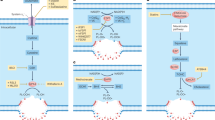
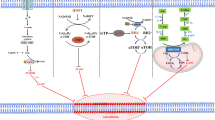
Abstract
Ferroptosis is an oxidative, lipid peroxidation-driven form of regulated cell death that occurs when antioxidant and organelle-protective systems are compromised. Increasing evidence implicates ferroptosis as a process that can exert both tumour-suppressive and tumour-promoting effects depending on cellular context at multiple stages of cancer evolution (from tumour initiation to metastatic colonization), sparking substantial interest in therapeutically exploiting this mechanism of cell death. Yet, despite rapid preclinical progress, clinical translation of ferroptosis-based strategies remains nascent. In this Review, we examine the major barriers to translation, including pharmacological limitations, tumour-intrinsic heterogeneity, microenvironmental and immune constraints, and gaps in current preclinical modelling. We also highlight emerging opportunities such as new ferroptosis-inducing agents, biomarker-guided patient selection and rational combinations with chemotherapy, radiotherapy, targeted agents or immunotherapies. Finally, we outline a translational roadmap for integrating ferroptosis-based therapies into oncology practice. By defining key challenges and future directions, this Review aims to position ferroptosis as a viable therapeutic paradigm and to accelerate progress towards clinical application.
Key points
-
Ferroptosis is a lipid peroxidation-driven cell death process shaped by antioxidant, metabolic and organelle-protective pathways, highlighting the adaptive plasticity that cancer cells use to maintain redox homeostasis.
-
Tumour-intrinsic heterogeneity, including variable genetic, metabolic, epigenetic and lineage programmes, creates diverse ferroptosis sensitivities, reinforcing the need for biomarker-guided patient selection rather than uniform therapeutic strategies.
-
The tumour microenvironment, including stromal buffering, hypoxia, acidosis, immune cell vulnerability and microbiota-derived metabolites, profoundly modulates ferroptosis, making therapeutic induction inherently context dependent.
-
First-generation ferroptosis inducers have limited drug-like properties, whereas new agents, including GPX4 and FSP1 inhibitors, degraders, prodrugs, nanocarriers and antibody–drug conjugates, offer improved potency and tumour selectivity.
-
Integrated genomic, transcriptomic, lipidomic and imaging biomarkers, including ACSL4, TFRC, various damage-associated molecular patterns and ferroptosis-focused PET probes, are essential for predicting ferroptosis sensitivity and pharmacodynamic monitoring pathway modulation.
-
Rational combinations of ferroptosis inducers with immunotherapy, radiotherapy, targeted therapies and metabolic inhibitors can exploit ferroptosis vulnerabilities to overcome therapeutic resistance and improve efficacy but require careful calibration to avoid immune or non-malignant tissue toxicity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galluzzi, L., Kepp, O., Zitvogel, L., Tang, D. & Kroemer, G. Cancer cell death: cell-autonomous and immunogenic dimensions. Cancer Cell 44, 281–305 (2026).
Dolma, S., Lessnick, S. L., Hahn, W. C. & Stockwell, B. R. Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells. Cancer Cell 3, 285–296 (2003).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012).
Dai, E. et al. A guideline on the molecular ecosystem regulating ferroptosis. Nat. Cell Biol. 26, 1447–1457 (2024).
Xue, Q. et al. Copper-dependent autophagic degradation of GPX4 drives ferroptosis. Autophagy 19, 1982–1996 (2023).
Chen, J. et al. Unusual iron-independent ferroptosis-like cell death induced by photoactivation of a typical iridium complex for hypoxia photodynamic therapy. ACS Appl. Mater. Interfaces 17, 5684–5694 (2025).
Stockwell, B. R. Ferroptosis turns 10: emerging mechanisms, physiological functions, and therapeutic applications. Cell 185, 2401–2421 (2022).
Song, X. et al. Cytosolic cytochrome c represses ferroptosis. Cell Metab. 37, 1326–1343.e10 (2025).
Tang, Q. et al. Bim- and Bax-mediated mitochondrial pathway dominates abivertinib-induced apoptosis and ferroptosis. Free. Radic. Biol. Med. 180, 198–209 (2022).
Hangauer, M. J. et al. Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature 551, 247–250 (2017).
Viswanathan, V. S. et al. Dependency of a therapy-resistant state of cancer cells on a lipid peroxidase pathway. Nature 547, 453–457 (2017).
Chen, X., Kang, R., Kroemer, G. & Tang, D. Broadening horizons: the role of ferroptosis in cancer. Nat. Rev. Clin. Oncol. 18, 280–296 (2021).
Wahida, A. & Conrad, M. Decoding ferroptosis for cancer therapy. Nat. Rev. Cancer 25, 910–924 (2025).
Friedmann Angeli, J. P. et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat. Cell Biol. 16, 1180–1191 (2014).
Doll, S. et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575, 693–698 (2019).
Bersuker, K. et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575, 688–692 (2019).
Mao, C. et al. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature 593, 586–590 (2021).
Soula, M. et al. Metabolic determinants of cancer cell sensitivity to canonical ferroptosis inducers. Nat. Chem. Biol. 16, 1351–1360 (2020).
Kraft, V. A. N. et al. GTP cyclohydrolase 1/tetrahydrobiopterin counteract ferroptosis through lipid remodeling. ACS Cent. Sci. 6, 41–53 (2020).
Mishima, E. et al. A non-canonical vitamin K cycle is a potent ferroptosis suppressor. Nature 608, 778–783 (2022).
Kolbrink, B. et al. Vitamin K1 inhibits ferroptosis and counteracts a detrimental effect of phenprocoumon in experimental acute kidney injury. Cell Mol. Life Sci. 79, 387 (2022).
Wu, Z. et al. Hydropersulfides inhibit lipid peroxidation and protect cells from ferroptosis. J. Am. Chem. Soc. 144, 15825–15837 (2022).
Chidley, C. et al. A CRISPRi/a screening platform to study cellular nutrient transport in diverse microenvironments. Nat. Cell Biol. 26, 825–838 (2024).
Yang, W. S. et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 156, 317–331 (2014).
Zou, Y. et al. A GPX4-dependent cancer cell state underlies the clear-cell morphology and confers sensitivity to ferroptosis. Nat. Commun. 10, 1617 (2019).
Xie, Y., Kang, R., Klionsky, D. J. & Tang, D. GPX4 in cell death, autophagy, and disease. Autophagy 19, 2621–2638 (2023).
Badgley, M. A. et al. Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 368, 85–89 (2020).
Zhou, N. et al. SLC7A11 is an unconventional H+ transporter in lysosomes. Cell 188, 3441–3458.e25 (2025).
Yang, J. S. et al. ALDH7A1 protects against ferroptosis by generating membrane NADH and regulating FSP1. Cell 188, 2569–2585.e20 (2025).
Dai, E. et al. AIFM2 blocks ferroptosis independent of ubiquinol metabolism. Biochem. Biophys. Res. Commun. 523, 966–971 (2020).
Lange, M. et al. FSP1-mediated lipid droplet quality control prevents neutral lipid peroxidation and ferroptosis. Nat. Cell Biol. 27, 1902–1913 (2025).
Palma, M. et al. Lymph node environment drives FSP1 targetability in metastasizing melanoma. Nature 649, 477–486 (2026).
Emmanuel, N., Li, H., Chen, J. & Zhang, Y. FSP1, a novel KEAP1/NRF2 target gene regulating ferroptosis and radioresistance in lung cancers. Oncotarget 13, 1136–1139 (2022).
Takahashi, N. et al. 3D culture models with CRISPR screens reveal hyperactive NRF2 as a prerequisite for spheroid formation via regulation of proliferation and ferroptosis. Mol. Cell 80, 828–844.e6 (2020).
Sun, X. et al. Activation of the p62–Keap1–NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells. Hepatology 63, 173–184 (2016).
Sun, X. et al. Metallothionein-1G facilitates sorafenib resistance through inhibition of ferroptosis. Hepatology 64, 488–500 (2016).
Yan, B. et al. Membrane damage during ferroptosis is caused by oxidation of phospholipids catalyzed by the oxidoreductases POR and CYB5R1. Mol. Cell 81, 355–369 (2021).
Zou, Y. et al. Cytochrome P450 oxidoreductase contributes to phospholipid peroxidation in ferroptosis. Nat. Chem. Biol. 16, 302–309 (2020).
Hou, W. et al. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 12, 1425–1428 (2016).
Gao, M. et al. Ferroptosis is an autophagic cell death process. Cell Res. 26, 1021–1032 (2016).
Muller, S. et al. CD44 regulates epigenetic plasticity by mediating iron endocytosis. Nat. Chem. 12, 929–938 (2020).
Wang, Y., Liu, Y., Liu, J., Kang, R. & Tang, D. NEDD4L-mediated LTF protein degradation limits ferroptosis. Biochem. Biophys. Res. Commun. 531, 581–587 (2020).
Li, J. et al. Tumor heterogeneity in autophagy-dependent ferroptosis. Autophagy 17, 3361–3374 (2021).
Liu, J. et al. NUPR1 is a critical repressor of ferroptosis. Nat. Commun. 12, 647 (2021).
Zhuang, X. et al. Ageing limits stemness and tumorigenesis by reprogramming iron homeostasis. Nature 637, 184–194 (2025).
Yu, Y. et al. Hepatic transferrin plays a role in systemic iron homeostasis and liver ferroptosis. Blood 136, 726–739 (2020).
Yuan, H., Li, X., Zhang, X., Kang, R. & Tang, D. Identification of ACSL4 as a biomarker and contributor of ferroptosis. Biochem. Biophys. Res. Commun. 478, 1338–1343 (2016).
Doll, S. et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 13, 91–98 (2017).
Kagan, V. E. et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 13, 81–90 (2017).
Zhang, H. L. et al. PKCbetaII phosphorylates ACSL4 to amplify lipid peroxidation to induce ferroptosis. Nat. Cell Biol. 24, 88–98 (2022).
Magtanong, L. et al. Exogenous monounsaturated fatty acids promote a ferroptosis-resistant cell state. Cell Chem. Biol. 26, 420–432.e29 (2019).
Luis, G. et al. Tumor resistance to ferroptosis driven by stearoyl-CoA desaturase-1 (SCD1) in cancer cells and fatty acid biding protein-4 (FABP4) in tumor microenvironment promote tumor recurrence. Redox Biol. 43, 102006 (2021).
Li, Z. et al. LPCAT1-mediated membrane phospholipid remodelling promotes ferroptosis evasion and tumour growth. Nat. Cell Biol. 26, 811–824 (2024).
Lin, Z. et al. The lipid flippase SLC47A1 blocks metabolic vulnerability to ferroptosis. Nat. Commun. 13, 7965 (2022).
Gao, M. et al. Role of mitochondria in ferroptosis. Mol. Cell 73, 354–363.e3 (2019).
Gao, M., Monian, P., Quadri, N., Ramasamy, R. & Jiang, X. Glutaminolysis and transferrin regulate ferroptosis. Mol. Cell 59, 298–308 (2015).
Qiu, B. et al. Phospholipids with two polyunsaturated fatty acyl tails promote ferroptosis. Cell 187, 1177–1190.e18 (2024).
Deshwal, S. et al. Mitochondria regulate intracellular coenzyme Q transport and ferroptotic resistance via STARD7. Nat. Cell Biol. 25, 246–257 (2023).
Mishima, E. et al. DHODH inhibitors sensitize to ferroptosis by FSP1 inhibition. Nature 619, E9–E18 (2023).
Guo, W. et al. Mitochondrial CCN1 drives ferroptosis via fatty acid β-oxidation. Dev. Cell 60, 2294–2312 (2025).
Liang, F. G. et al. OPA1 promotes ferroptosis by augmenting mitochondrial ROS and suppressing an integrated stress response. Mol. Cell 84, 3098–3114.e6 (2024).
Li, C., Liu, J., Hou, W., Kang, R. & Tang, D. STING1 promotes ferroptosis through MFN1/2-dependent mitochondrial fusion. Front. Cell Dev. Biol. 9, 698679 (2021).
Liu, X. et al. Endocytosis is essential for cysteine-deprivation-induced ferroptosis. Mol. Cell 85, 3333–3342.e4 (2025).
Caneque, T. et al. Activation of lysosomal iron triggers ferroptosis in cancer. Nature 642, 492–500 (2025).
Gao, H. et al. Ferroptosis is a lysosomal cell death process. Biochem. Biophys. Res. Commun. 503, 1550–1556 (2018).
Swanda, R. V. et al. Lysosomal cystine governs ferroptosis sensitivity in cancer via cysteine stress response. Mol. Cell 83, 3347–3359.e9 (2023).
Armenta, D. A. et al. Ferroptosis inhibition by lysosome-dependent catabolism of extracellular protein. Cell Chem. Biol. 29, 1588–1600.e7 (2022).
von Krusenstiern, A. N. et al. Identification of essential sites of lipid peroxidation in ferroptosis. Nat. Chem. Biol. 19, 719–730 (2023).
Sassano, M. L. et al. Endoplasmic reticulum-mitochondria contacts are prime hotspots of phospholipid peroxidation driving ferroptosis. Nat. Cell Biol. 27, 902–917 (2025).
Zou, Y. et al. Plasticity of ether lipids promotes ferroptosis susceptibility and evasion. Nature 585, 603–608 (2020).
Bai, Y. et al. Lipid storage and lipophagy regulates ferroptosis. Biochem. Biophys. Res. Commun. 508, 997–1003 (2019).
Riegman, M. et al. Ferroptosis occurs through an osmotic mechanism and propagates independently of cell rupture. Nat. Cell Biol. 22, 1042–1048 (2020).
Co, H. K. C., Wu, C. C., Lee, Y. C. & Chen, S. H. Emergence of large-scale cell death through ferroptotic trigger waves. Nature 631, 654–662 (2024).
Ramos, S., Hartenian, E., Santos, J. C., Walch, P. & Broz, P. NINJ1 induces plasma membrane rupture and release of damage-associated molecular pattern molecules during ferroptosis. EMBO J. 43, 1164–1186 (2024).
Kayagaki, N. et al. NINJ1 mediates plasma membrane rupture during lytic cell death. Nature 591, 131–136 (2021).
Hirata, Y. et al. Lipid peroxidation increases membrane tension, Piezo1 gating, and cation permeability to execute ferroptosis. Curr. Biol. 33, 1282–1294.e5 (2023).
Dai, E., Meng, L., Kang, R., Wang, X. & Tang, D. ESCRT-III-dependent membrane repair blocks ferroptosis. Biochem. Biophys. Res. Commun. 522, 415–421 (2020).
Wu, J. et al. Intercellular interaction dictates cancer cell ferroptosis via NF2-YAP signalling. Nature 572, 402–406 (2019).
Chen, X. et al. A noncanonical function of EIF4E limits ALDH1B1 activity and increases susceptibility to ferroptosis. Nat. Commun. 13, 6318 (2022).
Yang, W. S. & Stockwell, B. R. Synthetic lethal screening identifies compounds activating iron-dependent, nonapoptotic cell death in oncogenic-RAS-harboring cancer cells. Chem. Biol. 15, 234–245 (2008).
Eaton, J. K. et al. Selective covalent targeting of GPX4 using masked nitrile-oxide electrophiles. Nat. Chem. Biol. 16, 497–506 (2020).
Cheff, D. M. et al. The ferroptosis inducing compounds RSL3 and ML162 are not direct inhibitors of GPX4 but of TXNRD1. Redox Biol. 62, 102703 (2023).
Ren, X. et al. Programmable melanoma-targeted radio-immunotherapy via fusogenic liposomes functionalized with multivariate-gated aptamer assemblies. Nat. Commun. 15, 5035 (2024).
Herrick, W. G. et al. Potent ferroptosis agent RSL3 induces cleavage of pyroptosis-specific gasdermins in cancer cells. Sci. Rep. 15, 25249 (2025).
DeAngelo, S. L. et al. Recharacterization of the tumor suppressive mechanism of RSL3 identifies the selenoproteome as a druggable pathway in colorectal cancer. Cancer Res. 85, 2788–2804 (2025).
Zhang, Y. et al. Imidazole ketone erastin induces ferroptosis and slows tumor growth in a mouse lymphoma model. Cell Chem. Biol. 26, 623–633.e9 (2019).
Nakamura, T. et al. Phase separation of FSP1 promotes ferroptosis. Nature 619, 371–377 (2023).
Nakamura, T. et al. Integrated chemical and genetic screens unveil FSP1 mechanisms of ferroptosis regulation. Nat. Struct. Mol. Biol. 30, 1806–1815 (2023).
Hendricks, J. M. et al. Identification of structurally diverse FSP1 inhibitors that sensitize cancer cells to ferroptosis. Cell Chem. Biol. 30, 1090–1103.e7 (2023).
Salem, S. et al. Targeting FSP1 to induce ferroptosis in chromophobe renal cell carcinoma. Oncogene 44, 4075–4086 (2025).
Wu, K. et al. Targeting FSP1 triggers ferroptosis in lung cancer. Nature 649, 487–495 (2026).
Liu, J., Kang, R. & Tang, D. Adverse effects of ferroptotic therapy: mechanisms and management. Trends Cancer 10, 417–429 (2024).
Ge, C. et al. Emerging mechanisms and disease implications of ferroptosis: potential applications of natural products. Front. Cell Dev. Biol. 9, 774957 (2021).
Crippa, V. et al. Integrative analysis of KEAP1/NFE2L2 alterations across 3600+ tumors reveals an NRF2 expression signature as a prognostic biomarker in cancer. NPJ Precis. Oncol. 9, 291 (2025).
Wohlhieter, C. A. et al. Concurrent mutations in STK11 and KEAP1 promote ferroptosis protection and SCD1 Dependence in lung cancer. Cell Rep. 33, 108444 (2020).
Shimura, T. et al. The prognostic importance of the negative regulators of ferroptosis, GPX4 and HSPB1, in patients with colorectal cancer. Oncol. Lett. 29, 144 (2025).
Li, X., Zhu, M. & Dong, R. Ferroptosis’s kmaster switch GPX4 emerges as universal biomarker for precision immunotherapy: a pan-cancer study with in vitro experiments validation. Front. Oncol. 15, 1643235 (2025).
Koppula, P. et al. A targetable CoQ-FSP1 axis drives ferroptosis- and radiation-resistance in KEAP1 inactive lung cancers. Nat. Commun. 13, 2206 (2022).
Jiang, L. et al. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 520, 57–62 (2015).
Yang, X. et al. Regulation of VKORC1L1 is critical for p53-mediated tumor suppression through vitamin K metabolism. Cell Metab. 35, 1474–1490.e8 (2023).
Xie, Y. et al. The tumor suppressor p53 limits ferroptosis by blocking DPP4 activity. Cell Rep. 20, 1692–1704 (2017).
Su, Z. et al. Specific regulation of BACH1 by the hotspot mutant p53(R175H) reveals a distinct gain-of-function mechanism. Nat. Cancer 4, 564–581 (2023).
Yagoda, N. et al. RAS-RAF-MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 447, 864–868 (2007).
Andreani, C., Bartolacci, C. & Scaglioni, P. P. Ferroptosis: a specific vulnerability of RAS-driven cancers? Front. Oncol. 12, 923915 (2022).
Lei, G. et al. BRCA1 mediated dual regulation of ferroptosis exposes a vulnerability to GPX4 and PARP co-inhibition in BRCA1-deficient cancers. Cancer Discov. 14, 1476–1495 (2024).
Alborzinia, H. et al. MYCN mediates cysteine addiction and sensitizes neuroblastoma to ferroptosis. Nat. Cancer 3, 471–485 (2022).
Zhang, Y. et al. BAP1 links metabolic regulation of ferroptosis to tumour suppression. Nat. Cell Biol. 20, 1181–1192 (2018).
Liu, Y., Wang, Y., Liu, J., Kang, R. & Tang, D. Interplay between MTOR and GPX4 signaling modulates autophagy-dependent ferroptotic cancer cell death. Cancer Gene Ther. 28, 55–63 (2021).
Yi, J., Zhu, J., Wu, J., Thompson, C. B. & Jiang, X. Oncogenic activation of PI3K-AKT-mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis. Proc. Natl Acad. Sci. USA 117, 31189–31197 (2020).
Zhang, Y. et al. mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat. Commun. 12, 1589 (2021).
Conlon, M. et al. A compendium of kinetic modulatory profiles identifies ferroptosis regulators. Nat. Chem. Biol. 17, 665–674 (2021).
Hua, Y. et al. Targeting SLC7A11-mediated cysteine metabolism for the treatment of trastuzumab-resistant HER2-positive breast cancer. eLife 14, RP103953 (2025).
Zhang, M. et al. PRMT5-mediated arginine methylation of ACSL4 attenuates its stability and suppresses ferroptosis in renal cancer. Research 8, 0789 (2025).
Schmitt, A. et al. BRD4 inhibition sensitizes diffuse large B-cell lymphoma cells to ferroptosis. Blood 142, 1143–1155 (2023).
Wang, Y. et al. Histone demethylase KDM3B protects against ferroptosis by upregulating SLC7A11. FEBS Open Bio 10, 637–643 (2020).
Luo, M. et al. miR-137 regulates ferroptosis by targeting glutamine transporter SLC1A5 in melanoma. Cell Death Differ. 25, 1457–1472 (2018).
Song, K. et al. Plasticity of extrachromosomal and intrachromosomal BRAF amplifications in overcoming targeted therapy dosage challenges. Cancer Discov. 12, 1046–1069 (2022).
He, Y. et al. Multi-omics characterization and therapeutic liability of ferroptosis in melanoma. Signal. Transduct. Target. Ther. 7, 268 (2022).
Klasson, T. D. et al. ACSL3 regulates lipid droplet biogenesis and ferroptosis sensitivity in clear cell renal cell carcinoma. Cancer Metab. 10, 14 (2022).
Feng, H. et al. Transferrin receptor is a specific ferroptosis marker. Cell Rep. 30, 3411–3423.e7 (2020).
Singhal, R. et al. HIF-2α activation potentiates oxidative cell death in colorectal cancers by increasing cellular iron. J. Clin. Invest. 131, e143691 (2021).
Calhoon, D. et al. Glycosaminoglycan-driven lipoprotein uptake protects tumours from ferroptosis. Nature 644, 799–808 (2025).
Yang, C. et al. De novo pyrimidine biosynthetic complexes support cancer cell proliferation and ferroptosis defence. Nat. Cell Biol. 25, 836–847 (2023).
Gentric, G. et al. PML-regulated mitochondrial metabolism enhances chemosensitivity in human ovarian cancers. Cell Metab. 29, 156–173.e10 (2019).
Zhang, X. et al. Targeting ethylglyoxal metabolism to enhance ferroptosis sensitivity in tumor therapy. Adv. Sci. 12, e05356 (2025).
Li, Y. et al. 7-Dehydrocholesterol dictates ferroptosis sensitivity. Nature 626, 411–418 (2024).
Freitas, F. P. et al. 7-Dehydrocholesterol is an endogenous suppressor of ferroptosis. Nature 626, 401–410 (2024).
Urano, Y., Iwagaki, A., Takeishi, A., Uchiyama, N. & Noguchi, N. Downregulation of the SREBP pathways and disruption of redox status by 25-hydroxycholesterol predispose cells to ferroptosis. Free. Radic. Biol. Med. 228, 319–328 (2025).
Wu, R., Liu, J., Tang, D. & Kang, R. The dual role of ACOD1 in inflammation. J. Immunol. 211, 518–526 (2023).
Lin, H. et al. Itaconate transporter SLC13A3 impairs tumor immunity via endowing ferroptosis resistance. Cancer Cell 42, 2032–2044.e6 (2024).
Zhao, Y. et al. Neutrophils resist ferroptosis and promote breast cancer metastasis through aconitate decarboxylase 1. Cell Metab. 35, 1688–1703.e10 (2023).
Liu, K. et al. Induction of autophagy-dependent ferroptosis to eliminate drug-tolerant human retinoblastoma cells. Cell Death Dis. 13, 521 (2022).
Sharma, P. et al. Polyamines buffer labile iron to suppress ferroptosis. Preprint at https://www.biorxiv.org/content/10.1101/2025.06.30.662349v1 (2025).
Tsoi, J. et al. Multi-stage differentiation defines melanoma subtypes with differential vulnerability to drug-induced iron-dependent oxidative stress. Cancer Cell 33, 890–904.e5 (2018).
Graziani, V. et al. SLC7A11 protects amoeboid-disseminating cancer cells from oxidative stress. Cell Rep. 44, 115939 (2025).
Schwab, A. et al. Zeb1 mediates EMT/plasticity-associated ferroptosis sensitivity in cancer cells by regulating lipogenic enzyme expression and phospholipid composition. Nat. Cell Biol. 26, 1470–1481 (2024).
Tsai, T. et al. CD44-hyaluronan mediating endocytosis of iron-platinum alloy nanoparticles induces ferroptotic cell death in mesenchymal-state lung cancer cells with tyrosine kinase inhibitor resistance. Acta Biomater. 186, 396–410 (2024).
Yang, F. et al. Ferroptosis heterogeneity in triple-negative breast cancer reveals an innovative immunotherapy combination strategy. Cell Metab. 35, 84–100.e8 (2023).
Feng, J. et al. ACSL4 is a predictive biomarker of sorafenib sensitivity in hepatocellular carcinoma. Acta Pharmacol. Sin. 42, 160–170 (2021).
Liang, D. et al. Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones. Cell 186, 2748–2764.e22 (2023).
Yan, H. et al. Discovery of decreased ferroptosis in male colorectal cancer patients with KRAS mutations. Redox Biol. 62, 102699 (2023).
Liu, Z. et al. Systematic analysis of the aberrances and functional implications of ferroptosis in cancer. iScience 23, 101302 (2020).
Zhu, Y. et al. Cancer-associated fibroblasts reprogram cysteine metabolism to increase tumor resistance to ferroptosis in pancreatic cancer. Theranostics 14, 1683–1700 (2024).
Gurung, S. et al. Stromal lipid species dictate melanoma metastasis and tropism. Cancer Cell 43, 1108–1124.e11 (2025).
Hecht, F. et al. Catabolism of extracellular glutathione supplies amino acids to support tumor growth. Preprint at bioRxiv https://doi.org/10.1101/2024.10.10.617667 (2024).
Yang, Z. et al. HIF-1α drives resistance to ferroptosis in solid tumors by promoting lactate production and activating SLC1A1. Cell Rep. 42, 112945 (2023).
Chauhan, S. S. et al. Hypoxia induced lipid droplet accumulation promotes resistance to ferroptosis in prostate cancer. Oncotarget 16, 532–544 (2025).
Fan, Z. et al. Hypoxia blocks ferroptosis of hepatocellular carcinoma via suppression of METTL14 triggered YTHDF2-dependent silencing of SLC7A11. J. Cell Mol. Med. 25, 10197–10212 (2021).
Tan, S. K. et al. Obesity-dependent adipokine chemerin suppresses fatty acid oxidation to confer ferroptosis resistance. Cancer Discov. 11, 2072–2093 (2021).
Yang, Z. et al. OGT/HIF-2alpha axis promotes the progression of clear cell renal cell carcinoma and regulates its sensitivity to ferroptosis. iScience 26, 108148 (2023).
Minikes, A. M. et al. HIF-independent oxygen sensing via KDM6A regulates ferroptosis. Mol. Cell 85, 2973–2987.e6 (2025).
Song, J. et al. Hypoxia inhibits ferritinophagy-mediated ferroptosis in esophageal squamous cell carcinoma via the USP2-NCOA4 axis. Oncogene 43, 2000–2014 (2024).
Alvarez, S. W. et al. NFS1 undergoes positive selection in lung tumours and protects cells from ferroptosis. Nature 551, 639–643 (2017).
Dierge, E. et al. Peroxidation of n-3 and n-6 polyunsaturated fatty acids in the acidic tumor environment leads to ferroptosis-mediated anticancer effects. Cell Metab. 33, 1701–1715.e5 (2021).
Song, X. et al. PDK4 dictates metabolic resistance to ferroptosis by suppressing pyruvate oxidation and fatty acid synthesis. Cell Rep. 34, 108767 (2021).
Wen, Q., Liu, J., Kang, R., Zhou, B. & Tang, D. The release and activity of HMGB1 in ferroptosis. Biochem. Biophys. Res. Commun. 510, 278–283 (2019).
Efimova, I. et al. Vaccination with early ferroptotic cancer cells induces efficient antitumor immunity. J. Immunother. Cancer 8, e001369 (2020).
Liu, J. et al. DCN released from ferroptotic cells ignites AGER-dependent immune responses. Autophagy 18, 2036–2049 (2022).
Morotti, M. et al. PGE2 inhibits TIL expansion by disrupting IL-2 signalling and mitochondrial function. Nature 629, 426–434 (2024).
Liu, J. et al. Extracellular GPX4 impairs antitumor immunity via dendritic ZP3 receptors. Cell 189, 1056–1073 (2026).
Ma, X. et al. CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell Metab. 33, 1001–1012.e5 (2021).
Han, L. et al. PPARG-mediated ferroptosis in dendritic cells limits antitumor immunity. Biochem. Biophys. Res. Commun. 576, 33–39 (2021).
Wang, W. et al. CD8+ T cells regulate tumour ferroptosis during cancer immunotherapy. Nature 569, 270–274 (2019).
Liao, P. et al. CD8+ T cells and fatty acids orchestrate tumor ferroptosis and immunity via ACSL4. Cancer Cell 40, 365–378.e6 (2022).
Cui, W. et al. Gut microbial metabolite facilitates colorectal cancer development via ferroptosis inhibition. Nat. Cell Biol. 26, 124–137 (2024).
Fiore, A. et al. Kynurenine importation by SLC7A11 propagates anti-ferroptotic signaling. Mol. Cell 82, 920–932.e27 (2022).
Zitvogel, L., Fidelle, M. & Kroemer, G. Long-distance microbial mechanisms impacting cancer immunosurveillance. Immunity 57, 2013–2029 (2024).
Jia, T. et al. A Pseudomonas aeruginosa quorum-sensing metabolite manipulates macrophage ferroptosis through a methylation pathway. Nat. Commun. 16, 9992 (2025).
Dai, E. et al. Ferroptotic damage promotes pancreatic tumorigenesis through a TMEM173/STING-dependent DNA sensor pathway. Nat. Commun. 11, 6339 (2020).
Rademaker, G. et al. PCSK9 drives sterol-dependent metastatic organ choice in pancreatic cancer. Nature 643, 1381–1390 (2025).
Conche, C. et al. Combining ferroptosis induction with MDSC blockade renders primary tumours and metastases in liver sensitive to immune checkpoint blockade. Gut 72, 1774–1782 (2023).
He, F. et al. ATF4 suppresses hepatocarcinogenesis by inducing SLC7A11 (xCT) to block stress-related ferroptosis. J. Hepatol. 79, 362–377 (2023).
Guo, J. et al. The origin of hepatocellular carcinoma depends on metabolic zonation. Science 391, eadv7129 (2025).
Ubellacker, J. M. et al. Lymph protects metastasizing melanoma cells from ferroptosis. Nature 585, 113–118 (2020).
Hong, X. et al. The lipogenic regulator SREBP2 induces transferrin in circulating melanoma cells and suppresses ferroptosis. Cancer Discov. 11, 678–695 (2021).
Wang, Y. et al. ACSL4 and polyunsaturated lipids support metastatic extravasation and colonization. Cell 188, 412–429.e27 (2025).
Zhao, Z. et al. Sickle cell disease induces chromatin introversion and ferroptosis in CD8+ T cells to suppress anti-tumor immunity. Immunity 58, 1484–1501.e11 (2025).
Morgan, P. K. et al. A lipid atlas of human and mouse immune cells provides insights into ferroptosis susceptibility. Nat. Cell Biol. 26, 645–659 (2024).
Xu, S. et al. Uptake of oxidized lipids by the scavenger receptor CD36 promotes lipid peroxidation and dysfunction in CD8+ T cells in tumors. Immunity 54, 1561–1577.e7 (2021).
Tzeng, S. F. et al. PLT012, a humanized CD36-blocking antibody, is effective for unleashing antitumor immunity against liver cancer and liver metastasis. Cancer Discov. 15, 1676–1696 (2025).
Ping, Y. et al. PD-1 signaling limits expression of phospholipid phosphatase 1 and promotes intratumoral CD8+ T cell ferroptosis. Immunity 57, 2122–2139.e9 (2024).
Xiao, L. et al. IL-9/STAT3/fatty acid oxidation-mediated lipid peroxidation contributes to Tc9 cell longevity and enhanced antitumor activity. J. Clin. Invest. 132, e153247 (2022).
Xu, C. et al. The glutathione peroxidase Gpx4 prevents lipid peroxidation and ferroptosis to sustain Treg cell activation and suppression of antitumor immunity. Cell Rep. 35, 109235 (2021).
Poznanski, S. M. et al. Metabolic flexibility determines human NK cell functional fate in the tumor microenvironment. Cell Metab. 33, 1205–1220.e5 (2021).
Cui, J. X. et al. L-kynurenine induces NK cell loss in gastric cancer microenvironment via promoting ferroptosis. J. Exp. Clin. Cancer Res. 42, 52 (2023).
Kim, R. et al. Ferroptosis of tumour neutrophils causes immune suppression in cancer. Nature 612, 338–346 (2022).
Della Volpe, L. et al. Inhibiting ferroptosis enhances ex vivo expansion of human haematopoietic stem cells. Nat. Cell Biol. 27, 2214–2224 (2025).
Zhao, J. et al. Human hematopoietic stem cell vulnerability to ferroptosis. Cell 186, 732–747.e16 (2023).
Carlson, B. A. et al. Glutathione peroxidase 4 and vitamin E cooperatively prevent hepatocellular degeneration. Redox Biol. 9, 22–31 (2016).
Ferrer, M. et al. Ketogenic diet promotes tumor ferroptosis but induces relative corticosterone deficiency that accelerates cachexia. Cell Metab. 35, 1147–1162.e7 (2023).
Shi, L. et al. The DRD2 antagonist haloperidol mediates autophagy-induced ferroptosis to increase temozolomide sensitivity by promoting endoplasmic reticulum stress in glioblastoma. Clin. Cancer Res. 29, 3172–3188 (2023).
Yue, J. et al. Discovery of the inhibitor targeting the SLC7A11/xCT axis through in silico and in vitro experiments. Int. J. Mol. Sci. 25, 8284 (2024).
Cramer, S. L. et al. Systemic depletion of L-cyst(e)ine with cyst(e)inase increases reactive oxygen species and suppresses tumor growth. Nat. Med. 23, 120–127 (2017).
Agnello, G. et al. Development of a human therapeutic L-cyst(e)Ine-degrading enzyme for the treatment of hematological malignancies. Blood 128, 1587–1587 (2016).
Guerra, A., Parhiz, H. & Rivella, S. Novel potential therapeutics to modify iron metabolism and red cell synthesis in diseases associated with defective erythropoiesis. Haematologica 108, 2582–2593 (2023).
Liu, J., Tang, D. & Kang, R. Targeting GPX4 in ferroptosis and cancer: chemical strategies and challenges. Trends Pharmacol. Sci. 45, 666–670 (2024).
Dai, Q. et al. Inhibition of FSP1: a new strategy for the treatment of tumors (Review). Oncol. Rep. 52, 105 (2024).
Liu, H. et al. Small-molecule allosteric inhibitors of GPX4. Cell Chem. Biol. 29, 1680–1693.e9 (2022).
Sun, D. et al. Simvastatin inhibits PD-L1 via ILF3 to induce ferroptosis in gastric cancer cells. Cell Death Dis. 16, 208 (2025).
Li, J. et al. Tumor-specific GPX4 degradation enhances ferroptosis-initiated antitumor immune response in mouse models of pancreatic cancer. Sci. Transl. Med. 15, eadg3049 (2023).
Wang, C. et al. Dual degradation mechanism of GPX4 degrader in induction of ferroptosis exerting anti-resistant tumor effect. Eur. J. Med. Chem. 247, 115072 (2023).
Zhu, J. et al. PROTAC degraders of FSP1 act as potent GPX4 sensitizers to induce ferroptosis for hepatoma treatment. Chin. Chem. Lett. 37, 11185 (2026).
Szwed, M., Poczta-Krawczyk, A., Bukowski, K. & Marczak, A. Nanoparticle-mediated ferroptosis for cancer therapy: mechanisms and therapeutic strategies. Nanotechnol. Sci. Appl. 18, 445–470 (2025).
Huang, A. et al. An iron-containing nanomedicine for inducing deep tumor penetration and synergistic ferroptosis in enhanced pancreatic cancer therapy. Mater. Today Bio 27, 101132 (2024).
Zhang, Y., Du, J., Cui, X., Ling, Y. & Tang, C. Development of a bispecific CDH17-GUCY2C ADC bearing the ferroptosis inducer RSL3 for the treatment of colorectal cancer. Cell Death Discov. 11, 347 (2025).
Chung, T. T., Piao, Z. & Lee, S. J. Identification of ferroptosis-related signature predicting prognosis and therapeutic responses in pancreatic cancer. Sci. Rep. 15, 75 (2025).
Wenzel, S. E. et al. PEBP1 wardens ferroptosis by enabling lipoxygenase generation of lipid death signals. Cell 171, 628–641.e6 (2017).
Minami, J. K. et al. CDKN2A deletion remodels lipid metabolism to prime glioblastoma for ferroptosis. Cancer Cell 41, 1048–1060.e9 (2023).
Zhao, N. et al. Ferronostics: measuring tumoral ferrous iron with PET to predict sensitivity to iron-targeted cancer therapies. J. Nucl. Med. 62, 949–955 (2021).
Shibata, Y., Yasui, H., Higashikawa, K. & Kuge, Y. Transferrin-based radiolabeled probe predicts the sensitivity of human renal cancer cell lines to ferroptosis inducer erastin. Biochem. Biophys. Rep. 26, 100957 (2021).
Park, S. Y. et al. Clinical evaluation of (4S)-4-(3-[18F]fluoropropyl)-L-glutamate (18F-FSPG) for PET/CT imaging in patients with newly diagnosed and recurrent prostate cancer. Clin. Cancer Res. 26, 5380–5387 (2020).
Park, S. Y. et al. Initial evaluation of (4S)-4-(3-[18F]fluoropropyl)-L-glutamate (FSPG) PET/CT imaging in patients with head and neck cancer, colorectal cancer, or non-Hodgkin lymphoma. EJNMMI Res. 10, 100 (2020).
Zeng, F. et al. Ferroptosis MRI for early detection of anticancer drug-induced acute cardiac/kidney injuries. Sci. Adv. 9, eadd8539 (2023).
Cui, S. et al. Identification of hyperoxidized PRDX3 as a ferroptosis marker reveals ferroptotic damage in chronic liver diseases. Mol. Cell 83, 3931–3939 (2023).
Xu, S. et al. FXN targeting induces cell death in ovarian cancer stem-like cells through PRDX3-mediated oxidative stress. iScience 27, 110506 (2024).
Jiang, Y. Z. et al. Integrated multiomic profiling of breast cancer in the Chinese population reveals patient stratification and therapeutic vulnerabilities. Nat. Cancer 5, 673–690 (2024).
Tang, C. et al. Immunometabolic coevolution defines unique microenvironmental niches in ccRCC. Cell Metab. 35, 1424–1440.e5 (2023).
Chen, R., Zou, J., Liu, J., Kang, R. & Tang, D. DAMPs in the immunogenicity of cell death. Mol. Cell 85, 3874–3889 (2025).
Dai, E. et al. Autophagy-dependent ferroptosis drives tumor-associated macrophage polarization via release and uptake of oncogenic KRAS protein. Autophagy 16, 2069–2083 (2020).
Xiao, Y. et al. HEBP2-governed glutamine competition between tumor and macrophages dictates immunotherapy efficacy in triple-negative breast cancer. Cell Metab. 37, 2030–2047.e7 (2025).
Li, S. et al. Neutralization of acyl coenzyme A binding protein for the experimental prevention and treatment of hepatocellular carcinoma. Cell Rep. Med. 6, 102232 (2025).
Lei, G. et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 30, 146–162 (2020).
Lang, X. et al. Radiotherapy and immunotherapy promote tumoral lipid oxidation and ferroptosis via synergistic repression of SLC7A11. Cancer Discov. 9, 1673–1685 (2019).
Jin, X. et al. High-dose ionizing radiation-induced ferroptosis leads to radiotherapy sensitization of nasopharyngeal carcinoma. Radiother. Oncol. 213, 111166 (2025).
Mao, C. et al. Unraveling ETC complex I function in ferroptosis reveals a potential ferroptosis-inducing therapeutic strategy for LKB1-deficient cancers. Mol. Cell 84, 1964–1979.e6 (2024).
Zhang, T. et al. Targeting histone deacetylase enhances the therapeutic effect of erastin-induced ferroptosis in EGFR-activating mutant lung adenocarcinoma. Transl. Lung Cancer Res. 10, 1857–1872 (2021).
Lv, D. et al. EGFR promotes ALKBH5 nuclear retention to attenuate N6-methyladenosine and protect against ferroptosis in glioblastoma. Mol. Cell 83, 4334–4351.e7 (2023).
Bian, Y. et al. Targeting ALDH1A1 to enhance the efficacy of KRAS-targeted therapy through ferroptosis. Redox Biol. 77, 103361 (2024).
Bian, Y. et al. Targeting polyamine metabolism and ferroptosis enhances the efficacy of KRAS-targeted therapy depending on KEAP1 status. Nat. Commun. 16, 9923 (2025).
Zheng, H. et al. Targeted activation of ferroptosis in colorectal cancer via LGR4 targeting overcomes acquired drug resistance. Nat. Cancer 5, 572–589 (2024).
Guo, L., Hu, C., Yao, M. & Han, G. Mechanism of sorafenib resistance associated with ferroptosis in HCC. Front. Pharmacol. 14, 1207496 (2023).
Bruedigam, C. et al. Imetelstat-mediated alterations in fatty acid metabolism to induce ferroptosis as a therapeutic strategy for acute myeloid leukemia. Nat. Cancer 5, 47–65 (2024).
Sen, U., Coleman, C. & Sen, T. Stearoyl coenzyme A desaturase-1: multitasker in cancer, metabolism, and ferroptosis. Trends Cancer 9, 480–489 (2023).
Sen, U. et al. SCD1 inhibition blocks the AKT-NRF2-SLC7A11 pathway to induce lipid metabolism remodeling and ferroptosis priming in lung adenocarcinoma. Cancer Res. 85, 2485–2503 (2025).
Bartolacci, C. et al. Targeting de novo lipogenesis and the Lands cycle induces ferroptosis in KRAS-mutant lung cancer. Nat. Commun. 13, 4327 (2022).
Chen, Y. et al. Mevalonate pathway promotes liver cancer by suppressing ferroptosis through CoQ10 production and selenocysteine-tRNA modification. J. Hepatol. 83, 1338–1352 (2025).
Guo, D., Bell, E. H., Mischel, P. & Chakravarti, A. Targeting SREBP-1-driven lipid metabolism to treat cancer. Curr. Pharm. Des. 20, 2619–2626 (2014).
Wong, T. L., Kong, Y. & Ma, S. Lipid metabolism in cancer cells: its role in hepatocellular carcinoma progression and therapeutic resistance. Hepatol. Commun. 9, e0837 (2025).
Wang, X. et al. Adipocyte-derived ferroptotic signaling mitigates obesity. Cell Metab. 37, 673–691.e7 (2025).
Dixon, S. J. et al. Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. eLife 3, e02523 (2014).
Zhu, S. et al. HSPA5 regulates ferroptotic cell death in cancer cells. Cancer Res. 77, 2064–2077 (2017).
Fang, X. et al. Ferroptosis as a target for protection against cardiomyopathy. Proc. Natl Acad. Sci. USA 116, 2672–2680 (2019).
Tadokoro, T. et al. Mitochondria-dependent ferroptosis plays a pivotal role in doxorubicin cardiotoxicity. JCI Insight 5, e132747 (2020).
Xie, X. et al. Targeting GPX4-mediated ferroptosis protection sensitizes BRCA1-deficient cancer cells to PARP inhibitors. Redox Biol. 76, 103350 (2024).
Chen, Z. et al. PRDX6 contributes to selenocysteine metabolism and ferroptosis resistance. Mol. Cell 84, 4645–4659.e9 (2024).
Ito, J. et al. PRDX6 dictates ferroptosis sensitivity by directing cellular selenium utilization. Mol. Cell 84, 4629–4644.e9 (2024).
Hu, Y. et al. Targeting PRDX6-dependent localization and function of GPX4 enhances ferroptosis-mediated tumor suppression. Mol. Cell 85, 4602–4620.e9 (2025).
Tao, Y. et al. SCRN1 confers hepatocellular carcinoma resistance to ferroptosis by stabilizing GPX4 via STK38-mediated phosphorylation. Nat. Cancer 6, 1976–1993 (2025).
Wu, K. et al. Creatine kinase B suppresses ferroptosis by phosphorylating GPX4 through a moonlighting function. Nat. Cell Biol. 25, 714–725 (2023).
Zhou, L. et al. Palmitoylation of GPX4 via the targetable ZDHHC8 determines ferroptosis sensitivity and antitumor immunity. Nat. Cancer 6, 768–785 (2025).
Fan, Y. et al. PRMT5-mediated arginine methylation stabilizes GPX4 to suppress ferroptosis in cancer. Nat. Cell Biol. 27, 641–653 (2025).
Acknowledgements
The authors thank the many pioneers in the field and colleagues whose work has shaped current understanding of ferroptosis.
Author information
Authors and Affiliations
Contributions
D.T. conceived and supervised the overall framework of the Review. All authors contributed substantially to discussions of content, writing, and review and editing of all sections of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
G.K. declares research contracts with Daiichi Sankyo, Eleor, Kaleido Biosciences, Lytix Pharma, PharmaMar, Osasuna Therapeutics, Samsara Therapeutics, Sanofi, Sutro Biopharma, Tollys and VASCage; serves on the Board of Directors of the Bristol Myers Squibb Foundation France, and on scientific advisory boards for Hevolution, Institut Servier, Longevity Vision Funds and Rejuveron Life Sciences/Centenara Labs; is a scientific co-founder of everImmune, Osasuna Therapeutics, Samsara Therapeutics and Therafast Bio; and is the inventor of patents covering therapeutic targeting of ageing, cancer, cystic fibrosis and metabolic disorders, including one, ‘Methods for weight reduction’ (US11905330B1), relevant to this work. G.K.’s brother, Romano Kroemer, was an employee of Sanofi and now consults for Boehringer-Ingelheim. G.K.’s wife, Laurence Zitvogel, has held research contracts with GSK, Incyte, Lytix Pharma, Kaleido Biosciences, Innovate Pharma, Daiichi Sankyo, PiLeJe, Merus, Transgene SA, 9M Life Sciences, Tusk Therapeutic and Roche; was on the Board of Directors of Transgene SA; is a cofounder of everImmune; and holds patents covering the treatment of cancer and the therapeutic manipulation of the microbiota. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks E. Catanzaro, X. Chen, F. Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
US NIH ClinicalTrials.gov database: https://www.clinicaltrials.gov
US NIH PubChem database: https://pubchem.ncbi.nlm.nih.gov/
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kang, R., Liu, J., Wang, J. et al. Translating ferroptosis into oncology: challenges, opportunities and future directions. Nat Rev Clin Oncol (2026). https://doi.org/10.1038/s41571-026-01128-z
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41571-026-01128-z