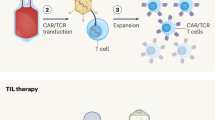
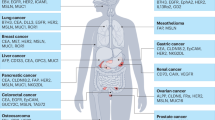
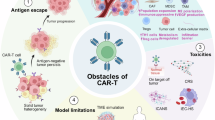
Abstract
Chimeric antigen receptor (CAR) T cells have become standard-of-care therapies for patients with certain relapsed and/or refractory haematological malignancies over the past decade. However, this approach remains largely ineffective in patients with solid tumours, in part owing to limited CAR T cell persistence, the immunosuppressive tumour microenvironment of many solid tumours and limited trafficking of CAR T cells into tumours. Central nervous system (CNS) tumours, many of which are associated with a poor prognosis and require new treatment approaches, present additional challenges such as the presence of the blood–brain barrier as well as concerns over treatment-related neurotoxicities. Despite these difficulties, clinical trials involving both adult and paediatric patients with primary CNS tumours have provided signals of efficacy. In this Review, we discuss completed, ongoing and anticipated trials testing CAR T cells in patients with CNS tumours. We also highlight the most promising preclinical developments that might lead to novel clinical approaches in this area.
Key points
-
Advances in chimeric antigen receptor (CAR) T cell engineering, combination treatment strategies and central nervous system (CNS)-specific targeting approaches have the potential to transform the landscape of cellular immunotherapy for CNS tumours.
-
CNS tumours harbour several novel tumour-specific, and tumour-associated targets for CAR T cell approaches.
-
The immunosuppressive effects of CNS solid tumours often limit the efficacy of CAR T cells.
-
Continued interdisciplinary efforts will be crucial to translating preclinical innovations into effective clinical applications to improve the outcomes of patients with CNS malignancies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 71, 7–33 (2021).
National Cancer Institute. Life after diagnosis: survival. Cancer Trends Progress Report. NCI https://progressreport.cancer.gov/after/survival (2024).
Sperduto, P. W. et al. Survival in patients with brain metastases: summary report on the updated diagnosis-specific graded prognostic assessment and definition of the eligibility quotient. J. Clin. Oncol. 38, 3773–3784 (2020).
Li, A. Y. et al. Association of brain metastases with survival in patients with limited or stable extracranial disease: a systematic review and meta-analysis. JAMA Netw. Open. 6, e230475 (2023).
Finck, A. V., Blanchard, T., Roselle, C. P., Golinelli, G. & June, C. H. Engineered cellular immunotherapies in cancer and beyond. Nat. Med. 28, 678–689 (2022).
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
Singh, N. et al. Mechanisms of resistance to CAR T cell therapies. Semin. Cancer Biol. 65, 91–98 (2020).
Del Bufalo, F. et al. GD2-CART01 for relapsed or refractory high-risk neuroblastoma. N. Engl. J. Med. 388, 1284–1295 (2023).
Li, C. H. et al. Long-term outcomes of GD2-directed CAR-T cell therapy in patients with neuroblastoma. Nat. Med. 31, 1125–1129 (2025).
Qi, C. et al. Claudin-18 isoform 2-specific CAR T-cell therapy (satri-cel) versus treatment of physician’s choice for previously treated advanced gastric or gastro-oesophageal junction cancer (CT041-ST-01): a randomised, open-label, phase 2 trial. Lancet 405, 2049–2060 (2025).
Price, M. et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2017-2021. Neuro Oncol. 26, vi1–vi85 (2024).
Louis, D. N. et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol. 23, 1231–1251 (2021).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Weller, M. et al. Glioma. Nat. Rev. Dis. Primers 10, 33 (2024).
Bagley, S. J. et al. Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results. Nat. Med. 30, 1320–1329 (2024).
Brown, C. E. et al. Locoregional delivery of IL-13Rα2-targeting CAR-T cells in recurrent high-grade glioma: a phase 1 trial. Nat. Med. 30, 1001–1012 (2024).
Choi, B. D. et al. Intraventricular CARv3-TEAM-E T cells in recurrent glioblastoma. N. Engl. J. Med. 390, 1290–1298 (2024).
Bartolomé, R. A. et al. IL13 receptor α2 signaling requires a scaffold protein, FAM120A, to activate the FAK and PI3K pathways in colon cancer metastasis. Cancer Res. 75, 2434–2444 (2015).
Joshi, B. H., Plautz, G. E. & Puri, R. K. Interleukin-13 receptor alpha chain: a novel tumor-associated transmembrane protein in primary explants of human malignant gliomas. Cancer Res. 60, 1168–1172 (2000).
Brown, C. E. et al. Glioma IL13Rα2 is associated with mesenchymal signature gene expression and poor patient prognosis. PLoS ONE 8, e77769 (2013).
Jaén, M., Martín-Regalado, Á, Bartolomé, R. A., Robles, J. & Casal, J. I. Interleukin 13 receptor alpha 2 (IL13Rα2): expression, signaling pathways and therapeutic applications in cancer. Biochim. Biophys. Acta Rev. Cancer 1877, 188802 (2022).
Brown, C. E. et al. Bioactivity and safety of IL13Rα2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma. Clin. Cancer Res. 21, 4062–4072 (2015).
Brown, C. E. et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N. Engl. J. Med. 375, 2561–2569 (2016).
Weller, M., Cloughesy, T., Perry, J. R. & Wick, W. Standards of care for treatment of recurrent glioblastoma — are we there yet? Neuro Oncol. 15, 4–27 (2013).
AACR Project GENIE Consortium AACR Project GENIE: powering precision medicine through an international consortium. Cancer Discov. 7, 818–831 (2017).
Brennan, C. W. et al. The somatic genomic landscape of glioblastoma. Cell 155, 462–477 (2013).
Hoogstrate, Y. et al. The EGFRvIII transcriptome in glioblastoma: a meta-omics analysis. Neuro Oncol. 24, 429–441 (2022).
Chi, A. S. et al. Exploring predictors of response to dacomitinib in EGFR-amplified recurrent glioblastoma. JCO Precis. Oncol. 4, 593–613 (2020).
Lin, B. et al. EGFR, the Lazarus target for precision oncology in glioblastoma. Neuro Oncol. 24, 2035–2062 (2022).
O’Rourke, D. M. et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci. Transl. Med. 9, eaaa0984 (2017).
Goff, S. L. et al. Pilot trial of adoptive transfer of chimeric antigen receptor-transduced T cells targeting EGFRvIII in patients with glioblastoma. J. Immunother. 42, 126–135 (2019).
Weller, M. et al. Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): a randomised, double-blind, international phase 3 trial. Lancet Oncol. 18, 1373–1385 (2017).
Bagley, S. J. et al. Repeated peripheral infusions of anti-EGFRvIII CAR T cells in combination with pembrolizumab show no efficacy in glioblastoma: a phase 1 trial. Nat. Cancer 5, 517–531 (2024).
Ahmed, N. et al. HER2-specific chimeric antigen receptor-modified virus-specific T cells for progressive glioblastoma: a phase 1 dose-escalation trial. JAMA Oncol. 3, 1094–1101 (2017).
Ramezani, M., Siami, S., Rezaei, M., Khazaei, S. & Sadeghi, M. An immunohistochemical study of HER2 expression in primary brain tumors. Biomedicine 10, 21–27 (2020).
Mount, C. W. et al. Potent antitumor efficacy of anti-GD2 CAR T cells in H3-K27M+ diffuse midline gliomas. Nat. Med. 24, 572–579 (2018).
Majzner, R. G. et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature 603, 934–941 (2022).
Liu, Z. et al. Safety and antitumor activity of GD2-specific 4SCAR-T cells in patients with glioblastoma. Mol. Cancer 22, 3 (2023).
Majzner, R. G. et al. CAR T cells targeting B7-H3, a pan-cancer antigen, demonstrate potent preclinical activity against pediatric solid tumors and brain tumors. Clin. Cancer Res. 25, 2560–2574 (2019).
Jin, L. et al. CD70, a novel target of CAR T-cell therapy for gliomas. Neuro Oncol. 20, 55–65 (2018).
Wang, D. et al. Chlorotoxin-directed CAR T cells for specific and effective targeting of glioblastoma. Sci. Transl. Med. 12, eaaw2672 (2020).
Li, G. et al. A phase 1 study of B7H3 CAR-T cells administered intracranially in recurrent glioblastoma [abstract]. J. Clin. Oncol. 43, 2018 (2025).
Eisenbarth, D. & Wang, Y. A. Glioblastoma heterogeneity at single cell resolution. Oncogene 42, 2155–2165 (2023).
Yabo, Y. A., Niclou, S. P. & Golebiewska, A. Cancer cell heterogeneity and plasticity: a paradigm shift in glioblastoma. Neuro Oncol. 24, 669–682 (2022).
Bagley, S. J. et al. Intracerebroventricular bivalent CAR T cells targeting EGFR and IL-13Rα2 in recurrent glioblastoma: a phase 1 trial. Nat. Med. 31, 2778–2787 (2025).
Reilly, E. B. et al. Characterization of ABT-806, a humanized tumor-specific anti-EGFR monoclonal antibody. Mol. Cancer Ther. 14, 1141–1151 (2015).
Orellana, L. et al. Oncogenic mutations at the EGFR ectodomain structurally converge to remove a steric hindrance on a kinase-coupled cryptic epitope. Proc. Natl Acad. Sci. USA 116, 10009–10018 (2019).
Lassman, A. B. et al. Comparison of biomarker assays for EGFR: implications for precision medicine in patients with glioblastoma. Clin. Cancer Res. 25, 3259–3265 (2019).
Mahdi, J. et al. Tumor inflammation-associated neurotoxicity. Nat. Med. 29, 803–810 (2023).
Choi, B. D. et al. CAR-T cells secreting BiTEs circumvent antigen escape without detectable toxicity. Nat. Biotechnol. 37, 1049–1058 (2019).
Gerstner, E. R. et al. Safety and tolerability of intraventricular CARv3-TEAM-E T cells following lymphodepleting chemotherapy in recurrent glioblastoma: INCIPIENT trial [abstract]. J. Clin. Oncol. 43, 2017 (2025).
Roybal, K. T. et al. Engineering T cells with customized therapeutic response programs using synthetic notch receptors. Cell 167, 419–432.e6 (2016).
Choe, J. H. et al. SynNotch-CAR T cells overcome challenges of specificity, heterogeneity, and persistence in treating glioblastoma. Sci. Transl. Med. 13, eabe7378 (2021).
Akingbemi, W. et al. Stereotactic radiotherapy and CART for treatment of secondary CNS lymphoma. Leuk. Lymphoma 65, 860–863 (2024).
Ryan, C. E. et al. Clinical efficacy and safety of chimeric antigen receptor T-cell therapy for mantle cell lymphoma with secondary central nervous system involvement. Br. J. Haematol. 203, 774–780 (2023).
Karschnia, P. et al. Toxicities and response rates of secondary CNS lymphoma after adoptive immunotherapy with CD19-directed chimeric antigen receptor T cells. Neurology 98, 884–889 (2022).
Choquet, S. et al. CAR T-cell therapy induces a high rate of prolonged remission in relapsed primary CNS lymphoma: real-life results of the LOC network. Am. J. Hematol. 99, 1240–1249 (2024).
Kline, K. et al. Treatment of secondary CNS lymphoma using CD19-targeted chimeric antigen receptor (CAR) T cells. Cancer Immunol. Immunother. 73, 45 (2024).
Cook, M. R. et al. Toxicity and efficacy of CAR T-cell therapy in primary and secondary CNS lymphoma: a meta-analysis of 128 patients. Blood Adv. 7, 32–39 (2023).
Frigault, M. J. et al. Tisagenlecleucel CAR T-cell therapy in secondary CNS lymphoma. Blood 134, 860–866 (2019).
Siddiqi, T. et al. CD19-directed CAR T-cell therapy for treatment of primary CNS lymphoma. Blood Adv. 5, 4059–4063 (2021).
Baker, D. J., Arany, Z., Baur, J. A., Epstein, J. A. & June, C. H. CAR T therapy beyond cancer: the evolution of a living drug. Nature 619, 707–715 (2023).
Nayak, L. et al. A pilot study of axicabtagene ciloleucel (axi-cel) for relapsed/refractory primary and secondary central nervous system lymphoma (PCNSL and SCNSL) [abstract]. J. Clin. Oncol. 42, 2006 (2024).
Frigault, M. J. et al. Safety and efficacy of tisagenlecleucel in primary CNS lymphoma: a phase 1/2 clinical trial. Blood 139, 2306–2315 (2022).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
Price, M. et al. Childhood, adolescent, and adult primary brain and central nervous system tumor statistics for practicing healthcare providers in neuro-oncology, CBTRUS 2015-2019. Neurooncol Pract. 11, 5–25 (2024).
Gust, J. et al. Locoregional infusion of EGFR806-CAR T cells for recurrent or refractory pediatric CNS tumors: results of the completed BrainChild02 phase 1 clinical trial. Neuro Oncol. 27, 2170–2181 (2025).
Gregorio, A. et al. Small round blue cell tumours: diagnostic and prognostic usefulness of the expression of B7-H3 surface molecule. Histopathology 53, 73–80 (2008).
Zhou, Z. et al. B7-H3, a potential therapeutic target, is expressed in diffuse intrinsic pontine glioma. J. Neurooncol 111, 257–264 (2013).
Kramer, K. et al. Compartmental intrathecal radioimmunotherapy: results for treatment for metastatic CNS neuroblastoma. J. Neurooncol 97, 409–418 (2010).
Souweidane, M. M. et al. Convection-enhanced delivery for diffuse intrinsic pontine glioma: a single-centre, dose-escalation, phase 1 trial. Lancet Oncol. 19, 1040–1050 (2018).
Vitanza, N. A. et al. Intraventricular B7-H3 CAR T cells for diffuse intrinsic pontine glioma: preliminary first-in-human bioactivity and safety. Cancer Discov. 13, 114–131 (2023).
Vitanza, N. A. et al. Intracerebroventricular B7-H3-targeting CAR T cells for diffuse intrinsic pontine glioma: a phase 1 trial. Nat. Med. 31, 861–868 (2025).
Cooney, T. et al. Contemporary survival endpoints: an international diffuse intrinsic pontine glioma registry study. Neuro-Oncol. 19, 1279–1280 (2017).
Vitanza, N. A. et al. Locoregional CAR T cells for the treatment of CNS tumors in children: investigational drug service pharmacy activities. J. Hematol. Oncol. Pharm. 14, 148–154 (2024).
Dhillon, S. Dinutuximab: first global approval. Drugs 75, 923–927 (2015).
Pule, M. A. et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat. Med. 14, 1264–1270 (2008).
Kaczanowska, S. et al. Immune determinants of CAR-T cell expansion in solid tumor patients receiving GD2 CAR-T cell therapy. Cancer Cell 42, 35–51.e8 (2024).
Louis, C. U. et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood 118, 6050–6056 (2011).
Monje, M. et al. Intravenous and intracranial GD2-CAR T cells for H3K27M+ diffuse midline gliomas. Nature 637, 708–715 (2025).
Lin, F. Y. et al. Phase I trial of GD2.CART cells augmented with constitutive interleukin-7 receptor for treatment of high-grade pediatric CNS tumors. J. Clin. Oncol. 42, 2769–2779 (2024).
Wang, L. et al. Expansion of endogenous T cells in CSF of pediatric CNS tumor patients undergoing locoregional delivery of IL13Rα2-targeting CAR T cells: an interim analysis. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-3454977/v1 (2023).
Vitanza, N. A. et al. Locoregional infusion of HER2-specific CAR T cells in children and young adults with recurrent or refractory CNS tumors: an interim analysis. Nat. Med. 27, 1544–1552 (2021).
Birley, K. et al. A novel anti-B7-H3 chimeric antigen receptor from a single-chain antibody library for immunotherapy of solid cancers. Mol. Ther. Oncolytics 26, 429–443 (2022).
Venkataraman, S. et al. Developing a safe and potent tumor-targeting gated CAR-T cell therapy for DIPG: a deadly pediatric brain tumor [abstract]. Cancer Res. 84, PR010 (2024).
Foster, J. B. et al. Development of GPC2-directed chimeric antigen receptors using mRNA for pediatric brain tumors. J. Immunother. Cancer 10, e004450 (2022).
Ciccone, R. et al. GD2-Targeting CAR T-cell therapy for patients with GD2+mMedulloblastoma. Clin. Cancer Res. 30, 2545–2557 (2024).
Agarwal, S. et al. Deletion of the inhibitory co-receptor CTLA-4 enhances and invigorates chimeric antigen receptor T cells. Immunity 56, 2388–2407.e9 (2023).
Chong, E. A. et al. Pembrolizumab for B-cell lymphomas relapsing after or refractory to CD19-directed CAR T-cell therapy. Blood 139, 1026–1038 (2022).
Heczey, A. et al. CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol. Ther. 25, 2214–2224 (2017).
Yin, Y. et al. Checkpoint blockade reverses anergy in IL-13Rα2 humanized scFv-based CAR T cells to treat murine and canine gliomas. Mol. Ther. Oncolytics 11, 20–38 (2018).
Tang, O. Y. et al. PD1 expression in EGFRvIII-directed CAR T cell infusion product for glioblastoma is associated with clinical response. Front. Immunol. 13, 872756 (2022).
Fan, J. et al. Potential synergy between radiotherapy and CAR T-cells — a multicentric analysis of the role of radiotherapy in the combination of CAR T cell therapy. Radiother. Oncol. 183, 109580 (2023).
Fang, P. Q. et al. Radiation and CAR T-cell therapy in lymphoma: future frontiers and potential opportunities for synergy. Front. Oncol. 11, 648655 (2021).
McGrath, K. & Dotti, G. Combining oncolytic viruses with chimeric antigen receptor t cell therapy. Hum. Gene Ther. 32, 150–157 (2021).
Suryadevara, C. M. et al. Temozolomide lymphodepletion enhances CAR abundance and correlates with antitumor efficacy against established glioblastoma. Oncoimmunology 7, e1434464 (2018).
Boardman, A. P. & Palomba, M. L. Does BTKi improve CAR T-cell therapy in MCL? Blood 143, 653–655 (2024).
Stock, S., Kluever, A. K., Endres, S. & Kobold, S. Enhanced chimeric antigen receptor T cell therapy through co-application of synergistic combination partners. Biomedicines 10, 307 (2022).
Steffin, D. et al. Interleukin-15-armoured GPC3 CAR T cells for patients with solid cancers. Nature 637, 940–946 (2025).
Liu, Y. et al. Armored inducible expression of IL-12 enhances antitumor activity of glypican-3-targeted chimeric antigen receptor-engineered T cells in hepatocellular carcinoma. J. Immunol. 203, 198–207 (2019).
Rosewell Shaw, A. et al. IL-12 encoding oNDV synergizes with CAR-T cells in orthotopic models of non-small cell lung cancer. Mol. Ther. Oncol. 32, 200899 (2024).
Zhu, Y. et al. Mesothelin CAR-T cells expressing tumor-targeted immunocytokine IL-12 yield durable efficacy and fewer side effects. Pharmacol. Res. 203, 107186 (2024).
Hou, A. J., Chang, Z. L., Lorenzini, M. H., Zah, E. & Chen, Y. Y. TGF-β-responsive CAR-T cells promote anti-tumor immune function. Bioeng. Transl. Med. 3, 75–86 (2018).
Hou, A. J. et al. IL-13Rα2/TGF-β bispecific CAR-T cells counter TGF-β-mediated immune suppression and potentiate anti-tumor responses in glioblastoma. Neuro Oncol. 26, 1850–1866 (2024).
Li, N. et al. Armored bicistronic CAR T cells with dominant-negative TGF-β receptor II to overcome resistance in glioblastoma. Mol. Ther. 32, 3522–3538 (2024).
Kloss, C. C. et al. Dominant-negative TGF-β receptor enhances PSMA-targeted human CAR T cell proliferation and augments prostate cancer eradication. Mol. Ther. 26, 1855–1866 (2018).
Foster, M. C. et al. Utility of a safety switch to abrogate CD19.CAR T-cell-associated neurotoxicity. Blood 137, 3306–3309 (2021).
Lin, H. et al. Anti-CD5 CAR-T cells with a tEGFR safety switch exhibit potent toxicity control. Blood Cancer J. 14, 98 (2024).
Mestermann, K. et al. The tyrosine kinase inhibitor dasatinib acts as a pharmacologic on/off switch for CAR T cells. Sci. Transl. Med. 11, eaau5907 (2019).
Binder, Z. A. et al. Epidermal growth factor receptor extracellular domain mutations in glioblastoma present opportunities for clinical imaging and therapeutic development. Cancer Cell 34, 163–177.e7 (2018).
Colardo, M., Segatto, M. & Di Bartolomeo, S. Targeting RTK-PI3K-mTOR axis in gliomas: an update. Int. J. Mol. Sci. 22, 4899 (2021).
Ohgaki, H. & Kleihues, P. Genetic alterations and signaling pathways in the evolution of gliomas. Cancer Sci. 100, 2235–2241 (2009).
Pearson, J. R. D. & Regad, T. Targeting cellular pathways in glioblastoma multiforme. Signal. Transduct. Target. Ther. 2, 17040 (2017).
Qin, A., Musket, A., Musich, P. R., Schweitzer, J. B. & Xie, Q. Receptor tyrosine kinases as druggable targets in glioblastoma: do signaling pathways matter? Neurooncol. Adv. 3, vdab133 (2021).
Stommel, J. M. et al. Coactivation of receptor tyrosine kinases affects the response of tumor cells to targeted therapies. Science 318, 287–290 (2007).
Tilak, M., Holborn, J., New, L. A., Lalonde, J. & Jones, N. Receptor tyrosine kinase signaling and targeting in glioblastoma multiforme. Int. J. Mol. Sci. 22, 1831 (2021).
Thokala, R. et al. High-affinity chimeric antigen receptor with cross-reactive scFv to clinically relevant EGFR oncogenic isoforms. Front. Oncol. 11, 664236 (2021).
Park, D. H. et al. Novel tri-specific T-cell engager targeting IL-13Rα2 and EGFRvIII provides long-term survival in heterogeneous GBM challenge and promotes antitumor cytotoxicity with patient immune cells. J. Immunother. Cancer 12, e009604 (2024).
Yin, Y. et al. Locally secreted BiTEs complement CAR T cells by enhancing killing of antigen heterogeneous solid tumors. Mol. Ther. 30, 2537–2553 (2022).
Dagher, O. K. et al. Preclinical efficacy of multi-targeting mRNA-based CAR T cell therapy in resection models of glioblastoma. Mol. Ther. Nucleic Acids 36, 102676 (2025).
Simic, M. S. et al. Programming tissue-sensing T cells that deliver therapies to the brain. Science 386, eadl4237 (2024).
Foeng, J., Comerford, I. & McColl, S. R. Harnessing the chemokine system to home CAR-T cells into solid tumors. Cell Rep. Med. 3, 100543 (2022).
Sun, R. et al. CXCR4-modified CAR-T cells suppresses MDSCs recruitment via STAT3/NF-κB/SDF-1α axis to enhance efficacy against pancreatic cancer. Mol. Ther. 31, 3193–3209 (2023).
Chongsathidkiet, P. et al. Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors. Nat. Med. 24, 1459–1468 (2018).
Dobersalske, C. et al. Cranioencephalic functional lymphoid units in glioblastoma. Nat. Med. 30, 2947–2956 (2024).
Smyth, L. C. D. et al. Identification of direct connections between the dura and the brain. Nature 627, 165–173 (2024).
Brown, C. E. et al. Off-the-shelf, steroid-resistant, IL13Rα2-specific CAR T cells for treatment of glioblastoma. Neuro Oncol. 24, 1318–1330 (2022).
Kang, I., Kim, Y. & Lee, H. K. γδ T cells as a potential therapeutic agent for glioblastoma. Front. Immunol. 14, 1273986 (2023).
Shaim, H. et al. Targeting the αv integrin/TGF-β axis improves natural killer cell function against glioblastoma stem cells. J. Clin. Invest. 131, e142116 (2021).
Shanley, M. et al. Interleukin-21 engineering enhances NK cell activity against glioblastoma via CEBPD. Cancer Cell 42, 1450–1466.e11 (2024).
Quintarelli, C. et al. Donor-derived GD2-specific CAR T cells in relapsed or refractory neuroblastoma. Nat. Med. 31, 849–860 (2025).
Lin, Q. et al. First-in-human trial of EphA2-redirected CAR T-cells in patients with recurrent glioblastoma: a preliminary report of three cases at the starting dose. Front. Oncol. 11, 694941 (2021).
Author information
Authors and Affiliations
Contributions
All authors made a substantial contribution to all aspects of the preparation of this article.
Corresponding author
Ethics declarations
Competing interests
Z.A.B., S.J.B. and D.M.O’R. have received research funding from Kite Pharma. J.B.F. declares no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks B. Badie, B. Choi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Binder, Z.A., Bagley, S.J., Foster, J.B. et al. The development of CAR T cells for patients with CNS malignancies. Nat Rev Clin Oncol 23, 137–150 (2026). https://doi.org/10.1038/s41571-025-01102-1
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41571-025-01102-1