Abstract
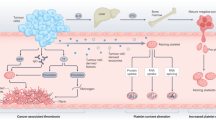
Accumulating evidence indicates that platelets promote cancer progression through direct interactions with malignant cells, the secretion of soluble mediators and the release of platelet-derived extracellular vesicles. In certain contexts, platelets can also suppress tumour progression by modulating immune responses, delivering antiproliferative microRNAs or releasing inhibitory factors. This dynamic and context-dependent interplay limits the effectiveness of strategies that solely inhibit or activate platelet activity and has driven the development of engineering approaches to reprogramme platelets with therapeutic intent. Unlike most cellular products, platelet-based approaches can be implemented in both autologous and allogeneic settings, providing more flexible approaches for developing new therapies. Over the past few years, advances in genetic and chemical engineering have enabled the multifunctional modification of platelets while preserving native properties essential for cancer therapy. Engineered platelets can act as targeted delivery vehicles to enhance local drug accumulation and release, or as active effector cells that directly modulate tumour progression. The clinical implementation of these engineered products will require control of platelet stability and activation, scalable manufacturing processes and rigorous safety evaluation. In this Review, we summarize the current understanding of platelet biology in cancer, examine engineering strategies for their therapeutic use, and outline opportunities and challenges for their clinical translation.
Key points
-
Platelets are active participants in cancer biology, with tumour-promoting or tumour-suppressive roles that are dynamic and context dependent, mediated through direct interactions, secretion of bioactive molecules and release of platelet-derived extracellular vesicles.
-
Platelets and their derivatives can be engineered into tumour-targeted drug delivery platforms by leveraging their cancer-associated biological properties and incorporating therapeutic payloads through internal encapsulation or surface conjugation.
-
Genetic or chemical engineering of platelets or their precursors generates hybrid products that integrate innate tumour-relevant characteristics with engineered functionalities, leading to enhanced or novel anticancer activities.
-
Translating platelet-based therapeutics into clinical applications will require scalable manufacturing processes and robust analytical strategies to ensure product consistency as well as support from regulatory agencies to facilitate widespread adoption.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
Brudno, J. N. & Kochenderfer, J. N. Current understanding and management of CAR T cell-associated toxicities. Nat. Rev. Clin. Oncol. 21, 501–521 (2024).
Myers, J. A. & Miller, J. S. Exploring the NK cell platform for cancer immunotherapy. Nat. Rev. Clin. Oncol. 18, 85–100 (2021).
D’Avanzo, C., Blaeschke, F., Lysandrou, M., Ingelfinger, F. & Zeiser, R. Advances in cell therapy: progress and challenges in hematological and solid tumors. Trends Pharmacol. Sci. 45, 1119–1134 (2024).
Tarannum, M. et al. Engineering innate immune cells for cancer immunotherapy. Nat. Biotechnol. 43, 516–533 (2025).
Leung, J., Cau, M. F. & Kastrup, C. J. Emerging gene therapies for enhancing the hemostatic potential of platelets. Transfusion 61, S275–S285 (2021).
van der Meijden, P. E. J. & Heemskerk, J. W. M. Platelet biology and functions: new concepts and clinical perspectives. Nat. Rev. Cardiol. 16, 166–179 (2019).
Haemmerle, M., Stone, R. L., Menter, D. G., Afshar-Kharghan, V. & Sood, A. K. the platelet lifeline to cancer: challenges and opportunities. Cancer Cell 33, 965–983 (2018).
Xu, X. R., Yousef, G. M. & Ni, H. Cancer and platelet crosstalk: opportunities and challenges for aspirin and other antiplatelet agents. Blood 131, 1777–1789 (2018).
Gordon, M. S. et al. A phase I trial of recombinant human interleukin-6 in patients with myelodysplastic syndromes and thrombocytopenia. Blood 85, 3066–3076 (1995).
Machlus, K. R. et al. CCL5 derived from platelets increases megakaryocyte proplatelet formation. Blood 127, 921–926 (2016).
Contursi, A. et al. Biology and pharmacology of platelet-type 12-lipoxygenase in platelets, cancer cells, and their crosstalk. Biochem. Pharmacol. 205, 115252 (2022).
Martling, A. et al. Low-dose aspirin for PI3K-altered localized colorectal cancer. N. Engl. J. Med. 393, 1051–1064 (2025).
Lucotti, S. et al. Aspirin blocks formation of metastatic intravascular niches by inhibiting platelet-derived COX-1/thromboxane A2. J. Clin. Invest. 129, 1845–1862 (2019).
Yang, J. et al. Aspirin prevents metastasis by limiting platelet TXA(2) suppression of T cell immunity. Nature 640, 1052–1061 (2025).
Benoy, I. et al. Serum interleukin 6, plasma VEGF, serum VEGF, and VEGF platelet load in breast cancer patients. Clin. Breast Cancer 2, 311–315 (2002).
Muller, K., Gilbertz, K. P. & Meineke, V. Serotonin and ionizing radiation synergistically affect proliferation and adhesion molecule expression of malignant melanoma cells. J. Dermatol. Sci. 68, 89–98 (2012).
Ma, C. et al. Platelets control liver tumor growth through P2Y12-dependent CD40L release in NAFLD. Cancer Cell 40, 986–998.e5 (2022).
Michael, J. V. et al. Platelet microparticles infiltrating solid tumors transfer miRNAs that suppress tumor growth. Blood 130, 567–580 (2017).
Sier, V. Q. et al. Cell-based tracers as trojan horses for image-guided surgery. Int. J. Mol. Sci. 22, 755 (2021).
Lu, Y., Hu, Q., Jiang, C. & Gu, Z. Platelet for drug delivery. Curr. Opin. Biotechnol. 58, 81–91 (2019).
Wang, C. et al. In situ activation of platelets with checkpoint inhibitors for post-surgical cancer immunotherapy. Nat. Biomed. Eng. 1, 0011 (2017).
Irvine, D. J., Maus, M. V., Mooney, D. J. & Wong, W. W. The future of engineered immune cell therapies. Science 378, 853–858 (2022).
Wang, Y. et al. Cell–drug conjugates. Nat. Biomed. Eng. 8, 1347–1365 (2024).
McPhedran, S. J., Carleton, G. A. & Lum, J. J. Metabolic engineering for optimized CAR-T cell therapy. Nat. Metab. 6, 396–408 (2024).
Li, Z., Wang, Y., Gu, Z. & Hu, Q. Engineering cells for therapy and diagnosis. Nat. Rev. Bioeng. 2, 770–784 (2024).
Martin, J. F., Kristensen, S. D., Mathur, A., Grove, E. L. & Choudry, F. A. The causal role of megakaryocyte–platelet hyperactivity in acute coronary syndromes. Nat. Rev. Cardiol. 9, 658–670 (2012).
Heijnen, H. F. G. & Korporaal, S. J. A. in Platelets in Thrombotic and Non-Thrombotic Disorders: Pathophysiology, Pharmacology and Therapeutics: an Update (eds Gresele, P. et al.) 21–37 (Springer, 2017).
Franco, A. T., Corken, A. & Ware, J. Platelets at the interface of thrombosis, inflammation, and cancer. Blood 126, 582–588 (2015).
Baaten, C., Nagy, M., Bergmeier, W., Spronk, H. M. H. & van der Meijden, P. E. J. Platelet biology and function: plaque erosion vs. rupture. Eur. Heart J. 45, 18–31 (2024).
Jiang, D. et al. RBCs regulate platelet function and hemostasis under shear conditions through biophysical and biochemical means. Blood 144, 1521–1531 (2024).
Rodrigues, M., Kosaric, N., Bonham, C. A. & Gurtner, G. C. Wound healing: a cellular perspective. Physiol. Rev. 99, 665–706 (2019).
Buka, R. J. et al. PF4 activates the c-Mpl–Jak2 pathway in platelets. Blood 143, 64–69 (2024).
Koupenova, M., Livada, A. C. & Morrell, C. N. Platelet and megakaryocyte roles in innate and adaptive immunity. Circ. Res. 130, 288–308 (2022).
Levin, J. & Conley, C. L. Thrombocytosis associated with malignant disease. Arch. Intern. Med. 114, 497–500 (1964).
Khorana, A. A., Francis, C. W., Culakova, E., Kuderer, N. M. & Lyman, G. H. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J. Thromb. Haemost. 5, 632–634 (2007).
Sallah, S., Wan, J. Y. & Nguyen, N. P. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb. Haemost. 87, 575–579 (2002).
Stein, P. D. et al. Incidence of venous thromboembolism in patients hospitalized with cancer. Am. J. Med. 119, 60–68 (2006).
Cho, M. S. et al. Platelets increase the proliferation of ovarian cancer cells. Blood 120, 4869–4872 (2012).
Hu, Q. et al. Role of platelet-derived TGFβ1 in the progression of ovarian cancer. Clin. Cancer Res. 23, 5611–5621 (2017).
Labelle, M., Begum, S. & Hynes, R. O. Direct signaling between platelets and cancer cells induces an epithelial–mesenchymal-like transition and promotes metastasis. Cancer Cell 20, 576–590 (2011).
Ho-Tin-Noe, B., Goerge, T., Cifuni, S. M., Duerschmied, D. & Wagner, D. D. Platelet granule secretion continuously prevents intratumor hemorrhage. Cancer Res. 68, 6851–6858 (2008).
De Palma, M., Biziato, D. & Petrova, T. V. Microenvironmental regulation of tumour angiogenesis. Nat. Rev. Cancer 17, 457–474 (2017).
Verheul, H. M. et al. Platelet and coagulation activation with vascular endothelial growth factor generation in soft tissue sarcomas. Clin. Cancer Res. 6, 166–171 (2000).
Janowska-Wieczorek, A., Marquez-Curtis, L. A., Wysoczynski, M. & Ratajczak, M. Z. Enhancing effect of platelet-derived microvesicles on the invasive potential of breast cancer cells. Transfusion 46, 1199–1209 (2006).
Janowska-Wieczorek, A. et al. Microvesicles derived from activated platelets induce metastasis and angiogenesis in lung cancer. Int. J. Cancer 113, 752–760 (2005).
Liang, H. et al. MicroRNA-223 delivered by platelet-derived microvesicles promotes lung cancer cell invasion via targeting tumor suppressor EPB41L3. Mol. Cancer 14, 58 (2015).
Helley, D. et al. Platelet microparticles: a potential predictive factor of survival in hormone-refractory prostate cancer patients treated with docetaxel-based chemotherapy. Eur. Urol. 56, 479–484 (2009).
Kim, H. K. et al. Elevated levels of circulating platelet microparticles, VEGF, IL-6 and RANTES in patients with gastric cancer: possible role of a metastasis predictor. Eur. J. Cancer 39, 184–191 (2003).
Cooke, N. M. et al. Increased platelet reactivity in patients with late-stage metastatic cancer. Cancer Med. 2, 564–570 (2013).
Wolny-Rokicka, E., Tukiendorf, A., Wydmański, J. & Zembroń-Łacny, A. The potential of the quick detection of selectins using raman spectroscopy to discriminate lung cancer patients from healthy subjects. J. Spectrosc. 2018, 7843208 (2018).
Lesurtel, M. et al. Platelet-derived serotonin mediates liver regeneration. Science 312, 104–107 (2006).
Nocito, A. et al. Serotonin regulates macrophage-mediated angiogenesis in a mouse model of colon cancer allografts. Cancer Res. 68, 5152–5158 (2008).
Sibilano, M. et al. Platelet-derived miR-126-3p directly targets AKT2 and exerts anti-tumor effects in breast cancer cells: further insights in platelet-cancer interplay. Int. J. Mol. Sci. 23, 5484 (2022).
Lazar, S. & Goldfinger, L. E. Platelets and extracellular vesicles and their cross talk with cancer. Blood 137, 3192–3200 (2021).
Tsoi, K. K. F., Ho, J. M. W., Chan, F. C. H. & Sung, J. J. Y. Long-term use of low-dose aspirin for cancer prevention: a 10-year population cohort study in Hong Kong. Int. J. Cancer 145, 267–273 (2019).
McNeil, J. J. et al. Effect of aspirin on cancer incidence and mortality in older adults. J. Natl Cancer Inst. 113, 258–265 (2021).
McNeil, J. J. et al. Effect of aspirin on all-cause mortality in the healthy elderly. N. Engl. J. Med. 379, 1519–1528 (2018).
Floyd, J. S. & Serebruany, V. L. Prasugrel as a potential cancer promoter: review of the unpublished data. Arch. Intern. Med. 170, 1078–1080 (2010).
Wiviott, S. D. et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N. Engl. J. Med. 357, 2001–2015 (2007).
Morrow, D. A. et al. Vorapaxar in the secondary prevention of atherothrombotic events. N. Engl. J. Med. 366, 1404–1413 (2012).
Wang, Y., Li, Z., Mo, F., Gu, Z. & Hu, Q. Engineered platelets: advocates for tumor immunotherapy. Nano Today 40, 101281 (2021).
Lv, Y., Wei, W. & Ma, G. Recent advances in platelet engineering for anti-cancer therapies. Particuology 64, 2–13 (2022).
Hu, Q. et al. Inhibition of post-surgery tumour recurrence via a hydrogel releasing CAR-T cells and anti-PDL1-conjugated platelets. Nat. Biomed. Eng. 5, 1038–1047 (2021).
Hu, Q. et al. Conjugation of haematopoietic stem cells and platelets decorated with anti-PD-1 antibodies augments anti-leukaemia efficacy. Nat. Biomed. Eng. 2, 831–840 (2018).
Yap, M. L. et al. Activated platelets in the tumor microenvironment for targeting of antibody-drug conjugates to tumors and metastases. Theranostics 9, 1154–1169 (2019).
Wang, X., Chen, J. & Shi, H. Platelets: novel biomaterials for cancer diagnosis and therapeutic delivery. MedComm – Biomater. Appl. 4, e70010 (2025).
Li, Z. et al. Cell-based delivery systems: emerging carriers for immunotherapy. Adv. Funct. Mater. 31, 2100088 (2021).
Sarkar, S., Alam, M. A., Shaw, J. & Dasgupta, A. K. Drug delivery using platelet cancer cell interaction. Pharm. Res. 30, 2785–2794 (2013).
Ortiz-Otero, N., Mohamed, Z. & King, M. R. in Biomechanics in Oncology (eds Dong, C. et al.) 235–251 (Springer, 2018).
Zhang, Y. et al. A platelet intelligent vehicle with navigation for cancer photothermal-chemotherapy. ACS Nano 16, 6359–6371 (2022).
Tang, S. et al. Enzyme-powered Janus platelet cell robots for active and targeted drug delivery. Sci. Robot. 5, eaba6137 (2020).
Zheng, Q. et al. The recent progress on metal-organic frameworks for phototherapy. Chem. Soc. Rev. 50, 5086–5125 (2021).
Stubelius, A., Lee, S. & Almutairi, A. The chemistry of boronic acids in nanomaterials for drug delivery. Acc. Chem. Res. 52, 3108–3119 (2019).
Yan, J. et al. Platelet pharmacytes for the hierarchical amplification of antitumor immunity in response to self-generated immune signals. Adv. Mater. 34, e2109517 (2022).
Li, H. et al. Disrupting tumour vasculature and recruitment of aPDL1-loaded platelets control tumour metastasis. Nat. Commun. 12, 2773 (2021).
Lu, Q. et al. Bioengineered platelets combining chemotherapy and immunotherapy for postsurgical melanoma treatment: internal core-loaded doxorubicin and external surface-anchored anti-PD-L1 antibody backpacks. Nano Lett. 22, 3141–3150 (2022).
Wang, Y. et al. Active recruitment of anti-PD-1-conjugated platelets through tumor-selective thrombosis for enhanced anticancer immunotherapy. Sci. Adv. 9, eadf6854 (2023).
Chen, Y., Pal, S. & Hu, Q. Cell-based relay delivery strategy in biomedical applications. Adv. Drug Deliv. Rev. 198, 114871 (2023).
Fan, X. et al. Surface-anchored tumor microenvironment-responsive protein nanogel-platelet system for cytosolic delivery of therapeutic protein in the post-surgical cancer treatment. Acta Biomater. 154, 412–423 (2022).
Wang, S. et al. Engineered platelets-based drug delivery platform for targeted thrombolysis. Acta Pharm. Sin. B 12, 2000–2013 (2022).
Zhou, L., Feng, W., Mao, Y., Chen, Y. & Zhang, X. Nanoengineered sonosensitive platelets for synergistically augmented sonodynamic tumor therapy by glutamine deprivation and cascading thrombosis. Bioact. Mater. 24, 26–36 (2023).
Fuentes, R. E. et al. A chimeric platelet-targeted urokinase prodrug selectively blocks new thrombus formation. J. Clin. Invest. 126, 483–494 (2016).
Ding, Y. F. et al. Supramolecularly functionalized platelets for rapid control of hemorrhage. Acta Biomater. 149, 248–257 (2022).
Shen, D. et al. Antibody-armed platelets for the regenerative targeting of endogenous stem cells. Nano Lett. 19, 1883–1891 (2019).
Zhao, J. et al. Glucose-decorated engineering platelets for active and precise tumor-targeted drug delivery. Biomater. Sci. 11, 3965–3975 (2023).
Yang, Y. et al. T cell-mimicking platelet-drug conjugates. Matter 6, 2340–2355 (2023).
Gao, Y. et al. Engineering platelets with PDL1 antibodies and iron oxide nanoparticles for postsurgical cancer immunotherapy. ACS Appl. Bio Mater. 6, 257–266 (2023).
Hansen, C. E. et al. Platelet-microcapsule hybrids leverage contractile force for targeted delivery of hemostatic agents. ACS Nano 11, 5579–5589 (2017).
Stephan, M. T., Moon, J. J., Um, S. H., Bershteyn, A. & Irvine, D. J. Therapeutic cell engineering with surface-conjugated synthetic nanoparticles. Nat. Med. 16, 1035–1041 (2010).
Lu, R., Li, Y., Xu, A., King, B. & Ruan, K. H. Reprogramming megakaryocytes for controlled release of platelet-like particles carrying a single-chain thromboxane A2 receptor-g-protein complex with therapeutic potential. Cells 12, 2775 (2023).
Ma, Y. et al. Bioengineering platelets presenting PD-L1, galectin-9 and BTLA to ameliorate type 1 diabetes. Adv. Sci. 12, e2501139 (2025).
He, J. et al. Platelet backpacking nanoparticles based on bacterial outer membrane vesicles enhanced photothermal-immune anti-tumor therapy. Nanoscale 17, 1510–1523 (2025).
Feng, Y. et al. Engineering supramolecular peptide nanofibers for in vivo platelet-hitchhiking beyond ligand-receptor recognition. Sci. Adv. 10, eadq2072 (2024).
Li, Y. T., Nishikawa, T. & Kaneda, Y. Platelet-cytokine complex suppresses tumour growth by exploiting intratumoural thrombin-dependent platelet aggregation. Sci. Rep. 6, 25077 (2016).
Xu, H. Z. et al. Targeted photodynamic therapy of glioblastoma mediated by platelets with photo-controlled release property. Biomaterials 290, 121833 (2022).
Cacic, D., Hervig, T. & Reikvam, H. Platelets for advanced drug delivery in cancer. Expert. Opin. Drug Deliv. 20, 673–688 (2023).
Xu, P. et al. Doxorubicin-loaded platelets conjugated with anti-CD22 mAbs: a novel targeted delivery system for lymphoma treatment with cardiopulmonary avoidance. Oncotarget 8, 58322–58337 (2017).
Lu, Q. et al. Long-acting bioengineered platelets with internal doxorubicin loaded and external quercetin liposomes anchored for post-surgical tumor therapy. J. Control. Rel. 381, 113546 (2025).
Dong, H., Gao, M., Lu, L., Gui, R. & Fu, Y. Doxorubicin-loaded platelet decoys for enhanced chemoimmunotherapy against triple-negative breast cancer in mice model. Int. J. Nanomed. 18, 3577–3593 (2023).
Xu, P. et al. Doxorubicin-loaded platelets as a smart drug delivery system: an improved therapy for lymphoma. Sci. Rep. 7, 42632 (2017).
Wu, Y. W. et al. Clinical-grade cryopreserved doxorubicin-loaded platelets: role of cancer cells and platelet extracellular vesicles activation loop. J. Biomed. Sci. 27, 45 (2020).
Tanaka, H. et al. Treatment of hepatocellular carcinoma with autologous platelets encapsulating sorafenib or lenvatinib: a novel therapy exploiting tumor–platelet interactions. Int. J. Cancer 150, 1640–1653 (2022).
Rao, L. et al. Platelet-facilitated photothermal therapy of head and neck squamous cell carcinoma. Angew. Chem. Int. Ed. Engl. 57, 986–991 (2018).
Nishikawa, T., Tung, L. Y. & Kaneda, Y. Systemic administration of platelets incorporating inactivated Sendai virus eradicates melanoma in mice. Mol. Ther. 22, 2046–2055 (2014).
Abdelgawwad, M. S. et al. Transfusion of platelets loaded with recombinant ADAMTS13 (A Disintegrin and Metalloprotease With Thrombospondin Type 1 Repeats-13) is efficacious for inhibiting arterial thrombosis associated with thrombotic thrombocytopenic purpura. Arterioscler. Thromb. Vasc. Biol. 38, 2731–2743 (2018).
Lv, Y. et al. Near-infrared light-triggered platelet arsenal for combined photothermal-immunotherapy against cancer. Sci. Adv. 7, eabd7614 (2021).
Zhao, Z. et al. Platelet-drug conjugates engineered via one-step fusion approach for metastatic and postoperative cancer treatment. Angew. Chem. Int. Ed. Engl. 63, e202403541 (2024).
Morales-Pacheco, M. et al. The role of platelets in cancer: from their influence on tumor progression to their potential use in liquid biopsy. Biomarker Res. 13, 27 (2025).
Geranpayehvaghei, M. et al. Targeting delivery of platelets inhibitor to prevent tumor metastasis. Bioconjug. Chem. 30, 2349–2357 (2019).
Demers, M. & Wagner, D. D. Targeting platelet function to improve drug delivery. Oncoimmunology 1, 100–102 (2012).
Flaumenhaft, R., Mairuhu, A. T. & Italiano, J. E. Platelet- and megakaryocyte-derived microparticles. Semin. Thromb. Hemost. 36, 881–887 (2010).
Heijnen, H. F., Schiel, A. E., Fijnheer, R., Geuze, H. J. & Sixma, J. J. Activated platelets release two types of membrane vesicles: microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and α-granules. Blood 94, 3791–3799 (1999).
Tao, S. C., Guo, S. C. & Zhang, C. Q. Platelet-derived extracellular vesicles: an emerging therapeutic approach. Int. J. Biol. Sci. 13, 828–834 (2017).
Soleymani, S., Yari, F., Bolhassani, A. & Bakhshandeh, H. Platelet microparticles: an effective delivery system for anti-viral drugs. J. Drug Deliv. Sci. Technol. 51, 290–296 (2019).
Gamonet, C. et al. Processing methods and storage duration impact extracellular vesicle counts in red blood cell units. Blood Adv. 4, 5527–5539 (2020).
Yao, C. & Wang, C. Platelet-derived extracellular vesicles for drug delivery. Biomater. Sci. 11, 5758–5768 (2023).
Meliciano, A., Salvador, D., Mendonca, P., Louro, A. F. & Serra, M. Clinically expired platelet concentrates as a source of extracellular vesicles for targeted anti-cancer drug delivery. Pharmaceutics 15, 953 (2023).
Kailashiya, J., Gupta, V. & Dash, D. Engineered human platelet-derived microparticles as natural vectors for targeted drug delivery. Oncotarget 10, 5835–5846 (2019).
Ning, S. et al. Platelet-derived exosomes hybrid liposomes facilitate uninterrupted singlet oxygen generation to enhance breast cancer immunotherapy. Cell Rep. Phys. Sci. 4, 101505 (2023).
Wang, H. et al. Platelet–membrane–biomimetic nanoparticles for targeted antitumor drug delivery. J. Nanobiotechnol. 17, 60 (2019).
Fang, R. H., Gao, W. & Zhang, L. Targeting drugs to tumours using cell membrane-coated nanoparticles. Nat. Rev. Clin. Oncol. 20, 33–48 (2023).
Hu, Q. et al. Anticancer platelet-mimicking nanovehicles. Adv. Mater. 27, 7043–7050 (2015).
Gu, Z. & Hu, Q. Platelet membrane-coated drug delivery system. US patent 10,363,226 (30 July 2019).
Hu, Q. et al. Engineered nanoplatelets for enhanced treatment of multiple myeloma and thrombus. Adv. Mater. 28, 9573–9580 (2016).
Mei, D. et al. Platelet membrane-cloaked paclitaxel-nanocrystals augment postoperative chemotherapeutical efficacy. J. Control. Rel. 324, 341–353 (2020).
Zhang, M. et al. Platelet-mimicking biotaxis targeting vasculature-disrupted tumors for cascade amplification of hypoxia-sensitive therapy. ACS Nano 13, 14230–14240 (2019).
Li, J. et al. Targeted drug delivery to circulating tumor cells via platelet membrane-functionalized particles. Biomaterials 76, 52–65 (2016).
Bahmani, B. et al. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat. Commun. 12, 1999 (2021).
Hubbell, J. A. & Langer, R. Translating materials design to the clinic. Nat. Mater. 12, 963–966 (2013).
Danhier, F. To exploit the tumor microenvironment: since the EPR effect fails in the clinic, what is the future of nanomedicine? J. Control. Rel. 244, 108–121 (2016).
Quader, S. & Kataoka, K. Nanomaterial-enabled cancer therapy. Mol. Ther. 25, 1501–1513 (2017).
Rosenblum, D., Joshi, N., Tao, W., Karp, J. M. & Peer, D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 9, 1410 (2018).
Au, J. L. S., Jang, S. H. & Wientjes, M. G. Clinical aspects of drug delivery to tumors. J. Control. Rel. 78, 81–95 (2002).
Minchinton, A. I. & Tannock, I. F. Drug penetration in solid tumours. Nat. Rev. Cancer 6, 583–592 (2006).
Wang, S. et al. Drug targeting via platelet membrane-coated nanoparticles. Small Struct. 1, 2000018 (2020).
Dash, P., Piras, A. M. & Dash, M. Cell membrane coated nanocarriers — an efficient biomimetic platform for targeted therapy. J. Control. Rel. 327, 546–570 (2020).
Schiffer, C. A. et al. Platelet transfusion for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J. Clin. Oncol. 36, 283–299 (2018).
Kogler, V. J. et al. Platelet dysfunction reversal with cold-stored vs room temperature-stored platelet transfusions. Blood 143, 2073–2088 (2024).
Haemmerle, M. et al. FAK regulates platelet extravasation and tumor growth after antiangiogenic therapy withdrawal. J. Clin. Invest. 126, 1885–1896 (2016).
Rachidi, S. et al. Platelets subvert T cell immunity against cancer via GARP–TGFβ axis. Sci. Immunol. 2, eaai7911 (2017).
Braun, A., Anders, H. J., Gudermann, T. & Mammadova-Bach, E. Platelet-cancer interplay: molecular mechanisms and new therapeutic avenues. Front. Oncol. 11, 665534 (2021).
Li, J., Sharkey, C. C., Wun, B., Liesveld, J. L. & King, M. R. Genetic engineering of platelets to neutralize circulating tumor cells. J. Control. Rel. 228, 38–47 (2016).
Zhang, X. et al. Engineering PD-1-presenting platelets for cancer immunotherapy. Nano Lett. 18, 5716–5725 (2018).
Strong, C. et al. Genetic engineering of transfusable platelets with mRNA-lipid nanoparticles is compatible with blood banking practices. Blood 144, 2223–2236 (2024).
Leung, J. et al. Genetically engineered transfusable platelets using mRNA lipid nanoparticles. Sci. Adv. 9, eadi0508 (2023).
Chatterjee, S., Kon, E., Sharma, P. & Peer, D. Endosomal escape: a bottleneck for LNP-mediated therapeutics. Proc. Natl Acad. Sci. USA 121, e2307800120 (2024).
Kong, H. et al. An antifouling membrane-fusogenic liposome for effective intracellular delivery in vivo. Nat. Commun. 15, 4267 (2024).
Novakowski, S., Jiang, K., Prakash, G. & Kastrup, C. Delivery of mRNA to platelets using lipid nanoparticles. Sci. Rep. 9, 552 (2019).
Strilchuk, A. W. et al. Sustained depletion of FXIII-A by inducing acquired FXIII-B deficiency. Blood 136, 2946–2954 (2020).
Hong, W. et al. Transfection of human platelets with short interfering RNA. Clin. Transl. Sci. 4, 180–182 (2011).
Lazar, S., Wurtzel, J. G. T., Chen, X., Ma, P. & Goldfinger, L. E. High-efficiency unassisted transfection of platelets with naked double-stranded miRNAs modulates signal-activated translation and platelet function. Platelets 32, 794–806 (2021).
Wang, Y. et al. Chemically engineering cells for precision medicine. Chem. Soc. Rev. 52, 1068–1102 (2023).
Hoffmeister, K. M. & Falet, H. Platelet clearance by the hepatic ashwell-morrell receptor: mechanisms and biological significance. Thromb. Res. 141 (Suppl. 2), S68–S72 (2016).
Reusswig, F., An, O. & Deppermann, C. Platelet life cycle during aging: function, production and clearance. Platelets 35, 2433750 (2024).
Li, J. et al. Desialylated platelet clearance in the liver is a novel mechanism of systemic immunosuppression. Research 6, 0236 (2023).
Papa, A. L. et al. Platelet decoys inhibit thrombosis and prevent metastatic tumor formation in preclinical models. Sci. Transl. Med. 11, eaau5898 (2019).
Tamura, T. & Hamachi, I. N-Acyl-N-alkyl/aryl sulfonamide chemistry assisted by proximity for modification and covalent inhibition of endogenous proteins in living systems. Acc. Chem. Res. 58, 87–100 (2025).
Chen, Y. et al. Engineered platelets as targeted protein degraders and application to breast cancer models. Nat. Biotechnol. 43, 1800–1812 (2024).
Tozzi, L. et al. Multi-channel silk sponge mimicking bone marrow vascular niche for platelet production. Biomaterials 178, 122–133 (2018).
Avanzi, M. P. et al. A novel bioreactor and culture method drives high yields of platelets from stem cells. Transfusion 56, 170–178 (2016).
Thon, J. N. et al. Platelet bioreactor-on-a-chip. Blood 124, 1857–1867 (2014).
Ito, Y. et al. Turbulence activates platelet biogenesis to enable clinical scale ex vivo production. Cell 174, 636–648.e18 (2018).
Sugimoto, N. et al. iPLAT1: the first-in-human clinical trial of iPSC-derived platelets as a phase 1 autologous transfusion study. Blood 140, 2398–2402 (2022).
Sugimoto, N. & Eto, K. Ex vivo production of platelets from iPSCs: the iPLAT1 study and beyond. Hemasphere 7, e884 (2023).
Schut, A. M., Kirschbaum, M., Adelmeijer, J., de Groot, P. G. & Lisman, T. In vitro uptake of recombinant factor VIIa by megakaryocytes with subsequent production of platelets containing functionally active drug. Br. J. Haematol. 178, 482–486 (2017).
Wilcox, D. A. et al. Induction of megakaryocytes to synthesize and store a releasable pool of human factor VIII. J. Thromb. Haemost. 1, 2477–2489 (2003).
Miao, H. Z. et al. Bioengineering of coagulation factor VIII for improved secretion. Blood 103, 3412–3419 (2004).
Figueiredo, C. et al. Generation of HLA-deficient platelets from hematopoietic progenitor cells. Transfusion 50, 1690–1701 (2010).
Suzuki, D. et al. iPSC-derived platelets depleted of HLA class I are inert to anti-HLA class I and natural killer cell immunity. Stem Cell Rep. 14, 49–59 (2020).
Fuentes, R. et al. Infusion of mature megakaryocytes into mice yields functional platelets. J. Clin. Invest. 120, 3917–3922 (2010).
Patel, A. et al. Pre-clinical development of a cryopreservable megakaryocytic cell product capable of sustained platelet production in mice. Transfusion 59, 3698–3713 (2019).
Kim, H. et al. Studies of infused megakaryocytes into mice support a “catch-and-release” model of pulmonary-centric thrombopoiesis. Preprint at bioRxiv https://doi.org/10.1101/2024.06.04.597316 (2024).
Wang, X. et al. Intraosseous delivery of lentiviral vectors targeting factor VIII expression in platelets corrects murine hemophilia A. Mol. Ther. 23, 617–626 (2015).
Wang, X. et al. Enhancing therapeutic efficacy of in vivo platelet-targeted gene therapy in hemophilia a mice. Blood Adv. 4, 5722–5734 (2020).
Basha, G. et al. Lipid nanoparticle delivery of siRNA to osteocytes leads to effective silencing of SOST and inhibition of sclerostin in vivo. Mol. Ther. Nucleic Acids 5, e363 (2016).
Sago, C. D. et al. Nanoparticles that deliver RNA to bone marrow identified by in vivo directed evolution. J. Am. Chem. Soc. 140, 17095–17105 (2018).
Labrosse, R. et al. Outcomes of hematopoietic stem cell gene therapy for Wiskott–Aldrich syndrome. Blood 142, 1281–1296 (2023).
Gratwohl, A. et al. Hematopoietic stem cell transplantation for hematological malignancies in Europe. Leukemia 17, 941–959 (2003).
Wojtukiewicz, M. Z., Sierko, E., Hempel, D., Tucker, S. C. & Honn, K. V. Platelets and cancer angiogenesis nexus. Cancer Metastasis Rev. 36, 249–262 (2017).
Borsig, L. et al. Heparin and cancer revisited: mechanistic connections involving platelets, P-selectin, carcinoma mucins, and tumor metastasis. Proc. Natl Acad. Sci. USA 98, 3352–3357 (2001).
Karpatkin, S., Pearlstein, E., Ambrogio, C. & Coller, B. S. Role of adhesive proteins in platelet tumor interaction in vitro and metastasis formation in vivo. J. Clin. Invest. 81, 1012–1019 (1988).
Herrmann, I. K., Wood, M. J. A. & Fuhrmann, G. Extracellular vesicles as a next-generation drug delivery platform. Nat. Nanotechnol. 16, 748–759 (2021).
Propper, D. J. & Balkwill, F. R. Harnessing cytokines and chemokines for cancer therapy. Nat. Rev. Clin. Oncol. 19, 237–253 (2022).
Anselmo, A. C. & Mitragotri, S. Nanoparticles in the clinic: an update. Bioeng. Transl. Med. 4, e10143 (2019).
Looby, M. et al. Application of quality by design principles to the development and technology transfer of a major process improvement for the manufacture of a recombinant protein. Biotechnol. Prog. 27, 1718–1729 (2011).
Kim, Y. J. et al. Quality by design characterization of the perfusion culture process for recombinant FVIII. Biologicals 59, 37–46 (2019).
Quach, M. E., Chen, W. & Li, R. Mechanisms of platelet clearance and translation to improve platelet storage. Blood 131, 1512–1521 (2018).
Cesar, J. M. & Vecino, A. M. Survival and function of transfused platelets. studies in two patients with congenital deficiencies of platelet membrane glycoproteins. Platelets 20, 158–162 (2009).
Rank, A. et al. Clearance of platelet microparticles in vivo. Platelets 22, 111–116 (2011).
Bashor, C. J., Hilton, I. B., Bandukwala, H., Smith, D. M. & Veiseh, O. Engineering the next generation of cell-based therapeutics. Nat. Rev. Drug Discov. 21, 655–675 (2022).
Solves Alcaina, P. Platelet transfusion: and update on challenges and outcomes. J. Blood Med. 11, 19–26 (2020).
Manduzio, P. Transfusion-associated graft-versus-host disease: a concise review. Hematol. Rep. 10, 7724 (2018).
Leitner, G. C. et al. Influence of human platelet antigen match on the success of stem cell transplantation after myeloablative conditioning. Bone Marrow Transpl. 32, 821–824 (2003).
Duquesnoy, R. J., Filip, D. J., Rodey, G. E., Rimm, A. A. & Aster, R. H. Successful transfusion of platelets “mismatched” for HLA antigens to alloimmunized thrombocytopenic patients. Am. J. Hematol. 2, 219–226 (1977).
Ward, S. R. et al. 984-30 use of electroporated platelets as a novel drug delivery system in preventing complications of coronary angioplasty. J. Am. Coll. Cardiol. 25, 303A–304A (1995).
Acknowledgements
The work of the authors is supported, in part, by an American Cancer Society Research Scholar Grant (grant number RSG-23-1140821-01-ET, to Q.H.), a METAVIVOR Foundation Early Career Research Grant Award (to Q.H.) and V Foundation Scholar Grant (to Q.H.). The authors also thankfully acknowledge support from the Pancreas Cancer Task Force and the University of Wisconsin Carbone Cancer Center Research Collaborative, and the start-up package from the University of Wisconsin–Madison.
Author information
Authors and Affiliations
Contributions
Y.C. and Q.H. researched data for the article. All authors wrote the article, contributed substantially to the discussion of the content, and reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
Z.G. is the co-founder of µZen Inc., ZCapsule Inc. and Zenomics Inc. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks H. Goubran, B. Ho-Tin-Noé and J. Shi for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Wolter, T., Gu, Z. et al. Engineering platelets as cancer therapeutics. Nat Rev Clin Oncol (2026). https://doi.org/10.1038/s41571-026-01122-5
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41571-026-01122-5