Abstract
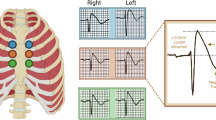
Brugada syndrome (BrS) is a cardiac channelopathy associated with an elevated risk of arrhythmias and sudden cardiac death compared with the general population. Since its initial description in 1992 by Pedro and Josep Brugada, there has been tremendous progress in our understanding and management of BrS. The condition is characterized by ‘coved’ ST segment elevations in the anterior precordial electrocardiogram leads, which occasionally requires additional pharmacological provocation for diagnosis. Substantial geographical variation in the prevalence, genetic characteristics and clinical behaviour of BrS exists. Improvements in the understanding of the genetic and molecular mechanisms of the condition have been made over the past 30 years, opening avenues for the discovery of diagnostic and management opportunities. In this Primer, we discuss the evolving epidemiology of BrS, the emerging genetic understanding of the condition, as well as its diagnosis and management. We summarize the major societal guideline recommendations pertaining to BrS and highlight the potential for technological advancements, such as digital health and machine learning, to improve patient care.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$119.00 per year
only $119.00 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Brugada, P. & Brugada, J. Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report. J. Am. Coll. Cardiol. 20, 1391–1396 (1992). The original and earliest description of BrS.
Kapplinger, J. D. et al. An international compendium of mutations in the SCN5A-encoded cardiac sodium channel in patients referred for Brugada syndrome genetic testing. Heart Rhythm 7, 33–46 (2010).
Brugada, J., Campuzano, O., Arbelo, E., Sarquella-Brugada, G. & Brugada, R. Present status of Brugada syndrome. J. Am. Coll. Cardiol. 72, 1046–1059 (2018).
Priori, S. G. et al. Executive summary: HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes. EP Eur. 15, 1389–1406 (2013).
Vutthikraivit, W. et al. Worldwide prevalence of Brugada syndrome: a systematic review and meta-analysis. Acta Cardiol. Sin. 34, 267–277 (2018). A meta-analysis outlining the geographical variations in the prevalence of BrS.
Quan, X. Q. et al. A meta-analytic review of prevalence for Brugada ECG patterns and the risk for death. Medicine 95, e5643 (2016).
Nademanee, K. Sudden unexplained death syndrome in Southeast Asia. Am. J. Cardiol. 79, 10–11 (1997).
Milman, A. et al. Profile of patients with Brugada syndrome presenting with their first documented arrhythmic event: data from the Survey on Arrhythmic Events in BRUgada Syndrome (SABRUS). Heart Rhythm 15, 716–724 (2018). A large multicentre international registry profiling clinical events in patients with BrS.
Probst, V. et al. Clinical aspects and prognosis of Brugada syndrome in children. Circulation 115, 2042–2048 (2007).
Matsuo, K., Akahoshi, M., Seto, S. & Yano, K. Disappearance of the Brugada-type electrocardiogram after surgical castration: a role for testosterone and an explanation for the male preponderance? Pacing Clin. Electrophysiol. 26, 1551–1553 (2003).
Yang, G. et al. Lack of influence of sex hormones on Brugada syndrome-associated mutant NaV1.5 sodium channel. J. Electrocardiol. 52, 82–87 (2019).
Milman, A. et al. Age of first arrhythmic event in Brugada syndrome: data from the SABRUS (Survey on Arrhythmic Events in Brugada Syndrome) in 678 patients. Circ. Arrhythm. Electrophysiol. 10, e005222 (2017).
Li, H. et al. Refined geographic distribution of the oriental ALDH2*504Lys (nee 487Lys) variant. Ann. Hum. Genet. 73, 335–345 (2009).
Zhao, D. et al. Long-term follow-up in patients with Brugada syndrome in South China. Ann. Noninvasive Electrocardiol. 26, e12823 (2021).
Crotti, L. et al. Spectrum and prevalence of mutations involving BrS1- through BrS12-susceptibility genes in a cohort of unrelated patients referred for Brugada syndrome genetic testing: implications for genetic testing. J. Am. Coll. Cardiol. 60, 1410–1418 (2012). This paper stratified patients by ethnicity to assess the prevalence and clinical implications of different BrS-related mutations.
Huang, M. H. & Marcus, F. I. Idiopathic Brugada-type electrocardiographic pattern in an octogenarian. J. Electrocardiol. 37, 109–111 (2004).
Milman, A. et al. Ethnic differences in patients with Brugada syndrome and arrhythmic events: new insights from survey on arrhythmic events in Brugada syndrome. Heart Rhythm 16, 1468–1474 (2019).
Huang, P. S. et al. Prevalence of sudden arrhythmic death syndrome-related genetic mutations in an asian cohort of whole genome sequence. EP Eur. 22, 1287–1297 (2020).
Bezzina, C. R. et al. Common sodium channel promoter haplotype in asian subjects underlies variability in cardiac conduction. Circulation 113, 338–344 (2006).
Li, H. et al. Geographically separate increases in the frequency of the derived ADH1B*47His allele in Eastern and Western Asia. Am. J. Hum. Genet. 81, 842–846 (2007).
Wu, Q. et al. Genetic variants of alcohol‐metabolizing enzymes in Brugada syndrome: insights into syncope after drinking alcohol. J. Arrhythm. 35, 547–554 (2019).
Suzuki, Y. et al. Alcohol dehydrogenase 2 variant is associated with cerebral infarction and lacunae. Neurology 63, 1711–1713 (2004).
El-Mas, M. M. & Abdel-Rahman, A. A. Role of alcohol oxidative metabolism in its cardiovascular and autonomic effects. Adv. Exp. Med. Biol. 1193, 1–33 (2019).
Michowitz, Y. et al. Fever-related arrhythmic events in the multicenter survey on arrhythmic events in Brugada syndrome. Heart Rhythm 15, 1394–1401 (2018).
Wilde, A. A. M. et al. The pathophysiological mechanism underlying Brugada syndrome: depolarization versus repolarization. J. Mol. Cell Cardiol. 49, 543–553 (2010).
Yan, G. X. & Antzelevitch, C. Cellular basis for the Brugada syndrome and other mechanisms of arrhythmogenesis associated with ST-segment elevation. Circulation 100, 1660–1666 (1999).
Coronel, R. et al. Right ventricular fibrosis and conduction delay in a patient with clinical signs of Brugada syndrome: a combined electrophysiological, genetic, histopathologic, and computational study. Circulation 112, 2769–2777 (2005).
Miles, C. et al. Subepicardial cardiomyopathy: a disease underlying J-wave syndromes and idiopathic ventricular fibrillation. Circulation 147, 1622–1633 (2023).
Behr, E. R., Ben-Haim, Y., Ackerman, M. J., Krahn, A. D. & Wilde, A. A. M. Brugada syndrome and reduced right ventricular outflow tract conduction reserve: a final common pathway? Eur. Heart J. 42, 1073–1081 (2021).
Hosseini, S. M. et al. Reappraisal of reported genes for sudden arrhythmic death: evidence-based evaluation of gene validity for Brugada syndrome. Circulation 138, 1195–1205 (2018). A study re-evaluating the validity of many previously reported genes.
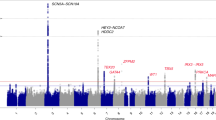
Barc, J. et al. Genome-wide association analyses identify new Brugada syndrome risk loci and highlight a new mechanism of sodium channel regulation in disease susceptibility. Nat. Genet. 54, 232–239 (2022).
Makarawate, P. et al. Common and rare susceptibility genetic variants predisposing to Brugada syndrome in Thailand. Heart Rhythm 17, 2145–2153 (2020).
Tarantino, A. et al. NaV1.5 autoantibodies in Brugada syndrome: pathogenetic implications. Eur. Heart J. 45, 4336–4348 (2024).
Jiang, D. et al. Structure of the cardiac sodium channel. Cell 134, 122–134.e10 (2020).
Catterall, W. A. From ionic currents to molecular mechanisms: the structure and function of voltage-gated sodium channels. Neuron 26, 13–25 (2000).
Yu, F. H. & Catterall, W. A. Overview of the voltage-gated sodium channel family. Genome Biol. 4, 207 (2003).
Fozzard, H. A. & Hanck, D. A. Structure and function of voltage-dependent sodium channels: comparison of brain II and cardiac isoforms. Physiol. Rev. 76, 887–926 (1996).
Monasky, M. M., Micaglio, E., Locati, E. T. & Pappone, C. Evaluating the use of genetics in Brugada syndrome risk stratification. Front. Cardiovasc. Med. 8, 652027 (2021).
Clancy, C. E. & Rudy, Y. Na+ channel mutation that causes both Brugada and long-QT syndrome phenotypes: a simulation study of mechanism. Circulation 105, 1208–1213 (2002).
Kapa, S. et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation 120, 1752–1760 (2009).
Wilde, A. A. M. & Behr, E. R. Genetic testing for inherited cardiac disease. Nat. Rev. Cardiol. 10, 571–583 (2013).
Wilde, A. A. M. & Amin, A. S. Clinical spectrum of SCN5A mutations. JACC Clin. Electrophysiol. 4, 569–579 (2018). A detailed overview of SCN5A mutations and their clinical implications.
Antzelevitch, C. et al. Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation 115, 442–449 (2007).
Monasky, M. M. et al. Calcium in Brugada syndrome: questions for future research. Front. Physiol. 9, 1088 (2018).
Valderrábano, M., Chen, P. S. & Lin, S. F. Spatial distribution of phase singularities in ventricular fibrillation. Circulation 108, 354–359 (2003).
Poelzing, S. et al. SCN5A polymorphism restores trafficking of a Brugada syndrome mutation on a separate gene. Circulation 114, 368–376 (2006).
Bezzina, C. R. et al. Common variants at SCN5A-SCN10A and HEY2 are associated with Brugada syndrome, a rare disease with high risk of sudden cardiac death. Nat. Genet. 45, 1044–1049 (2013).
Hu, D. et al. Mutations in SCN10A are responsible for a large fraction of cases of Brugada syndrome. J. Am. Coll. Cardiol. 64, 66–79 (2014).
Watanabe, H. et al. Sodium channel β1 subunit mutations associated with Brugada syndrome and cardiac conduction disease in humans. J. Clin. Invest. 118, 2260–2268 (2008).
J, D. M. et al. Characterization of sodium channel alpha- and beta-subunits in rat and mouse cardiac myocytes. Circulation 103, 1303–1310 (2001).
Riuró, H. et al. A missense mutation in the sodium channel β2 subunit reveals SCN2B as a new candidate gene for Brugada syndrome. Hum. Mutat. 34, 961–966 (2013).
Hu, D. et al. A mutation in the beta 3 subunit of the cardiac sodium channel associated with Brugada ECG phenotype. Circ. Cardiovasc. Genet. 2, 270–278 (2009).
Valdivia, C. R., Ueda, K., Ackerman, M. J. & Makielski, J. C. GPD1L links redox state to cardiac excitability by PKC-dependent phosphorylation of the sodium channel SCN5A. Am. J. Physiol. Heart Circ. Physiol. 297, H1446–H1452 (2009).
London, B. et al. Mutation in glycerol-3-phosphate dehydrogenase 1 like gene (GPD1-L) decreases cardiac Na+ current and causes inherited arrhythmias. Circulation 116, 2260–2268 (2007).
Cerrone, M. et al. Missense mutations in plakophilin-2 cause sodium current deficit and associate with a Brugada syndrome phenotype. Circulation 129, 1092–1103 (2014).
Giudicessi, J. R. et al. Transient outward current (Ito) gain-of-function mutations in the KCND3-encoded Kv4.3 potassium channel and Brugada syndrome. Heart Rhythm 8, 1024–1032 (2011).
Boczek, N. J. et al. Characterization of SEMA3A-encoded semaphorin as a naturally occurring Kv4.3 protein inhibitor and its contribution to Brugada syndrome. Circ. Res. 115, 460–469 (2014).
Nakajima, T. et al. KCNE3 T4A as the genetic basis of Brugada-pattern electrocardiogram. Circ. J. 76, 2763–2772 (2012).
Portero, V. et al. Dysfunction of the voltage‐gated K+ channel β2 subunit in a familial case of Brugada syndrome. J. Am. Heart Assoc. 5, e003122 (2016).
Medeiros-Domingo, A. et al. Gain-of-function mutation S422L in the KCNJ8-encoded cardiac KATP channel Kir6.1 as a pathogenic substrate for J-wave syndromes. Heart Rhythm 7, 1466–1471 (2010).
Hu, D. et al. ABCC9 is a novel Brugada and early repolarization syndrome susceptibility gene. Int. J. Cardiol. 171, 431–442 (2014).
Wang, Q. et al. Gain-of-function KCNH2 mutations in patients with Brugada syndrome. J. Cardiovasc. Electrophysiol. 25, 522–530 (2014).
Li, K. H. C. et al. Brugada syndrome: a comprehensive review of pathophysiological mechanisms and risk stratification strategies. IJC Heart Vasc. 26, 100468 (2020).
Burashnikov, E. et al. Mutations in the cardiac L-type calcium channel associated with inherited J-wave syndromes and sudden cardiac death. Heart Rhythm 7, 1872–1882 (2010).
Cordeiro, J. M. et al. Accelerated inactivation of the L-type calcium current due to a mutation in CACNB2b underlies Brugada syndrome. J. Mol. Cell Cardiol. 46, 695–703 (2009).
Catterall, W. A., Perez-Reyes, E., Snutch, T. P. & Striessnig, J. International Union of Pharmacology. XLVIII. Nomenclature and structure–function relationships of voltage-gated calcium channels. Pharmacol. Rev. 57, 411–425 (2005).
Abernethy, D. R. & Soldatov, N. M. Structure-functional diversity of human L-type Ca2+ channel: perspectives for new pharmacological targets. J. Pharmacol. Exp. Ther. 300, 724–728 (2002).
Fukuyama, M. et al. L-type calcium channel mutations in Japanese patients with inherited arrhythmias. Circ. J. 77, 1799–1806 (2013).
Baruscotti, M. et al. Deep bradycardia and heart block caused by inducible cardiac-specific knockout of the pacemaker channel gene HCN4. Proc. Natl Acad. Sci. USA 108, 1705–1710 (2011).
Ueda, K. et al. Role of HCN4 channel in preventing ventricular arrhythmia. J. Hum. Genet. 54, 115–121 (2009).
Liu, H. et al. Molecular genetics and functional anomalies in a series of 248 Brugada cases with 11 mutations in the TRPM4 channel. PLoS ONE 8, e54131 (2013).
Juang, J. M. J. et al. GSTM3 variant is a novel genetic modifier in Brugada syndrome, a disease with a risk of sudden cardiac death. eBioMedicine 57, 102843 (2020).
Postema, P. G. et al. Drugs and Brugada syndrome patients: review of the literature, recommendations, and an up-to-date website (www.brugadadrugs.org). Heart Rhythm 6, 1335–1341 (2009).
Macedo, P. G. et al. Sleep-disordered breathing in patients with the Brugada syndrome. Am. J. Cardiol. 107, 709–713 (2011).
Bigi, M. A. B., Aslani, A. & Aslani, A. Significance of cardiac autonomic neuropathy in risk stratification of Brugada syndrome. Europace 10, 821–824 (2008).
Adler, A. et al. Fever-induced Brugada pattern: how common is it and what does it mean? Heart Rhythm 10, 1375–1382 (2013).
Kamakura, S. et al. Long-term prognosis of probands with Brugada-pattern ST-elevation in leads V1–V 3. Circ. Arrhythm. Electrophysiol. 2, 495–503 (2009).
Takigawa, M. et al. Seasonal and circadian distributions of ventricular fibrillation in patients with Brugada syndrome. Heart Rhythm 5, 1523–1527 (2008).
Postema, P. G. & Wilde, A. A. M. Arrhythmias in Brugada syndrome: changing throughout day and season? Heart Rhythm 5, 1528–1529 (2008).
Luna, A. B. et al. Current electrocardiographic criteria for diagnosis of Brugada pattern: a consensus report. J. Electrocardiol. 45, 433–442 (2012).
Zeppenfeld, K. et al. 2022 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 43, 3997–4126 (2022).
Antzelevitch, C. et al. J-wave syndromes expert consensus conference report: emerging concepts and gaps in knowledge. Europace 19, 665–694 (2017).
Gaita, F. et al. Asymptomatic patients with Brugada ECG pattern: long-term prognosis from a large prospective study. Circulation 148, 1543–1555 (2023).
Veltmann, C. et al. A prospective study on spontaneous fluctuations between diagnostic and non-diagnostic ECGs in Brugada syndrome: implications for correct phenotyping and risk stratification. Eur. Heart J. 27, 2544–2552 (2006).
Antzelevitch, C. et al. Brugada syndrome: report of the second consensus conference: endorsed by the heart rhythm society and the European heart rhythm association. Circulation 111, 659–670 (2005).
Cerrato, N. et al. Prevalence of type 1 Brugada electrocardiographic pattern evaluated by twelve-lead twenty-four-hour holter monitoring. Am. J. Cardiol. 115, 52–56 (2015).
Wilde, A. A. M., Amin, A. S., Morita, H. & Tadros, R. Use, misuse, and pitfalls of the drug challenge test in the diagnosis of the Brugada syndrome. Eur. Heart J. 44, 2427–2439 (2023).
Anselm, D. D. Brugada phenocopy: a new electrocardiogram phenomenon. World J. Cardiol. 6, 81–6 (2014).
Chevallier, S. et al. New electrocardiographic criteria for discriminating between Brugada types 2 and 3 patterns and incomplete right bundle branch block. J. Am. Coll. Cardiol. 58, 2290–2298 (2011).
Serra, G. et al. New electrocardiographic criteria to differentiate the type-2 Brugada pattern from electrocardiogram of healthy athletes with r′-wave in leads V1/V2. Europace 16, 1639–1645 (2014).
Antzelevitch, C. et al. J-wave syndromes expert consensus conference report: emerging concepts and gaps in knowledge. J. Arrhythm. 32, 315–339 (2016).
Kawada, S. et al. Shanghai score system for diagnosis of Brugada syndrome: validation of the score system and system and reclassification of the patients. JACC Clin. Electrophysiol. 4, 724–730 (2018).
Brugada, R. et al. Sodium channel blockers identify risk for sudden death in patients with ST-segment elevation and right bundle branch block but structurally normal hearts. Circulation 101, 510–515 (2000). Original descriptions of pharmacological provocative testing.
Priori, S. G. et al. Clinical and genetic heterogeneity of right bundle branch block and ST-segment elevation syndrome: a prospective evaluation of 52 families. Circulation 102, 2509–2515 (2000).
Calvo, D. et al. Time-dependent responses to provocative testing with flecainide in the diagnosis of Brugada syndrome. Heart Rhythm 12, 350–357 (2015).
Conte, G. et al. Life-threatening ventricular arrhythmias during ajmaline challenge in patients with Brugada syndrome: incidence, clinical features, and prognosis. Heart Rhythm 10, 1869–1874 (2013).
Dobbels, B., Cleen, D. & Ector, J. Ventricular arrhythmia during ajmaline challenge for the Brugada syndrome. Europace 18, 1501–1506 (2016).
Monasky, M. M. et al. Comparable clinical characteristics in Brugada syndrome patients harboring SCN5A or novel SCN10A variants. Europace 21, 1550–1558 (2019).
Kinoshita, K. et al. SCN5A(K817E), a novel Brugada syndrome–associated mutation that alters the activation gating of NaV1.5 channel. Heart Rhythm 13, 1113–1120 (2016).
Papavassiliu, T. et al. Magnetic resonance imaging findings in patients with Brugada syndrome. J. Cardiovasc. Electrophysiol. 15, 1133–1138 (2004).
Takagi, M. Localized right ventricular morphological abnormalities detected by electron-beam computed tomography represent arrhythmogenic substrates in patients with the Brugada syndrome. Eur. Heart J. 22, 1032–1041 (2001).
Ohkubo, K. et al. Right ventricular histological substrate and conduction delay in patients with Brugada syndrome. Int. Heart J. 51, 17–23 (2010).
Papavassiliu, T. et al. Spontaneous type 1 electrocardiographic pattern is associated with cardiovascular magnetic resonance imaging changes in Brugada syndrome. Heart Rhythm 7, 1790–1796 (2010).
Ciconte, G. et al. Brugada syndrome genetics is associated with phenotype severity. Eur. Heart J. 42, 1082–1090 (2021).
Priori, S. G. et al. Natural history of Brugada syndrome: insights for risk stratification and management. Circulation 105, 1342–1347 (2002).
MA, B. B., A, A. & S, S. aVR sign as a risk factor for life-threatening arrhythmic events in patients with Brugada syndrome. Heart Rhythm 4, 1009–1012 (2007).
Morita, H. et al. Fragmented QRS as a marker of conduction abnormality and a predictor of prognosis of Brugada syndrome. Circulation 118, 1697–1704 (2008).
Maury, P. et al. Increased Tpeak–Tend interval is highly and independently related to arrhythmic events in Brugada syndrome. Heart Rhythm 12, 2469–2476 (2015).
J, C. H. et al. Tpeak–Tend and Tpeak–Tend dispersion as risk factors for ventricular tachycardia/ventricular fibrillation in patients with the Brugada syndrome. J. Am. Coll. Cardiol. 47, 1828–1834 (2006).
Adler, A. et al. Risk stratification in Brugada syndrome: clinical characteristics, electrocardiographic parameters, and auxiliary testing. Heart Rhythm 13, 299–310 (2016).
Huang, Z. et al. Role of signal-averaged electrocardiograms in arrhythmic risk stratification of patients with Brugada syndrome: a prospective study. Heart Rhythm 6, 1156–1162 (2009).
Smits, J. P. P. et al. Genotype–phenotype relationship in Brugada syndrome: electrocardiographic features differentiate SCN5A-related patients from non-SCN5A-related patients. J. Am. Coll. Cardiol. 40, 350–356 (2002).
Rattanawong, P. et al. SCN5A mutation status increases the risk of major arrhythmic events in Asian populations with Brugada syndrome: systematic review and meta-analysis. Ann. Noninvasive Electrocardiol. 24, e12589 (2019).
Maury, P. et al. Prevalence and prognostic role of various conduction disturbances in patients with the Brugada syndrome. Am. J. Cardiol. 112, 1384–1389 (2013).
Calò, L. et al. A new electrocardiographic marker of sudden death in Brugada syndrome. J. Am. Coll. Cardiol. 67, 1427–1440 (2016).
Sieira, J. et al. A score model to predict risk of events in patients with Brugada syndrome. Eur. Heart J. 38, 1756–1763 (2017).
Probst, V. et al. Long-term prognosis of patients diagnosed with Brugada syndrome: results from the FINGER Brugada syndrome registry. Circulation 121, 635–643 (2010).
Sroubek, J., Probst, V. & Mazzanti, A. Programmed ventricular stimulation for risk stratification in the Brugada syndrome: a pooled analysis. Circulation 133, 622–630 (2016).
Makimoto, H. et al. Clinical impact of the number of extrastimuli in programmed electrical stimulation in patients with Brugada type 1 electrocardiogram. Heart Rhythm 9, 242–248 (2012).
Pappone, C. et al. Assessing the malignant ventricular arrhythmic substrate in patients with Brugada syndrome. J. Am. Coll. Cardiol. 71, 1631–1646 (2018).
Gray, B. et al. Twelve-lead ambulatory electrocardiographic monitoring in Brugada syndrome: potential diagnostic and prognostic implications. Heart Rhythm 14, 866–874 (2017).
Liao, S. et al. Use of wearable technology and deep learning to improve the diagnosis of Brugada syndrome. JACC Clin. Electrophysiol. 8, 1010–1020 (2022).
Al-Khatib, S. M., Stevenson, W. G. & Ackerman, M. J. AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary: a report of the American college of Cardiology/American Heart Association task force on clinical practice guidelines and the heart rhythm society (published correction appears in Circulation). Circulation 138, e415–e418 (2017).
D’Imperio, S., Monasky, M. M., Micaglio, E., Negro, G. & Pappone, C. Impact of dietary factors on Brugada syndrome and long QT syndrome. Nutrients 13, 2482 (2021).
Sandhu, R. K. et al. Smoking, smoking cessation, and risk of sudden cardiac death in women. Circ. Arrhythm. Electrophysiol. 5, 1091–1097 (2012).
Mazzanti, A. et al. Efficacy and limitations of quinidine in patients with Brugada syndrome. Circ. Arrhythm. Electrophysiol. 12, e007143 (2019).
Shenthar, J., Chakali, S. S., Acharya, D., Parvez, J. & Banavalikar, B. Oral quinine sulfate for the treatment of electrical storm and prevention of recurrent shocks in Brugada syndrome after failed cilostazol therapy. HeartRhythm Case Rep. 3, 470–474 (2017).
Ohgo, T. et al. Acute and chronic management in patients with Brugada syndrome associated with electrical storm of ventricular fibrillation. Heart Rhythm 4, 695–700 (2007).
Maury, P., Hocini, M. & Haïssaguerre, M. Electrical storms in Brugada syndrome: review of pharmacologic and ablative therapeutic options. Indian Pacing Electrophysiol. J. 5, 25–34 (2005).
Brodie, O. T., Michowitz, Y. & Belhassen, B. Pharmacological therapy in Brugada syndrome. Arrhythm. Electrophysiol. Rev. 7, 135–142 (2018).
Schweizer, P. A., Becker, R., Katus, H. A. & Thomas, D. Successful acute and long-term management of electrical storm in Brugada syndrome using orciprenaline and quinine/quinidine. Clin. Res. Cardiol. 99, 467–470 (2010).
García-Izquierdo, E. et al. Arrhythmia detection using an implantable loop recorder after a negative electrophysiology study in Brugada syndrome: observations from a multicenter international registry. Heart Rhythm 21, 1317–1324 (2024).
Bergonti, M. et al. Implantable loop recorders in patients with Brugada syndrome: the BruLoop study. Eur. Heart J. 45, 1255–1265 (2024).
Pappone, C. et al. Electrical substrate elimination in 135 consecutive patients with Brugada syndrome. Circ. Arrhythm. Electrophysiol. 10, e005053 (2017). The largest study evaluating the effectiveness of catheter ablation in BrS.
Kotake, Y., Barua, S. & Kazi, S. Efficacy and safety of catheter ablation for Brugada syndrome: an updated systematic review (published correction appears in Clin. Res. Cardiol.). Clin. Res. Cardiol. 112, 853 (2023).
Nademanee, K. et al. Long-term outcomes of Brugada substrate ablation: a report from BRAVO (Brugada ablation of VF substrate ongoing multicenter registry). Circulation 147, 1568–1578 (2023).
Santinelli, V. et al. High-risk Brugada syndrome: factors associated with arrhythmia recurrence and benefits of epicardial ablation in addition to implantable cardioverter defibrillator implantation. Europace 26, euae019 (2023).
Pappone, C. & Santinelli, V. Brugada syndrome: progress in diagnosis and management. Arrhythm. Electrophysiol. Rev. 8, 13–18 (2019). A review article that provides an update on the diagnosis and management of BrS.
Fernandes, G. C. et al. Ablation strategies for the management of symptomatic Brugada syndrome: a systematic review. Heart Rhythm 15, 1140–1147 (2018).
Li, L. et al. Outcomes of catheter ablation in high-risk patients with Brugada syndrome refusing an implantable cardioverter defibrillator implantation. Europace 26, euad318 (2023).
Makhnoon, S., Shirts, B. H. & Bowen, D. J. Patients’ perspectives of variants of uncertain significance and strategies for uncertainty management. J. Genet. Couns. 28, 313–325 (2019).
Six, S. et al. Patient-reported outcome measures on mental health and psychosocial factors in patients with Brugada syndrome. Europace 25, euad205 (2023).
Passman, R. et al. Implantable cardioverter defibrillators and quality of life: results from the defibrillators in nonischemic cardiomyopathy treatment evaluation study. Arch. Intern. Med. 167, 2226–2232 (2007).
Chatterjee, D. et al. An autoantibody profile detects Brugada syndrome and identifies abnormally expressed myocardial proteins. Eur. Heart J. 41, 2878–2890 (2020).
Wilde, A. A. M. & Lodder, E. M. A highly specific biomarker for Brugada syndrome. Also too good to be true? Eur. Heart J. 41, 2891–2893 (2020).
Melo, L. et al. Deep learning unmasks the ECG signature of Brugada syndrome. Proc. Natl Acad. Sci. USA Nexus 2, pgad327 (2023).
Zanchi, B. et al. Identification of Brugada syndrome based on P-wave features: an artificial intelligence-based approach. Europace 25, euad334 (2023).
Tse, G., Lee, S. & Liu, T. Prediction of the presence of ventricular fibrillation from a Brugada electrocardiogram using artificial intelligence. Circ. J. 87, 1015 (2023).
Acknowledgements
The authors thank S. J. Asirvatham for his insightful suggestions and comments on this article.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
C.K. is the founder of HumanX, a company that focuses on artificial intelligence in precision medicine and innovation in health care; he declares that his affiliation with HumanX is not a competing interest. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks M. Hocini, who co-reviewed with J. Duchateau; H. Morita; K. Nademanee; K. Nakamura; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Narasimhan, B., Na, J., Monasky, M.M. et al. Brugada syndrome. Nat Rev Dis Primers 11, 38 (2025). https://doi.org/10.1038/s41572-025-00622-5
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41572-025-00622-5