the nephrologist needs to know. Clin. J. Am. Soc. Nephrol. 13, 796–798 (2018).
Gupta, S. et al. Acute kidney injury and electrolyte abnormalities after chimeric antigen receptor T-cell (CAR-T) therapy for diffuse large B-cell lymphoma. Am J Kidney Dis. 76, 63–71 (2020).
Gutgarts, V. et al. Acute kidney injury after CAR-T cell therapy: low incidence and rapid recovery. Biol. Blood Marrow Transplant. 26, 1071–1076 (2020).
Wudhikarn, K. et al. DLBCL patients treated with CD19 CAR T cells experience a high burden of organ toxicities but low nonrelapse mortality. Blood Adv. 4, 3024–3033 (2020).
Shah, N. N. et al. Clonal expansion of CAR T cells harboring lentivector integration in the CBL gene following anti-CD22 CAR T-cell therapy. Blood Adv. 3, 2317–2322 (2019).
Cornetta, K. et al. Absence of replication-competent lentivirus in the clinic: analysis of infused T cell products. Mol. Ther. 26, 280–288 (2018).
Cornetta, K. et al. Screening clinical cell products for replication competent retrovirus: the National Gene Vector Biorepository experience. Mol. Ther. Methods Clin. Dev. 10, 371–378 (2018).
US Food and Drug Administration. Long term follow-up after administration of human gene therapy products: guidance for industry (2020).
Britten, O., Ragusa, D., Tosi, S. & Kamel, Y. M. MLL-rearranged acute leukemia with t(4;11)(q21;q23)–current treatment options. Is there a role for CAR-T cell therapy? Cells. 8, 1341 (2019).
Gardner, R. et al. Acquisition of a CD19-negative myeloid phenotype allows immune escape of MLL-rearranged B-ALL from CD19 CAR-T-cell therapy. Blood. 127, 2406–2410 (2016).
Wolfl, M. et al. Spontaneous reversion of a lineage switch following an initial blinatumomab-induced ALL-to-AML switch in MLL-rearranged infant ALL. Blood Adv. 2, 1382–1385 (2018).
Rayes, A., McMasters, R. L. & O’Brien, M. M. Lineage switch in MLL-rearranged infant leukemia following CD19-directed therapy. Pediatr. Blood Cancer 63, 1113–1115 (2016).
Haddox, C. L. et al. Blinatumomab-induced lineage switch of B-ALL with t(4:11)(q21;q23) KMT2A/AFF1 into an aggressive AML: pre- and post-switch phenotypic, cytogenetic and molecular analysis. Blood Cancer J. 7, e607 (2017).
Oberley, M. J. et al. Myeloid lineage switch following chimeric antigen receptor T-cell therapy in a patient with TCF3-ZNF384 fusion-positive B-lymphoblastic leukemia. Pediatr. Blood Cancer 65, e27265 (2018).
Zoghbi, A., Zur Stadt, U., Winkler, B., Muller, I. & Escherich, G. Lineage switch under blinatumomab treatment of relapsed common acute lymphoblastic leukemia without MLL rearrangement. Pediatr Blood Cancer. 64, e26594 (2017).
Schmiegelow, K. et al. Second malignant neoplasms after treatment of childhood acute lymphoblastic leukemia. J. Clin. Oncol. 31, 2469–2476 (2013).
Aguilera, D. G. et al. Pediatric therapy-related myelodysplastic syndrome/acute myeloid leukemia: the MD Anderson Cancer Center experience. J. Pediatr. Hematol. Oncol. 31, 803–811 (2009).
Shouse, G. P. et al. MDS as a cause for prolonged hematologic toxicity after treatment with CD19 targeted CAR-T cell therapy in patients with relapsed refractory lymphoma. Hematological Oncol. 37, 507–508 (2019).
Good, M. L. et al. Intrahepatic cholangiocarcinoma as a rare secondary malignancy after allogeneic hematopoietic stem cell transplantation for childhood acute lymphoblastic leukemia: a case report. Pediatr. Transpl. 24, e13653 (2020).
Tichelli, A. et al. Evaluation of second solid cancers after hematopoietic stem cell transplantation in European patients. JAMA Oncol. 5, 229–235 (2019).
Inamoto, Y. et al. Secondary solid cancer screening following hematopoietic cell transplantation. Bone Marrow Transplant. 50, 1013–1023 (2015).
Maude, S. L., Teachey, D. T., Porter, D. L. & Grupp, S. A. CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood. 125, 4017–4023 (2015).
Lee, D. W. et al. Long-term outcomes following CD19 CAR T cell therapy for B-ALL are superior in patients receiving a fludarabine/cyclophosphamide preparative regimen and post-CAR hematopoietic stem cell transplantation [abstract]. Blood. 128, 218 (2016).
Summers, C. et al. Long term follow-up after SCRI-CAR19v1 reveals late recurrences as well as a survival advantage to consolidation with HCT after CAR T cell induced remission [abstract]. Blood. 132 (Suppl. 1), 967 (2018).
Asnani, M. et al. Retention of CD19 intron 2 contributes to CART-19 resistance in leukemias with subclonal frameshift mutations in CD19. Leukemia. 34, 1202–1207 (2020).
Bagashev, A. et al. CD19 alterations emerging after CD19-directed immunotherapy cause retention of the misfolded protein in the endoplasmic reticulum. Mol Cell Biol 38, e00383–18 (2018).
Sotillo, E. et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy. Cancer Discov. 5, 1282–1295 (2015).
Orlando, E. J. et al. Genetic mechanisms of target antigen loss in CAR19 therapy of acute lymphoblastic leukemia. Nat. Med. 24, 1504–1506 (2018).
Schultz, L. M. et al. Phase 1 study of CD19/CD22 bispecific chimeric antigen receptor (CAR) therapy in children and young adults with B cell acute lymphoblastic leukemia (ALL) [abstract]. Blood. 132 (Suppl. 1), 898 (2018).
Gardner, R. et al. Early clinical experience of CD19 x CD22 dual specific CAR T cells for enhanced anti-leukemic targeting of acute lymphoblastic leukemia [abstract]. Blood. 132 (Suppl. 1), 278 (2018).
Amrolia, P. J. et al. Simultaneous targeting of CD19 and CD22: phase I study of AUTO3, a bicistronic chimeric antigen receptor (CAR) T-cell therapy, in pediatric patients with relapsed/refractory B-cell acute lymphoblastic leukemia (r/r B-ALL): Amelia Study [abstract]. Blood. 132 (Suppl. 1), 279 (2018).
Ruella, M. et al. Dual CD19 and CD123 targeting prevents antigen-loss relapses after CD19-directed immunotherapies. J. Clin. Invest. 126, 3814–3826 (2016).
Qin, H. et al. CAR T cells targeting BAFF-R can overcome CD19 antigen loss in B cell malignancies. Sci Transl Med. 11, eaaw9414 (2019).
Qin, H. et al. Eradication of B-ALL using chimeric antigen receptor-expressing T cells targeting the TSLPR oncoprotein. Blood. 126, 629–639 (2015).
Frey, N. V. et al. Optimizing chimeric antigen receptor T-cell therapy for adults with acute lymphoblastic leukemia. J. Clin. Oncol. 38, 415–422 (2020).
Hay, K. A. et al. Factors associated with durable EFS in adult B-cell ALL patients achieving MRD-negative CR after CD19 CAR T-cell therapy. Blood. 133, 1652–1663 (2019).
Park, J. H. et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 378, 449–459 (2018).
Pulsipher, M. A. et al. Potential utility of minimal residual disease (MRD) to identify relapse in pediatric and young adult (AYA) B-cell acute lymphoblastic leukemia (B-ALL) patients treated with tisagenlecleucel [abstract]. Cancer Res. 79 (Suppl. 13), CT077 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03186118 (2020).
Annesley, C. et al. Novel CD19t T-antigen presenting cells expand CD19 CAR T cells in vivo [abstract]. Blood. 134 (Suppl. 1), 223 (2019).
Turtle, C. J. et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J. Clin. Invest. 126, 2123–2138 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT2374333 (2020).
Maude, S. L. et al. Durable remissions with humanized CD19-targeted chimeric antigen receptor (CAR)-modified T cells in CAR-naive and CAR-exposed children and young adults with relapsed/refractory acute lymphoblastic leukemia [abstract]. Blood. 130 (Suppl. 1), 1319 (2017).
Li, A. M. et al. Checkpoint inhibitors augment CD19-directed chimeric antigen receptor (CAR) T cell therapy in relapsed B-cell acute lymphoblastic leukemia. Blood. 132, 556–556 (2018).
Curran, K. J. et al. Toxicity and response after CD19-specific CAR T-cell therapy in pediatric/young adult relapsed/refractory B-ALL. Blood. 134, 2361–2368 (2019).
Yoon, D. H., Osborn, M. J., Tolar, J. & Kim, C. J. Incorporation of immune checkpoint blockade into chimeric antigen receptor T cells (CAR-Ts): combination or built-in CAR-T. Int. J. Mol. Sci. 19, 340 (2018).
Chong, E. A. et al. PD-1 blockade modulates chimeric antigen receptor (CAR)-modified T cells: refueling the CAR. Blood. 129, 1039–1041 (2017).
Hill, B. T., Roberts, Z. J., Rossi, J. M. & Smith, M. R. Marked re-expansion of chimeric antigen receptor (CAR) T cells and tumor regression following nivolumab treatment in a patient treated with axicabtagene ciloleucel (axi-cel; KTE-C19) for refractory diffuse large B cell lymphoma (DLBCL) [abstract]. Blood. 130 (Suppl. 1), 2825 (2017).
Qu, C. et al. Radiation priming chimeric antigen receptor T-cell therapy in relapsed/refractory diffuse large B-cell lymphoma with high tumor burden. J. Immunother. 43, 32–37 (2020).
DeSelm, C. et al. Low-dose radiation conditioning enables CAR T cells to mitigate antigen escape. Mol. Ther. 26, 2542–2552 (2018).
Wright, C. M. et al. Bridging radiation therapy before commercial chimeric antigen receptor T-cell therapy for relapsed or refractory aggressive B-cell lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 108, 178–188 (2020).
Sim, A. J. et al. Radiation therapy as a bridging strategy for CAR T cell therapy with axicabtagene ciloleucel in diffuse large B-cell lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 105, 1012–1021 (2019).
Imber, B. S. et al. Early experience using salvage radiotherapy for relapsed/refractory non-Hodgkin lymphomas after CD19 chimeric antigen receptor (CAR) T cell therapy. Br. J. Haematol. 190, 45–51 (2020).
Acknowledgements
The initial efforts from the CAR T cell Therapy Beyond the Storm Consortium were presented on 14 May 2020 in a conference jointly sponsored by the National Cancer Institute (NCI), and the Pediatric Transplantation and Cell Therapy Consortium (PTCTC). The authors acknowledge all the speakers and the >200 international participants. The audio recording from the conference can be found on the conference website. The authors acknowledge the NCI and the PTCTC for their support, and specifically L. Schultz for sharing her efforts on CAR T cell Real World consortia studies. PTCTC receives support through a Johnny Crisstopher Children’s Charitable Foundation St. Baldrick’s Consortium Grant and a NHLBI/NCI grant (2UG1HL069254). Additional funding is from National Institute of Neurological Disorders and Stroke Child Neurology Career Development Program K12 (1K12NS098482-02) (Gust). The work of N.N.S. is supported in part by the Intramural Research Program of the NCI and NIH Clinical Center (ZIA BC 011823).
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of the manuscript, and reviewed and approved the content prior to submission. H.S., J.G., A.T., A.B.L., R.A.G., C.A., C.N.D., S.A.G., M.A.P. and N.N.S. comprised the steering committee leading this effort.
Corresponding author
Ethics declarations
Competing interests
J.G. is a consultant for Johnson & Johnson. T.W.L. has consulted for Bayer, Cellectis and Novartis; and his institution has research funding from Bayer, Novartis and Pfizer. R.A.G. has received honoraria from Novartis. J.A.H. has consulted for Allogene Therapeutics. K.J.C. has received research support from Juno Therapeutics and Novartis and has consulted and participated in advisory boards or educational seminars for Juno Therapeutics, Mesoblast and Novartis. M.C.P. has received research support from BMS, Kite and Novartis, and has been a consultant for Amgen and BMS. S.A.G. receives research support from Kite, Novartis and Servier; is a consultant for CBMG, GSK, Humanigen, Janssen/Johnson & Johnson, Novartis and Roche; is a study steering committee or scientific advisory board member for Adaptimmune, Allogene, Cellectis, Jazz, Juno, Novartis, TCR2 and Vertex/CRISPR; and has a patent (WO 2014011984 A1) that is managed according to the University of Pennsylvania patent policy. M.A.P. has received fees from Novartis, grants and personal fees from Miltenyi, grants from Adaptive, and personal fees from Mesoblast. The remaining authors declare no competing interests.
Additional information
Disclaimer
The content of this publication does not necessarily reflect the views and policies of the US Department of Health and Human Services, nor does mention of trade names, commercial products or organizations imply endorsement by the US Government.
Peer review information
Nature Reviews Clinical Oncology thanks J. Gauthier, N. Kapoor, R. Phelan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
CAR T-Cell Therapy: Beyond The Storm conference: https://ncifrederick.cancer.gov/events/conferences/car-t-cell-therapy-beyond-storm
Supplementary information
Rights and permissions
About this article
Cite this article
Shalabi, H., Gust, J., Taraseviciute, A. et al. Beyond the storm — subacute toxicities and late effects in children receiving CAR T cells. Nat Rev Clin Oncol 18, 363–378 (2021). https://doi.org/10.1038/s41571-020-00456-y
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41571-020-00456-y
This article is cited by
-
Endocrine late-effects in survivors of childhood and adolescence cancer: clinical management
Journal of Endocrinological Investigation (2026)
-
The quintessential role for CAR T cell therapy in children, adolescents and young adults with cancer
Nature Reviews Clinical Oncology (2026)
-
CAR T-cell therapy in hematologic and solid malignancies: mechanisms, clinical applications, and future directions
Medical Oncology (2025)
-
New insights into CAR T-cell hematological toxicities: manifestations, mechanisms, and effective management strategies
Experimental Hematology & Oncology (2024)
-
Programmable synthetic receptors: the next-generation of cell and gene therapies
Signal Transduction and Targeted Therapy (2024)