Abstract
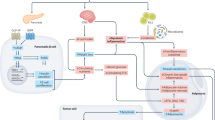
Glucagon-like peptide 1 receptor agonists (GLP1RAs) are widely used for the treatment of type 2 diabetes mellitus (T2DM) and, at higher doses, obesity. Both T2DM and obesity are associated with a higher risk of cancer, which can be reduced by intentional weight loss, whereas effects of a reduction in hyperglycaemia are uncertain. GLP1RAs might have further direct effects, either beneficial or detrimental, on the development of specific malignancies. Evidence from preclinical and clinical studies suggests heterogeneous effects of GLP1RAs on cancer risk: the incidence of hepatocellular, oesophageal, endometrial, ovarian and prostate cancers might be reduced, whereas safety concerns persist with respect to thyroid (both medullary and non-medullary) carcinomas. Conversely, initial concerns on the risk of pancreatic cancer have not been confirmed. Nonetheless, the interpretation of current data is limited by detection and prescription biases in observational studies as well as insufficient follow-up and number of events in randomized trials. In this Review, we summarize current preclinical and clinical evidence, showing that the risk–benefit profile of GLP1RAs remains favourable in individuals with T2DM and obesity, although caution is warranted in those with a low cardiometabolic risk, for whom the potential risks of cancer might outweigh any expected benefits; conversely, the potential use of GLP1RAs as adjuvant therapies for certain forms of cancer needs to be further investigated.
Key points
-
The weight loss induced by glucagon-like peptide receptor 1 agonists (GLP1RAs) should reduce the risk of some malignancies, although stimulation of GLP1 receptors might have either detrimental or beneficial direct effects on cancer risk.
-
Early experimental data raised concerns of a possible risk of medullary thyroid carcinoma, whereas data from randomized trials suggest a possible increase in risk of non-medullary thyroid carcinoma.
-
Previous concerns regarding an association between GLP1RAs and pancreatic cancer have not been confirmed by data from epidemiological studies or randomized trials.
-
Accumulating evidence supports a possible reduction in risk of hepatocellular carcinoma as well as oesophageal, endometrial, prostate and ovarian cancers in those receiving GLP1RAs.
-
Methodological limitations of the available observational studies and randomized trials preclude definitive conclusions regarding the effects of GLP1RAs on the risk of various different malignancies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Drucker, D. J. GLP-1-based therapies for diabetes, obesity and beyond. Nat. Rev. Drug Discov. 24, 631–650 (2025). A comprehensive and updated review on GLP-1 receptor agonists in a clinical perspective.
Lincoff, A. M. et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N. Engl. J. Med. 389, 2221–2232 (2023).
World Cancer Research Fund and American Institute for Cancer Research. Diet, nutrition, physical activity and cancer: a global perspective — a summary of the Third Expert Report. https://www.wcrf.org/wp-content/uploads/2021/02/Summary-of-Third-Expert-Report-2018.pdf (2018).
Mahamat-Saleh, Y. et al. Association of metabolic obesity phenotypes with risk of overall and site-specific cancers: a systematic review and meta-analysis of cohort studies. Br. J. Cancer 131, 1480–1495 (2024).
Gallagher, E. J. & LeRoith, D. Hyperinsulinaemia in cancer. Nat. Rev. Cancer 20, 629–644 (2020).
Wahner Hendrickson, A. E. et al. Expression of insulin receptor isoform A and insulin-like growth factor-1 receptor in human acute myelogenous leukemia: effect of the dual-receptor inhibitor BMS-536924 in vitro. Cancer Res. 69, 7635–7643 (2009).
Sciacca, L. et al. Insulin analogues differently activate insulin receptor isoforms and post-receptor signalling. Diabetologia 53, 1743–1753 (2010).
Szablewski, L. Insulin resistance: the increased risk of cancers. Curr. Oncol. 31, 998–1027 (2024).
Hopkins, B. D., Goncalves, M. D. & Cantley, L. C. Insulin-PI3K signalling: an evolutionarily insulated metabolic driver of cancer. Nat. Rev. Endocrinol. 16, 276–283 (2020).
Soták, M., Clark, M., Suur, B. E. & Börgeson, E. Inflammation and resolution in obesity. Nat. Rev. Endocrinol. 21, 45–61 (2025).
Zahid, H. et al. Leptin regulation of the p53-HIF1α/PKM2-aromatase axis in breast adipose stromal cells: a novel mechanism for the obesity-breast cancer link. Int. J. Obes. 42, 711–720 (2018).
Nahmias-Blank, D. et al. Excess body weight and postmenopausal breast cancer: emerging molecular mechanisms and perspectives. Semin. Cancer Biol. 96, 26–35 (2023).
Shi, X. et al. Role of body mass index and weight change in the risk of cancer: a systematic review and meta-analysis of 66 cohort studies. J. Glob. Health 29, 04067 (2024).
Teras, L. R. et al. Sustained weight loss and risk of breast cancer in women 50 years and older: a pooled analysis of prospective data. J. Natl Cancer Inst. 112, 929–937 (2020).
Moy, F. M., Greenwood, D. C. & Cade, J. E. Associations of clothing size, adiposity and weight change with risk of postmenopausal breast cancer in the UK Women’s Cohort Study (UKWCS). BMJ Open 8, e022599 (2018).
Luo, J. et al. Intentional weight loss and obesity-related cancer risk. JNCI Cancer Spectr. 3, pkz054 (2019).
Wiggins, T., Antonowicz, S. S. & Markar, S. R. Cancer risk following bariatric surgery-systematic review and meta-analysis of national population-based cohort studies. Obes. Surg. 29, 1031–1039 (2019). A meta-analysis summarizing epidemiological data on the effects of weight loss on cancer risk.
Wilson, R. B., Lathigara, D. & Kaushal, D. Systematic review and meta-analysis of the impact of bariatric surgery on future cancer risk. Int. J. Mol. Sci. 24, 6192 (2023).
Sjöholm, K. et al. Association of bariatric surgery with cancer incidence in patients with obesity and diabetes: long-term results from the Swedish Obese Subjects study. Diabetes Care 45, 444–450 (2022).
Kristensson, F. M. et al. Breast cancer risk after bariatric surgery and influence of insulin levels: a nonrandomized controlled trial. JAMA Surg. 159, 856–863 (2024).
Ma, C. et al. Effects of weight loss interventions for adults who are obese on mortality, cardiovascular disease, and cancer: systematic review and meta-analysis. BMJ 359, j4849 (2017).
Crawley, D. J. et al. Serum glucose and risk of cancer: a meta-analysis. BMC Cancer 14, 985 (2014).
Ling, S. et al. Association of type 2 diabetes with cancer: a meta-analysis with bias analysis for unmeasured confounding in 151 cohorts comprising 32 million people. Diabetes Care 43, 2313–2322 (2020).
Johnson, J. A., Bowker, S. L., Richardson, K. & Marra, C. A. Time-varying incidence of cancer after the onset of type 2 diabetes: evidence of potential detection bias. Diabetologia 54, 2263–2271 (2011).
De Bruijn, K. M. et al. Detection bias may be the main cause of increased cancer incidence among diabetics: results from the Rotterdam Study. Eur. J. Cancer 50, 2449–2455 (2014).
Harding, J. L., Shaw, J. E., Peeters, A., Cartensen, B. & Magliano, D. J. Cancer risk among people with type 1 and type 2 diabetes: disentangling true associations, detection bias, and reverse causation. Diabetes Care 38, 264–270 (2015).
Lega, I. C. & Lipscombe, L. L. Review: diabetes, obesity, and cancer-pathophysiology and clinical implications. Endocr. Rev. 41, bnz014 (2020).
Onitilo, A. A. et al. Breast cancer incidence before and after diagnosis of type 2 diabetes mellitus in women: increased risk in the prediabetes phase. Eur. J. Cancer Prev. 23, 76–83 (2014).
Pannala, R., Basu, A., Petersen, G. M. & Chari, S. T. New-onset diabetes: a potential clue to the early diagnosis of pancreatic cancer. Lancet Oncol. 10, 88–95 (2009).
Colmers, I. N. et al. Detection bias and overestimation of bladder cancer risk in type 2 diabetes: a matched cohort study. Diabetes Care 36, 3070–3075 (2013).
Ghareghomi, S. et al. Hyperglycemia-driven signaling bridges between diabetes and cancer. Biochem. Pharmacol. 229, 116450 (2024).
Weinberg Sibony, R., Segev, O., Dor, S. & Raz, I. Overview of oxidative stress and inflammation in diabetes. J. Diabetes 16, e70014 (2024).
Lee, H., Kim, M. J., Lee, I. K., Hong, C. W. & Jeon, J. H. Impact of hyperglycemia on immune cell function: a comprehensive review. Diabetol. Int. 15, 745–760 (2024).
Sona, M. F., Myung, S. K., Park, K. & Jargalsaikhan, G. Type 1 diabetes mellitus and risk of cancer: a meta-analysis of observational studies. Jpn. J. Clin. Oncol. 48, 426–433 (2018).
Zhong, W. & Mao, Y. Daily insulin dose and cancer risk among patients with type 1 diabetes. JAMA Oncol. 8, 1356–1358 (2022).
Rentsch, C. T. et al. Risk of 16 cancers across the full glycemic spectrum: a population-based cohort study using the UK Biobank. BMJ Open Diabetes Res. Care 8, e001600 (2020).
Johnson, J. A. & Bowker, S. L. Intensive glycaemic control and cancer risk in type 2 diabetes: a meta-analysis of major trials. Diabetologia 54, 25–31 (2011).
O’Connor, L. et al. Association of metformin use and cancer incidence: a systematic review and meta-analysis. J. Natl Cancer Inst. 116, 518–529 (2024).
Monami, M., Dicembrini, I. & Mannucci, E. Thiazolidinediones and cancer: results of a meta-analysis of randomized clinical trials. Acta Diabetol. 51, 91–101 (2014).
Chen, Y. et al. Diabetes medications and cancer risk associations: a systematic review and meta-analysis of evidence over the past 10 years. Sci. Rep. 13, 11844 (2023).
Jain, C., Ansarullah, Bilekova, S. & Lickert, H. Targeting pancreatic β cells for diabetes treatment. Nat. Metab. 4, 1097–1108 (2022).
Marzioni, M. et al. Glucagon-like peptide-1 and its receptor agonist exendin-4 modulate cholangiocyte adaptive response to cholestasis. Gastroenterology 133, 244–255 (2007).
Tong, G. et al. Effects of GLP-1 receptor agonists on biological behavior of colorectal cancer cells by regulating PI3k/Akt/mTOR signaling pathway. Front. Pharmacol. 13, 901559 (2022).
Ligumsky, H. et al. Glucagon-like peptide-1 analogs activate AMP kinase leading to reversal of the Warburg metabolic switch in breast cancer cells. Med. Oncol. 41, 138 (2024).
Alhajahjeh, A. et al. From diabetes to oncology: glucagon-like peptide-1 (GLP-1) receptor agonist’s dual role in prostate cancer. Cancers 16, 1538 (2024).
Nomiyama, T. et al. Exendin-4, a GLP-1 receptor agonist, attenuates prostate cancer growth. Diabetes 63, 3891–3905 (2014).
Iwaya, C. et al. Exendin-4, a glucagonlike peptide-1 receptor agonist, attenuates breast cancer growth by inhibiting NF-κB activation. Endocrinology 158, 4218–4232 (2017).
Lin, A. et al. Glucagon-like peptide 1 receptor agonists and cancer risk: advancing precision medicine through mechanistic understanding and clinical evidence. Biomark. Res. 13, 50 (2025).
US Food and Drug Administration. Highlights of prescribing information OZEMPIC. FDA.gov https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl.pdf (accessed 2 September 2025).
European Medicines Agency. Product information and details OZEMPIC. EMA https://www.ema.europa.eu/en/medicines/human/EPAR/ozempic (accessed 2 September 2025).
Bjerre, K. L. et al. Glucagonlike peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation. Endocrinology 151, 1473–1486 (2010).
Crespel, A., De Boisvilliers, F., Gros, L. & Kervran, A. Effects of glucagon and glucagon-like peptide-1-(7-36) amide on C cells from rat thyroid and medullary thyroid carcinoma CA-77 cell line. Endocrinology 137, 3674–3680 (1996).
Bjerre Knudsen, L. et al. Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation. Endocrinology 151, 1473–1486 (2010).
Hollingshead, B. D. & Radi, Z. A. Human relevance of pharmaceutical drug-induced thyroid tumors in rats, labeling implications, and carcinogenicity study requirements. J. Appl. Toxicol. 45, 1426–1439 (2025).
Boess, F. et al. Effect of GLP1R agonists taspoglutide and liraglutide on primary thyroid C-cells from rodent and man. J. Mol. Endocrinol. 50, 325–336 (2013).
Bethel, M. A. et al. Changes in serum calcitonin concentrations, incidence of medullary thyroid carcinoma, and impact of routine calcitonin concentration monitoring in the EXenatide Study of Cardiovascular Event Lowering (EXSCEL). Diabetes Care 42, 1075–1080 (2019).
Kamrul-Hasan, A. B. M. et al. Tirzepatide and cancer risk in individuals with and without diabetes: a systematic review and meta-analysis. Endocrinol. Metab. 40, 112–124 (2025).
Silverii, G. A. et al. Glucagon-like peptide-1 receptor agonists and risk of thyroid cancer: a systematic review and meta-analysis of randomized controlled trials. Diabetes Obes. Metab. 26, 891–900 (2024).
Ko, A. et al. Risk for cancer with glucagon-like peptide-1 receptor agonists and dual agonists: a systematic review and meta-analysis. Ann. Intern. Med. 179, 216–229 (2025).
He, L. et al. Effects of insulin analogs and glucagon-like peptide-1 receptor agonists on proliferation and cellular energy metabolism in papillary thyroid cancer. OncoTargets Ther. 10, 5621–5631 (2017).
Gier, B. et al. Glucagon like peptide-1 receptor expression in the human thyroid gland. J. Clin. Endocrinol. Metab. 97, 121–131 (2012).
Wang, L. et al. Semaglutide reprograms macrophages via the GLP-1R/PPARG/ACSL1 pathway to suppress papillary thyroid carcinoma growth. J. Clin. Endocrinol. Metab. 110, 2777–2789 (2025).
Sencar, M. E. et al. The effect of exenatide on thyroid-stimulating hormone and thyroid volume. Eur. Thyroid J. 8, 307–311 (2019).
Tee, S. A., Tsatlidis, V. & Razvi, S. The GLP-1 receptor agonist exenatide reduces serum TSH by its effect on body weight in people with type 2 diabetes. Clin. Endocrinol. 99, 401–408 (2023).
Hisan, U. K., Myung, S. K. & Nguyen, G. V. Associations between obesity and risk of thyroid cancer: a meta-analysis of cohort studies. Nutr. Cancer 77, 288–298 (2025).
Miller, J. R. et al. Cancer incidence, type, and survival after bariatric surgery. Surg. Obes. Relat. Dis. 20, 644–651 (2024).
Abdul-Maksoud, R. S. et al. GLP-1R polymorphism (rs1042044) and expression are associated with the risk of papillary thyroid cancer among the Egyptian population. Gene 834, 146597 (2022). A genetic study supporting a relationship between glucagon-like peptide 1 receptor stimulation and the risk for non-medullary thyroid cancer.
Mali, G., Ahuja, V. & Dubey, K. Glucagon-like peptide-1 analogues and thyroid cancer: an analysis of cases reported in the European pharmacovigilance database. J. Clin. Pharm. Ther. 46, 99–105 (2021).
Funch, D. et al. Risk of thyroid cancer associated with use of liraglutide and other antidiabetic drugs in a US commercially insured population. Diabetes Metab. Syndr. Obes. 14, 2619–2629 (2021).
Bezin, J. et al. GLP-1 receptor agonists and the risk of thyroid cancer. Diabetes Care 46, 384–390 (2023).
Baxter, S. M. et al. Glucagon-like peptide 1 receptor agonists and risk of thyroid cancer: an international multisite cohort study. Thyroid 35, 69–78 (2025).
Morales, D. R. et al. Risk of thyroid tumors with GLP-1 receptor agonists: a retrospective cohort study. Diabetes Care 48, 1386–1394 (2025).
Pasternak, B. et al. Glucagon-like peptide 1 receptor agonist use and risk of thyroid cancer: Scandinavian cohort study. BMJ 385, e078225 (2024).
Bea, S. et al. Risk of thyroid cancer associated with glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in patients with type 2 diabetes: a population-based cohort study. Diabetes Obes. Metab. 26, 108–117 (2024).
Liang, C. et al. Exenatide use and incidence of pancreatic and thyroid cancer: a retrospective cohort study. Diabetes Obes. Metab. 21, 1037–1042 (2019).
Balachandra, S. et al. Evaluating thyroid cancer risk in glucagon-like peptide-1 analog users with thyroid nodules. J. Surg. Res. 312, 104–110 (2025).
Levy, S. et al. Differential effects of GLP-1 receptor agonists on cancer risk in obesity: a nationwide analysis of 1.1 million patients. Cancers 17, 78 (2024).
Brito, J. P. et al. GLP-1RA use and thyroid cancer risk. JAMA Otolaryngol. Head Neck Surg. 151, 243–252 (2025).
McGovern, A. et al. Comparison of medication adherence and persistence in type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes. Metab. 20, 1040–1043 (2018).
Silverii, G. A. et al. GLP-1 receptor agonists and the risk for cancer: a meta-analysis of randomized controlled trials. Diabetes Obes. Metab. 27, 4454–4468 (2025). A comprehensive meta-analysis collecting all available data from randomized trials on the effects of glucagon-like peptide 1 receptor agonists on the risk of different malignancies.
Duchemin, L. et al. Risk of thyroid cancer related to glucagon-like peptide-1 receptor agonists: a systematic review with meta-analysis of harms of randomized controlled trials. Diabetes Obes. Metab. 27, 4607–4610 (2025).
Elashoff, M., Matveyenko, A. V., Gier, B., Elashoff, R. & Butler, P. C. Pancreatitis, pancreatic, and thyroid cancer with glucagon-like peptide-1-based therapies. Gastroenterology 141, 150–156 (2011). An early analysis of pharmacovigilance data suggesting the association of glucagon-like peptide 1 receptor agonist use with an increased risk of some malignancies.
Nachnani, J. S. et al. Biochemical and histological effects of exendin-4 (exenatide) on the rat pancreas. Diabetologia 53, 153–159 (2010).
Gier, B. et al. Chronic GLP-1 receptor activation by exendin-4 induces expansion of pancreatic duct glands in rats and accelerates formation of dysplastic lesions and chronic pancreatitis in the Kras(G12D) mouse model. Diabetes 61, 1250–1262 (2012).
Monami, M. et al. Safety issues with glucagon-like peptide-1 receptor agonists (pancreatitis, pancreatic cancer and cholelithiasis): data from randomized controlled trials. Diabetes Obes. Metab. 19, 1233–1241 (2017).
Jura, N., Archer, H. & Bar-Sagi, D. Chronic pancreatitis, pancreatic adenocarcinoma and the black box in-between. Cell Res. 15, 72–77 (2005).
Nyborg, N. C., Mølck, A. M., Madsen, L. W. & Knudsen, L. B. The human GLP-1 analog liraglutide and the pancreas: evidence for the absence of structural pancreatic changes in three species. Diabetes 61, 1243–1249 (2012).
Yang, Z. et al. GLP-1 receptor agonist-associated tumor adverse events: a real-world study from 2004 to 2021 based on FAERS. Front. Pharmacol. 13, 925377 (2022).
Wang, T. et al. Using real-world data to evaluate the association of incretin-based therapies with risk of acute pancreatitis: a meta-analysis of 1,324,515 patients from observational studies. Diabetes Obes. Metab. 17, 32–41 (2015).
Montvida, O., Green, J. B., Atherton, J. & Paul, S. K. Treatment with incretins does not increase the risk of pancreatic diseases compared to older anti-hyperglycaemic drugs, when added to metformin: real world evidence in people with Type 2 diabetes. Diabet. Med. 36, 491–498 (2019).
Singh, S. et al. Glucagon-like peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes mellitus: a population-based matched case-control study. JAMA Intern. Med. 173, 534–539 (2013).
US Food and Drug Administration. Drug approval package for application 215256Orig1s011 Wegovy (semaglutide). FDA.gov https://www.accessdata.fda.gov/drugsatfda_docs/nda/2024/215256Orig1s011.pdf (accessed 26 July 2025).
Abd El Aziz, M., Cahyadi, O., Meier, J. J., Schmidt, W. E. & NaucK, M. A. Incretin-based glucose-lowering medications and the risk of acute pancreatitis and malignancies: a meta-analysis based on cardiovascular outcomes trials. Diabetes Obes. Metab. 22, 699–704 (2020).
Hidayat, K. et al. A systematic review and meta-analysis of observational studies of the association between the use of incretin-based therapies and the risk of pancreatic cancer. Pharmacoepidemiol. Drug Saf. 32, 107–125 (2023).
Figlioli, G. et al. Glucagon-like peptide-1 receptor agonists and risk of gastrointestinal cancers: a systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 208, 107401 (2024).
Ayoub, M. et al. The use of glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes mellitus does not increase the risk of pancreatic cancer: a U.S.-based cohort study. Cancers 16, 1625 (2024).
Dankner, R. et al. Glucagon-like peptide-1 receptor agonists and pancreatic cancer risk in patients with type 2 diabetes. JAMA Netw. Open 7, e2350408 (2024).
Gamborg, M. et al. Long-term cancer risk in users of GLP-1 agonists in Denmark: a nationwide emulated trial. Lancet Reg. Health Eur. 55, 101346 (2025).
Wang, L., Xu, R., Kaelber, D. C. & Berger, N. A. Glucagon-like peptide 1 receptor agonists and 13 obesity-associated cancers in patients with type 2 diabetes. JAMA Netw. Open 7, e2421305 (2024). A retrospective cohort study on the effects of glucagon-like peptide 1 receptor agonist use on the risk of different obesity-associated malignancies.
Wang, L., Wang, Q., Li, L., Kaelber, D. C. & Xu, R. Glucagon-like peptide-1 receptor agonists and pancreatic cancer risk: target trial emulation using real-world data. J. Natl Cancer Inst. 117, 476–485 (2025).
Krishnan, A., Hadi, Y., Hutson, W. R., Thakkar, S. & Singh, S. Glucagon-like peptide 1-based therapies and risk of pancreatic cancer in patients with diabetes and obesity. Pancreas 51, 1398–1403 (2022).
Zhao, H. J. et al. Activation of GLP-1 receptor enhances the chemosensitivity of pancreatic cancer cells. J. Mol. Endocrinol. 64, 103–113 (2020).
Cencioni, C. et al. The GLP-1R agonist semaglutide reshapes pancreatic cancer associated fibroblasts reducing collagen proline hydroxylation and favoring T lymphocyte infiltration. J. Exp. Clin. Cancer Res. 44, 18 (2025).
Clements, O. et al. Risk factors for intrahepatic and extrahepatic cholangiocarcinoma: a systematic review and meta-analysis. J. Hepatol. 72, 95–103 (2020).
Giorda, C. B. et al. Incretin-based therapy and risk of cholangiocarcinoma: a nested case-control study in a population of subjects with type 2 diabetes. Acta Diabetol. 57, 401–408 (2020).
Krishnan, A. et al. Association between incretin-based drugs and risk of cholangiocarcinoma among patients with type 2 diabetes: a large population-based matched cohort study. J. Clin. Transl. Endocrinol. 38, 100370 (2024).
Ueda, P. et al. Use of incretin-based drugs and risk of cholangiocarcinoma: Scandinavian cohort study. Diabetologia 64, 2204–2214 (2021).
Wang, L., Berger, N. A., Kaelber, D. C. & Xu, R. Association of GLP-1 receptor agonists and hepatocellular carcinoma incidence and hepatic decompensation in patients with type 2 diabetes. Gastroenterology 167, 689–703 (2024).
Newsome, P. N. & Loomba, R. Therapeutic horizons in metabolic dysfunction-associated steatohepatitis. J. Clin. Invest. 135, e186425 (2025).
Romero-Gómez, M., Zelber-Sagi, S. & Trenell, M. Treatment of NAFLD with diet, physical activity and exercise. J. Hepatol. 67, 829–846 (2017).
Gupta, N. A. et al. Glucagon-like peptide-1 receptor is present on human hepatocytes and has a direct role in decreasing hepatic steatosis in vitro by modulating elements of the insulin signaling pathway. Hepatology 51, 1584–1592 (2010).
Targher, G., Byrne, C. D. & Tilg, H. MASLD: a systemic metabolic disorder with cardiovascular and malignant complications. Gut 73, 691–702 (2024).
Zhou, M. et al. The anti-diabetic drug exenatide, a glucagon-like peptide-1 receptor agonist, counteracts hepatocarcinogenesis through cAMP-PKA-EGFR-STAT3 axis. Oncogene 36, 4135–4149 (2017).
Yamada, N. et al. GLP-1 reduces the migration of hepatocellular carcinoma cells via suppression of the stress-activated protein kinase/c-Jun N-terminal kinase pathway. Arch. Biochem. Biophys. 703, 108851 (2021).
Chen, D., Liang, H., Huang, L., Zhou, H. & Wang, Z. Liraglutide enhances the effect of checkpoint blockade in lung and liver cancers through the inhibition of neutrophil extracellular traps. FEBS Open Bio 14, 1365–1377 (2024).
Pasta, A. et al. Effects of glucagon-like peptide-1 receptor agonists on incidence of hepatocellular carcinoma and liver decompensation in patients with diabetes: a systematic review and meta-analysis. Eur. J. Clin. Invest. 55, e70000 (2025). A meta-analysis collecting clinical data on the possible protective effect of glucagon-like peptide 1 receptor agonists with respect to hepatic adverse events, including hepatocellular carcinoma.
Passos, P. R. C. et al. Influence of glucagon-like peptide-1 receptor agonists on hepatic events in type 2 diabetes: a systematic review and meta-analysis. J. Gastroenterol. Hepatol. 40, 67–77 (2025).
Dalbeni, A. et al. Glucagon-like peptide-1 receptor agonists and hepatocellular carcinoma prevention: a meta-analysis and clinical decision framework. Cancer Med. 14, e71434 (2025).
Lim, P. W. et al. Bariatric surgery and longitudinal cancer risk: a review. JAMA Surg. 159, 331–338 (2024).
Noh, Y., Yin, H., Yu, O. H. Y., Bitton, A., & Azoulaym L. Glucagon-like peptide-1 receptor agonists and risk for gastroesophageal reflux disease in patients with type 2 diabetes: a population-based cohort study. Ann. Intern. Med. 178, 1268–1278 (2025).
Åkerström, J. H. et al. Glucagon-like peptide-1 receptor agonist treatment and risk of esophageal cancer. Am. J. Gastroenterol. https://doi.org/10.14309/ajg.0000000000003885 (2025).
Li, W., Gu, Y., Liu, S., Ruan, F. & Lv, W. GLP1R inhibits the progression of endometrial carcinoma through activation of cAMP/PKA pathway. J. Clin. Lab. Anal. 36, e24604 (2022).
Kanda, R. et al. Expression of the glucagon-like peptide-1 receptor and its role in regulating autophagy in endometrial cancer. BMC Cancer 18, 657 (2018).
Dai, H. et al. GLP-1 receptor agonists and cancer risk in adults with obesity. JAMA Oncol. 11, 1186–1193 (2025).
Rothman, S. M., Yin, H., Yu, O. H. Y., Pollak, M., Azoulay, L. Incretin-based drugs and the incidence of endometrial cancer among people with type 2 diabetes: active comparator new-user design. Drug Saf. 48, 1023–1033 (2025).
Discacciati, A., Orsini, N. & Wolk, A. Body mass index and incidence of localized and advanced prostate cancer-a dose-response meta-analysis of prospective studies. Ann. Oncol. 23, 1665–1671 (2012).
Salvio, G. et al. Effects of glucagon-like peptide 1 receptor agonists on testicular dysfunction: a systematic review and meta-analysis. Andrology 13, 2022–2034 (2025).
Li, X. N. et al. Glucagon-like peptide-1 analogues inhibit proliferation and increase apoptosis of human prostate cancer cells in vitro. Exp. Clin. Endocrinol. Diabetes 125, 91–97 (2017).
Sharma, N. et al. Effect of GLP-1 receptor agonists on prostate cancer risk reduction: a systematic review and meta-analysis. Int. Urol. Nephrol. 57, 1039–1049 (2025).
He, W. et al. Exendin-4 inhibits growth and augments apoptosis of ovarian cancer cells. Mol. Cell. Endocrinol. 436, 240–249 (2016).
Kosowska, A. et al. Exenatide modulates tumor-endothelial cell interactions in human ovarian cancer cells. Endocr. Connect. 6, 856–865 (2017).
Wang, M. et al. Association between glucagon-like peptide-1 receptor agonists and ovarian cancer survival: a population-based cohort study. Gynecol. Oncol. 199, 57–63 (2025).
Ziller, S. G. et al. Adiposity associations with risk of colorectal cancer: a systematic review. Obesity 33, 1416–1431 (2025).
Janik, M. R., Clapp, B., Sroczyński, P. & Ghanem, O. The effect of bariatric surgery on reducing the risk of colorectal cancer: a meta-analysis of 3,233,044 patients. Surg. Obes. Relat. Dis. 19, 328–334 (2023).
Stenberg, E., Ottosson, J., Cao, Y., Lagstam, I. & Näslund, E. Risk for cancer after bariatric surgery compared with the general population: a nationwide matched cohort study. Obesity 33, 1725–1733 (2025).
Koehler, J. A., Kain, T. & Drucker, D. J. Glucagon-like peptide-1 receptor activation inhibits growth and augments apoptosis in murine CT26 colon cancer cells. Endocrinology 152, 3362–3372 (2011).
Kuo, C. C. et al. Glucagon-like peptide-1 receptor agonists and gastrointestinal cancer risk in individuals with type 2 diabetes. Diabetologia 68, 1924–1936 (2025).
Bushi, G. et al. Association between GLP1 RAs use and risk of colorectal cancer: a systematic review and meta-analysis. Health Sci. Rep. 8, e70490 (2025).
Hung, C. M. et al. The different colorectal tumor risk related to GLP-1 receptor agonists and SGLT2 inhibitors use: a network meta-analysis of 68 randomized controlled trials. Int. J. Surg. 112, 443–459 (2025).
Anveden, Å et al. Long-term incidence of female-specific cancer after bariatric surgery or usual care in the Swedish Obese Subjects study. Gynecol. Oncol. 145, 224–229 (2017).
Liu, Z. Z. et al. Glucagon-like peptide-1 receptor activation by liraglutide promotes breast cancer through NOX4/ROS/VEGF pathway. Life Sci. 294, 120370 (2022).
Hicks, B. M. et al. Glucagon-like peptide-1 analogues and risk of breast cancer in women with type 2 diabetes: population based cohort study using the UK Clinical Practice Research Datalink. BMJ. 355, i5340 (2016).
Santella, C. et al. Weight-lowering effects of glucagon-like peptide-1 receptor agonists and detection of breast cancer among obese women with diabetes. Epidemiology 31, 559–566 (2020). A study that poses an important methodological issue on the possible detection bias induced by glucagon-like peptide 1 receptor agonist treatment.
Piccoli, G. F. et al. Do GLP-1 receptor agonists increase the risk of breast cancer? A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 106, 912–921 (2021).
Grandhi, N. et al. Association between glucagon-like peptide-1 receptor agonist use and progression of monoclonal gammopathy of uncertain significance to multiple myeloma among patients with diabetes. JNCI Cancer Spectr. 8, pkae095 (2024).
Ashruf, O. S. et al. Hematologic cancers among patients with type 2 diabetes prescribed GLP-1 receptor agonists. JAMA Netw. Open 8, e250802 (2025).
Tabernacki, T., Wang, L., Kaelber, D. C., Xu, R. & Berger, N. A. Non-insulin antidiabetic agents and lung cancer risk in drug-naive patients with type 2 diabetes mellitus: a nationwide retrospective cohort study. Cancers 16, 2377 (2024).
Song, J. et al. Association between GLP-1 receptor agonists and incidence of lung cancer in treatment-naïve type 2 diabetes. J. Gen. Intern. Med. 40, 973–976 (2025).
Rouette, J. et al. Incretin-based drugs and risk of lung cancer among individuals with type 2 diabetes. Diabet. Med. 37, 868–875 (2020).
Lord, S. R. & Harris, A. L. Is it still worth pursuing the repurposing of metformin as a cancer therapeutic? Br. J. Cancer 128, 958–966 (2023).
Ungvari, Z. et al. Prognostic impact of glucagon-like peptide-1 receptor (GLP1R) expression on cancer survival and its implications for GLP-1R agonist therapy: an integrative analysis across multiple tumor types. GeroScience 47, 4413–4427 (2025).
Radwan, R. M. et al. GLP-1 RA use and survival among older adults with cancer and type 2 diabetes. JAMA Netw. Open 8, e2521887 (2025).
Petrelli, F. et al. Association of obesity with survival outcomes in patients with cancer: a systematic review and meta-analysis. JAMA Netw. Open 4, e213520 (2021).
Chen, L. et al. Impact of body mass index in therapeutic response for HER2 positive breast cancer treated with neoadjuvant targeted therapy: a multi-center study and meta-analysis. npg Breast Cancer 9, 46 (2023).
Adams, T. D. et al. Long-term cancer outcomes after bariatric surgery. Obesity 31, 2386–2397 (2023).
Evans, S. J., Waller, P. C. & Davis, S. Use of proportional reporting ratios (PRRs) for signal generation from spontaneous adverse drug reaction reports. Pharmacoepidemiol. Drug Saf. 10, 483–486 (2001).
Klungel, O. H. et al. Methods to assess intended effects of drug treatment in observational studies are reviewed. J. Clin. Epidemiol. 57, 1223–1231 (2004).
Sharma, A. M. et al. Determinants of adherence to obesity medication: a narrative review. Obes. Rev. 26, e13885 (2025).
West, S. et al. Weight regain after cessation of medication for weight management: systematic review and meta-analysis. BMJ 392, e085304 (2026).
Heckman-Stoddard, B. M. et al. Randomized study of metformin and intensive lifestyle intervention on cancer incidence over 21 years of follow-up in the diabetes prevention program. Cancer Prev. Res. 18, 401–411 (2025).
Shiwani, H., Clement, N. S., Daniels, J. P. & Atiomo, W. Metformin for endometrial hyperplasia. Cochrane Database Syst. Rev. 5, CD012214 (2024).
Liu, L., Cui, J., Neidecker, M. V. & Nahata, M. C. Tirzepatide vs semaglutide and liraglutide for weight loss in patients with overweight or obesity without diabetes: a short-term cost-effectiveness analysis in the United States. J. Manag. Care Spec. Pharm. 31, 441–450 (2025).
Lazzini, G. et al. Raman spectroscopy based diagnosis of pancreatic ductal adenocarcinoma. Sci. Rep. 15, 13240 (2025).
Huang, D. Q. et al. Metabolic dysfunction-associated steatotic liver disease in adults. Nat. Rev. Dis. Primers 11, 14 (2025).
Author information
Authors and Affiliations
Contributions
Both authors made a substantial contribution to all aspects of the preparation of this manuscript.
Corresponding author
Ethics declarations
Competing interests
E.M. has acted as a consultant of Boehringer Ingelheim, Dexcom, Eli Lilly, Novo Nordisk and Sanofi and received speaking fees from Eli Lilly, Novo Nordisk and Sanofi. I.D. has acted as a consultant of Medtronic, Novo Nordisk and Sanofi and received speaking fees from Abbott, Eli Lilly, Medtronic, MOVI, Novo Nordisk and Sanofi.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks M. Gunter, P.T. Tseng, J. Yarmolinsky and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mannucci, E., Dicembrini, I. Glucagon-like peptide 1 receptor agonists and cancer risk: the good, the bad and the unknown. Nat Rev Clin Oncol (2026). https://doi.org/10.1038/s41571-026-01135-0
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41571-026-01135-0