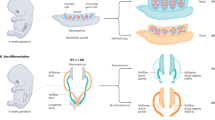
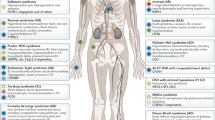
Abstract
Differences of sex development (DSD) represent a group of congenital conditions that affect human sex development and maturation owing to discrepancies of chromosomal, gonadal and phenotypic sex. The Chicago consensus classifies DSD as sex chromosome DSD, 46,XY DSD and 46,XX DSD, with subclassifications according to gonadal determination into testes and ovaries and hormone-dependent differentiation of Müllerian and Wolffian embryonic structures into female-typical or male-typical internal and external sex organs. DSD may occur as an isolated condition or as part of a complex syndrome. Diagnosis is based on clinical characteristics, imaging studies, hormonal measurements and genetic investigations. Management includes lifelong psychosocial support, hormonal treatments and surgical interventions that require personalization for each case as DSD encompasses a wide variety of aetiologies and presentations. This personalization must also consider individual values and preferences to ensure that clinical care is tailored to meet the unique needs and circumstances of each person, ideally provided by a care team with diverse specialities. This care involves psycho-educational counselling on the condition and its consequences, considering family and cultural norms. Additional efforts are needed to bridge gaps in knowledge related to diagnosis, management and long-term outcomes. Enhancing our understanding of the distinctions between sex and gender in societies is essential as greater awareness will inform and enrich public debates.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$119.00 per year
only $119.00 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout












Similar content being viewed by others
References
Melmed, S., Koenig, R., Rosen, C. J., Auchus, R. J. & Goldfine, A. B. Williams Textbook of Endocrinology (Elsevier, 2019).
Cools, M. et al. Caring for individuals with a difference of sex development (DSD): a consensus statement. Nat. Rev. Endocrinol. 14, 415–429 (2018).
Hughes, I. A., Houk, C., Ahmed, S. F. & Lee, P. A. Lawson Wilkins pediatric endocrine society/European society for paediatric endocrinology consensus g. consensus statement on management of intersex disorders. J. Pediatr. Urol. 2, 148–162 (2006).
Lee, P. A. et al. Global disorders of sex development update since 2006: perceptions, approach and care. Horm. Res. Paediatr. 85, 158–180 (2016).
Kouri, C. et al. Clinical and genetic characteristics of a large international cohort of individuals with rare NR5A1/SF-1 variants of sex development. eBioMedicine 99, 104941 (2024).
Division of Family and Reproductive Health. Gender and Health: Technical Paper (World Health Organization, 1998).
Claahsen-van der Grinten, H. L. et al. Congenital adrenal hyperplasia-current insights in pathophysiology, diagnostics, and management. Endocr. Rev. 43, 91–159 (2022).
Gravholt, C. H. et al. Clinical practice guidelines for the care of girls and women with Turner syndrome: proceedings from the 2016 Cincinnati international turner syndrome meeting. Eur. J. Endocrinol. 177, G1–G70 (2017).
Gravholt, C. H. et al. New developments and future trajectories in supernumerary sex chromosome abnormalities: a summary of the 2022 3rd International workshop on klinefelter syndrome, Trisomy X, and XYY. Endocr. Connect. 12, e220500 (2023).
Gravholt, C. H., Viuff, M. H., Brun, S., Stochholm, K. & Andersen, N. H. Turner syndrome: mechanisms and management. Nat. Rev. Endocrinol. 15, 601–614 (2019).
Kanakatti Shankar, R., Gravholt, C. H. & Backeljauw, P. F. Evolution of health care in turner syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 199, e32124 (2024).
Zitzmann, M. et al. European Academy of Andrology guidelines on Klinefelter syndrome endorsing organization: European Society of Endocrinology. Andrology 9, 145–167 (2021).
Speiser, P. W. et al. Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 103, 4043–4088 (2018).
Shankar, R. K. & Backeljauw, P. F. Current best practice in the management of Turner syndrome. Ther. Adv. Endocrinol. Metab. 9, 33–40 (2018).
Merke, D. P. & Auchus, R. J. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. N. Engl. J. Med. 383, 1248–1261 (2020).
Ratcliffe, S. Long-term outcome in children of sex chromosome abnormalities. Arch. Dis. Child. 80, 192–195 (1999).
Zhao, Y. et al. Detection and characterization of male sex chromosome abnormalities in the UK biobank study. Genet. Med. 24, 1909–1919 (2022).
Jacobs, P. A., Melville, M., Ratcliffe, S., Keay, A. J. & Syme, J. A cytogenetic survey of 11,680 newborn infants. Ann. Hum. Genet. 37, 359–376 (1974).
Nielsen, J. & Wohlert, M. Chromosome abnormalities found among 34,910 newborn children: results from a 13-year incidence study in Arhus, Denmark. Hum. Genet. 87, 81–83 (1991).
Lucas-Herald, A. K. et al. The outcome of prenatal identification of sex chromosome abnormalities. Arch. Dis. Child. Fetal Neonatal Ed. 101, F423–F427 (2016).
Ahmed, S. F. et al. Prevalence of hypospadias and other genital anomalies among singleton births, 1988-1997, in Scotland. Arch. Dis. Child. Fetal Neonatal Ed. 89, F149–F151 (2004).
Rodie, M. E. et al. A nationwide study of the prevalence and initial management of atypical genitalia in the newborn in Scotland. Sex. Dev. 16, 11–18 (2022).
Acerini, C. L., Miles, H. L., Dunger, D. B., Ong, K. K. & Hughes, I. A. The descriptive epidemiology of congenital and acquired cryptorchidism in a UK infant cohort. Arch. Dis. Child. 94, 868–872 (2009).
Virtanen, H. E. & Toppari, J. Epidemiology and pathogenesis of cryptorchidism. Hum. Reprod. Update 14, 49–58 (2008).
Nixon, R. et al. Prevalence of endocrine and genetic abnormalities in boys evaluated systematically for a disorder of sex development. Hum. Reprod. 32, 2130–2137 (2017).
Tang, Y. et al. Clinical characteristics and genetic expansion of 46,XY disorders of sex development children in a Chinese prospective study. Endocr. Connect. 12, e230029 (2023).
Gazdagh, G., Tobias, E. S., Ahmed, S. F. & McGowan, R.; DDD Study Group. Novel genetic associations and range of phenotypes in children with disorders of sex development and neurodevelopment: insights from the deciphering developmental disorders study. Sex. Dev. 10, 130–135 (2016).
Cox, K. et al. Novel associations in disorders of sex development: findings from the I-DSD registry. J. Clin. Endocrinol. Metab. 99, E348–E355 (2014).
Ahmed, S. F. et al. Society for endocrinology UK guidance on the initial evaluation of a suspected difference or disorder of sex development (Revised 2021). Clin. Endocrinol. 95, 818–840 (2021).
Berglund, A. et al. Incidence, prevalence, diagnostic delay, and clinical presentation of female 46,XY disorders of sex development. J. Clin. Endocrinol. Metab. 101, 4532–4540 (2016).
Batista, R. L. et al. Androgen insensitivity syndrome: a review. Arch. Endocrinol. Metab. 62, 227–235 (2018).
Callens, N. et al. Sexual quality of life after hormonal and surgical treatment, including phalloplasty, in men with micropenis: a review. J. Sex. Med. 10, 2890–2903 (2013).
Lucas-Herald, A. et al. The long-term outcome of boys with partial androgen insensitivity syndrome and a mutation in the androgen receptor gene. J. Clin. Endocrinol. Metab. 101, 3959–3967 (2016).
Falhammar, H. et al. Health status in 1040 adults with disorders of sex development (DSD): a European multicenter study. Endocr. Connect. 7, 466–478 (2018).
Lucas-Herald, A. K. et al. Vascular dysfunction and increased cardiovascular risk in hypospadias. Eur. Heart J. 43, 1832–1845 (2022).
Phillips, L. et al. Increased androgen-related comorbidity in adolescents and adults born with hypospadias: a population-based study. Andrology 10, 1376–1386 (2022).
Reyes, A. P., Leon, N. Y., Frost, E. R. & Harley, V. R. Genetic control of typical and atypical sex development. Nat. Rev. Urol. 20, 434–451 (2023).
Rotgers, E., Jorgensen, A. & Yao, H. H. At the crossroads of fate-somatic cell lineage specification in the fetal gonad. Endocr. Rev. 39, 739–759 (2018).
Stevant, I. & Nef, S. Genetic control of gonadal sex determination and development. Trends Genet. 35, 346–358 (2019).
Nef, S., Stevant, I. & Greenfield, A. Characterizing the bipotential mammalian gonad. Curr. Top. Dev. Biol. 134, 167–194 (2019).
Garcia-Alonso, L. et al. Single-cell roadmap of human gonadal development. Nature 607, 540–547 (2022).
Croft, B. et al. Human sex reversal is caused by duplication or deletion of core enhancers upstream of SOX9. Nat. Commun. 9, 5319 (2018).
Vining, B., Ming, Z., Bagheri-Fam, S. & Harley, V. Diverse regulation but conserved function: SOX9 in vertebrate sex determination. Genes 12, 486 (2021).
Ming, Z., Vining, B., Bagheri-Fam, S. & Harley, V. SOX9 in organogenesis: shared and unique transcriptional functions. Cell Mol. Life Sci. 79, 522 (2022).
Sreenivasan, R., Gonen, N. & Sinclair, A. SOX genes and their role in disorders of sex development. Sex. Dev. 16, 80–91 (2022).
Leon, N. Y. & Harley, V. R. ATR-X syndrome: genetics, clinical spectrum, and management. Hum. Genet. 140, 1625–1634 (2021).
Biason-Lauber, A. & Chaboissier, M. C. Ovarian development and disease: the known and the unexpected. Semin. Cell Dev. Biol. 45, 59–67 (2015).
He, J. et al. Unveiling the role of FOXL2 in female differentiation and disease: a comprehensive reviewdagger. Biol. Reprod. 112, 600–613 (2025).
Barbaux, S. et al. Donor splice-site mutations in WT1 are responsible for Frasier syndrome. Nat. Genet. 17, 467–470 (1997).
Bradford, S. T. et al. A cell-autonomous role for WT1 in regulating Sry in vivo. Hum. Mol. Genet. 18, 3429–3438 (2009).
Elzaiat, M., McElreavey, K. & Bashamboo, A. Genetics of 46,XY gonadal dysgenesis. Best Pract. Res. Clin. Endocrinol. Metab. 36, 101633 (2022).
McCann-Crosby, B. et al. State of the art review in gonadal dysgenesis: challenges in diagnosis and management. Int. J. Pediatr. Endocrinol. 2014, 4 (2014).
Sreenivasan, R. et al. Mutant NR5A1/SF-1 in patients with disorders of sex development shows defective activation of the SOX9 TESCO enhancer. Hum. Mutat. 39, 1861–1874 (2018).
Sato, N. S. et al. Partial duplication of DHH causes minifascicular neuropathy: a novel mutation detection of DHH. Ann. Clin. Transl. Neurol. 4, 415–421 (2017).
Croft, B. et al. FGF9 variant in 46,XY DSD patient suggests a role for dimerization in sex determination. Clin. Genet. 103, 277–287 (2023).
Bagheri-Fam, S. et al. FGFR2 mutation in 46,XY sex reversal with craniosynostosis. Hum. Mol. Genet. 24, 6699–6710 (2015).
Jameson, S. A., Lin, Y. T. & Capel, B. Testis development requires the repression of Wnt4 by Fgf signaling. Dev. Biol. 370, 24–32 (2012).
Ferrari, M. T. M. et al. Testicular differentiation in 46,XX DSD: an overview of genetic causes. Front. Endocrinol. 15, 1385901 (2024).
Hiort, M. et al. Testicular architecture of men with 46,XX testicular disorders of sex development. Sex. Dev. 17, 32–42 (2023).
Grinspon, R. P. & Rey, R. A. Disorders of sex development with testicular differentiation in SRY-negative 46,XX individuals: clinical and genetic aspects. Sex. Dev. 10, 1–11 (2016).
Bashamboo, A. et al. Loss of function of the nuclear receptor NR2F2, encoding COUP-TF2, causes testis development and cardiac defects in 46,XX children. Am. J. Hum. Genet. 102, 487–493 (2018).
Wankanit, S. et al. Evidence for NR2F2/COUP-TFII involvement in human testis development. Sci. Rep. 14, 17869 (2024).
Franca, M. M. & Mendonca, B. B. Genetics of ovarian insufficiency and defects of folliculogenesis. Best Pract. Res. Clin. Endocrinol. Metab. 36, 101594 (2022).
Roly, Z. Y. et al. The cell biology and molecular genetics of Mullerian duct development. Wiley Interdiscip. Rev. Dev. Biol. 7, e310 (2018).
Josso, N. & Picard, J. Y. Genetics of anti-Mullerian hormone and its signaling pathway. Best Pract. Res. Clin. Endocrinol. Metab. 36, 101634 (2022).
Josso, N. & Rey, R. A. What does AMH tell us in pediatric disorders of sex development? Front. Endocrinol. 11, 619 (2020).
Connan-Perrot, S. et al. Six decades of research on human fetal gonadal steroids. Int. J. Mol. Sci. 22, 6681 (2021).
Miller, W. L. & Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 32, 81–151 (2011).
Flueck C. E. & Pandey A. V. in Endocrinology of the Testis and Male Reproduction (eds Simoni, M. & Huhtaniemi, I. T.) 1–29 (Springer Cham, 2017).
Favorito, L. A. & Sampaio, F. J. B. in Translational Research in Pediatric Urology Basic and Clinical Aspects (ed. Favorito, L. A.) 27–48 (Springer, 2020).
Favorito, L. A. in Translational Research in Pediatric Urology Basic and Clinical Aspects (ed. Favorito, L. A.) 49–57 (Springer, 2020).
Rey, R. A. Biomarkers of male hypogonadism in childhood and adolescence. Adv. Lab. Med. 1, 20200024 (2020).
Bay, K., Main, K. M., Toppari, J. & Skakkebaek, N. E. Testicular descent: INSL3, testosterone, genes and the intrauterine milieu. Nat. Rev. Urol. 8, 187–196 (2011).
Bergada, I. et al. Time course of the serum gonadotropin surge, inhibins, and anti-Mullerian hormone in normal newborn males during the first month of life. J. Clin. Endocrinol. Metab. 91, 4092–4098 (2006).
Busch, A. S. et al. Male minipuberty in human and non-human primates: planting the seeds of future fertility. Reproduction 166, R63–R72 (2023).
Becker, M. & Hesse, V. Minipuberty: why does it happen? Horm. Res. Paediatr. 93, 76–84 (2020).
Johannsen, T. H. et al. Sex differences in reproductive hormones during mini-puberty in infants with normal and disordered sex development. J. Clin. Endocrinol. Metab. 103, 3028–3037 (2018).
Aksglaede, L., Davis, S. M., Ross, J. L. & Juul, A. Minipuberty in Klinefelter syndrome: current status and future directions. Am. J. Med. Genet. C. Semin. Med. Genet. 184, 320–326 (2020).
Fluck, C. E. et al. Why boys will be boys: two pathways of fetal testicular androgen biosynthesis are needed for male sexual differentiation. Am. J. Hum. Genet. 89, 201–218 (2011).
O’Shaughnessy, P. J. et al. Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. 17, e3000002 (2019).
Miller, W. L., Fluck, C. E., Breault, D. T. & Feldman, B. J. in Sperling – Pediatric Endocrinology 5th ed (eds Sperling, M. A. et al.) 425–490 (Elsevier, 2021).
Hornig, N. C. & Holterhus, P. M. Molecular basis of androgen insensitivity syndromes. Mol. Cell Endocrinol. 523, 111146 (2021).
Knerr, J. et al. Formin-mediated nuclear actin at androgen receptors promotes transcription. Nature 617, 616–622 (2023).
Boettcher, C. & Fluck, C. E. Rare forms of genetic steroidogenic defects affecting the gonads and adrenals. Best Pract. Res. Clin. Endocrinol. Metab. 36, 101593 (2022).
Pandey, A. V. & Fluck, C. E. NADPH P450 oxidoreductase: structure, function, and pathology of diseases. Pharmacol. Ther. 138, 229–254 (2013).
Grinspon, R. P., Bergada, I. & Rey, R. A. Male hypogonadism and disorders of sex development. Front. Endocrinol. 11, 211 (2020).
Latronico, A. C. & Arnhold, I. J. Gonadotropin resistance. Endocr. Dev. 24, 25–32 (2013).
Oosterhuis, J. W. & Looijenga, L. H. J. Human germ cell tumours from a developmental perspective. Nat. Rev. Cancer 19, 522–537 (2019).
Stoop, H. et al. Stem cell factor as a novel diagnostic marker for early malignant germ cells. J. Pathol. 216, 43–54 (2008).
Kersemaekers, A. M. et al. Identification of germ cells at risk for neoplastic transformation in gonadoblastoma: an immunohistochemical study for OCT3/4 and TSPY. Hum. Pathol. 36, 512–521 (2005).
Cools, M., Drop, S. L., Wolffenbuttel, K. P., Oosterhuis, J. W. & Looijenga, L. H. Germ cell tumors in the intersex gonad: old paths, new directions, moving frontiers. Endocr. Rev. 27, 468–484 (2006).
Cools, M. et al. Gonadoblastoma arising in undifferentiated gonadal tissue within dysgenetic gonads. J. Clin. Endocrinol. Metab. 91, 2404–2413 (2006).
Pyle, L. C. & Nathanson, K. L. A practical guide for evaluating gonadal germ cell tumor predisposition in differences of sex development. Am. J. Med. Genet. C. Semin. Med. Genet. 175, 304–314 (2017).
Cools, M. et al. Malignant testicular germ cell tumors in postpubertal individuals with androgen insensitivity: prevalence, pathology and relevance of single nucleotide polymorphism-based susceptibility profiling. Hum. Reprod. 32, 2561–2573 (2017).
Cools, M. et al. Morphological and immunohistochemical differences between gonadal maturation delay and early germ cell neoplasia in patients with undervirilization syndromes. J. Clin. Endocrinol. Metab. 90, 5295–5303 (2005).
Humphrey, P. A., Moch, H., Cubilla, A. L., Ulbright, T. M. & Reuter, V. E. The 2016 WHO classification of tumours of the urinary system and male genital organs-part B: prostate and bladder tumours. Eur. Urol. 70, 106–119 (2016).
Cools, M. et al. Gonadal pathology and tumor risk in relation to clinical characteristics in patients with 45,X/46,XY mosaicism. J. Clin. Endocrinol. Metab. 96, E1171–E1180 (2011).
Cools, M., Wolffenbuttel, K. P., Drop, S. L., Oosterhuis, J. W. & Looijenga, L. H. Gonadal development and tumor formation at the crossroads of male and female sex determination. Sex. Dev. 5, 167–180 (2011).
Oram, S. W., Liu, X. X., Lee, T. L., Chan, W. Y. & Lau, Y. F. TSPY potentiates cell proliferation and tumorigenesis by promoting cell cycle progression in HeLa and NIH3T3 cells. BMC Cancer 6, 154 (2006).
Lip, S. Z., Murchison, L. E., Cullis, P. S., Govan, L. & Carachi, R. A meta-analysis of the risk of boys with isolated cryptorchidism developing testicular cancer in later life. Arch. Dis. Child. 98, 20–26 (2013).
Tack, L. J. W. et al. Management of gonads in adults with androgen insensitivity: an international survey. Horm. Res. Paediatr. 90, 236–246 (2018).
van der Zwan, Y. G., Biermann, K., Wolffenbuttel, K. P., Cools, M. & Looijenga, L. H. Gonadal maldevelopment as risk factor for germ cell cancer: towards a clinical decision model. Eur. Urol. 67, 692–701 (2015).
Dorssers, L. C. J. et al. Molecular heterogeneity and early metastatic clone selection in testicular germ cell cancer development. Br. J. Cancer 120, 444–452 (2019).
Nogales, F. F. & Jimenez, R. E. Pathology and Biology of Human Germ Cell Tumors (Springer, 2017).
Audi, L. et al. Genetics in endocrinology: approaches to molecular genetic diagnosis in the management of differences/disorders of sex development (DSD): position paper of EU COST action BM 1303 ‘DSDnet’. Eur. J. Endocrinol. 179, R197–R206 (2018).
Kulle, A. et al. Steroid hormone analysis in diagnosis and treatment of DSD: position paper of EU COST Action BM 1303 ‘DSDnet’. Eur. J. Endocrinol. 176, P1–P9 (2017).
Lucas-Herald, A. K., Rodie, M. E. & Ahmed, S. F. Update on the management of a newborn with a suspected difference of sex development. Arch Dis Child. 107, 866–871 (2022).
Johannsen, T. H. et al. Peptide hormone analysis in diagnosis and treatment of differences of sex development: joint position paper of EU COST action ‘DSDnet’ and European reference network on rare endocrine conditions. Eur. J. Endocrinol. 182, P1–P15 (2020).
Délot, E. C. & Vilain, E. Towards improved genetic diagnosis of human differences of sex development. Nat. Rev. Genet. 22, 588–602 (2021).
Leon, N. Y., Reyes, A. P. & Harley, V. R. A clinical algorithm to diagnose differences of sex development. Lancet Diabetes Endocrinol. 7, 560–574 (2019).
Guerrero-Fernandez, J. et al. Management guidelines for disorders / different sex development (DSD) [Spanish]. An. Pediatr. 89, 315.e1–315.e19 (2018).
Fluck, C. E. & Guran, T. Ambiguous genitalia in the newborn. Endotext https://www.ncbi.nlm.nih.gov/books/NBK279168/ (2023).
Prader, A. Genital findings in the female pseudo-hermaphroditism of the congenital adrenogenital syndrome; morphology, frequency, development and heredity of the different genital forms [German]. Helv. Paediatr. Acta 9, 231–248 (1954).
Quigley, C. A. et al. Androgen receptor defects: historical, clinical, and molecular perspectives. Endocr. Rev. 16, 271–321 (1995).
Ahmed, S. F., Khwaja, O. & Hughes, I. A. The role of a clinical score in the assessment of ambiguous genitalia. BJU Int. 85, 120–124 (2000).
van der Straaten, S. et al. The external genitalia score (EGS): a European multicenter validation study. J. Clin. Endocrinol. Metab. 105, dgz142 (2020).
Sathyanarayana, S. et al. Anogenital distance and penile width measurements in the infant development and the environment study (TIDES): methods and predictors. J. Pediatr. Urol. 11, 76.e1–76.e6 (2015).
Thankamony, A., Ong, K. K., Dunger, D. B., Acerini, C. L. & Hughes, I. A. Anogenital distance from birth to 2 years: a population study. Env. Health Perspect. 117, 1786–1790 (2009).
Hryhorczuk, A. L., Phelps, A. S., Yu, R. N. & Chow, J. S. The radiologist’s role in assessing differences of sex development. Pediatr. Radiol. 52, 752–764 (2022).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Storup, G. Fetal sex determination during ultrasonography: should we focus on how rather than why? Acta Obstet. Gynecol. Scand. 98, 1483–1484 (2019).
Lubusky, M., Studnickova, M., Skrivanek, A., Vomackova, K. & Prochazka, M. Ultrasound evaluation of fetal gender at 12-14 weeks. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 156, 324–329 (2012).
Guo, N. et al. Positive predictive value of noninvasive prenatal testing for sex chromosome abnormalities. Mol. Biol. Rep. 49, 9251–9256 (2022).
Kyriakou, A. et al. Current models of care for disorders of sex development — results from an International survey of specialist centres. Orphanet J. Rare Dis. 11, 155 (2016).
Cools, M., Looijenga, L. H., Wolffenbuttel, K. P. & T’Sjoen, G. Managing the risk of germ cell tumourigenesis in disorders of sex development patients. Endocr. Dev. 27, 185–196 (2014).
Wolffenbuttel, K. P. et al. Gonadal dysgenesis in disorders of sex development: diagnosis and surgical management. J. Pediatr. Urol. 12, 411–416 (2016).
Dieckmann, K. P. et al. MicroRNAs miR-371-3 in serum as diagnostic tools in the management of testicular germ cell tumours. Br. J. Cancer 107, 1754–1760 (2012).
Gillis, A. J. et al. Targeted serum miRNA (TSmiR) test for diagnosis and follow-up of (testicular) germ cell cancer patients: a proof of principle. Mol. Oncol. 7, 1083–1092 (2013).
van Agthoven, T. & Looijenga, L. H. J. Accurate primary germ cell cancer diagnosis using serum based microRNA detection (ampTSmiR test). Oncotarget 8, 58037–58049 (2017).
Kalfa, N., Philibert, P., Baskin, L. S. & Sultan, C. Hypospadias: interactions between environment and genetics. Mol. Cell Endocrinol. 335, 89–95 (2011).
Jorgensen, A. et al. Environmental impacts on male reproductive development: lessons from experimental models. Horm. Res. Paediatr. 96, 190–206 (2023).
Hurtado-Gonzalez, P. & Mitchell, R. T. Analgesic use in pregnancy and male reproductive development. Curr. Opin. Endocrinol. Diabetes Obes. 24, 225–232 (2017).
Wilkins, L., Jones, H. W. Jr., Holman, G. H. & Stempfel, R. S. Jr Masculinization of the female fetus associated with administration of oral and intramuscular progestins during gestation: non-adrenal female pseudohermaphrodism. J. Clin. Endocrinol. Metab. 18, 559–585 (1958).
Troisi, R. et al. Prenatal diethylstilbestrol exposure and high-grade squamous cell neoplasia of the lower genital tract. Am. J. Obstet. Gynecol. 215, 322 e1–322 e8 (2016).
Stillman, R. J. In utero exposure to diethylstilbestrol: adverse effects on the reproductive tract and reproductive performance and male and female offspring. Am. J. Obstet. Gynecol. 142, 905–921 (1982).
Armstrong, K. et al. We all have a role to play: redressing inequities for children living with CAH and other chronic health conditions of childhood in resource-poor settings. Int. J. Neonatal Screen. 6, 76 (2020).
Armstrong, K. L., Henderson, C., Hoan, N. T. & Warne, G. L. Living with congenital adrenal hyperplasia in Vietnam: a survey of parents. J. Pediatr. Endocrinol. Metab. 19, 1207–1223 (2006).
Ediati, A., Maharani, N. & Utari, A. Sociocultural aspects of disorders of sex development. Birth Defects Res. C., Embryo Today 108, 380–383 (2016).
Khan, A. et al. Barriers in access to healthcare services for individuals with disorders of sex differentiation in Bangladesh: an analysis of regional representative cross-sectional data. BMC Public Health 20, 1261 (2020).
Joseph, A. A. et al. Gender issues and related social stigma affecting patients with a disorder of sex development in India. Arch. Sex. Behav. 46, 361–367 (2017).
Yarhere, I. & Ahmed, S. F. in Practical Pediatric Endocrinology in a Limited Resource Setting (ed. Zacharin, M.) 123–134 (Academic, 2013).
Warne, G. L. & Raza, J. Disorders of sex development (DSDs), their presentation and management in different cultures. Rev. Endocr. Metab. Disord. 9, 227–236 (2008).
Utari, A. et al. Challenges in the treatment of late-identified untreated congenital adrenal hyperplasia due to CYP11B1 deficiency: lessons from a developing country. Front. Endocrinol. 13, 1015973 (2022).
Lucas-Herald, A. K., Scougall, K. & Ahmed, S. F. Delivery of multidisciplinary care in the field of differences and disorders of sex development (DSD). Expert Rev. Endocrinol. Metab. 17, 225–234 (2022).
Cools, M. et al. Multi-stakeholder opinion statement on the care of individuals born with differences of sex development: common ground and opportunities for improvement. Horm. Res. Paediatr. 98, 226–242 (2025).
Gardner, M. et al. Decisional support needed when facing tough decisions: survey of parents with children having differences of sex development. Front. Urol. 3, 1089077 (2023).
Lightfoot, S. et al. Co-creating a suite of patient decision aids for parents of an infant or young child with differences of sex development: a methods roadmap. Front. Urol. 2, 1020152 (2023).
Ernst, M. M., Liao, L. M., Baratz, A. B. & Sandberg, D. E. Disorders of sex development/intersex: gaps in psychosocial care for children. Pediatrics 142, e20174045 (2018).
Atlas, G. et al. Challenges in care provision for children and adolescents with differences of sex development. Clin. Endocrinol. 102, 539–546 (2025).
Gramc, M., Streuli, J. & de Clercq, E. Multidisciplinary teams caring for people with variations of sex characteristics: a scoping review. BMJ Paediatr. Open 5, e001257 (2021).
Bennecke, E. et al. Subjective need for psychological support (PsySupp) in parents of children and adolescents with disorders of sex development (DSD). Eur. J. Pediatr. 174, 1287–1297 (2015).
Ernst, M. M. et al. Psychosocial screening in disorders/differences of sex development: psychometric evaluation of the psychosocial assessment tool. Horm. Res. Paediatr. 90, 368–380 (2018).
Sandberg, D. E., Gardner, M., Callens, N. & Mazur, T. DSD-TRN Psychosocial Workgroup, the DSD-TRN Advocacy Advisory Network, and Accord Alliance. Interdisciplinary care in disorders/differences of sex development (DSD): the psychosocial component of the DSD-translational research network. Am. J. Med. Genet. C. Semin. Med. Genet. 175, 279–292 (2017).
Mateo-Arriero, I., Perry, Y., Gilbey, D. & Ohan, J. L. Parents’ disclosure of their child’s health and neurodevelopmental conditions: a systematic review and qualitative metasynthesis. J. Fam. Psychol. 39, 503–514 (2025).
[No authors listed]. Disclosure of illness status to children and adolescents with HIV infection. American Academy of Pediatrics Committee on Pediatrics AIDS. Pediatrics 103, 164–166 (1999).
Slepian, M. L. The new psychology of secrecy. Curr. Dir. Psychol. Sci. https://doi.org/10.1177/09637214241226676 (2024).
Sisk, B. A., Bluebond-Langner, M., Wiener, L., Mack, J. & Wolfe, J. Prognostic disclosures to children: a historical perspective. Pediatrics 138, e20161278 (2016).
Moyer, D. N., Suorsa-Johnson, K. I., Weidler, E. M. & Ernst, M. M. DSD-TRN psychosocial workgroup. Information sharing in differences of sex development: the creation of a caregiver-support tool. Fam. Syst. Health 41, 256–264 (2023).
Baratz, A. B., Sharp, M. K., & Sandberg, D. E. in Endocrine Development. Understanding Differences and Disorders of Sex Development (DSD) Endocrine Development (eds Hiort, O. & Ahmed, S.) 99–112 (Karger, 2014).
de Brouwer, I. J., Suijkerbuijk, M., van de Grift, T. C. & Kreukels, B. P. C. First adolescent romantic and sexual experiences in individuals with differences of sex development/intersex conditions. J. Adolesc. Health 71, 688–695 (2022).
Mediå, L. M., Sigurdardottir, S., Fauske, L. & Waehre, A. Understanding sexual health concerns among adolescents and young adults with differences of sex development: a qualitative study. Int. J. Qual. Stud. Health Well-being 18, 2204635 (2023).
Engelen, M. M., Knoll, J. L., Rabsztyn, P. R. I., Maas-van Schaaijk, N. M. & van Gaal, B. G. I. Sexual health communication between healthcare professionals and adolescents with chronic conditions in western countries: an integrative review. Sexuality Disabil. 38, 191–216 (2020).
Bungener, S. L. et al. Talking about sexuality with youth: a taboo in psychiatry? J. Sex. Med. 19, 421–429 (2022).
MedlinePlus. NIH https://medlineplus.gov/ (2025).
Orphanet. Orphanet https://www.orpha.net/ (2025).
How sex development works. University Health Network & The Hospital for Sick Children https://pie.med.utoronto.ca/htbw/module.html?module=sex-development (2015).
Intersex-Affirming Hospital Policy Guide: Providing Ethical and Compassionate Health Care to Intersex Patients (Lambda Legal, 2018).
Cools, M. et al. Response to the Council of Europe Human Rights Commissioner’s issue paper on human rights and intersex people. Eur. Urol. 70, 407–409 (2016).
Gardner, M. & Sandberg, D. E. Navigating surgical decision making in disorders of sex development (DSD). Front. Pediatr. 6, 339 (2018).
Indig, G., Serrano, M., Dalke, K. B., Ejiogu, N. I. & Grimstad, F. Clinician advocacy and intersex health: a history of intersex health care and the role of the clinician advocate past, present, and future. Pediatr. Ann. 50, e359–e365 (2021).
“I want to be like nature made me.” Medically Unnecessary Surgeries on Intersex Children in the US (Human Rights Watch & InterACT, 2017).
Légaré, F. & Witteman, H. O. Shared decision making: examining key elements and barriers to adoption into routine clinical practice. Health Aff. 32, 276–284 (2013).
Scalia, P. et al. The impact and utility of encounter patient decision aids: systematic review, meta-analysis and narrative synthesis. Patient Educ. Couns. 102, 817–841 (2019).
Niburski, K., Guadagno, E., Mohtashami, S. & Poenaru, D. Shared decision making in surgery: a scoping review of the literature. Health Expect. 23, 1241–1249 (2020).
Stacey, D. et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst. Rev. 1, CD001431 (2024).
Stacey, D. et al. Are patient decision aids used in clinical practice after rigorous evaluation? A survey of trial authors. Med. Decis. Mak. 39, 805–815 (2019).
Sandberg, D. E. et al. Development of a decision support tool in pediatric differences/disorders of sex development. Semin. Pediatr. Surg. 28, 150838 (2019).
Ediati, A. et al. Social stigmatisation in late identified patients with disorders of sex development in Indonesia. BMJ Paediatr. Open 1, e000130 (2017).
Kuhnle, U. & Krahl, W. The impact of culture on sex assignment and gender development in intersex patients. Perspect. Biol. Med. 45, 85–103 (2002).
Duguid, A. et al. The psychological impact of genital anomalies on the parents of affected children. Acta Paediatr. 96, 348–352 (2007).
Pasterski, V., Mastroyannopoulou, K., Wright, D., Zucker, K. J. & Hughes, I. A. Predictors of posttraumatic stress in parents of children diagnosed with a disorder of sex development. Arch. Sex. Behav. 43, 369–375 (2014).
Traino, K. A. et al. Stigma, intrusiveness, and distress in parents of children with a disorder/difference of sex development. J. Dev. Behav. Pediatr. 43, e473–e482 (2022).
Weidler, E. M. & Peterson, K. E. The impact of culture on disclosure in differences of sex development. Semin. Pediatr. Surg. 28, 150840 (2019).
Auer, M. K., Nordenstrom, A., Lajic, S. & Reisch, N. Congenital adrenal hyperplasia. Lancet 401, 227–244 (2023).
Stancampiano, M. R. et al. Congenital micropenis: etiology and management. J. Endocr. Soc. 6, bvab172 (2022).
Fu, X. H., Zhang, W. Q. & Qu, X. S. Correlation of androgen receptor and SRD5A2 gene mutations with pediatric hypospadias in 46, XY DSD children. Genet. Mol. Res. 15, 15018232 (2016).
Nordenstrom, A. et al. Pubertal induction and transition to adult sex hormone replacement in patients with congenital pituitary or gonadal reproductive hormone deficiency: an Endo-ERN clinical practice guideline. Eur. J. Endocrinol. 186, G9–G49 (2022).
Andrade, J. G. R. et al. Clinical findings and follow-up of 46,XY and 45,X/46,XY testicular dysgenesis. Sex. Dev. 13, 171–177 (2019).
Ljubicic, M. L. et al. Clinical but not histological outcomes in males with 45,X/46,XY mosaicism vary depending on reason for diagnosis. J. Clin. Endocrinol. Metab. 104, 4366–4381 (2019).
Tantawy, S. et al. Testosterone production during puberty in two 46,XY patients with disorders of sex development and novel NR5A1 (SF-1) mutations. Eur. J. Endocrinol. 167, 125–130 (2012).
Chang, S., Skakkebaek, A., Davis, S. M. & Gravholt, C. H. Morbidity in Klinefelter syndrome and the effect of testosterone treatment. Am. J. Med. Genet. C Semin. Med. Genet. 184, 344–355 (2020).
Vorona, E., Zitzmann, M., Gromoll, J., Schuring, A. N. & Nieschlag, E. Clinical, endocrinological, and epigenetic features of the 46,XX male syndrome, compared with 47,XXY Klinefelter patients. J. Clin. Endocrinol. Metab. 92, 3458–3465 (2007).
Lek, N. et al. Predicting puberty in partial androgen insensitivity syndrome: use of clinical and functional androgen receptor indices. eBioMedicine 36, 401–409 (2018).
Hughes, I. A. et al. Androgen insensitivity syndrome. Lancet 380, 1419–1428 (2012).
Kim, Y. M., Oh, A., Kim, K. S., Yoo, H. W. & Choi, J. H. Pubertal outcomes and sex of rearing of patients with ovotesticular disorder of sex development and mixed gonadal dysgenesis. Ann. Pediatr. Endocrinol. Metab. 24, 231–236 (2019).
Bernard, V. et al. Spontaneous fertility and pregnancy outcomes amongst 480 women with Turner syndrome. Hum. Reprod. 31, 782–788 (2016).
Stancampiano, M. R. et al. Testosterone therapy and its monitoring in adolescent boys with hypogonadism: results of an international survey from the I-DSD registry. Sex. Dev. 15, 236–243 (2021).
Gawlik, A. M. et al. Late-onset puberty induction by transdermal estrogen in Turner syndrome girls — a longitudinal study. Front. Endocrinol. 9, 23 (2018).
Lawrence, S. E., Faught, K. A., Vethamuthu, J. & Lawson, M. L. Beneficial effects of raloxifene and tamoxifen in the treatment of pubertal gynecomastia. J. Pediatr. 145, 71–76 (2004).
Saito, R. et al. Tamoxifen treatment for pubertal gynecomastia in two siblings with partial androgen insensitivity syndrome. Horm. Res. Paediatr. 81, 211–216 (2014).
Patjamontri, S. et al. Gynecomastia and its management in boys with partial androgen insensitivity syndrome. J. Clin. Endocrinol. Metab. 110, e2018–e2025 (2025).
Almasri, J. et al. Genital reconstructive surgery in females with congenital adrenal hyperplasia: a systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 103, 4089–4096 (2018).
Baskin, A. et al. Post-operative complications following feminizing genitoplasty in moderate to severe genital atypia: results from a multicenter, observational prospective cohort study. J. Pediatr. Urol. 16, 568–575 (2020).
Dangle, P. P., Lee, A., Chaudhry, R. & Schneck, F. X. Surgical complications following early genitourinary reconstructive surgery for congenital adrenal hyperplasia-interim analysis at 6 years. Urology 101, 111–115 (2017).
Nordenstrom, A. et al. Sexual function and surgical outcome in women with congenital adrenal hyperplasia due to CYP21A2 deficiency: clinical perspective and the patients’ perception. J. Clin. Endocrinol. Metab. 95, 3633–3640 (2010).
Rapp, M. et al. Self- and proxy-reported outcomes after surgery in people with disorders/differences of sex development (DSD) in Europe (dsd-LIFE). J. Pediatr. Urol. 17, 353–365 (2021).
Stites, J., Bernabe, K. J., Galan, D., Felsen, D. & Poppas, D. P. Urinary continence outcomes following vaginoplasty in patients with congenital adrenal hyperplasia. J. Pediatr. Urol. 13, 38.e1–e7 (2017).
Krege, S. et al. Long-term results of surgical treatment and patient-reported outcomes in congenital adrenal hyperplasia — a multicenter European registry study. J. Clin. Med. 11, 4629 (2022).
Meyer-Bahlburg, H. F. L. The timing of genital surgery in somatic intersexuality: surveys of patients’ preferences. Horm. Res. Paediatr. 95, 12–20 (2022).
Cools, M., Verhagen, E., Hoebeke, P., Van Hoecke, E. & Cannoot, P. Working towards convergence of the clinical management of differences of sex development/intersex conditions and the human rights framework: a case study. Clin. Endocrinol. 101, 499–506 (2024).
Sandberg, D. E. & Vilain, E. Decision making in differences of sex development/intersex care in the USA: bridging advocacy and family-centred care. Lancet Diabetes Endocrinol. 10, 381–383 (2022).
Crouch, N. S., Minto, C. L., Laio, L. M., Woodhouse, C. R. & Creighton, S. M. Genital sensation after feminizing genitoplasty for congenital adrenal hyperplasia: a pilot study. BJU Int. 93, 135–138 (2004).
Minto, C. L., Liao, L. M., Woodhouse, C. R., Ransley, P. G. & Creighton, S. M. The effect of clitoral surgery on sexual outcome in individuals who have intersex conditions with ambiguous genitalia: a cross-sectional study. Lancet 361, 1252–1257 (2003).
Poppas, D. P. et al. Nerve sparing ventral clitoroplasty preserves dorsal nerves in congenital adrenal hyperplasia. J. Urol. 178, 1802–1806 (2007).
Rink, R. C. et al. Reconstruction of the high urogenital sinus: early perineal prone approach without division of the rectum. J. Urol. 158, 1293–1297 (1997).
Salle, J. L. et al. Surgical treatment of high urogenital sinuses using the anterior sagittal transrectal approach: a useful strategy to optimize exposure and outcomes. J. Urol. 187, 1024–1031 (2012).
Bangalore Krishna, K. et al. Individualized care for patients with intersex (differences of sex development): part 4/4. Considering the ifs, whens, and whats regarding sexual-reproductive system surgery. J. Pediatr. Urol. 17, 338–345 (2021).
Money, J. Ablatio penis: normal male infant sex-reassigned as a girl. Arch. Sex. Behav. 4, 65–71 (1975).
Johnson, E. K., Whitehead, J. & Cheng, E. Y. Differences of sex development: current issues and controversies. Urol. Clin. North Am. 50, 433–446 (2023).
Baskin, L. S. et al. Anatomical studies of the human clitoris. J. Urol. 162, 1015–1020 (1999).
Pena, A., Levitt, M. A., Hong, A. & Midulla, P. Surgical management of cloacal malformations: a review of 339 patients. J. Pediatr. Surg. 39, 470–479 (2004).
Rink, R. C. et al. Partial urogenital mobilization: a limited proximal dissection. J. Pediatr. Urol. 2, 351–356 (2006).
Gong, E. M. & Cheng, E. Y. Current challenges with proximal hypospadias: we have a long way to go. J. Pediatr. Urol. 13, 457–467 (2017).
Long, C. J. et al. Post-operative complications following masculinizing genitoplasty in moderate to severe genital atypia: results from a multicenter, observational prospective cohort study. J. Pediatr. Urol. 17, 379–386 (2021).
Long, C. J. et al. Intermediate-term followup of proximal hypospadias repair reveals high complication rate. J. Urol. 197, 852–858 (2017).
McNamara, E. R. et al. Management of proximal hypospadias with 2-stage repair: 20-year experience. J. Urol. 194, 1080–1085 (2015).
Saltzman, A. F. et al. Patients with disorders of sex development and proximal hypospadias are at high risk for reoperation. World J. Urol. 36, 2051–2058 (2018).
Johal, N. S., Nitkunan, T., O’Malley, K. & Cuckow, P. M. The two-stage repair for severe primary hypospadias. Eur. Urol. 50, 366–371 (2006).
Husmann, D. A. Erectile dysfunction in patients undergoing multiple attempts at hypospadias repair: etiologies and concerns. J. Pediatr. Urol. 17, 166 e1–e7 (2021).
Cheng, L. et al. Testicular cancer. Nat. Rev. Dis. Primers 4, 29 (2018).
van de Grift, T. C.; dsd-LIFE.Condition openness is associated with better mental health in individuals with an intersex/differences of sex development condition: structural equation modeling of European multicenter data. Psychol. Med. 53, 2229–2240 (2023).
Berra, M., Liao, L. M., Creighton, S. M. & Conway, G. S. Long-term health issues of women with XY karyotype. Maturitas 65, 172–178 (2010).
Dati, E. et al. Body composition and metabolic profile in women with complete androgen insensitivity syndrome. Sex. Dev. 3, 188–193 (2009).
Bertelloni, S., Meriggiola, M. C., Dati, E., Balsamo, A. & Baroncelli, G. I. Bone mineral density in women living with complete androgen insensitivity syndrome and intact testes or removed gonads. Sex. Dev. 11, 182–189 (2017).
Bojesen, A., Juul, S., Birkebaek, N. H. & Gravholt, C. H. Morbidity in Klinefelter syndrome: a Danish register study based on hospital discharge diagnoses. J. Clin. Endocrinol. Metab. 91, 1254–1260 (2006).
Bojesen, A. et al. Bone mineral density in Klinefelter syndrome is reduced and primarily determined by muscle strength and resorptive markers, but not directly by testosterone. Osteoporos. Int. 22, 1441–1450 (2011).
Ferlin, A. et al. Bone mass in subjects with Klinefelter syndrome: role of testosterone levels and androgen receptor gene CAG polymorphism. J. Clin. Endocrinol. Metab. 96, E739–E745 (2011).
Vena, W. et al. Prevalence and determinants of radiological vertebral fractures in patients with Klinefelter syndrome. Andrology 8, 1699–1704 (2020).
Batista, R. L. et al. Sexuality and fertility desire in a large cohort of individuals with 46, XY differences in sex development. Clinics 78, 100185 (2023).
Kreukels, B. P. C. et al. Gender dysphoria and gender change in disorders of sex development/intersex conditions: results from the dsd-LIFE study. J. Sex. Med. 15, 777–785 (2018).
Loch Batista, R. et al. Psychosexual aspects, effects of prenatal androgen exposure, and gender change in 46,XY disorders of sex development. J. Clin. Endocrinol. Metab. 104, 1160–1170 (2019).
Wisniewski, A. B. et al. Management of 46,XY differences/disorders of sex development (DSD) throughout life. Endocr. Rev. 40, 1547–1572 (2019).
Gomes, N. L. et al. Long-term outcomes and molecular analysis of a large cohort of patients with 46,XY disorder of sex development due to partial gonadal dysgenesis. Clin. Endocrinol. 89, 164–177 (2018).
Callens, N. et al. Recalled and current gender role behavior, gender identity and sexual orientation in adults with disorders/differences of sex development. Horm. Behav. 86, 8–20 (2016).
Slowikowska-Hilczer, J. et al. Fertility outcome and information on fertility issues in individuals with different forms of disorders of sex development: findings from the dsd-LIFE study. Fertil. Steril. 108, 822–831 (2017).
Juul, A., Gravholt, C. H., De Vos, M., Koledova, E. & Cools, M. Individuals with numerical and structural variations of sex chromosomes: interdisciplinary management with focus on fertility potential. Front. Endocrinol. 14, 1160884 (2023).
Giwercman, A. et al. Preserved male fertility despite decreased androgen sensitivity caused by a mutation in the ligand-binding domain of the androgen receptor gene. J. Clin. Endocrinol. Metab. 85, 2253–2259 (2000).
Islam, R. et al. Establishing reproductive potential and advances in fertility preservation techniques for XY individuals with differences in sex development. Clin. Endocrinol. 91, 237–244 (2019).
Tordjman, K. M. et al. Fertility after high-dose testosterone and intracytoplasmic sperm injection in a patient with androgen insensitivity syndrome with a previously unreported androgen receptor mutation. Andrologia 46, 703–706 (2014).
Brännström, M., Bokström, H., Hagberg, H. & Carlsson, Y. Maternal and perinatal outcomes of live births after uterus transplantation: a systematic review. Acta Obstet. Gynecol. Scand. 104, 559–578 (2025).
Johannesson, L. et al. The first 5 years of uterus transplant in the US: a report from the United States uterus transplant consortium. JAMA Surg. 157, 790–797 (2022).
Rapp, M. et al. Multicentre cross-sectional clinical evaluation study about quality of life in adults with disorders/differences of sex development (DSD) compared to country specific reference populations (dsd-LIFE). Health Qual. Life Outcomes 16, 54 (2018).
Thyen, U. et al. Quality of health care in adolescents and adults with disorders/differences of sex development (DSD) in six European countries (dsd-LIFE). BMC Health Serv. Res. 18, 527 (2018).
Erens, B. et al. Nonprobability web surveys to measure sexual behaviors and attitudes in the general population: a comparison with a probability sample interview survey. J. Med. Internet Res. 16, e276 (2014).
Hines, M. Brain Gender (Oxford Univ. Press, 2004).
Hines, M. in Hormones, Brain and Behavior (ed. Pfaff, D. W.) 425–461 (Academic, 2002).
Berenbaum, S. A., Korman, K. & Leveroni, C. Early hormones and sex differences in cognitive abilities. Learn. Individ. Differ. 7, 303–321 (1995).
Chase, C. What is the agenda of the intersex patient advocacy movement? Endocrinologist 13, 240–242 (2003).
Lee, P. A., Houk, C. P., Ahmed, S. F. & Hughes, I. A.; International Consensus Conference on Intersex organized by the Lawson Wilkins Pediatric Endocrine Society and the European Society for Paediatric Endocrinology. Consensus statement on management of intersex disorders. International Consensus Conference on Intersex. Pediatrics 118, e488–500 (2006).
Engberg, H. et al. Increased psychiatric morbidity in women with complete androgen insensitivity syndrome or complete gonadal dysgenesis. J. Psychosom. Res. 101, 122–127 (2017).
Sorouri Khorashad, B. et al. Psychiatric comorbidities in women with complete androgen insensitivity syndrome or mullerian aplasia/agenesis. J. Clin. Endocrinol. Metab. 110, 1906–1914 (2025).
Liedmeier, A. et al. The influence of psychosocial and sexual wellbeing on quality of life in women with differences of sexual development. Compr. Psychoneuroendocrinol. 8, 100087 (2021).
Ellerkamp, V. et al. Surgical therapy after failed feminizing genitoplasty in young adults with disorders of sex development: retrospective analysis and review of the literature. J. Sex. Med. 18, 1797–1806 (2021).
van de Grift, T. C., Rapp, M., Holmdahl, G., Duranteau, L. & Nordenskjold, A.; on behalf of the dsd-LIFE group.Masculinizing surgery in disorders/differences of sex development: clinician- and participant-evaluated appearance and function. BJU Int. 129, 394–405 (2022).
Sandberg D. E., Mazur T. in Gender Dysphoria and Disorders of Sex Development. Focus on Sexuality Research (eds Kreukels, B. P. C. et al.) 93–114 (Springer Science+Business Media; 2014).
Traino, K. A. et al. Adverse birth experiences and parent adjustment associated with atypical genital appearance due to differences of sex development. J. Pediatr. Psychol. 48, 759–767 (2023).
Boucher, N. A., Alkazemi, M. H., Tejwani, R. & Routh, J. C. Parents of children with newly diagnosed disorders of sex development identify major concerns: a qualitative study. Urology 164, 218–223 (2022).
Verma, P. et al. Evaluation of a nurse-led counselling intervention on selected outcome variables for parents of children with congenital adrenal hyperplasia. J. Pediatr. Endocrinol. Metab. 36, 248–254 (2023).
Nordenström, A. et al. Hormone therapy and patient satisfaction with treatment, in a large cohort of diverse disorders of sex development. Clin. Endocrinol. 88, 397–408 (2018).
Cheng, J. W. et al. Integration of child life services in the delivery of multi-disciplinary differences in sexual development (DSD) and congenital adrenal hyperplasia (CAH) care. J. Pediatr. Urol. 18, 612.e1–e6 (2022).
Dwiggins, M. et al. Multidimensional aspects of female sexual function in congenital adrenal hyperplasia: a case-control study. J. Endocr. Soc. 4, bvaa131 (2020).
Szymanski, K. M. et al. Growing up with clitoromegaly: experiences of North American women with congenital adrenal hyperplasia. J. Pediatr. Urol. 18, 775–783 (2022).
Weijenborg, P. T. M., Kluivers, K. B., Dessens, A. B., Kate-Booij, M. J. & Both, S. Sexual functioning, sexual esteem, genital self-image and psychological and relational functioning in women with Mayer-Rokitansky-Kuster-Hauser syndrome: a case-control study. Hum. Reprod. 34, 1661–1673 (2019).
Nowotny, H. F. & Reisch, N. Challenges waiting for an adult with DSD. Horm. Res. Paediatr. 96, 207–221 (2023).
Chulani, V. L., Gomez-Lobo, V., Kielb, S. J. & Grimsby, G. M. Healthcare transition for patients with differences of sexual development and complex urogenital conditions. Semin. Pediatr. Surg. 28, 150846 (2019).
Auchus, R. J. & Quint, E. H. Adolescents with disorders of sex development (DSD)-lost in transition? Horm. Metab. Res. 47, 367–374 (2015).
Crouch, N. S. & Creighton, S. M. Transition of care for adolescents with disorders of sex development. Nat. Rev. Endocrinol. 10, 436–442 (2014).
Mediå, L. M. et al. ‘It was supposed to be a secret’: a study of disclosure and stigma as experienced by adults with differences of sex development. Health Psychol. Behav. Med. 10, 579–595 (2022).
Meyer-Bahlburg, H. F., Reyes-Portillo, J. A., Khuri, J., Ehrhardt, A. A. & New, M. I. Syndrome-related stigma in the general social environment as reported by women with classical congenital adrenal hyperplasia. Arch. Sex. Behav. 46, 341–351 (2017).
Meyer-Bahlburg, H. F. L., Khuri, J., Reyes-Portillo, J., Ehrhardt, A. A. & New, M. I. Stigma associated with classical congenital adrenal hyperplasia in women’s sexual lives. Arch. Sex. Behav. 47, 943–951 (2018).
Meyer-Bahlburg, H. F. L., Khuri, J., Reyes-Portillo, J. & New, M. I. Stigma in medical settings as reported retrospectively by women with congenital adrenal hyperplasia (CAH) for their childhood and adolescence. J. Pediatr. Psychol. 42, 496–503 (2017).
Logie, C. H. & Nyblade, L. Recognizing and responding to stigma-related barriers in health care. Nat. Rev. Dis. Primers 10, 70 (2024).
Hiort, O. et al. Addressing gaps in care of people with conditions affecting sex development and maturation. Nat. Rev. Endocrinol. 15, 615–622 (2019).
Lucas-Herald, A. K. et al. I-DSD: the first 10 years. Horm. Res. Paediatr. 96, 238–246 (2023).
Sanders, C. et al. Involving individuals with disorders of sex development and their parents in exploring new models of shared learning: proceedings from a DSDnet COST action workshop. Sex. Dev. 12, 225–231 (2018).
World Health Organization. WHO Framework for Meaningful Engagement of People Living with Noncommunicable Diseases, and Mental Health and Neurological Conditions (WHO, 2023).
Syryn, H., De Baere, E. & Cools, M. Approach to the patient with a difference of sex development. J. Clin. Endocrinol. Metab. https://doi.org/10.1210/clinem/dgaf386 (2025).
Acknowledgements
C.E.F. acknowledges the support by Swiss National Science Foundation grant 320030_197725. The authors thank Melissa D. Gardner for supporting manuscript editing.
Author information
Authors and Affiliations
Contributions
Introduction (S.F.A.); Epidemiology (S.F.A.); Mechanisms/pathophysiology (M.C., V.H. and R.R.); Diagnosis, screening and prevention (M.C. and C.E.F.); Management (E.Y.C., B.B.M., A.N. and D.E.S.); Quality of life (D.E.S., K.A. and A.U.); Outlook (S.F.A., K.A., A.U. and C.E.F.); overview of the Primer (S.F.A. and C.E.F.).
Corresponding author
Ethics declarations
Competing interests
Authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks O. Hiort, M. Fukami and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Informed consent
The authors affirm that patient participants provided informed consent for publication of their experiences.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Adrenoleukodystrophy
-
Rare X-linked recessive inherited disorder, characterized by neurological symptoms and dysfunctional adrenal glands.
- Androgen insensitivity syndrome
-
Differences of sex development condition characterized by decreased or absent androgen signalling due to a genetic defect affecting the binding of testosterone or dihydrotestosterone to the androgen receptor.
- Bipotential gonads
-
Undifferentiated gonads that have the capacity to develop into either ovaries or testes.
- Chordee
-
A congenital condition that affects penile function and appearance, causing a curvature in the penis during erection.
- Cryptorchidism
-
Failure of testes to descend into the scrotum.
- Dysgerminoma
-
Malignant tumour arising from primordial germ cells in the ovary or dysgenetic gonad.
- Gender identity
-
A person’s basic subjective sense of self as a male, as a female or some other gendered identity, for example, an ‘intersex’ identity. Gender identity develops over time and is not directly a function of genes or prenatal sex hormone exposure.
- Gender role
-
Behaviours, attitudes and personality traits that a society, in a given culture and historical period, designates as masculine or feminine, and that are more ‘appropriate’ to or typical of the male or female social role.
- Gonadal dysgenesis
-
Differences of sex development condition characterized by the loss of primordial germ cells and abnormal gonadal development during embryonic development.
- Granulosa cells
-
Female-supporting cell lineage in the ovary.
- Klinefelter syndrome
-
A condition affecting only males that occurs when an extra X chromosome is present (47,XXY).
- Labioscrotal folds
-
A paired structure that is present during the indifferent stage of external genitalia development and is responsible for the formation of the labia majora in females and the scrotum in males.
- Leydig cells
-
Steroid-producing cells in the testis.
- Minifascicular peripheral neuropathy
-
A rare autosomal recessive inherited condition characterized by the presence of small fascicles in the peripheral nervous system.
- Non-seminoma
-
Malignant tumour derived from seminoma or its precursor lesion through reprogramming of the seminomatous cells into an embryonal carcinoma cell.
- Sertoli cells
-
Male-supporting cell lineage in the testis.
- Turner syndrome
-
A condition affecting only females that occurs when one of the X chromosomes (sex chromosomes) is missing or partially missing.
- Urethrovaginal fistula
-
A condition characterized by urine leakage from the urethra into the vagina due to an abnormal connection between the urethra and the vagina.
- Vaginal stenosis
-
An abnormal narrowing of the vaginal canal.
- Virilization
-
A condition in which females develop exaggerated male traits such as excessive facial hair.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmed, S.F., Armstrong, K., Cheng, E.Y. et al. Differences of sex development. Nat Rev Dis Primers 11, 54 (2025). https://doi.org/10.1038/s41572-025-00637-y
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41572-025-00637-y
This article is cited by
-
Eph-ephrin signaling and its potential role in female reproductive tract development
Molecular Biology Reports (2025)