Abstract
Schizophrenia is a challenging and diverse mental health condition with a lifetime prevalence of 0.4%. Schizophrenia usually manifests in late adolescence or early adulthood and is associated with high disability and a reduced life expectancy. Risk factors include genetic predisposition, prenatal and birth complications, infections and immune dysfunction, and cannabis use as well as psychosocial factors such as childhood trauma or migration. The first psychotic episode is often preceded by a long prodromal phase that can last for several years. No markers are yet available for clinical use that allow prediction of disease development or a diagnosis to be established. A leading theory postulates that excitatory–inhibitory (that is, glutamate–GABA) imbalance in the cortex ultimately leads to dysfunction of the dopaminergic system. Schizophrenia is a heterogeneous disease with different manifestations, including psychotic symptoms as well as negative symptoms and global cognitive deficit, that do not respond to antipsychotic drugs, making management very difficult. Pharmacological treatment coupled with psychotherapeutic interventions, such as cognitive behavioural therapy, cognitive remediation and psychoeducation, remains the mainstay of treatment; however, treatment resistance is frequent. The first medication that targets neurotransmitter systems other than dopamine has been approved for use. Current attempts to use virtual reality and avatars to improve psychotic symptoms and smartphone applications to prevent relapses seem promising.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$119.00 per year
only $119.00 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Solmi, M. et al. Incidence, prevalence, and global burden of schizophrenia — data, with critical appraisal, from the global burden of disease (GBD) 2019. Mol. Psychiatry https://doi.org/10.1038/s41380-023-02138-4 (2023).
McCutcheon, R. A., Keefe, R. S. E. & McGuire, P. K. Cognitive impairment in schizophreniaaetiology, pathophysiology, and treatment. Mol. Psychiatry 28, 1902–1918 (2023).
Sullivan, P. F., Kendler, K. S. & Neale, M. C. Schizophrenia as a complex traitevidence from a meta-analysis of twin studies. Arch. Gen. Psychiatry 60, 1187–1192 (2003).
Gottesman, I. I. & Shields, J. A polygenic theory of schizophrenia. Proc. Natl Acad. Sci. USA 58, 199–205 (1967).
Davies, C. et al. Prenatal and perinatal risk and protective factors for psychosis: a systematic review and meta-analysis. Lancet Psychiatry 7, 399–410 (2020).
D’Souza, D. C. et al. Consensus paper of the WFSBP task force on cannabis, cannabinoids and psychosis. World J. Biol. Psychiatry 23, 719–742 (2022).
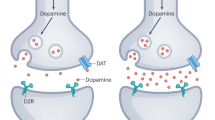
Howes, O. D. & Onwordi, E. C. The synaptic hypothesis of schizophrenia version IIIa master mechanism. Mol. Psychiatry 28, 1843–1856 (2023).
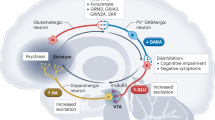
Howes, O. D. & Shatalina, E. Integrating the neurodevelopmental and dopamine hypotheses of schizophrenia and the role of cortical excitation-inhibition balance. Biol. Psychiatry 92, 501–513 (2022). An important theory of how diverse factors combine to cause schizophrenia.
Schultze-Lutter, F., Ruhrmann, S., Berning, J., Maier, W. & Klosterkotter, J. Basic symptoms and ultrahigh risk criteriasymptom development in the initial prodromal state. Schizophr. Bull. 36, 182–191 (2010).
Haijma, S. V. et al. Brain volumes in schizophrenia: a meta-analysis in over 18 000 subjects. Schizophr. Bull. 39, 1129–1138 (2013).
American Psychiatric Association. The American Psychiatric Association Practice Guideline for the Treatment of Patients with Schizophrenia 3rd edn (American Psychiatric Association Publishing, 2020).
Hansen, H. G. et al. Clinical recovery among individuals with a first-episode schizophrenia: an updated systematic review and meta-analysis. Schizophr. Bull. 49, 297–308 (2023).
Holm, M., Taipale, H., Tanskanen, A., Tiihonen, J. & Mitterdorfer-Rutz, E. Employment among people with schizophrenia or bipolar disorder: a population-based study using nationwide registers. Acta Psychiatr. Scand. 143, 61–71 (2021).
Marwaha, S. & Johnson, S. Schizophrenia and employment — a review. Soc. Psychiatry Psychiatr. Epidemiol. 39, 337–349 (2004).
Chan, J. K. N. et al. Life expectancy and years of potential life lost in people with mental disorders: a systematic review and meta-analysis. EClinicalMedicine 65, 102294 (2023).
Feber, L. et al. Antipsychotic drugs and cognitive function: a systematic review and network meta-analysis. JAMA Psychiatry 82, 47–56 (2025).
McGrath, J. et al. A systematic review of the incidence of schizophrenia: the distribution of rates and the influence of sex, urbanicity, migrant status and methodology. BMC Med. 2, 13 (2004).
Jongsma, H. E., Turner, C., Kirkbride, J. B. & Jones, P. B. International incidence of psychotic disorders, 2002-17: a systematic review and meta-analysis. Lancet Public Health 4, e229–e244 (2019).
Hogerzeil, S. J., Hoek, H. W. & van Hemert, A. M. The impact of study design on schizophrenia incidence estimates: a systematic review of Northern European studies 2008-2019. Schizophr. Res. 231, 134–141 (2021).
Saha, S., Chant, D. C., Welham, J. L. & McGrath, J. J. The incidence and prevalence of schizophrenia varies with latitude. Acta Psychiatr. Scand. 114, 36–39 (2006).
Solmi, M. et al. Age at onset of mental disorders worldwide: large-scale meta-analysis of 192 epidemiological studies. Mol. Psychiatry 27, 281–295 (2022).
Pedersen, C. B. et al. A comprehensive nationwide study of the incidence rate and lifetime risk for treated mental disorders. JAMA Psychiatry 71, 573–581 (2014).
Culbert, K. M., Thakkar, K. N. & Klump, K. L. Risk for midlife psychosis in women: critical gaps and opportunities in exploring perimenopause and ovarian hormones as mechanisms of risk. Psychol. Med. 52, 1612–1620 (2022).
McGrath, J., Saha, S., Chant, D. & Welham, J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol. Rev. 30, 67–76 (2008). A concise overview of incidence, prevalence and mortality in schizophrenia.
Aleman, A., Kahn, R. S. & Selten, J. P. Sex differences in the risk of schizophreniaevidence from meta-analysis. Arch. Gen. Psychiatry 60, 565–571 (2003).
Lewis, S. W. & Murray, R. M. Obstetric complications, neurodevelopmental deviance, and risk of schizophrenia. J. Psychiatr. Res. 21, 413–421 (1987).
Weinberger, D. R. Implications of normal brain development for the pathogenesis of schizophrenia. Arch. Gen. Psychiatry 44, 660–669 (1987).
McGrath, J. J. et al. Age of onset and cumulative risk of mental disorders: a cross-national analysis of population surveys from 29 countries. Lancet Psychiatry 10, 668–681 (2023).
Correll, C. U. et al. Mortality in people with schizophrenia: a systematic review and meta-analysis of relative risk and aggravating or attenuating factors. World Psychiatry 21, 248–271 (2022).
Hjorthoj, C., Sturup, A. E., McGrath, J. J. & Nordentoft, M. Years of potential life lost and life expectancy in schizophrenia: a systematic review and meta-analysis. Lancet Psychiatry 4, 295–301 (2017).
Saha, S., Chant, D. & McGrath, J. A systematic review of mortality in schizophrenia — is the differential mortality gap worsening over time? Arch. Gen. Psychiatry 64, 1123–1131 (2007).
Walker, E. R., McGee, R. E. & Druss, B. G. Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis. JAMA Psychiatry 72, 334–341 (2015).
Karlsson, H. & Dalman, C. Epidemiological studies of prenatal and childhood infection and schizophrenia. Curr. Top. Behav. Neurosci. 44, 35–47 (2020).
Laursen, T. M. et al. Cause-specific life years lost among persons diagnosed with schizophrenia: is it getting better or worse? Schizophr. Res. 206, 284–290 (2019).
Ali, S., Santomauro, D., Ferrari, A. J. & Charlson, F. Excess mortality in severe mental disorders: a systematic review and meta-regression. J. Psychiatr. Res. 149, 97–105 (2022).
Olfson, M., Gerhard, T., Huang, C., Crystal, S. & Stroup, T. S. Premature mortality among adults with schizophrenia in the United States. JAMA Psychiatry 72, 1172–1181 (2015).
Plana-Ripoll, O. et al. Exploring comorbidity within mental disorders among a Danish national population. JAMA Psychiatry 76, 259–270 (2019).
Leucht, S., Burkard, T., Henderson, J., Maj, M. & Sartorius, N. Physical illness and schizophrenia: a review of the literature. Acta Psychiatr. Scand. 116, 317–333 (2007). An overview of the multiple concomitant physical illnesses associated with schizophrenia.
Momen, N. C. et al. Mortality associated with mental disorders and comorbid general medical conditions. JAMA Psychiatry 79, 444–453 (2022).
Momen, N. C. et al. Association between mental disorders and subsequent medical conditions. N. Engl. J. Med. 382, 1721–1731 (2020).
Eaton, W. W. et al. Association of schizophrenia and autoimmune diseases: linkage of Danish national registers. Am. J. Psychiatry 163, 521–528 (2006).
Jester, D. J. et al. Review of major social determinants of health in schizophrenia-spectrum psychotic disorders: I. Clinical outcomes. Schizophr. Bull. 49, 837–850 (2023).
Beary, M., Hodgson, R. & Wildgust, H. J. A critical review of major mortality risk factors for all-cause mortality in first-episode schizophrenia: clinical and research implications. J. Psychopharmacol. 26, 52–61 (2012).
Veeneman, R. R. et al. Exploring the relationship between schizophrenia and cardiovascular disease: a genetic correlation and multivariable Mendelian randomization study. Schizophr. Bull. 48, 463–473 (2022).
Murray, R. M. & Lewis, S. W. Is schizophrenia a neurodevelopmental disorder? Br. Med J. 295, 681–682 (1987).
Cannon, M., Jones, P. B. & Murray, R. M. Obstetric complications and schizophrenia: historical and meta-analytic review. Am. J. Psychiatry 159, 1080–1092 (2002).
Cheslack-Postava, K. & Brown, A. S. Prenatal infection and schizophrenia: a decade of further progress. Schizophr. Res. 247, 7–15 (2022).
McGrath, J. J. et al. Neonatal vitamin D status and risk of schizophrenia: a population-based case-control study. Arch. Gen. Psychiatry 67, 889–894 (2010).
Eyles, D. W. et al. The association between neonatal vitamin D status and risk of schizophrenia. Sci. Rep. 8, 17692 (2018).
Brown, A. S. & Susser, E. S. Prenatal nutritional deficiency and risk of adult schizophrenia. Schizophr. Bull. 34, 1054–1063 (2008).
Freedman, R. et al. Choline, folic acid, vitamin D, and fetal brain development in the psychosis spectrum. Schizophr. Res. 247, 16–25 (2022).
Maxwell, A. M. & Rao, R. B. Perinatal iron deficiency as an early risk factor for schizophrenia. Nutr. Neurosci. 25, 2218–2227 (2022).
Albinana, C. et al. Developmental exposure to vitamin D deficiency and subsequent risk of schizophrenia. Schizophr. Res. 247, 26–32 (2022).
Horsdal, H. T. et al. Convergent evidence linking neonatal vitamin D status and risk of neurodevelopmental disorders: a Danish case-cohort study. Lancet Psychiatry 12, 410–420 (2025).
Ursini, G. et al. Convergence of placenta biology and genetic risk for schizophrenia. Nat. Med. 24, 792–801 (2018).
Ursini, G. et al. Prioritization of potential causative genes for schizophrenia in placenta. Nat. Commun. 14, 2613 (2023).
Hsu, C. W. et al. Month of birth and mental disorders: a population-based study and validation using global meta-analysis. Acta Psychiatr. Scand. 144, 153–167 (2021).
Rosenfield, P. J., Jiang, D. & Pauselli, L. Childhood adversity and psychotic disordersEpidemiological evidence, theoretical models and clinical considerations. Schizophr. Res. 247, 55–66 (2022).
Hailes, H. P., Yu, R., Danese, A. & Fazel, S. Long-term outcomes of childhood sexual abuse: an umbrella review. Lancet Psychiatry 6, 830–839 (2019).
Birnbaum, R. & Weinberger, D. R. The genesis of schizophrenia: an origin story. Am. J. Psychiatry 181, 482–492 (2024).
Gage, S. H., Hickman, M. & Zammit, S. Association between cannabis and psychosis: epidemiologic evidence. Biol. Psychiatry 79, 549–556 (2016).
Di Forti, M. et al. The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): a multicentre case-control study. Lancet Psychiatry 6, 427–436 (2019).
Cantor-Graae, E. & Selten, J. P. Schizophrenia and migration: a meta-analysis and review. Am. J. Psychiatry 162, 12–24 (2005).
Dykxhoorn, J. et al. Risk of schizophrenia, schizoaffective, and bipolar disorders by migrant status, region of origin, and age-at-migration: a national cohort study of 1.8 million people. Psychol. Med. 49, 2354–2363 (2019).
Selten, J. P., van der Ven, E. & Termorshuizen, F. Migration and psychosis: a meta-analysis of incidence studies. Psychol. Med. 50, 303–313 (2020).
van der Ven, E. & Selten, J. P. Migrant and ethnic minority status as risk indicators for schizophrenia: new findings. Curr. Opin. Psychiatry 31, 231–236 (2018).
Malaspina, D. et al. Advancing paternal age and the risk of schizophrenia. Arch. Gen. Psychiatry 58, 361–367 (2001).
Khachadourian, V., Zaks, N., Lin, E., Reichenberg, A. & Janecka, M. Advanced paternal age and risk of schizophrenia in offspring — review of epidemiological findings and potential mechanisms. Schizophr. Res. 233, 72–79 (2021).
Goriely, A., McGrath, J. J., Hultman, C. M., Wilkie, A. O. & Malaspina, D. “Selfish spermatogonial selection”: a novel mechanism for the association between advanced paternal age and neurodevelopmental disorders. Am. J. Psychiatry 170, 599–608 (2013).
Janecka, M. et al. Advanced paternal age effects in neurodevelopmental disorders — review of potential underlying mechanisms. Transl. Psychiatry 7, e1019 (2017).
Mortensen, P. B. et al. Effects of family history and place and season of birth on the risk of schizophrenia. N. Engl. J. Med. 340, 603–608 (1999).
Pedersen, C. B. & Mortensen, P. B. Evidence of a dose-response relationship between urbanicity during upbringing and schizophrenia risk. Arch. Gen. Psychiatry 58, 1039–1046 (2001).
Paksarian, D. et al. The role of genetic liability in the association of urbanicity at birth and during upbringing with schizophrenia in Denmark. Psychological Med. 48, 305–314 (2018).
Fan, C. C. et al. Spatial fine-mapping for gene-by-environment effects identifies risk hot spots for schizophrenia. Nat. Commun. 9, 5296 (2018).
Colodro-Conde, L. et al. Association between population density and genetic risk for schizophrenia. JAMA Psychiatry 75, 901–910 (2018).
Plana-Ripoll, O., Pedersen, C. B. & McGrath, J. J. Urbanicity and risk of schizophrenia — new studies and old hypotheses. JAMA Psychiatry 75, 687–688 (2018).
Galderisi, S. et al. EPA guidance on treatment of negative symptoms in schizophrenia. Eur. Psychiatry 64, e21 (2021).
Trubetskoy, V. et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 604, 502–508 (2022).
Kallmann, F. J. The Genetics of Schizophrenia: A Study of Heredity and Environment (J. J. Augustin, 1938).
van de Leemput, J., Hess, J. L., Glatt, S. J. & Tsuang, M. T. Genetics of schizophrenia: historical insights and prevailing evidence. Adv. Genet. 96, 99–141 (2016).
Skene, N. G. et al. Genetic identification of brain cell types underlying schizophrenia. Nat. Genet. 50, 825–833 (2018).
Gandal, M. J. et al. Transcriptome-wide isoform-level dysregulation in ASD, schizophrenia, and bipolar disorder. Science 362, eaat8127 (2018).
Kushima, I. et al. Comparative analyses of copy-number variation in autism spectrum disorder and schizophrenia reveal etiological overlap and biological insights. Cell Rep. 24, 2838–2856 (2018).
Zarrei, M. et al. A large data resource of genomic copy number variation across neurodevelopmental disorders. npj Genom. Med. 4, 26 (2019).
Legge, S. E. et al. Genetic architecture of schizophrenia: a review of major advancements. Psychol. Med. 51, 2168–2177 (2021).
Zoghbi, A. W. et al. High-impact rare genetic variants in severe schizophrenia. Proc. Natl. Acad. Sci. USA 118, e2112560118 (2021).
Sullivan, P. et al. Psychiatric genomics: an update and an agenda. Am. J. Psychiatry 175, 15–27 (2017).
Reichard, J. & Zimmer-Bensch, G. The epigenome in neurodevelopmental disorders. Front. Neurosci. 15, 776809 (2021).
Inyang, B. et al. The role of childhood trauma in psychosis and schizophrenia: a systematic review. Cureus 14, e21466 (2022).
Tang, W. et al. Adverse perinatal pregnancy outcomes in women with schizophrenia: a systematic review and meta-analysis. Schizophr. Res. 262, 156–167 (2023).
Saatci, D., van Nieuwenhuizen, A. & Handunnetthi, L. Maternal infection in gestation increases the risk of non-affective psychosis in offspring: a meta-analysis. J. Psychiatr. Res. 139, 125–131 (2021).
Murray, R. M., Bhavsar, V., Tripoli, G. & Howes, O. 30 Years on: how the neurodevelopmental hypothesis of schizophrenia morphed into the developmental risk factor model of psychosis. Schizophr. Bull. 43, 1190–1196 (2017).
Schmitt, A., Falkai, P. & Papiol, S. Neurodevelopmental disturbances in schizophreniaevidence from genetic and environmental factors. J. Neural Transm. 130, 195–205 (2023).
Dalmau, J. et al. An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: mechanisms and models. Lancet Neurol. 18, 1045–1057 (2019).
Sutterland, A. L. et al. Toxoplasma gondii infection and clinical characteristics of patients with schizophrenia: a systematic review and meta-analysis. Schizophrenia Bull. Open 1, sgaa042 (2020).
Coury, S. M. et al. Systematic review and meta-analysis: season of birth and schizophrenia risk. Schizophr. Res. 252, 244–252 (2023).
Severance, E. G. & Yolken, R. H. Deciphering microbiome and neuroactive immune gene interactions in schizophrenia. Neurobiol. Dis. 135, 104331 (2020).
Otero, A. M. & Antonson, A. M. At the crux of maternal immune activation: viruses, microglia, microbes, and IL-17A. Immunol. Rev. 311, 205–223 (2022).
Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Prinz, M., Jung, S. & Priller, J. Microglia biologyone century of evolving concepts. Cell 179, 292–311 (2019).
Sekar, A. et al. Schizophrenia risk from complex variation of complement component 4. Nature 530, 177–183 (2016).
Dudeck, L. et al. Differences in blood leukocyte subpopulations in schizophrenia: a systematic review and meta-analysis. JAMA Psychiatry 82, 492–504 (2025).
Gangadin, S. S., Enthoven, A. D., van Beveren, N. J. M., Laman, J. D. & Sommer, I. E. C. Immune dysfunction in schizophrenia spectrum disorders. Annu. Rev. Clin. Psychol. 20, 229–257 (2024).
Nasib, L. G. et al. The effect of prednisolone on symptom severity in schizophrenia: a placebo-controlled, randomized controlled trial. Schizophr. Res. 230, 79–86 (2021).
Hansen, N. & Malchow, B. Monoclonal antibody therapy in autoantibody-associated psychotic disorders and schizophrenia: narrative review of past and current clinical trials. Psychiatr. Danub. 35, 8–15 (2023).
McCutcheon, R. A., Abi-Dargham, A. & Howes, O. D. Schizophrenia, dopamine and the striatumfrom biology to symptoms. Trends Neurosci. 42, 205–220 (2019).
Connell, P. H. Amphetamine psychosis. Br. Med. J. 1, 582 (1957).
Leucht, S. et al. Antipsychotic drugs versus placebo for relapse prevention in schizophrenia: a systematic review and meta-analysis. Lancet 379, 2063–2071 (2012). A systematic review summarizing all important aspects of the efficacy of antipsychotics compared with placebo for relapse prevention of schizophrenia.
Benjamin, K. J. M. et al. Analysis of the caudate nucleus transcriptome in individuals with schizophrenia highlights effects of antipsychotics and new risk genes. Nat. Neurosci. 25, 1559–1568 (2022).
Kumakura, Y. & Cumming, P. PET studies of cerebral levodopa metabolism: a review of clinical findings and modeling approaches. Neuroscientist 15, 635–650 (2009).
Volkow, N. D. et al. PET evaluation of the dopamine system of the human brain. J. Nucl. Med. 37, 1242–1256 (1996).
Howes, O. D. et al. The nature of dopamine dysfunction in schizophrenia and what this means for treatment: meta-analysis of imaging studies. Arch. Gen. Psychiatry 69, 776–786 (2012).
McCutcheon, R., Beck, K., Jauhar, S. & Howes, O. D. Defining the locus of dopaminergic dysfunction in schizophrenia: a meta-analysis and test of the mesolimbic hypothesis. Schizophrenia Bull. 44, 1301–1311 (2018).
Brandl, F. et al. Negative symptoms, striatal dopamine and model-free reward decision-making in schizophrenia. Brain 146, 767–777 (2023).
Kambeitz, J., Abi-Dargham, A., Kapur, S. & Howes, O. D. Alterations in cortical and extrastriatal subcortical dopamine function in schizophrenia: systematic review and meta-analysis of imaging studies. Br. J. Psychiatry 204, 420–429 (2014).
Howes, O. D. et al. Midbrain dopamine function in schizophrenia and depression: a post-mortem and positron emission tomographic imaging study. Brain 136, 3242–3251 (2013).
Laruelle, M., Abi-Dargham, A., Gil, R., Kegeles, L. & Innis, R. Increased dopamine transmission in schizophrenia: relationship to illness phases. Biol. Psychiatry 46, 56–72 (1999).
Howes, O. D. et al. Dopamine synthesis capacity before onset of psychosis: a prospective [18 F]-DOPA PET imaging study. Am. J. Psychiatry 168, 1311–1317 (2011).
Cassidy, C. M. et al. A perceptual inference mechanism for hallucinations linked to striatal dopamine. Curr. Biol. 28, 503–514.e4 (2018).
Schmack, K., Bosc, M., Ott, T., Sturgill, J. & Kepecs, A. Striatal dopamine mediates hallucination-like perception in mice. Science 372, eabf4740 (2021).
Howes, O. D., Hird, E. J., Adams, R. A., Corlett, P. R. & McGuire, P. Aberrant salience, information processing, and dopaminergic signaling in people at clinical high risk for psychosis. Biol. Psychiatry 88, 304–314 (2020).
Avram, M. et al. Reduced striatal dopamine synthesis capacity in patients with schizophrenia during remission of positive symptoms. Brain 142, 1813–1826 (2019).
Galderisi, S., Mucci, A., Buchanan, R. W. & Arango, C. Negative symptoms of schizophrenia: new developments and unanswered research questions. Lancet Psychiatry 5, 664–677 (2018).
Engel, M., Fritzsche, A. & Lincoln, T. M. Anticipation and experience of emotions in patients with schizophrenia and negative symptoms. An experimental study in a social context. Schizophr. Res. 170, 191–197 (2016).
Anis, N. A., Berry, S. C., Burton, N. R. & Lodge, D. The dissociative anesthetics, ketamine and phencyclidine, selectively reduce excitation of central mammalian neurones by N-methyl-D-aspartate. Br. J. Pharmacol. 79, 565–575 (1983).
Lahti, A. C., Weiler, M. A., Tamara Michaelidis, B. A., Parwani, A. & Tamminga, C. A. Effects of ketamine in normal and schizophrenic volunteers. Neuropsychopharmacology 25, 455–467 (2001).
Holcomb, H. H., Lahti, A. C., Medoff, D. R., Weiler, M. & Tamminga, C. A. Sequential regional cerebral blood flow brain scans using PET with H215O demonstrate ketamine actions in CNS dynamically. Neuropsychopharmacology 25, 165–172 (2001).
Coyle, J. T., Basu, A., Benneyworth, M., Balu, D. & Konopaske, G. Glutamatergic synaptic dysregulation in schizophrenia: therapeutic implications. Handb. Exp. Pharmacol. 213, 267–295 (2012).
Amitai, N., Kuczenski, R., Behrens, M. M. & Markou, A. Repeated phencyclidine administration alters glutamate release and decreases GABA markers in the prefrontal cortex of rats. Neuropharmacology 62, 1422–1431 (2012).
Lewis, D. A., Curley, A. A., Glausier, J. R. & Volk, D. W. Cortical parvalbumin interneurons and cognitive dysfunction in schizophrenia. Trends Neurosci. 35, 57–67 (2012).
Dienel, S. J., Schoonover, K. E. & Lewis, D. A. Cognitive dysfunction and prefrontal cortical circuit alterations in schizophrenia: developmental trajectories. Biol. Psychiatry 92, 450–459 (2022).
Goldman-Rakic, P. S. Cellular basis of working memory. Neuron 14, 477–485 (1995).
Datta, D. & Arnsten, A. F. T. Unique molecular regulation of higher-order prefrontal cortical circuits: insights into the neurobiology of schizophrenia. ACS Chem. Neurosci. 9, 2127–2145 (2018).
Cools, R. & Arnsten, A. F. T. Neuromodulation of prefrontal cortex cognitive function in primates: the powerful roles of monoamines and acetylcholine. Neuropsychopharmacology 47, 309–328 (2022).
Tamminga, C. A., Stan, A. D. & Wagner, A. D. The hippocampal formation in schizophrenia. Am. J. Psychiatry 167, 1178–1193 (2010). An excellent review of the role of the hippocampus in schizophrenia.
Harrison, P. J. The hippocampus in schizophrenia: a review of the neuropathological evidence and its pathophysiological implications. Psychopharmacology 174, 151–162 (2004).
Honea, R., Crow, T. J., Passingham, D. & Mackay, C. E. Regional deficits in brain volume in schizophrenia: a meta-analysis of voxel-based morphometry studies. Am. J. Psychiatry 162, 2233–2245 (2005).
Wang, L. et al. Progressive deformation of deep brain nuclei and hippocampal-amygdala formation in schizophrenia. Biol. Psychiatry 64, 1060–1068 (2008).
Ruzicka, W. B., Subburaju, S. & Benes, F. M. Variability of DNA methylation within schizophrenia risk loci across subregions of human hippocampus. Genes 8, 143 (2017).
Ruzicka, W. B., Subburaju, S. & Benes, F. M. Circuit- and diagnosis-specific DNA methylation changes at γ-aminobutyric acid-related genes in postmortem human hippocampus in schizophrenia and bipolar disorder. JAMA Psychiatry 72, 541–551 (2015).
Konradi, C. & Heckers, S. Molecular aspects of glutamate dysregulation: implications for schizophrenia and its treatment. Pharmacol. Ther. 97, 153–179 (2003).
Falkai, P. et al. Decreased oligodendrocyte and neuron number in anterior hippocampal areas and the entire hippocampus in schizophrenia: a stereological postmortem study. Schizophr. Bull. 42 (Suppl. 1), S4–s12 (2016).
Li, W. et al. Synaptic proteins in the hippocampus indicative of increased neuronal activity in CA3 in schizophrenia. Am. J. Psychiatry 172, 373–382 (2015).
Perez, J. M. et al. Hippocampal subfield transcriptome analysis in schizophrenia psychosis. Mol. Psychiatry 26, 2577–2589 (2021).
Jaffe, A. et al. Profiling gene expression in the human dentate gyrus granule cell layer reveals insights into schizophrenia and its genetic risk. Nat. Neurosci. 23, 510–519 (2020).
Medoff, D. R., Holcomb, H. H., Lahti, A. C. & Tamminga, C. A. Probing the human hippocampus using rCBF: contrasts in schizophrenia. Hippocampus 11, 543–550 (2001).
Heckers, S., Zalesak, M., Weiss, A. P., Ditman, T. & Titone, D. Hippocampal activation during transitive inference in humans. Hippocampus 14, 153–162 (2004).
Schobel, S. A. et al. Differential targeting of the CA1 subfield of the hippocampal formation by schizophrenia and related psychotic disorders. Arch. Gen. Psychiatry 66, 938–946 (2009).
Schobel, S. A. et al. Imaging patients with psychosis and a mouse model establishes a spreading pattern of hippocampal dysfunction and implicates glutamate as a driver. Neuron 78, 81–93 (2013).
Segev, A. et al. Reduced GluN1 in mouse dentate gyrus is associated with CA3 hyperactivity and psychosis-like behaviors. Mol. Psychiatry 25, 2832–2843 (2020).
Javitt, D. C. & Freedman, R. Sensory processing dysfunction in the personal experience and neuronal machinery of schizophrenia. Am. J. Psychiatry 172, 17–31 (2015).
Van Haren, N. E. et al. Changes in cortical thickness during the course of illness in schizophrenia. Arch. Gen. Psychiatry 68, 871–880 (2011).
Cannon, T. D. et al. Progressive reduction in cortical thickness as psychosis develops: a multisite longitudinal neuroimaging study of youth at elevated clinical risk. Biol. Psychiatry 77, 147–157 (2015).
Ramanathan, S. et al. Longitudinal trajectories of cortical thickness as a biomarker for psychosis in individuals with 22q11. 2 deletion syndrome. Schizophrenia Res. 188, 35–41 (2017).
Nelson, E. A. et al. A prospective longitudinal investigation of cortical thickness and gyrification in schizophrenia. Can. J. Psychiatry 65, 381–391 (2020).
Glausier, J. R. & Lewis, D. A. Dendritic spine pathology in schizophrenia. Neuroscience 251, 90–107 (2013).
Onwordi, E. C. et al. Synaptic density marker SV2A is reduced in schizophrenia patients and unaffected by antipsychotics in rats. Nat. Commun. 11, 246 (2020).
Yoon, J. H. et al. Reductions in synaptic marker SV2A in early-course schizophrenia. J. Psychiatr. Res. 161, 213–217 (2023).
Pillinger, T., D’Ambrosio, E., McCutcheon, R. & Howes, O. D. Is psychosis a multisystem disorder? A meta-review of central nervous system, immune, cardiometabolic, and endocrine alterations in first-episode psychosis and perspective on potential models. Mol. Psychiatry 24, 776–794 (2019).
Yohn, S. E., Weiden, P. J., Felder, C. C. & Stahl, S. M. Muscarinic acetylcholine receptors for psychotic disorders: bench-side to clinic. Trends Pharmacol. Sci. 43, 1098–1112 (2022).
Paul, S. M., Yohn, S. E., Popiolek, M., Miller, A. C. & Felder, C. C. Muscarinic acetylcholine receptor agonists as novel treatments for schizophrenia. Am. J. Psychiatry 179, 611–627 (2022).
Steeds, H., Carhart-Harris, R. L. & Stone, J. M. Drug models of schizophrenia. Ther. Adv. Psychopharmacol. 5, 43–58 (2015).
Krystal, J. H. et al. Emraclidine, a novel positive allosteric modulator of cholinergic M4 receptors, for the treatment of schizophrenia: a two-part, randomised, double-blind, placebo-controlled, phase 1b trial. Lancet 400, 2210–2220 (2022).
Brannan, S. K. et al. Muscarinic cholinergic receptor agonist and peripheral antagonist for schizophrenia. N. Engl. J. Med. 384, 717–726 (2021).
Horan, W. P. et al. The impact of xanomeline and trospium chloride on cognitive impairment in acute schizophrenia: replication in pooled data from two phase 3 trials. Am. J. Psychiatry 182, 297–306 (2025).
Ribolsi, M., Daskalakis, Z. J., Siracusano, A. & Koch, G. Abnormal asymmetry of brain connectivity in schizophrenia. Front. Hum. Neurosci. 8, 1010 (2014).
Schijven, D. et al. Large-scale analysis of structural brain asymmetries in schizophrenia via the ENIGMA consortium. Proc. Natl Acad. Sci. USA 120, e2213880120 (2023).
Morita, K., Miura, K., Kasai, K. & Hashimoto, R. Eye movement characteristics in schizophrenia: a recent update with clinical implications. Neuropsychopharmacol. Rep. 40, 2–9 (2020).
Bramon, E., Rabe-Hesketh, S., Sham, P., Murray, R. M. & Frangou, S. Meta-analysis of the P300 and P50 waveforms in schizophrenia. Schizophr. Res. 70, 315–329 (2004).
Earls, H. A., Curran, T. & Mittal, V. A meta-analytic review of auditory event-related potential components as endophenotypes for schizophrenia: perspectives from first-degree relatives. Schizophr. Bull. 42, 1504–1516 (2016).
Haigh, S. M., Coffman, B. A. & Salisbury, D. F. Mismatch negativity in first-episode schizophrenia: a meta-analysis. Clin. EEG Neurosci. 48, 3–10 (2017).
Harikumar, A. et al. Revisiting functional dysconnectivity: a review of three model frameworks in schizophrenia. Curr. Neurol. Neurosci. Rep. https://doi.org/10.1007/s11910-023-01325-8 (2023).
Friston, K., Brown, H. R., Siemerkus, J. & Stephan, K. E. The dysconnection hypothesis (2016). Schizophr. Res. 176, 83–94 (2016).
Tandon, R. et al. The schizophrenia syndrome, circa 2024: what we know and how that informs its nature. Schizophr. Res. 264, 1–28 (2024).
Stroup, T. S. Heterogeneity of treatment effects in schizophrenia. Am. J. Med. 120, S26–S31 (2007).
McCutcheon, R. A. et al. The efficacy and heterogeneity of antipsychotic response in schizophrenia: a meta-analysis. Mol. Psychiatry 26, 1310–1320 (2021).
Brugger, S. P. et al. Heterogeneity of striatal dopamine function in schizophrenia: meta-analysis of variance. Biol. Psychiatry 87, 215–224 (2020).
Syvälahti, E., Räkköläinen, V., Aaltonen, J., Lehtinen, V. & Hietala, J. Striatal D2 dopamine receptor density and psychotic symptoms in schizophrenia: a longitudinal study. Schizophrenia Res. 43, 159–161 (2000).
Thompson, J. L. et al. Striatal dopamine release in schizophrenia comorbid with substance dependence. Mol. Psychiatry 18, 909–915 (2013).
Kraepelin, E. Einführung in die psychiatrische Klinik [German] 4th edn (Johann Ambrosius Barth, 1921).
Bleuler, E. Dementia praecox oder die Gruppe der Schizophrenien [German] (Deuticke, 1911).
Schneider, K. Klinische Psychopathologie (Thieme, 1971) [German].
Kirkpatrick, B., Fenton, W. S., Carpenter, W. T. Jr. & Marder, S. R. The NIMH-MATRICS consensus statement on negative symptoms. Schizophr. Bull. 32, 214–219 (2006).
Bobes, J., Arango, C., Garcia-Garcia, M. & Rejas, J. CLAMORS Study Collaborative Group. Prevalence of negative symptoms in outpatients with schizophrenia spectrum disorders treated with antipsychotics in routine clinical practicefindings from the CLAMORS study. J. Clin. Psychiatry 71, 280–286 (2010).
Rabinowitz, J. et al. Negative symptoms in schizophrenia — the remarkable impact of inclusion definitions in clinical trials and their consequences. Schizophr. Res. 150, 334–338 (2013).
Correll, C. U. & Schooler, N. R. Negative symptoms in schizophrenia: a review and clinical guide for recognition, assessment, and treatment. Neuropsychiatr. Dis. Treat. 16, 519–534 (2020).
Buchanan, R. W. et al. The FDA-NIMH-MATRICS guidelines for clinical trial design of cognitive-enhancing drugs: what do we know 5 years later? Schizophr. Bull. 37, 1209–1217 (2011).
Oehl, M., Hummer, M. & Fleischhacker, W. W. Compliance with antipsychotic treatment. Acta Psychiatr. Scand. Suppl. 407, 83–86 (2000).
Heckers, S. et al. Structure of the psychotic disorders classification in DSM-5. Schizophr. Res. 150, 11–14 (2013).
Leucht, S., van Os, J., Jager, M. & Davis, J. M. Prioritization of psychopathological symptoms and clinical characterization in psychiatric diagnoses: a narrative review. JAMA Psychiatry 81, 1149–1158 (2024).
Doherty, J. L. & Owen, M. J. Genomic insights into the overlap between psychiatric disorders: implications for research and clinical practice. Genome Med. 6, 29 (2014).
Staines, L. et al. Psychotic experiences in the general population, a review; definition, risk factors, outcomes and interventions. Psychol. Med. 52, 1–12 (2022).
McGrath, J. J. et al. The bidirectional associations between psychotic experiences and DSM-IV mental disorders. Am. J. Psychiatry 173, 997–1006 (2016).
Insel, T. et al. Research domain criteria (RDoC): toward a new classification framework for research on mental disorders. Am. J. Psychiatry 167, 748–751 (2010).
Fried, E. I. et al. Mental disorders as networks of problems: a review of recent insights. Soc. Psychiatry Psychiatr. Epidemiol. 52, 1–10 (2017).
Contreras, A., Nieto, I., Valiente, C., Espinosa, R. & Vazquez, C. The study of psychopathology from the network analysis perspective: a systematic review. Psychother. Psychosom. 88, 71–83 (2019).
Buchwald, K. et al. Centrality statistics of symptom networks of schizophrenia: a systematic review. Psychol. Med. 54, 1061–1073 (2024).
Kotov, R. et al. The hierarchical taxonomy of psychopathology (HiTOP): a quantitative nosology based on consensus of evidence. Annu. Rev. Clin. Psychol. 17, 83–108 (2021).
Jonas, K. G. et al. Psychosis superspectrum I: nosology, etiology, and lifespan development. Mol. Psychiatry 29, 1005–1019 (2024).
Haeffel, G. J. et al. The hierarchical taxonomy of psychopathology (HiTOP) is not an improvement over the DSM. Clin. Psychol. Sci. 10, 285–290 (2022).
Haeffel, G. J. et al. Folk classification and factor rotations: whales, sharks, and the problems with the hierarchical taxonomy of psychopathology (HiTOP). Clin. Psychol. Sci. 10, 259–278 (2022).
Mihura, J. L. et al. Improving dependability in science: a critique on the psychometric qualities of the HiTOP psychosis superspectrum. Schizophr. Res. 270, 433–440 (2024).
McGorry, P. D. & Hickie, I. B. Clinical Staging in Psychiatry Making Diagnosis Work for Research and Treatment (Cambridge University Press, 2019). A book describing in detail the staging model used in this Primer.
Kane, J. M. et al. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch. Gen. Psychiatry 45, 789–796 (1988).
Leucht, S. et al. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet 373, 31–41 (2009).
Dong, S. et al. A network meta-analysis of efficacy, acceptability, and tolerability of antipsychotics in treatment-resistant schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. https://doi.org/10.1007/s00406-023-01654-2 (2023).
Zohar, J. et al. A review of the current nomenclature for psychotropic agents and an introduction to the Neuroscience-based Nomenclature. Eur. Neuropsychopharmacol. 25, 2318–2325 (2015).
McCutcheon, R. A. et al. Data-driven taxonomy for antipsychotic medication: a new classification system. Biol. Psychiatry https://doi.org/10.1016/j.biopsych.2023.04.004 (2023).
Neufeld, N. H. & Blumberger, D. M. An update on the use of neuromodulation strategies in the treatment of schizophrenia. Am. J. Psychiatry 182, 332–340 (2025).
Sanghani, S. N., Petrides, G. & Kellner, C. H. Electroconvulsive therapy (ECT) in schizophrenia: a review of recent literature. Curr. Opin. Psychiatry 31, 213–222 (2018).
Kennedy, N. I., Lee, W. H. & Frangou, S. Efficacy of non-invasive brain stimulation on the symptom dimensions of schizophrenia: a meta-analysis of randomized controlled trials. Eur. Psychiatry 49, 69–77 (2018).
Tseng, P. T. et al. Assessment of noninvasive brain stimulation interventions for negative symptoms of schizophrenia: a systematic review and network meta-analysis. JAMA Psychiatry 79, 770–779 (2022).
Li, X., Dai, J., Liu, Q., Zhao, Z. & Zhang, X. Efficacy and safety of non-invasive brain stimulation on cognitive function for cognitive impairment associated with schizophrenia: a systematic review and meta-analysis. J. Psychiatr. Res. 170, 174–186 (2024).
Dechartres, A., Trinquart, L., Boutron, I. & Ravaud, P. Influence of trial sample size on treatment effect estimates: meta-epidemiological study. BMJ 346, f2304 (2013).
Vita, A. et al. European Psychiatric Association guidance on treatment of cognitive impairment in schizophrenia. Eur. Psychiatry 65, e57 (2022).
Bighelli, I. et al. Psychological interventions to reduce positive symptoms in schizophrenia: systematic review and network meta-analysis. World Psychiatry 17, 316–329 (2018). A network meta-analysis demonstrating the efficacy of CBT added to antipsychotics for positive symptoms of schizophrenia.
Bighelli, I. et al. Effects of psychological treatments on functioning in people with schizophrenia: a systematic review and meta-analysis of randomized controlled trials. Eur. Arch. Psychiatry Clin. Neurosci. 273, 779–810 (2023).
Hasan, A. et al. Revised S3 guidelines on schizophrenia developmental process and selected recommendations [German]. Nervenarzt 91, 26–33 (2020).
National Institute for Health and Care Excellence. Psychosis and Schizophrenia in Adults: Prevention and Management. NICE Clinical Guidelines, CG178 (2014).
Galletly, C. et al. Royal Australian and New Zealand College of Psychiatrists schizophrenia clinical practice guidelines. Aust. N. Z. J. Psychiatry 50, 410–472 (2016).
Bighelli, I. et al. Psychosocial and psychological interventions for relapse prevention in schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatry 8, 969–980 (2021).
Rodolico, A. et al. Family interventions for relapse prevention in schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatry 9, 211–221 (2022).
Vita, A. et al. Effectiveness, core elements, and moderators of response of cognitive remediation for schizophrenia: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatry 78, 848–858 (2021).
van Oosterhout, B. et al. Metacognitive training for schizophrenia spectrum patients: a meta-analysis on outcome studies. Psychol. Med. 46, 47–57 (2016).
Lutgens, D., Gariepy, G. & Malla, A. Psychological and psychosocial interventions for negative symptoms in psychosis: systematic review and meta-analysis. Br. J. Psychiatry 210, 324–332 (2017).
Turner, D. T., van der Gaag, M., Karyotaki, E. & Cuijpers, P. Psychological interventions for psychosis: a meta-analysis of comparative outcome studies. Am. J. Psychiatry 171, 523–538 (2014).
de Winter, L. et al. Who benefits from individual placement and support? A meta-analysis. Epidemiol. Psychiatr. Sci. 31, e50 (2022).
Hodann-Caudevilla, R. M., Diaz-Silveira, C., Burgos-Julian, F. A. & Santed, M. A. Mindfulness-based interventions for people with schizophrenia: a systematic review and meta-analysis. Int. J. Environ. Res. Public Health 17, 4690 (2020).
Dellazizzo, L., Potvin, S., Luigi, M. & Dumais, A. Evidence on virtual reality-based therapies for psychiatric disorders: meta-review of meta-analyses. J. Med. Internet Res. 22, e20889 (2020).
Craig, T. K. et al. AVATAR therapy for auditory verbal hallucinations in people with psychosis: a single-blind, randomised controlled trial. Lancet Psychiatry 5, 31–40 (2018).
Pot-Kolder, R. M. C. A. et al. Virtual-reality-based cognitive behavioural therapy versus waiting list control for paranoid ideation and social avoidance in patients with psychotic disorders: a single-blind randomised controlled trial. Lancet Psychiatry 5, 217–226 (2018).
Leucht, S. et al. The response of subgroups of patients with schizophrenia to different antipsychotic drugs: a systematic review and meta-analysis. Lancet Psychiatry 9, 884–893 (2022).
Huhn, M. et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet 394, 939–951 (2019). The most up-to-date meta-analysis of the effects of antipsychotic drugs in acutely ill people with schizophrenia.
Hamann, J. et al. Shared decision making for in-patients with schizophrenia. Acta Psychiatr. Scand. 114, 265–273 (2006).
World Health Organization. World Health Organization Model List of Essential Medicines. 23rd List (WHO, 2023).
Krause, M. et al. Antipsychotic drugs for patients with schizophrenia and predominant or prominent negative symptoms: a systematic review and meta-analysis. Eur. Arch. Psychiatry Clin. Neurosci. 268, 625–639 (2018).
Helfer, B. et al. Efficacy and safety of antidepressants added to antipsychotics for schizophrenia: a systematic review and meta-analysis. Am. J. Psychiatry 173, 876–886 (2016).
Samara, M. T. et al. Early improvement as a predictor of later response to antipsychotics in schizophrenia: a diagnostic test review. Am. J. Psychiatry 172, 617–629 (2015).
Leucht, S. Measurements of response, remission, and recovery in schizophrenia and examples for their clinical application. J. Clin. Psychiatry 75 (Suppl. 1), 8–14 (2014).
Andreasen, N. et al. Remission in schizophrenia: proposed criteria and rationale for consensus. Am. J. Psychiatry 62, 441–449 (2005).
Siafis, S. et al. Relapse in clinically stable adult patients with schizophrenia or schizoaffective disorder: evidence-based criteria derived by equipercentile linking and diagnostic test accuracy meta-analysis. Lancet Psychiatry 11, 36–46 (2024).
Leucht, S. et al. Dose-response meta-analysis of antipsychotic drugs for acute schizophrenia. Am. J. Psychiatry 177, 342–353 (2020).
Leucht, S., Priller, J. & Davis, J. M. Antipsychotic drugs: a concise review of history, classification, indications, mechanism, efficacy, side effects, dosing, and clinical application. Am. J. Psychiatry 181, 865–878 (2024).
Uchida, H. et al. Dopamine D2 receptor occupancy and clinical effects: a systematic review and pooled analysis. J. Clin. Psychopharmacol. 31, 497–502 (2011).
Samara, M. T. et al. Increasing antipsychotic dose for non response in schizophrenia. Cochrane Database Syst. Rev. 5, CD011883 (2018).
Samara, M. T. et al. Switching antipsychotics versus continued current treatment in people with non-responsive schizophrenia. Cochrane Database Syst. Rev. 4, CD011885 (2025).
Howes, O. D. et al. Treatment-resistant schizophrenia: treatment response and resistance in psychosis (TRRIP) working group consensus guidelines on diagnosis and terminology. Am. J. Psychiatry 174, 216–229 (2017).
Siskind, D., McCartney, L., Goldschlager, R. & Kisely, S. Clozapine v. first- and second-generation antipsychotics in treatment-refractory schizophreniasystematic review and meta-analysis. Br. J. Psychiatry https://doi.org/10.1192/bjp.bp.115.177261 (2016)
Samara, M. T. et al. Efficacy, acceptability, and tolerability of antipsychotics in treatment-resistant schizophrenia: a network meta-analysis. JAMA Psychiatry 73, 199–210 (2016).
Meltzer, H. Y. et al. Clozapine treatment for suicidality in schizophrenia: International Suicide Prevention Trial (InterSePT). Arch. Gen. Psychiatry 60, 82–91 (2003).
Correll, C. U. et al. Efficacy of 42 pharmacologic cotreatment strategies added to antipsychotic monotherapy in schizophrenia: systematic overview and quality appraisal of the meta-analytic evidence. JAMA Psychiatry 74, 675–684 (2017). A systematic review showing that, currently, no cotreatment strategy can be recommended for persistent positive symptoms.
Galling, B. et al. Antipsychotic augmentation vs. monotherapy in schizophrenia: systematic review, meta-analysis and meta-regression analysis. World Psychiatry 16, 77–89 (2017).
Yeh, T. C. et al. Pharmacological and nonpharmacological augmentation treatments for clozapine-resistant schizophrenia: a systematic review and network meta-analysis with normalized entropy assessment. Asian J. Psychiatry 79, 103375 (2023).
Zheng, W. et al. Electroconvulsive therapy added to non-clozapine antipsychotic medication for treatment resistant schizophrenia: meta-analysis of randomized controlled trials. PLoS ONE 11, e0156510 (2016).
Ceraso, A. et al. Maintenance treatment with antipsychotic drugs in schizophrenia: a Cochrane systematic review and meta-analysis. Schizophr. Bull. 48, 738–740 (2022).
Leucht, S., Hierl, S., Kissling, W., Dold, M. & Davis, J. M. Putting the efficacy of psychiatric and general medicine medication into perspectivereview of meta-analyses. Br. J. Psychiatry 200, 97–106 (2012).
Siskind, D. et al. Rates of treatment-resistant schizophrenia from first-episode cohortssystematic review and meta-analysis. Br. J. Psychiatry 220, 115–120 (2022).
Robinson, D. et al. Predictors of relapse following response from a first episode of schizophrenia or schizoaffective disorder. Arch. Gen. Psychiatry 56, 241–247 (1999).
Samaha, A. N., Seeman, P., Stewart, J., Rajabi, H. & Kapur, S. “Breakthrough” dopamine supersensitivity during ongoing antipsychotic treatment leads to treatment failure over time. J. Neurosci. 27, 2979–2986 (2007).
Brandt, L. et al. Adverse events after antipsychotic discontinuation: an individual participant data meta-analysis. Lancet Psychiatry 9, 232–242 (2022).
Takeuchi, H. et al. Does relapse contribute to treatment resistance? Antipsychotic response in first- vs. second-episode schizophrenia. Neuropsychopharmacology 44, 1036–1042 (2019).
Leucht, S. et al. Examination of dosing of antipsychotic drugs for relapse prevention in patients with stable schizophrenia: a meta-analysis. JAMA Psychiatry 78, 1238–1248 (2021).
Rodolico, A. et al. Antipsychotic dose reduction compared to dose continuation for people with schizophrenia. Cochrane Database Syst. Rev. 11, CD014384 (2022).
Tiihonen, J., Tanskanen, A. & Taipale, H. 20-year nationwide follow-up study on discontinuation of antipsychotic treatment in first-episode schizophrenia. Am. J. Psychiatry 175, 765–773 (2018).
Schneider-Thoma, J. et al. Comparative efficacy and tolerability of 32 oral and long-acting injectable antipsychotics for the maintenance treatment of adults with schizophrenia: a systematic review and network meta-analysis. Lancet 399, 824–836 (2022).
Carbon, M., Kane, J. M., Leucht, S. & Correll, C. U. Tardive dyskinesia risk with first- and second-generation antipsychotics in comparative randomized controlled trials: a meta-analysis. World Psychiatry 17, 330–340 (2018).
Sampson, S., Mansour, M., Maayan, N., Soares-Weiser, K. & Adams, C. E. Intermittent drug techniques for schizophrenia. Cochrane Database Syst. Rev. 7, CD006196 (2013).
Acosta, F. J., Hernandez, J. L., Pereira, J., Herrera, J. & Rodriguez, C. J. Medication adherence in schizophrenia. World J. Psychiatry 2, 74–82 (2012).
Leucht, S. & Heres, S. Epidemiology, clinical consequences, and psychosocial treatment of nonadherence in schizophrenia. J. Clin. Psychiatry 67 (Suppl. 5), 3–8 (2006).
Semahegn, A. et al. Psychotropic medication non-adherence and its associated factors among patients with major psychiatric disorders: a systematic review and meta-analysis. Syst. Rev. 9, 17 (2020).
Kishimoto, T., Hagi, K., Kurokawa, S., Kane, J. M. & Correll, C. U. Long-acting injectable versus oral antipsychotics for the maintenance treatment of schizophrenia: a systematic review and comparative meta-analysis of randomised, cohort, and pre-post studies. Lancet Psychiatry 8, 387–404 (2021).
Wu, H. et al. Antipsychotic-induced weight gain: dose-response meta-analysis of randomized controlled trials. Schizophr. Bull. 48, 643–654 (2022).
Siafis, S. et al. Antipsychotic dose, dopamine D2 receptor occupancy and extrapyramidal side-effects: a systematic review and dose-response meta-analysis. Mol. Psychiatry 28, 3267–3277 (2023).
Lin, X. et al. Antipsychotic-related prolactin changes: a systematic review and dose-response meta-analysis. CNS Drugs 39, 937–947 (2025).
Wu, H. et al. Antipsychotic-induced akathisia in adults with acute schizophrenia: a systematic review and dose-response meta-analysis. Eur. Neuropsychopharmacol. 72, 40–49 (2023).
Firth, J. et al. The Lancet Psychiatry Commission: a blueprint for protecting physical health in people with mental illness. Lancet Psychiatry 6, 675–712 (2019).
De Hert, M. et al. Physical illness in patients with severe mental disorders. II. Barriers to care, monitoring and treatment guidelines, plus recommendations at the system and individual level. World Psychiatry 10, 138–151 (2011).
Solmi, M. et al. Disparities in screening and treatment of cardiovascular diseases in patients with mental disorders across the world: systematic review and meta-analysis of 47 observational studies. Am. J. Psychiatry 178, 793–803 (2021).
Lawrence, D. M., Holman, C. D., Jablensky, A. V. & Hobbs, M. S. Death rate from ischaemic heart disease in Western Australian psychiatric patients 1980-1998. Br. J. Psychiatry 182, 31–36 (2003).
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease study 2019. Lancet 396, 1204–1222 (2020).
GBD 2019 Mental Disorders Collaborators. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990-2019: a systematic analysis for the global burden of disease study 2019. Lancet Psychiatry 9, 137–150 (2022).
Bellack, A. S. Scientific and consumer models of recovery in schizophrenia: concordance, contrasts, and implications. Schizophr. Bull. 32, 432–442 (2006).
Chan, R. C. H., Mak, W. W. S., Chio, F. H. N. & Tong, A. C. Y. Flourishing with psychosis: a prospective examination on the interactions between clinical, functional, and personal recovery processes on well-being among individuals with schizophrenia spectrum disorders. Schizophr. Bull. 44, 778–786 (2018).
Awad, A. G. & Voruganti, L. N. P. Measuring quality of life in patients with schizophrenia: an update. Pharmacoeconomics 30, 183–195 (2012).
Karow, A., Wittmann, L., Schöttle, D., Schäfer, I. & Lambert, M. The assessment of quality of life in clinical practice in patients with schizophrenia. Dialogues Clin. Neurosci. 16, 185–195 (2014).
Development of the World Health Organization WHOQOL-BREF quality of life assessment. The WHOQOL Group. Psychological Med. 28, 551–558 (1998).
World Health Organization. Health Promotion Glossary of Terms 2021 (WHO, 2021).
van Krugten, F. C. W., Feskens, K., Busschbach, J. J. V., Hakkaart-van Roijen, L. & Brouwer, W. B. F. Instruments to assess quality of life in people with mental health problems: a systematic review and dimension analysis of generic, domain- and disease-specific instruments. Health Qual. Life Outcomes 19, 249 (2021).
Attepe Özden, S., Tekindal, M. & Tekindal, M. A. Quality of life of people with schizophrenia: a meta-analysis. Int. J. Soc. Psychiatry 69, 1444–1452 (2023).
Alessandrini, M. et al. A structural equation modelling approach to explore the determinants of quality of life in schizophrenia. Schizophr. Res. 171, 27–34 (2016).
van Rooijen, G. et al. Longitudinal evidence for a relation between depressive symptoms and quality of life in schizophrenia using structural equation modeling. Schizophrenia Res. 208, 82–89 (2019).
Van Eck, R. M., Burger, T. J., Vellinga, A., Schirmbeck, F. & de Haan, L. The relationship between clinical and personal recovery in patients with schizophrenia spectrum disorders: a systematic review and meta-analysis. Schizophr. Bull. 44, 631–642 (2018).
Lysaker, P. H., Pattison, M. L., Leonhardt, B. L., Phelps, S. & Vohs, J. L. Insight in schizophrenia spectrum disorders: relationship with behavior, mood and perceived quality of life, underlying causes and emerging treatments. World Psychiatry 17, 12–23 (2018).
Davis, B. J., Lysaker, P. H., Salyers, M. P. & Minor, K. S. The insight paradox in schizophrenia: a meta-analysis of the relationship between clinical insight and quality of life. Schizophr. Res. 223, 9–17 (2020).
Staring, A. B., Van der Gaag, M., Van den Berge, M., Duivenvoorden, H. J. & Mulder, C. L. Stigma moderates the associations of insight with depressed mood, low self-esteem, and low quality of life in patients with schizophrenia spectrum disorders. Schizophr. Res. 115, 363–369 (2009).
Morgan, A. J., Reavley, N. J., Ross, A., San Too, L. & Jorm, A. F. Interventions to reduce stigma towards people with severe mental illness: systematic review and meta-analysis. J. Psychiatr. Res. 103, 120–133 (2018).
Thornicroft, G. et al. The Lancet Commission on Ending Stigma and Discrimination in Mental Health. Lancet 400, 1438–1480 (2022).
Sudlow, C. et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Murray, G. K. et al. Could polygenic risk scores be useful in psychiatry?: A review. JAMA Psychiatry https://doi.org/10.1001/jamapsychiatry.2020.3042 (2020).
Zhang, M. J. et al. Polygenic enrichment distinguishes disease associations of individual cells in single-cell RNA-seq data. Nat. Genet. 54, 1572–1580 (2022).
Leucht, S. et al. Towards a name change for schizophrenia: positive and negative symptoms disorder (PND). Eur. Arch. Psychiatry Clin. Neurosci. 275, 963–971 (2025).
Bristol Myers Squibb. Bristol Myers Squibb Announces Topline Results from Phase 3 ARISE Trial Evaluating Cobenfy (Xanomeline and Trospium Chloride) as an Adjunctive Treatment to Atypical Antipsychotics in Adults with Schizophrenia. Bristol Myers Squibb https://news.bms.com/news/details/2025/Bristol-Myers-Squibb-Announces-Topline-Results-from-Phase-3-ARISE-Trial-Evaluating-Cobenfy-xanomeline-and-trospium-chloride-as-an-Adjunctive-Treatment-to-Atypical-Antipsychotics-in-Adults-with-Schizophrenia/default.aspx (2025).
Alvarez-Jimenez, M. et al. Online, social media and mobile technologies for psychosis treatment: a systematic review on novel user-led interventions. Schizophrenia Res. 156, 96–106 (2014).
Siafis, S. et al. Evidence-based shared-decision-making assistant (SDM-assistant) for choosing antipsychotics: protocol of a cluster-randomized trial in hospitalized patients with schizophrenia. BMC Psychiatry 22, 406 (2022).
Gumley, A. I. et al. Digital smartphone intervention to recognise and manage early warning signs in schizophrenia to prevent relapse: the EMPOWER feasibility cluster RCT. Health Technol. Assess. 26, 1 (2022).
Tiihonen, J., Mittendorfer-Rutz, E., Torniainen, M., Alexanderson, K. & Tanskanen, A. Mortality and cumulative exposure to antipsychotics, antidepressants, and benzodiazepines in patients with schizophrenia: an observational follow-up study. Am. J. Psychiatry 173, 600–606 (2016).
Correll, C. U. et al. Comparison of early intervention services vs treatment as usual for early-phase psychosis: a systematic review, meta-analysis, and meta-regression. JAMA Psychiatry 75, 555–565 (2018).
Krause, M. et al. Antipsychotic drugs for elderly patients with schizophrenia: a systematic review and meta-analysis. Eur. Neuropsychopharmacol. 28, 1360–1370 (2018).
Furukawa, T. A. et al. Initial severity of schizophrenia and efficacy of antipsychotics: participant-level meta-analysis of 6 placebo-controlled studies. JAMA Psychiatry 72, 14–21 (2015).
Rabinowitz, J. et al. Determinants of antipsychotic response in schizophrenia: implications for practice and future clinical trials. J. Clin. Psychiatry 75, e308–e316 (2014).
Cipriani, A. et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet 378, 1306–1315 (2011).
Rogers, J. P. et al. Evidence-based consensus guidelines for the management of catatonia: recommendations from the British Association for Psychopharmacology. J. Psychopharmacol. 37, 327–369 (2023).
Siddiqui, S. et al. Psychosocial interventions for individuals with comorbid psychosis and substance use disorders: systematic review and meta-analysis of randomized studies. Schizophr. Bull. https://doi.org/10.1093/schbul/sbae101 (2024).
Mortazavi, M. et al. Impact of long-acting injectable versus oral antipsychotic treatment on all-cause discontinuation risk in people with early phase schizophrenia and comorbid substance use disorder: a secondary analysis of the EULAST randomized trial. CNS Drugs https://doi.org/10.1007/s40263-025-01225-0 (2025).
Tundo, A. & Necci, R. Cognitive-behavioural therapy for obsessive-compulsive disorder co-occurring with psychosis: systematic review of evidence. World J. Psychiatry 6, 449–455 (2016).
Schirmbeck, F. & Zink, M. Obsessive-compulsive symptoms in psychotic disorderspathogenesis and treatment [German]. Nervenarzt 93, 688–694 (2022).
Krakowski, M. I., Czobor, P., Citrome, L., Bark, N. & Cooper, T. B. Atypical antipsychotic agents in the treatment of violent patients with schizophrenia and schizoaffective disorder. Arch. Gen. Psychiatry 63, 622–629 (2006).
Salazar de Pablo, G. et al. What is the duration of untreated psychosis worldwide? — A meta-analysis of pooled mean and median time and regional trends and other correlates across 369 studies. Psychol. Med. 54, 652–662 (2024).
Fabiano, N. et al. Efficacy, tolerability, and safety of xanomeline-trospium chloride for schizophrenia: a systematic review and meta-analysis. Eur. Neuropsychopharmacol. 92, 62–73 (2025).
Fusar-Poli, P. et al. The psychosis high-risk state: a comprehensive state-of-the-art review. JAMA Psychiatry 70, 107–120 (2013).
Davies, C. et al. Lack of evidence to favor specific preventive interventions in psychosis: a network meta-analysis. World Psychiatry 17, 196–209 (2018).
Salazar de Pablo, G. et al. Probability of transition to psychosis in individuals at clinical high risk: an updated meta-analysis. JAMA Psychiatry 78, 970–978 (2021).
O’Keeffe, D., Kinsella, A., Waddington, J. L. & Clarke, M. 20-Year prospective, sequential follow-up study of heterogeneity in associations of duration of untreated psychosis with symptoms, functioning, and quality of life following first-episode psychosis. Am. J. Psychiatry 179, 288–297 (2022).
Acknowledgements
The authors acknowledge A. Rodolico for providing Supplementary Table 3; F. Milosavljević for his work on Fig. 1 and Fig. 4; D. Kim for his work on Box 3; and C. Mei for her help in producing this manuscript. The authors also thank W. Strube, I. Sommer, S. Gangadin and J.m Steiner for reviewing some sections outside the formal review process.
Author information
Authors and Affiliations
Contributions
Introduction (S.L., J.M.D., S.S. and J.P.); Epidemiology (S.L., J.J.M. and J.P.); Mechanisms/pathophysiology (S.L., O.D.H., S.S., C.T., R.C. and J.P.); Diagnosis, screening and prevention (S.L., P.M. and J.P.); Management (S.L., J.M.D., I.B., J.S.-T., S.S. and J.P.); Quality of life (S.L., S.S. and J.P.); Outlook (S.L., J.M.D., J.J.M., O.D.H., P.M., I.B., J.S.-T., S.S., C.T., R.C. and J.P.); overview of Primer (S.L.).
Corresponding author
Ethics declarations
Competing interests
In the last 3 years, S.L. has received honoraria as an adviser and/or for lectures and/or for educational material from Alkermes, Angelini, Apsen, Eisai, Gedeon Richter, Janssen, Karuna, Kynexis, Lundbeck, Medichem, Medscape, Merck Sharp and Dohme, Mitsubishi, Neurotorium, NovoNordisk, Otsuka, Recordati, Roche, Rovi, Sanofi Aventis, and TEVA. C.T. is on the Scientific Advisory Board for Karuna, owns stock and is an adviser for KyNexis, and she reviews for the National Institute of Mental Health. O.D.H. has received investigator-initiated research funding from and/or participated in advisory/speaker meetings organized by Angellini, Autifony, Biogen, Boehringer-Ingelheim, Eli Lilly, Elysium, Heptares, Global Medical Education, Invicro, Janssen, Karuna, Lundbeck, Merck, Neurocrine, Ontrack/Pangea, Otsuka, Sunovion, Recordati, Roche, Rovi, and Viatris/Mylan. He was previously a part-time employee of Lundbeck A/v. O.D.H. has a patent for the use of dopaminergic imaging. J.P. has a patent on EPO variants and is a member of the SINAPPS2 TSC. All other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks S. Lawrie, D. Malaspina, W. Fleischhacker and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Leucht, S., Siafis, S., McGrath, J.J. et al. Schizophrenia. Nat Rev Dis Primers 11, 83 (2025). https://doi.org/10.1038/s41572-025-00667-6
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41572-025-00667-6