Abstract
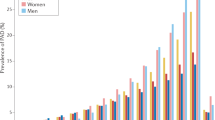
Peripheral artery disease (PAD) is characterized by blockage of the arteries that supply the lower extremities, often occurring as a result of atherosclerosis and thrombosis. PAD affects approximately 230 million people worldwide, with a growing prevalence owing to population ageing and concomitant cardiovascular risk factors, including smoking, diabetes mellitus, hypertension and dyslipidaemia. Patients with PAD have an increased risk of major cardiovascular and limb events, and substantially poorer walking performance compared with those without PAD. The screening and identification of PAD involves clinical and imaging assessments of disease extent and severity and stratification of individual risk to ensure appropriate management. Patients with PAD should be treated with guideline-directed medical therapy (GDMT), including antithrombotic, lipid-lowering, glucose-lowering and anti-hypertensive therapies, and exercise therapies that aim to improve function as well as cardiovascular and limb outcomes. For patients with compromised limb viability, such as acute and chronic limb-threatening ischaemia, or severe functional impairment that does not improve with exercise training, lower extremity revascularization is recommended. Given the complexity of PAD management, a multidisciplinary vascular team is required to achieve the best individualized treatment. Further research efforts should focus on reducing ischaemic events and health disparities and on optimizing the implementation of GDMT and exercise therapy, as well as improving the quality of life in patients with PAD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$119.00 per year
only $119.00 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hiatt, W. R., Armstrong, E. J., Larson, C. J. & Brass, E. P. Pathogenesis of the limb manifestations and exercise limitations in peripheral artery disease. Circ. Res. 116, 1527–1539 (2015).
Fowkes, F. G. R. et al. Peripheral artery disease: epidemiology and global perspectives. Nat. Rev. Cardiol. 14, 156–170 (2017). This paper describes the epidemiology of PAD.
Mazzolai, L. et al. 2024 ESC guidelines for the management of peripheral arterial and aortic diseases. Eur. Heart J. 45, 3538–3700 (2024). PAD guidelines from the ESC.
Gornik, H. L. et al. 2024 ACC/AHA/AACVPR/APMA/ABC/SCAI/SVM/SVN/SVS/SIR/VESS guideline for the management of lower extremity peripheral artery disease: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines. Circulation 149, e1313–e1410 (2024). PAD guidelines from the USA.
Criqui, M. H. et al. Mortality over a period of 10 years in patients with peripheral arterial disease. N. Engl. J. Med. 326, 381–386 (1992).
Criqui, M. H. & Aboyans, V. Epidemiology of peripheral artery disease. Circ. Res. 116, 1509–1526 (2015).
Criqui, M. H. et al. Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the American heart association. Circulation 144, e171–e191 (2021). Scientific statement by the AHA on PAD.
Bonaca, M. P. et al. Rivaroxaban in peripheral artery disease after revascularization. N. Engl. J. Med. 382, 1994–2004 (2020). VOYAGER-PAD study results including patients with PAD who underwent LER.
Farber, A. et al. Surgery or endovascular therapy for chronic limb-threatening ischemia. N. Engl. J. Med. 387, 2305–2316 (2022).
Allison, M. A. et al. Ethnic-specific prevalence of peripheral arterial disease in the United States. Am. J. Prev. Med. 32, 328–333 (2007).
Fowkes, F. G. R. et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet 382, 1329–1340 (2013).
Song, P. et al. Global, regional, and national prevalence and risk factors for peripheral artery disease in 2015: an updated systematic review and analysis. Lancet Glob. Health 7, e1020–e1030 (2019).
Adou, C. et al. Global epidemiology of lower extremity artery disease in the 21st century (2000-21): a systematic review and meta-analysis. Eur. J. Prev. Cardiol. 31, 803–811 (2024).
Polonsky, T. S. & McDermott, M. M. Lower extremity peripheral artery disease without chronic limb-threatening ischemia. JAMA 325, 2188 (2021).
Madabhushi, V. et al. Revascularization of intermittent claudicants leads to more chronic limb-threatening ischemia and higher amputation rates. J. Vasc. Surg. 74, 771–779 (2021).
Jarosinski, M. C. et al. Contemporary National incidence and outcomes of acute limb ischemia. Ann. Vasc. Surg. 110, 224–235 (2025).
Roth, G. A., Mensah, G. A. & Fuster, V. The global burden of cardiovascular diseases and risks. J. Am. Coll. Cardiol. 76, 2980–2981 (2020).
Kochar, A. et al. Cause of death among patients with peripheral artery disease. Circ. Cardiovasc. Qual. Outcomes 13, e006550 (2020).
Murray, C. J. L. et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the global burden of disease study 2019. Lancet 396, 1223–1249 (2020).
Allison, M. A. et al. Health disparities in peripheral artery disease: a scientific statement from the American heart association. Circulation 148, 286–296 (2023).
McDermott, M. M. et al. Racial differences in functional decline in peripheral artery disease and associations with socioeconomic status and education. J. Vasc. Surg. 66, 826–834 (2017).
Kim, E. S. H. et al. Sex differences in peripheral vascular disease: a scientific statement from the American heart association. Circulation 151, e877–e904 (2025). Scientific statement by the AHA on sex differences in PAD.
Aboyans, V. et al. Intrinsic contribution of gender and ethnicity to normal ankle-brachial index values: the multi-ethnic study of atherosclerosis (MESA). J. Vasc. Surg. 45, 319–327 (2007).
Thomas, V. E. & Beckman, J. A. Racial and socioeconomic health disparities in peripheral artery disease. J. Am. Heart Assoc. 13, e031446 (2024). This paper describes racial and socioeconomic health disparities in PAD.
Khaleghi, M., Isseh, I. N., Bailey, K. R. & Kullo, I. J. Family history as a risk factor for peripheral arterial disease. Am. J. Cardiol. 114, 928–932 (2014).
Klarin, D., Tsao, P. S. & Damrauer, S. M. Genetic determinants of peripheral artery disease. Circ. Res. 128, 1805–1817 (2021).
Joosten, M. M. et al. Associations between conventional cardiovascular risk factors and risk of peripheral artery disease in men. JAMA 308, 1660 (2012).
Chen, T. et al. Risk factors for peripheral artery disease and diabetic peripheral neuropathy among patients with type 2 diabetes. Diabetes Res. Clin. Pract. 207, 111079 (2024).
Fitridge, R. et al. The intersocietal IWGDF, ESVS, SVS guidelines on peripheral artery disease in people with diabetes mellitus and a foot ulcer. J. Vasc. Surg. 78, 1101–1131 (2023). Intersocietal guidelines on PAD and diabetes mellitus.
Wadström, B. N. et al. Elevated remnant and LDL cholesterol and the risk of peripheral artery disease. J. Am. Coll. Cardiol. 85, 1353–1368 (2025).
Wan, D. et al. Diet and nutrition in peripheral artery disease: a systematic review. Can. J. Cardiol. 38, 672–680 (2022).
Wan, D. et al. Dietary intake and cardiovascular outcomes in patients with chronic vascular disease: insights from the COMPASS trial cohort. Eur. J. Prev. Cardiol. 30, 709–718 (2023).
Narula, N. et al. Pathology of peripheral artery disease in patients with critical limb ischemia. J. Am. Coll. Cardiol. 72, 2152–2163 (2018). This paper discusses the pathophysiology of CLTI.
Torii, S. et al. Histopathologic characterization of peripheral arteries in subjects with abundant risk factors: correlating imaging with pathology. JACC Cardiovasc. Imaging 12, 1501–1513 (2019).
Narula, N., Olin, J. W. & Narula, N. Pathologic disparities between peripheral artery disease and coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 40, 1982–1989 (2020). This paper describes the differences in pathophysiology between CAD and PAD.
Caradu, C. et al. Restoring endothelial function by targeting desert Hedgehog downstream of Klf2 improves critical limb ischemia in adults. Circ. Res. 123, 1053–1065 (2018).
Vink, A., Schoneveld, A. H., Borst, C. & Pasterkamp, G. The contribution of plaque and arterial remodeling to de novo atherosclerotic luminal narrowing in the femoral artery. J. Vasc. Surg. 36, 1194–1197 (2002).
Hamburg, N. M. & Creager, M. A. Pathophysiology of intermittent claudication in peripheral artery disease. Circulation J. 81, 281–289 (2017).
Karayannacos, P. E., Talukder, N., Nerem, R. M., Roshon, S. & Vasko, J. S. The role of multiple noncritical arterial stenoses in the pathogenesis of ischemia. J. Thorac. Cardiovasc. Surg. 73, 458–469 (1977).
Vita, J. A. & Hamburg, N. M. Does endothelial dysfunction contribute to the clinical status of patients with peripheral arterial disease? Can. J. Cardiol. 26, 45A–50A (2010).
Alexander, Y. et al. Endothelial function in cardiovascular medicine: a consensus paper of the European Society of Cardiology working groups on atherosclerosis and vascular biology, aorta and peripheral vascular diseases, coronary pathophysiology and microcirculation, and thrombosis. Cardiovasc. Res. 117, 29–42 (2021).
Brevetti, G., Schiano, V. & Chiariello, M. Endothelial dysfunction: a key to the pathophysiology and natural history of peripheral arterial disease? Atherosclerosis 197, 1–11 (2008).
Tousoulis, D. et al. Endothelial dysfunction in conduit arteries and in microcirculation. Novel therapeutic approaches. Pharmacol. Ther. 144, 253–267 (2014).
Wang, M., Hao, H., Leeper, N. J. & Zhu, L. Thrombotic regulation from the endothelial cell perspectives. Arterioscler. Thromb. Vasc. Biol. 38, e90–e95 (2018).
Carmeliet, P. & Jain, R. K. Molecular mechanisms and clinical applications of angiogenesis. Nature 473, 298–307 (2011). This paper describes angiogenesis mechanisms.
Potente, M., Gerhardt, H. & Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 146, 873–887 (2011).
McDermott, M. M. et al. Effect of granulocyte-macrophage colony-stimulating factor with or without supervised exercise on walking performance in patients with peripheral artery disease. JAMA 318, 2089 (2017).
Deindl, E. & Schaper, W. The art of arteriogenesis. Cell Biochem. Biophys. 43, 1–15 (2005).
Krishnasamy, K. et al. Blood vessel control of macrophage maturation promotes arteriogenesis in ischemia. Nat. Commun. 8, 952 (2017).
McDermott, M. M. et al. Proximal superficial femoral artery occlusion, collateral vessels, and walking performance in peripheral artery disease. JACC Cardiovasc. Imaging 6, 687–694 (2013).
Kiani, S. et al. Peripheral artery disease is associated with severe impairment of vascular function. Vasc. Med. 18, 72–78 (2013).
Meneses, A. L. et al. Leg blood flow and skeletal muscle microvascular perfusion responses to submaximal exercise in peripheral arterial disease. Am. J. Physiol. Heart Circ. Physiol. 315, H1425–H1433 (2018).
Meredith, I. T. et al. Postischemic vasodilation in human forearm is dependent on endothelium-derived nitric oxide. Am. J. Physiol. 270, H1435–H1440 (1996).
Gokce, N. et al. Effect of exercise on upper and lower extremity endothelial function in patients with coronary artery disease. Am. J. Cardiol. 90, 124–127 (2002).
Foussard, N. et al. Praliciguat promotes ischemic leg reperfusion in leptin receptor-deficient mice. Circ. Res. 132, 34–48 (2023).
Tzoulaki, I. et al. Inflammatory, haemostatic, and rheological markers for incident peripheral arterial disease: Edinburgh artery study. Eur. Heart J. 28, 354–362 (2007). This paper discusses the inflammation component of PAD.
Kosmac, K. et al. Correlations of calf muscle macrophage content with muscle properties and walking performance in peripheral artery disease. J. Am. Heart Assoc. 9, e015929 (2020).
Payvandi, L. et al. Physical activity during daily life and brachial artery flow-mediated dilation in peripheral arterial disease. Vasc. Med. 14, 193–201 (2009).
Behm, C. Z. et al. Molecular imaging of endothelial vascular cell adhesion molecule-1 expression and inflammatory cell recruitment during vasculogenesis and ischemia-mediated arteriogenesis. Circulation 117, 2902–2911 (2008).
Signorelli, S. S. et al. Pathophysiology of chronic peripheral ischemia: new perspectives. Ther. Adv. Chronic Dis. 11, 204062231989446 (2020).
McDermott, M. M. et al. Skeletal muscle pathology in peripheral artery disease. Arterioscler. Thromb. Vasc. Biol. 40, 2577–2585 (2020).
Pradhan, A. D., Rifai, N. & Ridker, P. M. Soluble intercellular adhesion molecule-1, soluble vascular adhesion molecule-1, and the development of symptomatic peripheral arterial disease in men. Circulation 106, 820–825 (2002).
Pradhan, A. D. et al. Symptomatic peripheral arterial disease in women: nontraditional biomarkers of elevated risk. Circulation 117, 823–831 (2008).
McDermott, M. M. et al. Circulating blood markers and functional impairment in peripheral arterial disease. J. Am. Geriatr. Soc. 56, 1504–1510 (2008).
Habib, A., Petrucci, G. & Rocca, B. Pathophysiology of thrombosis in peripheral artery disease. Curr. Vasc. Pharmacol. 18, 204–214 (2020).
Aboyans, V. et al. Antithrombotic therapies in aortic and peripheral arterial diseases in 2021: a consensus document from the ESC working group on aorta and peripheral vascular diseases, the ESC working group on thrombosis, and the ESC working group on cardiovascular pharmacotherapy. Eur. Heart J. 42, 4013–4024 (2021).
Cooke, J. P. & Meng, S. Vascular regeneration in peripheral artery disease. Arterioscler. Thromb. Vasc. Biol. 40, 1627–1634 (2020).
White, S. H. et al. Walking performance is positively correlated to calf muscle fiber size in peripheral artery disease subjects, but fibers show aberrant mitophagy: an observational study. J. Transl. Med. 14, 284 (2016).
McDermott, M. M. Lower extremity manifestations of peripheral artery disease. Circ. Res. 116, 1540–1550 (2015).
McDermott, M. M. et al. Impairments of muscles and nerves associated with peripheral arterial disease and their relationship with lower extremity functioning: the InCHIANTI study. J. Am. Geriatr. Soc. 52, 405–410 (2004).
McDermott, M. M. et al. Lower extremity nerve function in patients with lower extremity ischemia. Arch. Intern. Med. 166, 1986–1992 (2006).
Garg, P. K. et al. Lower extremity nerve function, calf skeletal muscle characteristics, and functional performance in peripheral arterial disease. J. Am. Geriatr. Soc. 59, 1855–1863 (2011).
Roustit, M., Loader, J., Deusenbery, C., Baltzis, D. & Veves, A. Endothelial dysfunction as a link between cardiovascular risk factors and peripheral neuropathy in diabetes. J. Clin. Endocrinol. Metab. 101, 3401–3408 (2016).
Chapouly, C. et al. Impaired hedgehog signalling-induced endothelial dysfunction is sufficient to induce neuropathy: implication in diabetes. Cardiovasc. Res. 109, 217–227 (2016).
Renault, M.-A. et al. Desert hedgehog promotes ischemia-induced angiogenesis by ensuring peripheral nerve survival. Circ. Res. 112, 762–770 (2013).
Pasini, F. L. et al. Peripheral neuropathy associated with ischemic vascular disease of the lower limbs. Angiology 47, 569–577 (1996).
Lend, G. C. & Fowkes, F. G. R. The Edinburgh Claudication Questionnaire: an improved version of the WHO/Rose Questionnaire for use in epidemiological surveys. J. Clin. Epidemiol. 45, 1101–1109 (1992).
McGrae McDermott, M. et al. Leg symptoms in peripheral arterial disease. JAMA 286, 1599 (2001).
Aboyans, V. et al. 2017 ESC guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the European society for vascular surgery (ESVS). Eur. Heart J. 39, 763–816 (2018).
Coral- Rivera, N. S. & Tascón-Barona, A. H. Leriche syndrome: importance of diagnostic imaging for early detection and management. Radiol. Case Rep. 20, 2070–2074 (2025).
Munakomi, S., Foris, L. A. & Varacallo, M. A. Spinal Stenosis and Neurogenic Claudication (StatPearls, 2025).
Norgren, L. et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J. Vasc. Surg. 45, S5–S67 (2007). Inter-society consensus on PAD management.
Hess, C. N. et al. Acute limb ischemia in peripheral artery disease. Circulation 140, 556–565 (2019). This paper discusses ALI in PAD.
Abou-Zamzam, A. M. et al. A prospective analysis of critical limb ischemia: factors leading to major primary amputation versus revascularization. Ann. Vasc. Surg. 21, 458–463 (2007).
Herraiz-Adillo, Á, Cavero-Redondo, I., Álvarez-Bueno, C., Pozuelo-Carrascosa, D. P. & Solera-Martínez, M. The accuracy of toe brachial index and ankle brachial index in the diagnosis of lower limb peripheral arterial disease: a systematic review and meta-analysis. Atherosclerosis 315, 81–92 (2020).
Aboyans, V. et al. Measurement and interpretation of the ankle-brachial index. Circulation 126, 2890–2909 (2012). This article describes the measurement and interpretation of ABI.
Ichihashi, S. et al. Accuracy and reliability of the ankle brachial index measurement using a multicuff oscillometric device versus the doppler method. Eur. J. Vasc. Endovasc. Surg. 60, 462–468 (2020).
Tehan, P. E., Santos, D. & Chuter, V. H. A systematic review of the sensitivity and specificity of the toe-brachial index for detecting peripheral artery disease. Vasc. Med. 21, 382–389 (2016).
Conte, M. S. et al. Global vascular guidelines on the management of chronic limb-threatening ischemia. J. Vasc. Surg. 69, 3S–125S.e40 (2019).
McDermott, M. M. et al. The ankle brachial index is associated with leg function and physical activity: the walking and leg circulation study. Ann. Intern. Med. 136, 873 (2002).
Gardner, A. W., Montgomery, P. S. & Killewich, L. A. Natural history of physical function in older men with intermittent claudication. J. Vasc. Surg. 40, 73–78 (2004).
McDermott, M. M. et al. Six-minute walk is a better outcome measure than treadmill walking tests in therapeutic trials of patients with peripheral artery disease. Circulation 130, 61–68 (2014). The role of the 6-MWT in PAD.
McDermott, M. M. et al. Baseline functional performance predicts the rate of mobility loss in persons with peripheral arterial disease. J. Am. Coll. Cardiol. 50, 974–982 (2007).
McDermott, M. M. et al. Prognostic value of functional performance for mortality in patients with peripheral artery disease. J. Am. Coll. Cardiol. 51, 1482–1489 (2008).
McDermott, M. M. et al. Comparing 6-minute walk versus treadmill walking distance as outcomes in randomized trials of peripheral artery disease. J. Vasc. Surg. 71, 988–1001 (2020).
McDermott, M. M. et al. Effect of low-intensity vs high-intensity home-based walking exercise on walk distance in patients with peripheral artery disease. JAMA 325, 1266 (2021).
Woessner, M. et al. Beet the best? Circ. Res. 123, 654–659 (2018).
McDermott, M. M. et al. Cocoa to improve walking performance in older people with peripheral artery disease. Circ. Res. 126, 589–599 (2020).
Gardner, A. W., Montgomery, P. S. & Wang, M. Minimal clinically important differences in treadmill, 6-minute walk, and patient-based outcomes following supervised and home-based exercise in peripheral artery disease. Vasc. Med. 23, 349–357 (2018).
Bonaca, M. P. et al. Design and baseline characteristics of the STRIDE trial: evaluating semaglutide in people with symptomatic peripheral artery disease and type 2 diabetes. Eur. Heart J. Cardiovasc. Pharmacother. 10, 728–737 (2025).
Collins, R. et al. A systematic review of duplex ultrasound, magnetic resonance angiography and computed tomography angiography for the diagnosis and assessment of symptomatic, lower limb peripheral arterial disease. Health Technol. Assess. 11, 1–184 (2007).
Bolt, L. J. J. et al. Duplex-guided versus conventional percutaneous transluminal angioplasty of iliac TASC II A and B lesion: a randomized controlled trial. Ann. Vasc. Surg. 55, 138–147 (2019).
Met, R., Bipat, S., Legemate, D. A., Reekers, J. A. & Koelemay, M. J. W. Diagnostic performance of computed tomography angiography in peripheral arterial disease. JAMA 301, 415 (2009). The role of CTA in PAD.
Ramalho, J. et al. Gadolinium-based contrast agent accumulation and toxicity: an update. Am. J. Neuroradiol. 37, 1192–1198 (2016).
Menke, J. Meta-analysis: accuracy of contrast-enhanced magnetic resonance angiography for assessing steno-occlusions in peripheral arterial disease. Ann. Intern. Med. 153, 325 (2010).
Rose, G. A. & Blackburn, H. Cardiovascular Survey Methods WHO Monograph Series No. 56 (WHO, 1968).
Criqui, M. H. et al. The correlation between symptoms and non-invasive test results in patients referred for peripheral arterial disease testing. Vasc. Med. 1, 65–71 (1996).
US Preventive Services Task Force. Screening for peripheral artery disease and cardiovascular disease risk assessment with the ankle-brachial index: US Preventive Services Task Force recommendation statement. JAMA 320, 177–183 (2018).
Pande, R. L., Perlstein, T. S., Beckman, J. A. & Creager, M. A. Secondary prevention and mortality in peripheral artery disease: National health and nutrition examination study, 1999 to 2004. Circulation 124, 17–23 (2011).
Lindholt, J. S. & Søgaard, R. Population screening and intervention for vascular disease in Danish men (VIVA): a randomised controlled trial. Lancet 390, 2256–2265 (2017).
Lindholt, J. S. et al. Five-year outcomes of the Danish cardiovascular screening (DANCAVAS) trial. N. Engl. J. Med. 387, 1385–1394 (2022).
Canonico, M. E., Hess, C. N., Rogers, R. K. & Bonaca, M. P. Medical therapy for peripheral artery disease. Curr. Cardiol. Rep. 26, 651–659 (2024).
Abramson, B. L. et al. Canadian Cardiovascular Society 2022 guidelines for peripheral arterial disease. Can. J. Cardiol. 38, 560–587 (2022). PAD guidelines from the CCS.
Morrison, J. T. et al. Low-dose rivaroxaban plus aspirin in patients with peripheral artery disease undergoing lower extremity revascularization with and without concomitant coronary artery disease: insights from VOYAGER PAD. Circulation 149, 1536–1539 (2024).
Hsia, J. et al. Rivaroxaban in patients with recent peripheral artery revascularization and renal impairment. J. Am. Coll. Cardiol. 78, 757–759 (2021).
Canonico, M. E. et al. Low-dose rivaroxaban plus aspirin in fragile patients after lower extremity revascularization. J. Am. Coll. Cardiol. 84, 801–811 (2024).
Bonaca, M. P. et al. Polyvascular disease, type 2 diabetes, and long-term vascular risk: a secondary analysis of the IMPROVE-IT trial. Lancet Diabetes Endocrinol. 6, 934–943 (2018).
Bonaca, M. P. et al. Low-density lipoprotein cholesterol lowering with evolocumab and outcomes in patients with peripheral artery disease. Circulation 137, 338–350 (2018). A study exploring the role of evolocumab in patients with PAD.
Jukema, J. W. et al. Alirocumab in patients with polyvascular disease and recent acute coronary syndrome. J. Am. Coll. Cardiol. 74, 1167–1176 (2019). A study exploring role of alirocumab in patients with PAD.
Schwartz, G. G. et al. Peripheral artery disease and venous thromboembolic events after acute coronary syndrome. Circulation 141, 1608–1617 (2020).
Nissen, S. E. et al. Bempedoic acid and cardiovascular outcomes in statin-intolerant patients. N. Engl. J. Med. 388, 1353–1364 (2023).
Dhatariya, K. et al. The impact of liraglutide on diabetes-related foot ulceration and associated complications in patients with type 2 diabetes at high risk for cardiovascular events: results from the LEADER trial. Diabetes Care 41, 2229–2235 (2018).
Verma, S. et al. Cardiovascular efficacy of liraglutide and semaglutide in individuals with diabetes and peripheral artery disease. Diabetes Obes. Metab. 24, 1288–1299 (2022).
Bonaca, M. P. et al. Semaglutide and walking capacity in people with symptomatic peripheral artery disease and type 2 diabetes (STRIDE): a phase 3b, double-blind, randomised, placebo-controlled trial. Lancet 405, 1580–1593 (2025). STRIDE trial results including patients with PAD and T2DM.
McGuire, D. K. et al. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. N. Engl. J. Med. 392, 2001–2012 (2025).
Verma, S. et al. Cardiovascular outcomes and safety of empagliflozin in patients with type 2 diabetes mellitus and peripheral artery disease. Circulation 137, 405–407 (2018).
Barraclough, J. Y. et al. Cardiovascular and renal outcomes with canagliflozin in patients with peripheral arterial disease: data from the CANVAS Program and CREDENCE trial. Diabetes Obes. Metab. 24, 1072–1083 (2022).
Bonaca, M. P. et al. Dapagliflozin and cardiac, kidney, and limb outcomes in patients with and without peripheral artery disease in DECLARE-TIMI 58. Circulation 142, 734–747 (2020).
Östergren, J. Impact of ramipril in patients with evidence of clinical or subclinical peripheral arterial disease. Eur. Heart J. 25, 17–24 (2004).
The ONTARGET Investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N. Engl. J. Med. 358, 1547–1559 (2008).
Mazzolai, L. et al. Exercise therapy for chronic symptomatic peripheral artery disease. Eur. Heart J. 45, 1303–1321 (2024). Consensus document by ESC on exercise therapy.
Ambelu, T. & Teferi, G. The impact of exercise modalities on blood glucose, blood pressure and body composition in patients with type 2 diabetes mellitus. BMC Sports Sci. Med. Rehabil. 15, 153 (2023).
McDermott, M. M. et al. Home-based walking exercise intervention in peripheral artery disease: a randomized clinical trial. JAMA 310, 57–65 (2013).
Gardner, A. W., Parker, D. E., Montgomery, P. S., Scott, K. J. & Blevins, S. M. Efficacy of quantified home-based exercise and supervised exercise in patients with intermittent claudication: a randomized controlled trial. Circulation 123, 491–498 (2011).
King, R. W., Canonico, M. E., Bonaca, M. P. & Hess, C. N. Management of peripheral arterial disease: lifestyle modifications and medical therapies. J. Soc. Cardiovasc. Angiogr. Interv. 1, 100513 (2022).
Murphy, T. P. et al. Supervised exercise versus primary stenting for claudication resulting from aortoiliac peripheral artery disease. Circulation 125, 130–139 (2012).
Fakhry, F. et al. Endovascular revascularization and supervised exercise for peripheral artery disease and intermittent claudication. JAMA 314, 1936 (2015).
Cetlin, M. D. et al. Barriers to participation in supervised exercise therapy reported by people with peripheral artery disease. J. Vasc. Surg. 77, 506–514 (2023).
Magalhães, S. et al. Effect of arm-ergometry versus treadmill supervised exercise on cardiorespiratory fitness and walking distances in patients with peripheral artery disease: the ARMEX randomized clinical trial. J. Cardiopulm. Rehabil. Prev. 44, 353–360 (2024).
Jansen, S. C. et al. Modes of exercise training for intermittent claudication. Cochrane Database Syst. Rev. 8, CD009638 (2020).
Klein, A. J. et al. SCAI expert consensus statement for aorto-iliac arterial intervention appropriate use. Catheter. Cardiovasc. Interv. 84, 520–528 (2014).
Beckman, J. A., Schneider, P. A. & Conte, M. S. Advances in revascularization for peripheral artery disease: revascularization in PAD. Circ. Res. 128, 1885–1912 (2021).
Li, J. et al. Below-the-Knee endovascular revascularization. JACC Cardiovasc. Interv. 17, 589–607 (2024).
Banerjee, S. et al. A percutaneous crossing algorithm for femoropopliteal and tibial artery chronic total occlusions (PCTO algorithm). J. Invasive Cardiol. 31, 111–119 (2019).
Tsai, S. et al. Comparative outcomes of interventions for femoropopliteal chronic total occlusion versus non–chronic total occlusion lesions from the multicenter XLPAD registry. J. Am. Heart Assoc. 12, e028425 (2023).
Almasri, J. et al. A systematic review and meta-analysis of revascularization outcomes of infrainguinal chronic limb-threatening ischemia. Eur. J. Vasc. Endovasc. Surg. 58, S110–S119 (2019).
Shishehbor, M. H. & Jaff, M. R. Percutaneous therapies for peripheral artery disease. Circulation 134, 2008–2027 (2016).
Shah, P. S. et al. Full metal jacket stenting of the superficial femoral artery: a retrospective review. Ann. Vasc. Surg. 25, 127–131 (2011).
Bausback, Y. et al. Drug-eluting stent versus drug-coated balloon revascularization in patients with femoropopliteal arterial disease. J. Am. Coll. Cardiol. 73, 667–679 (2019).
Schneider, P. A., Varcoe, R. L., Secemsky, E., Schermerhorn, M. & Holden, A. Update on paclitaxel for femoral-popliteal occlusive disease in the 15 months following a summary level meta-analysis demonstrated increased risk of late mortality and dose response to paclitaxel. J. Vasc. Surg. 73, 311–322 (2021).
Byrne, R. A., Joner, M., Alfonso, F. & Kastrati, A. Drug-coated balloon therapy in coronary and peripheral artery disease. Nat. Rev. Cardiol. 11, 13–23 (2014).
Komshian, S., Lu, K., Pike, S. & Siracuse, J. Infrainguinal open reconstruction: a review of surgical considerations and expected outcomes. Vasc. Health Risk Manag. 13, 161–168 (2017).
Bradbury, A. W. et al. A vein bypass first versus a best endovascular treatment first revascularisation strategy for patients with chronic limb threatening ischaemia who required an infra-popliteal, with or without an additional more proximal infra-inguinal revascularisation procedure to restore limb perfusion (BASIL-2): an open-label, randomised, multicentre, phase 3 trial. Lancet 401, 1798–1809 (2023).
Pereira, C. E., Albers, M., Romiti, M., Brochado-Neto, F. C. & Pereira, C. A. B. Meta-analysis of femoropopliteal bypass grafts for lower extremity arterial insufficiency. J. Vasc. Surg. 44, 510–517 (2006).
Lyden, S. P. et al. DETOUR2 trial outcomes demonstrate clinical utility of percutaneous transmural bypass for the treatment of long segment, complex femoropopliteal disease. J. Vasc. Surg. 79, 1420–1427.e2 (2024).
Shishehbor, M. H. et al. Transcatheter arterialization of deep veins in chronic limb-threatening ischemia. N. Engl. J. Med. 388, 1171–1180 (2023).
Changal, K. et al. Drug-eluting stents versus conventional endovascular therapies in symptomatic infrapopliteal peripheral artery disease: a meta-analysis. J. Soc. Cardiovasc. Angiogr. Interv. 1, 100024 (2022).
Chandra, V. et al. Thirty-day outcomes from the Disrupt PAD BTK II study of the shockwave intravascular lithotripsy system for treatment of calcified below-the-knee peripheral arterial disease. J. Vasc. Surg. 81, 710–719.e2 (2025).
Duff, S., Mafilios, M. S., Bhounsule, P. & Hasegawa, J. T. The burden of critical limb ischemia: a review of recent literature. Vasc. Health Risk Manag. 15, 187–208 (2019).
Aber, A. et al. Themes that determine quality of life in patients with peripheral arterial disease: a systematic review. Patient 11, 489–502 (2018).
Diehm, C. et al. Mortality and vascular morbidity in older adults with asymptomatic versus symptomatic peripheral artery disease. Circulation 120, 2053–2061 (2009).
Joyce, C. R. B., Hickey, A., McGee, H. M. & O’Boyle, C. A. A theory-based method for the evaluation of individual quality of life: the SEIQoL. Qual. Life Res. 12, 275–280 (2003).
Treat-Jacobson, D. et al. The PADQOL: development and validation of a PAD-specific quality of life questionnaire. Vasc. Med. 17, 405–415 (2012).
Hageman, D. et al. Vascular quality of life questionnaire-6 before and after supervised exercise therapy in patients with intermittent claudication. Eur. J. Vasc. Endovasc. Surg. 63, 457–463 (2022).
Steunenberg, S. L., Raats, J. W., te Slaa, A., de Vries, J. & van der Laan, L. Quality of life in patients suffering from critical limb ischemia. Ann. Vasc. Surg. 36, 310–319 (2016).
Donker, J. et al. Review: quality of life in lower limb peripheral vascular surgery. Vascular 24, 88–95 (2016).
Breek, J. C., Hamming, J. F., De Vries, J., Aquarius, A. E. A. M. & van Berge Henegouwen, D. P. Quality of life in patients with intermittent claudication using the world health organisation (WHO) questionnaire. Eur. J. Vasc. Endovasc. Surg. 21, 118–122 (2001).
McDermott, M. M. et al. Asymptomatic peripheral arterial disease is associated with more adverse lower extremity characteristics than intermittent claudication. Circulation 117, 2484–2491 (2008).
Campia, U., Gerhard-Herman, M., Piazza, G. & Goldhaber, S. Z. Peripheral artery disease: past, present, and future. Am. J. Med. 132, 1133–1141 (2019).
Calanca, L., Lanzi, S., Ney, B., Berchtold, A. & Mazzolai, L. Multimodal supervised exercise significantly improves walking performances without changing hemodynamic parameters in patients with symptomatic lower extremity peripheral artery disease. Vasc. Endovasc. Surg. 54, 605–611 (2020).
Smolderen, K. G. et al. Health status and cognitive function for risk stratification in chronic coronary and peripheral artery disease. Eur. J. Prev. Cardiol. 30, 535–545 (2023).
Thomas, P. E., Vedel-Krogh, S., Nielsen, S. F., Nordestgaard, B. G. & Kamstrup, P. R. Lipoprotein(a) and risks of peripheral artery disease, abdominal aortic aneurysm, and major adverse limb events. J. Am. Coll. Cardiol. 82, 2265–2276 (2023). This paper discusses the role of lipoprotein(a) in PAD.
Brevetti, G., Giugliano, G., Brevetti, L. & Hiatt, W. R. Inflammation in peripheral artery disease. Circulation 122, 1862–1875 (2010).
Russell, K. S. et al. A randomized, placebo-controlled trial of canakinumab in patients with peripheral artery disease. Vasc. Med. 24, 414–421 (2019).
Khachigian, L. M., Varcoe, R. L., Suoranta, T., Laham-Karam, N. & Ylä-Herttuala, S. Gene therapeutic strategies for peripheral artery disease and new opportunities provided by adeno-associated virus vectors. Arterioscler. Thromb. Vasc. Biol. 43, 836–851 (2023).
Creager, M. A. et al. Effect of hypoxia-inducible factor-1α gene therapy on walking performance in patients with intermittent claudication. Circulation 124, 1765–1773 (2011).
Canonico, M. E., Hsia, J., Hess, C. N. & Bonaca, M. P. Sex differences in guideline-directed medical therapy in 2021–22 among patients with peripheral artery disease. Vasc. Med. 28, 233–235 (2023).
Aboyans, V. & Chastaingt, L. What LEADs to the under-treatment of patients with lower-extremity artery disease? Eur. J. Prev. Cardiol. 30, 1090–1091 (2023).
Hess, C. N. et al. Randomized trial of a vascular care team vs education for patients with peripheral artery disease. J. Am. Coll. Cardiol. 83, 2658–2670 (2024). An implementation trial of lipid management in patients with PAD.
Saxon, J. T. et al. Adherence to guideline-recommended therapy—including supervised exercise therapy referral—across peripheral artery disease specialty clinics: insights from the international PORTRAIT registry. J. Am. Heart Assoc. 9, e012541 (2020).
Bashir, A. Z. Exploring telehealth interventions to monitor rehabilitation in patients with peripheral artery disease. SAGE Open Med. 11, 205031212311755 (2023).
Tchankoni, M. K. et al. Epidemiology of non-traumatic lower extremities amputations in West Africa: nationwide data from Togo. Eur. J. Vasc. Endovasc. Surg. 67, 959–968 (2024).
Fontaine, R., Kim, M. & Kieny, R. Surgical treatment of peripheral circulation disorders [German]. Helv. Chir. Acta 21, 499–533 (1954).
Author information
Authors and Affiliations
Contributions
Introduction (V.A., M.E.C., L.C. and M.P.B.); Epidemiology (V.A., M.E.C., L.C. and M.P.B.); Mechanisms/pathophysiology (V.A., M.E.C., S.S.A. and M.P.B.); Diagnosis, screening and prevention (V.A., M.E.C., S.S.A., M.B., T.C. and M.P.B.); Management (V.A., M.E.C., M.B., T.C., M.H.C. and M.P.B.); Quality of life (V.A., M.E.C., M.H.C., E.S.D., L.M., M.M.M. and M.P.B.); Outlook (V.A., M.E.C., E.S.D., L.M., M.M.M. and M.P.B.); overview of the Primer (V.A.).
Corresponding author
Ethics declarations
Competing interests
V.A. has received, over the past 3 years, honoraria from: Amarin, Amgen, AstraZeneca, Bayer, Boehringer-Ingelheim and Novo-Nordisk. M.E.C. and M.P.B. receive salary support from CPC, a non-profit academic research organization affiliated with the University of Colorado that receives or has received research grant and/or consulting funding between August 2021 and the present from: Abbott Laboratories, Agios Pharmaceuticals, Inc., Alexion Pharma, Alnylam Pharmaceuticals, Inc., Amgen, Inc., Angionetics, Inc., Anthos Therapeutics, Array BioPharma, Inc., AstraZeneca and affiliates, Atentiv LLC, Audentes Therapeutics, Inc., Bayer and affiliates, Bristol-Meyers Squibb, Cambrian Biopharma, Inc., Cardiol Therapeutics, Inc., CellResearch Corp., Cleerly, Inc., Cook Regentec LLC, CSL Behring LLC, Eidos Therapeutics, Inc., EP Trading Co. Ltd, Epizon Pharma, Inc., Esperion Therapeutics, Inc., Everly Well, Inc., Exicon Consulting Pvt. Ltd, Faraday Pharmaceuticals, Inc., Foresee Pharmaceuticals Co. Ltd, Fortress Biotech, Inc., HDL Therapeutics, Inc., HeartFlow, Inc., Hummingbird Bioscience, Insmed, Inc., Ionis Pharmaceuticals, Janssen and affiliates, Kowa Research Institute, Inc., Lexicon Pharmaceuticals, Inc., MedImmune Ltd, Merck & affiliates, Nectero Medical, Inc., Novartis Pharmaceuticals Corp., Novo-Nordisk, Inc., Osiris Therapeutics, Inc., Pfizer, Inc., PhaseBio Pharmaceuticals, Inc., Prairie Education and Research Cooperative, Prothena Biosciences Ltd, Regeneron Pharmaceuticals, Inc., Regio Biosciences, Inc., Sanofi–Aventis Group, Silence Therapeutics PLC, Smith & Nephew plc, Stealth BioTherapeutics, Inc., Tourmaline Bio, Inc., VarmX, and Virta Health Corporation. M.M.M. receives research support from Mars and ACI Medical. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks Mehdi Shishehbor, Nelson Wolosker, Raphael Ritti-Dias and Subhash Banerjee for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aboyans, V., Canonico, M.E., Chastaingt, L. et al. Peripheral artery disease. Nat Rev Dis Primers 11, 68 (2025). https://doi.org/10.1038/s41572-025-00651-0
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41572-025-00651-0