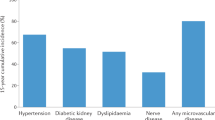
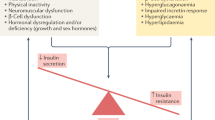
Abstract
Type 2 diabetes mellitus (T2DM) is a chronic, progressive disease driven by a complex interplay of genetic, biological, behavioural and social factors. The epidemiology of T2DM has shifted considerably, largely attributable to increasing obesity rates. Furthermore, T2DM prevalence is increasing in younger people (diagnosis <40 years of age; early-onset T2DM), which is associated with more aggressive disease progression, higher risk factor burden, earlier and more severe complications, and greater lifetime morbidity than later-onset T2DM. T2DM is traditionally associated with a high risk of microvascular and macrovascular complications, although rates of cardiovascular complications have reduced in some high-income countries. Currently, emerging and non-traditional diabetes complications, such as those related to mental health and cognitive function, are being recognized, and people with T2DM increasingly experience multimorbidity and reduced quality of life. Additionally, a growing prevalence of obesity has resulted in high rates of obesity-related complications. Novel therapies and technologies may offer considerable benefit, although socioeconomic disparities may exacerbate barriers to effective prevention and equitable access. The complex nature of T2DM and its comorbidities underscores the urgent need for a person-centred, holistic approach that integrates glucose and weight management with broader attention to comorbidities, 24-h physical behaviours, psychosocial well-being and social determinants of health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$119.00 per year
only $119.00 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Lascar, N. et al. Type 2 diabetes in adolescents and young adults. Lancet Diabetes Endocrinol. 6, 69–80 (2018). This review examines the epidemiology, pathophysiology and clinical challenges of T2DM in adolescents and young adults.
IDF Diabetes Atlas 2025 https://diabetesatlas.org/resources/idf-diabetes-atlas-2025/ (International Diabetes Federation, 2025).
Goldney, J. et al. Burden of vascular risk factors by age, sex, ethnicity and deprivation in young adults with and without newly diagnosed type 2 diabetes. Diabetes Res. Clin. Pract. 220, 112002 (2025).
Barker, M. M. et al. Age at type 2 diabetes diagnosis and cause-specific mortality: observational study of primary care patients in England. Diabetes Care 46, 1965–1972 (2023).
Barker, M. M. et al. Age at diagnosis of type 2 diabetes and cardiovascular risk factor profile: a pooled analysis. World J. Diabetes 13, 260–271 (2022).
Petrak, F., Baumeister, H., Skinner, T. C., Brown, A. & Holt, R. I. G. Depression and diabetes: treatment and health-care delivery. Lancet Diabetes Endocrinol. 3, 472–485 (2015).
Srikanth, V., Sinclair, A. J., Hill-Briggs, F., Moran, C. & Biessels, G. J. Type 2 diabetes and cognitive dysfunction-towards effective management of both comorbidities. Lancet Diabetes Endocrinol. 8, 535–545 (2020).
Harding, J. L., Pavkov, M. E., Magliano, D. J., Shaw, J. E. & Gregg, E. W. Global trends in diabetes complications: a review of current evidence. Diabetologia 62, 3–16 (2019).
Gregg, E. W. et al. Multiple long-term conditions as the next transition in the global diabetes epidemic. Commun. Med. 5, 42 (2025).
Gregg, E. W. et al. The burden of diabetes-associated multiple long-term conditions on years of life spent and lost. Nat. Med. 30, 2830–2837 (2024).
Multiple long-term conditions (multimorbidity): a priority for global health research. The Academy of Medical Sciences https://acmedsci.ac.uk/policy/policy-projects/multimorbidity (2026).
Khunti, K. et al. Diabetes and multiple long-term conditions: a review of our current global health challenge. Diabetes Care 46, 2092–2101 (2023).
Coles, B. et al. Rates and estimated cost of primary care consultations in people diagnosed with type 2 diabetes and comorbidities: a retrospective analysis of 8.9 million consultations. Diabetes Obes. Metab. 23, 1301–1310 (2021).
Sancho-Mestre, C. et al. Pharmaceutical cost and multimorbidity with type 2 diabetes mellitus using electronic health record data. BMC Health Serv. Res. 16, 394 (2016).
Ling, S. et al. Association of type 2 diabetes with cancer: a meta-analysis with bias analysis for unmeasured confounding in 151 cohorts comprising 32 million people. Diabetes Care 43, 2313–2322 (2020).
Lingvay, I., Sumithran, P., Cohen, R. V. & Le Roux, C. W. Obesity management as a primary treatment goal for type 2 diabetes: time to reframe the conversation. Lancet 399, 394–405 (2022).
Anjana, R. M. et al. Contrasting associations between diabetes and cardiovascular mortality rates in low-, middle-, and high-income countries: cohort study data from 143,567 individuals in 21 countries in the PURE study. Diabetes Care 43, 3094–3101 (2020).
Shivashankar, R. et al. Quality of diabetes care in low- and middle-income Asian and Middle Eastern countries (1993-2012): 20-year systematic review. Diabetes Res. Clin. Pract. 107, 203–223 (2015).
Agardh, E., Allebeck, P., Hallqvist, J., Moradi, T. & Sidorchuk, A. Type 2 diabetes incidence and socio-economic position: a systematic review and meta-analysis. Int. J. Epidemiol. 40, 804–818 (2011).
Davies, M. J. et al. Management of hyperglycemia in type 2 diabetes, 2022. a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 45, 2753–2786 (2022). This consensus provides updated global recommendations for management of hyperglycaemia in T2DM from the American Diabetes Association and the European Association for the Study of Diabetes.
Melson, E., Ashraf, U., Papamargaritis, D. & Davies, M. J. What is the pipeline for future medications for obesity? Int. J. Obes. 49, 433–451 (2025). This review summarizes the therapeutic pipeline for future obesity treatments.
Goldney, J. & Davies, M. J. GLP1 agonists: current and future landscape of clinical trials for patients with metabolic dysfunction. Nat. Rev. Gastroenterol. Hepatol. 21, 664–666 (2024). This review outlines the current and future clinical trial landscape for GLP1RAs.
American Diabetes Association Professional Practice Committee. 13. Older adults: standards of care in diabetes — 2025. Diabetes Care 48, S266–S282 (2024).
Laiteerapong, N. & Huang, E. S. in Diabetes in America (eds Cowie, C. C. et al.) Ch. 16 (National Institute of Diabetes and Digestive and Kidney Diseases, 2018).
National Diabetes Statistics Report https://www.cdc.gov/diabetes/php/data-research/index.html (US Centers for Disease Control and Prevention, 2024).
Golden, S. H., Yajnik, C., Phatak, S., Hanson, R. L. & Knowler, W. C. Racial/ethnic differences in the burden of type 2 diabetes over the life course: a focus on the USA and India. Diabetologia 62, 1751–1760 (2019).
Phelps, N. H. et al. Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet 403, 1027–1050 (2024).
Jaacks, L. M. et al. The obesity transition: stages of the global epidemic. Lancet Diabetes Endocrinol. 7, 231–240 (2019).
Zhou, B. et al. Worldwide trends in diabetes prevalence and treatment from 1990 to 2022: a pooled analysis of 1108 population-representative studies with 141 million participants. Lancet 404, 2077–2093 (2024). This study provides a global pooled analysis of 141 million participants, showing long-term trends in diabetes prevalence and treatment.
Luk, A. et al. Early-onset type 2 diabetes: the next major diabetes transition. Lancet 405, 2313–2326 (2025).
Butler, A. M. & Eddington, A. Disparities in youth-onset type 2 diabetes. Endocrinol. Metab. Clin. N. Am. 54, 225–232 (2025).
Perng, W., Conway, R., Mayer-Davis, E. & Dabelea, D. Youth-onset type 2 diabetes: the epidemiology of an awakening epidemic. Diabetes Care 46, 490–499 (2023).
Narayan, K. M. V. et al. Copenhagen Declaration: a transformative vision for global diabetes. Lancet Diabetes Endocrinol. 13, 543–545 (2025). This report sets out the Copenhagen Declaration, a global vision for transformative diabetes prevention and care.
Beyh, Y. S. et al. Copenhagen Declaration: A Blueprint for Diabetes Care and Research in an Inter-connected World http://diabetes.emory.edu/copenhagen-diabetes-declaration/index.html (Global Diabetes Forum, 2024).
Hill-Briggs, F. et al. Social determinants of health and diabetes: a scientific review. Diabetes Care 44, 258–279 (2020).
den Braver, N. R. et al. Built environmental characteristics and diabetes: a systematic review and meta-analysis. BMC Med. 16, 12 (2018).
Chan, J. C. N. et al. The lancet commission on diabetes: using data to transform diabetes care and patient lives. Lancet 396, 2019–2082 (2020).
O’Hearn, M. et al. Incident type 2 diabetes attributable to suboptimal diet in 184 countries. Nat. Med. 29, 982–995 (2023).
Goff, L. M. Ethnicity and type 2 diabetes in the UK. Diabet. Med. 36, 927–938 (2019).
Diabetes. World Health Organization https://www.who.int/news-room/fact-sheets/detail/diabetes (2024).
Institute for Health Metrics and Evaluation. Global Burden of Disease Study 2019. Global Health Data Exchange https://vizhub.healthdata.org/gbd-results (2020).
Balooch Hasankhani, M., Mirzaei, H. & Karamoozian, A. Global trend analysis of diabetes mellitus incidence, mortality, and mortality-to-incidence ratio from 1990 to 2019. Sci. Rep. 13, 21908 (2023).
Rawshani, A. et al. Association between socioeconomic status and mortality, cardiovascular disease, and cancer in patients with type 2 diabetes. JAMA Intern. Med. 176, 1146–1154 (2016).
Gregg, E. W. et al. Trends in lifetime risk and years of life lost due to diabetes in the USA, 1985-2011: a model. study. Lancet Diabetes Endocrinol. 2, 867–874 (2014).
Kaptoge, S. et al. Life expectancy associated with different ages at diagnosis of type 2 diabetes in high-income countries: 23 million person-years of observation. Lancet Diabetes Endocrinol. 11, 731–742 (2023).
Chen, L. et al. A systematic review of trends in all-cause mortality among people with diabetes. Diabetologia 63, 1718–1735 (2020).
Gregg, E. W. et al. Trends in cause-specific mortality among adults with and without diagnosed diabetes in the USA: an epidemiological analysis of linked national survey and vital statistics data. Lancet 391, 2430–2440 (2018).
Holman, N. et al. Trends in mortality and hospitalisations for cardiovascular, kidney and liver disease in people with type 2 diabetes in England, 2009–2019. Diabetes Obes. Metab. 27, 6341–6352 (2025).
Huang, G. et al. Decomposing the differences in healthy life expectancy between migrants and natives: the ‘healthy migrant effect’ and its age variations in Australia. J. Pop. Res. 41, 3 (2023).
Anand, S. S. et al. Reducing inequalities in cardiovascular disease: focus on marginalized populations considering ethnicity and race. Lancet Reg. Health Eur. 56, 101371 (2025).
Harding, J. L. et al. Mortality trends among people with type 1 and type 2 diabetes in Australia: 1997–2010. Diabetes Care 37, 2579–2586 (2014).
Pearson-Stuttard, J. et al. Trends in predominant causes of death in individuals with and without diabetes in England from 2001 to 2018: an epidemiological analysis of linked primary care records. Lancet Diabetes Endocrinol. 9, 165–173 (2021).
Bjornsdottir, H. H. et al. A national observation study of cancer incidence and mortality risks in type 2 diabetes compared to the background population over time. Sci. Rep. 10, 17376 (2020).
European Association for the Study of Obesity. Study of UK biobank reveals link between new-onset type 2 diabetes and some but not all obesity-related cancers. EurekAlert! https://www.eurekalert.org/news-releases/1077674 (22 March 2025).
Renehan, A. G., Tipping, O. & Wang, M. Diabetes and cancer: doubts of a causal link. Int. J. Cancer 154, 1875–1876 (2024).
Zaccardi, F. et al. Trajectories of type 2 diabetes and cancer in 330 000 individuals with prediabetes: 20-year observational study in England. Lancet Diabetes Endocrinol. 14, 41–49 (2026).
Zhang, A. M. Y., Wellberg, E. A., Kopp, J. L. & Johnson, J. D. Hyperinsulinemia in obesity, inflammation, and cancer. Diabetes Metab. J. 45, 285–311 (2021).
Lincoff, A. M. et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N. Engl. J. Med. 389, 2221–2232 (2023).
Butler, A. E. et al. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 52, 102–110 (2003).
Moon, J. H., Choe, H. J. & Lim, S. Pancreatic beta-cell mass and function and therapeutic implications of using antidiabetic medications in type 2 diabetes. J. Diabetes Investig. 15, 669–683 (2024).
Ahmad, E., Lim, S., Lamptey, R., Webb, D. R. & Davies, M. J. Type 2 diabetes. Lancet 400, 1803–1820 (2022).
Hanley, S. Pancreatic β-cell mass as a pharmacologic target in diabetes. McGill J. Med. 12, 51 (2009).
Donath, M. Y. & Halban, P. A. Decreased beta-cell mass in diabetes: significance, mechanisms and therapeutic implications. Diabetologia 47, 581–589 (2004).
Fiaschi-Taesch, N. M. et al. Induction of human beta-cell proliferation and engraftment using a single G1/S regulatory molecule, CDK6. Diabetes 59, 1926–1936 (2010).
Ackeifi, C. et al. GLP-1 receptor agonists synergize with DYRK1A inhibitors to potentiate functional human β cell regeneration. Sci. Transl. Med. 12, eaaw9996 (2020).
Wang, P. et al. A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication. Nat. Med. 21, 383–388 (2015).
Wang, P. et al. Combined Inhibition of DYRK1A, SMAD, and trithorax pathways synergizes to induce robust replication in adult human beta cells. Cell Metab. 29, 638–652.e5 (2019).
Talchai, C., Xuan, S., Lin, H. V., Sussel, L. & Accili, D. Pancreatic β cell dedifferentiation as a mechanism of diabetic β cell failure. Cell 150, 1223–1234 (2012).
Zhou, Y. et al. METRNL represses beta-to-alpha cell trans-differentiation to maintain beta cell function under diabetic metabolic stress in mice. Diabetologia 68, 1769–1788 (2025).
Nguyen, H. T. et al. Palmitate impairs autophagic degradation via oxidative stress/perilysosomal Ca2+ overload/mTORC1 activation pathway in pancreatic β cells. JCI Insight 10, e192827 (2025).
Alanazi, Y. A. et al. Role of autophagy in type 2 diabetes mellitus: the metabolic clash. J. Cell Mol. Med. 28, e70240 (2024).
Uchida, M. et al. Multiorgan fibrosis and risk of type 2 diabetes: genetic and observational evidence highlighting a causal role of pancreatic fibrosis. Diabetes https://doi.org/10.2337/db25-0629 (2026).
Cheng, C.-W. et al. Fasting-mimicking diet promotes ngn3-driven β-cell regeneration to reverse. Diabetes Cell 168, 775–788.e12 (2017).
Taylor, R. Calorie restriction and reversal of type 2 diabetes. Expert Rev. Endocrinol. Metab. 11, 521–528 (2016).
Accili, D., Deng, Z. & Liu, Q. Insulin resistance in type 2 diabetes mellitus. Nat. Rev. Endocrinol. 21, 413–426 (2025). This review synthesizes current understanding of insulin resistance mechanisms in T2DM.
Roden, M. & Shulman, G. I. The integrative biology of type 2 diabetes. Nature 576, 51–60 (2019).
Rabiee, A., Hossain, M. A. & Poojari, A. Adipose tissue insulin resistance: a key driver of metabolic syndrome pathogenesis. Biomedicines 13, 2376 (2025).
Saltiel, A. R. & Olefsky, J. M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Invest. 127, 1–4 (2017).
Neeland, I. J. et al. Metabolic syndrome. Nat. Rev. Dis. Primers 10, 77 (2024).
Chavakis, T., Alexaki, V. I. & Ferrante, A. W. Macrophage function in adipose tissue homeostasis and metabolic inflammation. Nat. Immunol. 24, 757–766 (2023).
Tchernof, A. & Després, J.-P. Pathophysiology of human visceral obesity: an update. Physiol. Rev. 93, 359–404 (2013).
Sylow, L., Tokarz, V. L., Richter, E. A. & Klip, A. The many actions of insulin in skeletal muscle, the paramount tissue determining glycemia. Cell Metab. 33, 758–780 (2021).
Kim, H.-K. et al. Association of visceral fat obesity, sarcopenia, and myosteatosis with non-alcoholic fatty liver disease without obesity. Clin. Mol. Hepatol. 29, 987–1001 (2023).
Callahan, H. S. et al. Postprandial suppression of plasma ghrelin level is proportional to ingested caloric load but does not predict intermeal interval in humans. J. Clin. Endocrinol. Metab. 89, 1319–1324 (2004).
Huang, Y. et al. Low thigh muscle strength in relation to myosteatosis in patients with type 2 diabetes mellitus. Sci. Rep. 13, 1957 (2023).
Lu, X. et al. Thigh muscle fat fraction is independently associated with impaired glucose metabolism in individuals with obesity. Endocr. Connect. 12, e230248 (2023).
Choe, H. J., Chang, W., Blüher, M., Heymsfield, S. B. & Lim, S. Independent association of thigh muscle fat density with vascular events in Korean adults. Cardiovasc. Diabetol. 23, 44 (2024).
Savage, D. B. et al. Accumulation of saturated intramyocellular lipid is associated with insulin resistance. J. Lipid Res. 60, 1323–1332 (2019).
Szendroedi, J. et al. Role of diacylglycerol activation of PKCθ in lipid-induced muscle insulin resistance in humans. Proc. Natl Acad. Sci. USA 111, 9597–9602 (2014).
Goodpaster, B. H., Kelley, D. E., Wing, R. R., Meier, A. & Thaete, F. L. Effects of weight loss on regional fat distribution and insulin sensitivity in obesity. Diabetes 48, 839–847 (1999).
Manini, T. M. et al. Reduced physical activity increases intermuscular adipose tissue in healthy young adults. Am. J. Clin. Nutr. 85, 377–384 (2007).
Mehta, M., Shah, J. & Joshi, U. Understanding insulin resistance in NAFLD: a systematic review and meta-analysis focused on HOMA-IR in South Asians. Cureus 16, e70768 (2024).
Shulman, G. I. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N. Engl. J. Med. 371, 1131–1141 (2014).
James, D. E., Stöckli, J. & Birnbaum, M. J. The aetiology and molecular landscape of insulin resistance. Nat. Rev. Mol. Cell Biol. 22, 751–771 (2021).
Samuel, V. T. & Shulman, G. I. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J. Clin. Invest. 126, 12–22 (2016).
Koliaki, C. & Roden, M. Alterations of mitochondrial function and insulin sensitivity in human obesity and diabetes mellitus. Annu. Rev. Nutr. 36, 337–367 (2016).
Xourafa, G., Korbmacher, M. & Roden, M. Inter-organ crosstalk during development and progression of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 20, 27–49 (2024).
Kubota, N. et al. Differential hepatic distribution of insulin receptor substrates causes selective insulin resistance in diabetes and obesity. Nat. Commun. 7, 12977 (2016).
Lim, S., Taskinen, M.-R. & Borén, J. Crosstalk between nonalcoholic fatty liver disease and cardiometabolic syndrome. Obes. Rev. 20, 599–611 (2019).
Rinella, M. E. et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 78, 1966–1986 (2023).
Younossi, Z. M. et al. Global consensus recommendations for metabolic dysfunction-associated steatotic liver disease and steatohepatitis. Gastroenterology 169, 1017–1032.e2 (2025).
Huang, D. Q. et al. Metabolic dysfunction-associated steatotic liver disease in adults. Nat. Rev. Dis. Primers 11, 14 (2025).
Holst, J. J., Vilsbøll, T. & Deacon, C. F. The incretin system and its role in type 2 diabetes mellitus. Mol. Cell. Endocrinol. 297, 127–136 (2009).
Młynarska, E. et al. Exploring the significance of gut microbiota in diabetes pathogenesis and management — a narrative review. Nutrients 16, 1938 (2024).
Mueller, N. T. et al. Metformin affects gut microbiome composition and function and circulating short-chain fatty acids: a randomized trial. Diabetes Care 44, 1462–1471 (2021).
Lassenius, M. I. et al. Bacterial endotoxin activity in human serum is associated with dyslipidemia, insulin resistance, obesity, and chronic inflammation. Diabetes Care 34, 1809–1815 (2011).
Liu, S., Tao, Z., Qiao, M. & Shi, L. The functions of major gut microbiota in obesity and type 2 diabetes. Metabolites 15, 167 (2025).
DeFronzo, R. A. et al. Inadequately controlled type 2 diabetes and hypercortisolism: improved glycemia with mifepristone treatment. Diabetes Care https://doi.org/10.2337/dc25-1055 (2025).
Buse, J. B. et al. Prevalence of hypercortisolism in difficult-to-control type 2 diabetes. Diabetes Care https://doi.org/10.2337/dc24-2841 (2025).
Suzuki, K. et al. Genetic drivers of heterogeneity in type 2 diabetes pathophysiology. Nature 627, 347–357 (2024). This study defines genetic drivers of heterogeneity in T2DM pathophysiology using large-scale genomic analyses.
Bonnefond, A., Florez, J. C., Loos, R. J. F. & Froguel, P. Dissection of type 2 diabetes: a genetic perspective. Lancet Diabetes Endocrinol. 13, 149–164 (2025).
Bonnefond, A. et al. Monogenic diabetes. Nat. Rev. Dis. Primers 9, 12 (2023).
Hinault, C., Caroli-Bosc, P., Bost, F. & Chevalier, N. Critical overview on endocrine disruptors in diabetes mellitus. Int. J. Mol. Sci. 24, 4537 (2023).
Khaledi, M., Haghighatdoost, F., Feizi, A. & Aminorroaya, A. The prevalence of comorbid depression in patients with type 2 diabetes: an updated systematic review and meta-analysis on huge number of observational studies. Acta Diabetol. 56, 631–650 (2019). This meta-analysis shows a high prevalence of comorbid depression in people with T2DM.
Perrin, N. E., Davies, M. J., Robertson, N., Snoek, F. J. & Khunti, K. The prevalence of diabetes-specific emotional distress in people with type 2 diabetes: a systematic review and meta-analysis. Diabet. Med. 34, 1508–1520 (2017).
Brownlee, M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54, 1615–1625 (2005).
Madonna, R., Balistreri, C. R., Geng, Y.-J. & De Caterina, R. Diabetic microangiopathy: pathogenetic insights and novel therapeutic approaches. Vasc. Pharmacol. 90, 1–7 (2017).
Pushparani, D. S., Varalakshmi, J., Roobini, K., Hamshapriya, P. & Livitha, A. Diabetic retinopathy — a review. Curr. Diabetes Rev. 21, 43–55 (2025).
Tang, S. C. W. & Yiu, W. H. Innate immunity in diabetic kidney disease. Nat. Rev. Nephrol. 16, 206–222 (2020).
Aziz, N. et al. New horizons in diabetic neuropathies: an updated review on their pathology, diagnosis, mechanism, screening techniques, pharmacological, and future approaches. Curr. Diabetes Rev. 20, e201023222416 (2024).
Islam, K. et al. Diabetes mellitus and associated vascular disease: pathogenesis, complications, and evolving treatments. Adv. Ther. 42, 2659–2678 (2025).
Attiq, A., Afzal, S., Ahmad, W. & Kandeel, M. Hegemony of inflammation in atherosclerosis and coronary artery disease. Eur. J. Pharmacol. 966, 176338 (2024).
Agarwala, A. et al. Biomarkers and degree of atherosclerosis are independently associated with incident atherosclerotic cardiovascular disease in a primary prevention cohort: the ARIC study. Atherosclerosis 253, 156–163 (2016).
Pasut, A., Lama, E., Van Craenenbroeck, A. H., Kroon, J. & Carmeliet, P. Endothelial cell metabolism in cardiovascular physiology and disease. Nat. Rev. Cardiol. 22, 923–943 (2025).
Umpierrez, G. E. et al. Hyperglycemic crises in adults with diabetes: a consensus report. Diabetes Care 47, 1257–1275 (2024).
Sattar, N., Presslie, C., Rutter, M. K. & McGuire, D. K. Cardiovascular and kidney risks in individuals with type 2 diabetes: contemporary understanding with greater emphasis on excess adiposity. Diabetes Care 47, 531–543 (2024).
Luo, J. et al. Features, functions, and associated diseases of visceral and ectopic fat: a comprehensive review. Obesity 33, 825–838 (2025).
Sewaybricker, L. E., Huang, A., Chandrasekaran, S., Melhorn, S. J. & Schur, E. A. The significance of hypothalamic inflammation and gliosis for the pathogenesis of obesity in humans. Endocr. Rev. 44, 281–296 (2023).
Choe, H. J., Almas, T., Neeland, I. J., Lim, S. & Després, J.-P. Obesity phenotypes and atherogenic dyslipidemias. Eur. J. Clin. Invest. https://doi.org/10.1111/eci.70151 (2025).
Levelt, E. et al. Ectopic and visceral fat deposition in lean and obese patients with type 2 diabetes. J. Am. Coll. Cardiol. 68, 53–63 (2016).
Taylor, R., Al-Mrabeh, A. & Sattar, N. Understanding the mechanisms of reversal of type 2 diabetes. Lancet Diabetes Endocrinol. 7, 726–736 (2019). This review discusses biological mechanisms underlying reversal of T2DM following weight loss.
Lim, S. & Meigs, J. B. Ectopic fat and cardiometabolic and vascular risk. Int. J. Cardiol. 169, 166–176 (2013).
Piché, M.-E., Tchernof, A. & Després, J.-P. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ. Res. 126, 1477–1500 (2020).
Petersen, M. C. & Shulman, G. I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 98, 2133–2223 (2018).
Chaurasia, B. & Summers, S. A. Ceramides — lipotoxic inducers of metabolic disorders. Trends Endocrinol. Metab. 26, 538–550 (2015).
Blagov, A. et al. Mitochondrial dysfunction as a factor of energy metabolism disorders in type 2 diabetes mellitus. Front. Biosci. 16, 5 (2024).
Pinti, M. V. et al. Mitochondrial dysfunction in type 2 diabetes mellitus: an organ-based analysis. Am. J. Physiol. Endocrinol. Metab. 316, E268–E285 (2019).
Rovira-Llopis, S. et al. Mitochondrial dynamics in type 2 diabetes: pathophysiological implications. Redox Biol. 11, 637–645 (2017).
Kuchay, M. S., Choudhary, N. S. & Ramos-Molina, B. Pathophysiological underpinnings of metabolic dysfunction-associated steatotic liver disease. Am. J. Physiol. Cell Physiol. 328, C1637–C1666 (2025).
Steinberg, G. R., Valvano, C. M., De Nardo, W. & Watt, M. J. Integrative metabolism in MASLD and MASH: pathophysiology and emerging mechanisms. J. Hepatol. 83, 584–595 (2025).
Larsson, S. C., Bäck, M., Rees, J. M. B., Mason, A. M. & Burgess, S. Body mass index and body composition in relation to 14 cardiovascular conditions in UK Biobank: a Mendelian randomization study. Eur. Heart J. 41, 221–226 (2020).
Sjöström, L. et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 311, 2297–2304 (2014).
Garvey, W. T. Long-term health benefits of intensive lifestyle intervention in the Look AHEAD study. Obesity 29, 1242–1243 (2021). This study highlights long-term health benefits of intensive lifestyle intervention in people with T2DM.
Wing, R. R.; Look AHEAD Research Group. Does lifestyle intervention improve health of adults with overweight/obesity and type 2 diabetes? Findings from the Look AHEAD randomized trial. Obesity 29, 1246–1258 (2021).
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844 (2016).
Bergman, M. et al. International Diabetes Federation position statement on the 1-hour post-load plasma glucose for the diagnosis of intermediate hyperglycaemia and type 2 diabetes. Diabetes Res. Clin. Pract. 209, 111589 (2024).
Meijnikman, A. S. et al. Not performing an OGTT results in significant underdiagnosis of (pre)diabetes in a high risk adult caucasian population. Int. J. Obes. 41, 1615–1620 (2017).
Holt, R. I. G. et al. The management of type 1 diabetes in adults. a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 44, 2589–2625 (2021).
Wagner, R. et al. Adult-onset type 1 diabetes: early detection, differential diagnosis, and emerging disease-modifying therapies. Diabetes Res. Clin. Pract. 231, 113047 (2025).
Olesen, S. S., Toledo, F. G. S. & Hart, P. A. The spectrum of diabetes in acute and chronic pancreatitis. Curr. Opin. Gastroenterol. 38, 509–515 (2022).
Fitipaldi, H. & Franks, P. W. Ethnic, gender and other sociodemographic biases in genome-wide association studies for the most burdensome non-communicable diseases: 2005-2022. Hum. Mol. Genet. 32, 520–532 (2023).
Smith, K. et al. Multi-ancestry polygenic mechanisms of type 2 diabetes. Nat. Med. 30, 1065–1074 (2024).
Udler, M. S. et al. Type 2 diabetes genetic loci informed by multi-trait associations point to disease mechanisms and subtypes: a soft clustering analysis. PLoS Med. 15, e1002654 (2018).
Dias, J.-A. et al. Evaluating multi-ancestry genome-wide association methods: statistical power, population structure, and practical implications. Am. J. Hum. Genet. 112, 2493–2508 (2025).
Redondo, M. J. & Balasubramanyam, A. Toward an improved classification of type 2 diabetes: lessons from research into the heterogeneity of a complex disease. J. Clin. Endocrinol. Metab. 106, e4822–e4833 (2021).
Ahlqvist, E. et al. Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol. 6, 361–369 (2018). This study identifies novel subgroups of adult-onset diabetes through cluster analysis and links them to clinical outcomes.
Ahlqvist, E., Prasad, R. B. & Groop, L. Subtypes of type 2 diabetes determined from clinical parameters. Diabetes 69, 2086–2093 (2020). This study demonstrates the potential clinical utility of subtyping T2DM using routine clinical parameters.
Anjana, R. M. et al. Novel subgroups of type 2 diabetes and their association with microvascular outcomes in an Asian Indian population: a data-driven cluster analysis: the INSPIRED study. BMJ Open Diabetes Res. Care 8, e001506 (2020).
Slieker, R. C. et al. Replication and cross-validation of type 2 diabetes subtypes based on clinical variables: an IMI-RHAPSODY study. Diabetologia 64, 1982–1989 (2021).
Zaharia, O. P. et al. Risk of diabetes-associated diseases in subgroups of patients with recent-onset diabetes: a 5-year follow-up study. Lancet Diabetes Endocrinol. 7, 684–694 (2019).
Prasad, R. B. et al. Subgroups of patients with young-onset type 2 diabetes in India reveal insulin deficiency as a major driver. Diabetologia 65, 65–78 (2022).
Schön, M. et al. Analysis of type 2 diabetes heterogeneity with a tree-like representation: insights from the prospective German diabetes study and the LURIC cohort. Lancet Diabetes Endocrinol. 12, 119–131 (2024).
Dennis, J. M., Shields, B. M., Henley, W. E., Jones, A. G. & Hattersley, A. T. Disease progression and treatment response in data-driven subgroups of type 2 diabetes compared with models based on simple clinical features: an analysis using clinical trial data. Lancet Diabetes Endocrinol. 7, 442–451 (2019).
Gray, L. J. et al. The Leicester Risk Assessment score for detecting undiagnosed type 2 diabetes and impaired glucose regulation for use in a multiethnic UK setting. Diabet. Med. 27, 887–895 (2010).
Barber, S. R., Dhalwani, N. N., Davies, M. J., Khunti, K. & Gray, L. J. External national validation of the Leicester self-assessment score for type 2 diabetes using data from the English Longitudinal Study of Ageing. Diabet. Med. 34, 1575–1583 (2017). This study validates the Leicester Self-Assessment risk score for T2DM in a national cohort.
Lindström, J. et al. The finnish diabetes prevention study (DPS): lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care 26, 3230–3236 (2003). This trial shows that lifestyle intervention prevents progression to T2DM in individuals at high risk.
Chadha, C. et al. Reproducibility of a prediabetes classification in a contemporary population. Metab. Open 6, 100031 (2020).
Wang, J. et al. Asians have lower body mass index (BMI) but higher percent body fat than do whites: comparisons of anthropometric measurements. Am. J. Clin. Nutr. 60, 23–28 (1994).
Aggarwal, R. et al. Diabetes screening by race and ethnicity in the United States: equivalent body mass index and age thresholds. Ann. Intern. Med. 175, 765–773 (2022).
Tillin, T. et al. Ethnicity-specific obesity cut-points in the development of type 2 diabetes — a prospective study including three ethnic groups in the United Kingdom. Diabet. Med. 32, 226–234 (2015).
Mayne, S. L., Auchincloss, A. H. & Michael, Y. L. Impact of policy and built environment changes on obesity-related outcomes: a systematic review of naturally occurring experiments. Obes. Rev. 16, 362–375 (2015).
Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the diabetes prevention program outcomes study. Lancet Diabetes Endocrinol. 3, 866–875 (2015). This trial confirms durable benefits of lifestyle and metformin on diabetes incidence and microvascular outcomes over 15 years.
Gong, Q. et al. Long-term effects of a randomised trial of a 6-year lifestyle intervention in impaired glucose tolerance on diabetes-related microvascular complications: the China Da Qing Diabetes Prevention Outcome Study. Diabetologia 54, 300–307 (2011).
Gong, Q. et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the da qing diabetes prevention outcome study. Lancet Diabetes Endocrinol. 7, 452–461 (2019). This trial demonstrates sustained reductions in diabetes incidence and mortality after 30 years of lifestyle intervention.
Knowler, W. C. et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 346, 393–403 (2002).
Hamman, R. F. et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 29, 2102–2107 (2006).
Henson, J. et al. Waking up to the importance of sleep in type 2 diabetes management: a narrative review. Diabetes Care 47, 331–343 (2024). This paper reviews evidence showing that poor sleep quality and duration significantly affect blood glucose control and overall management of T2DM, highlighting sleep as a critical but often overlooked component of diabetes care.
Taheri, S. Sleep and cardiometabolic health-not so strange bedfellows. Lancet Diabetes Endocrinol. 11, 532–534 (2023).
Jastreboff, A. M. et al. Tirzepatide for obesity treatment and diabetes prevention. N. Engl. J. Med. 392, 958–971 (2025).
Riddle, M. C. et al. Consensus report: definition and interpretation of remission in type 2 diabetes. Diabetes Care 44, 2438–2444 (2021).
Khunti, K., Papamargaritis, D., Aroda, V. R., Anjana, R. M. & Kashyap, S. R. Re-evaluating the concept of remission in type 2 diabetes: a call for patient-centric approaches. Lancet Diabetes Endocrinol. 13, 615–634 (2025).
Rubino, F. et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol. 13, 221–262 (2025).
Kanbour, S., Ageeb, R. A., Malik, R. A. & Abu-Raddad, L. J. Impact of bodyweight loss on type 2 diabetes remission: a systematic review and meta-regression analysis of randomised controlled trials. Lancet Diabetes Endocrinol. 13, 294–306 (2025).
Taheri, S. Type 2 diabetes remission: a new mission in diabetes care. Diabetes Care 47, 47–49 (2023).
Khunti, K. et al. Glycaemic control is still central in the hierarchy of priorities in type 2 diabetes management. Diabetologia 68, 17–28 (2025).
Powers, M. A. et al. Diabetes self-management education and support in adults with type 2 diabetes: a consensus report of the American Diabetes Association, the Association of Diabetes Care & Education Specialists, the Academy of Nutrition and Dietetics, the American Academy of Family Physicians, the American Academy of PAs, the American Association of Nurse Practitioners, and the American Pharmacists Association. Diabetes Care 43, 1636–1649 (2020).
American Diabetes Association Professional Practice Committee. 5. Facilitating positive health behaviors and well-being to improve health outcomes: standards of care in diabetes — 2025. Diabetes Care 48, S86–S127 (2024).
Zhao, F.-F., Suhonen, R., Koskinen, S. & Leino-Kilpi, H. Theory-based self-management educational interventions on patients with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. J. Adv. Nurs. 73, 812–833 (2017).
Bekele, B. B. et al. Effect of diabetes self-management education (DSME) on glycated hemoglobin (HbA1c) level among patients with T2DM: systematic review and meta-analysis of randomized controlled trials. Diabetes Metab. Syndr. 15, 177–185 (2021).
Davies, M. J. et al. Effectiveness of the diabetes education and self management for ongoing and newly diagnosed (DESMOND) programme for people with newly diagnosed type 2 diabetes: cluster randomised controlled trial. BMJ https://doi.org/10.1136/bmj.39474.922025.BE (2008).
Chatterjee, S. et al. Diabetes structured self-management education programmes: a narrative review and current innovations. Lancet Diabetes Endocrinol. 6, 130–142 (2018).
Philis-Tsimikas, A., Fortmann, A., Lleva-Ocana, L., Walker, C. & Gallo, L. C. Peer-led diabetes education programs in high-risk Mexican Americans improve glycemic control compared with standard approaches: a project dulce promotora randomized trial. Diabetes Care 34, 1926–1931 (2011).
Hadjiconstantinou, M. et al. Do web-based interventions improve well-being in type 2 diabetes? A systematic review and meta-analysis. J. Med. Internet Res. 18, e270 (2016).
Barker, M. M. et al. User retention and engagement in the digital-based diabetes education and self-management for ongoing and newly diagnosed (myDESMOND) program: descriptive longitudinal study. JMIR Diabetes 8, e44943 (2023).
Moschonis, G. et al. Effectiveness, reach, uptake, and feasibility of digital health interventions for adults with type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials. Lancet Digit. Health 5, e125–e143 (2023).
Philis-Tsimikas, A. et al. Dulce digital-me: results of a randomized comparative trial of static versus adaptive digital interventions for Latine adults with diabetes. Ann. Behav. Med. 59, kaae077 (2025).
Gershkowitz, B. D., Hillert, C. J. & Crotty, B. H. Digital coaching strategies to facilitate behavioral change in type 2 diabetes: a systematic review. J. Clin. Endocrinol. Metab. 106, e1513–e1520 (2021).
Shippee, N. D., Shah, N. D., May, C. R., Mair, F. S. & Montori, V. M. Cumulative complexity: a functional, patient-centered model of patient complexity can improve research and practice. J. Clin. Epidemiol. 65, 1041–1051 (2012).
Boehmer, K. R. et al. Patient capacity and constraints in the experience of chronic disease: a qualitative systematic review and thematic synthesis. BMC Fam. Pract. 17, 127 (2016).
May, C. R. et al. Rethinking the patient: using burden of treatment theory to understand the changing dynamics of illness. BMC Health Serv. Res. 14, 281 (2014).
Sargeant, J. A. et al. A review of the effects of glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors on lean body mass in humans. Endocrinol. Metab. 34, 247–262 (2019).
Ahmad, E., Sargeant, J. A., Yates, T., Webb, D. R. & Davies, M. J. Type 2 diabetes and impaired physical function: a growing problem. Diabetology 3, 30–45 (2022).
Boulé, N. G., Haddad, E., Kenny, G. P., Wells, G. A. & Sigal, R. J. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA 286, 1218–1227 (2001).
Rowlands, A. et al. Wrist-worn accelerometers: recommending ~1.0 mg as the minimum clinically important difference (MCID) in daily average acceleration for inactive adults. Br. J. Sports Med. 55, 814–815 (2021).
Yates, T. et al. Association between change in daily ambulatory activity and cardiovascular events in people with impaired glucose tolerance (NAVIGATOR trial): a cohort analysis. Lancet 383, 1059–1066 (2014).
Homer, A. R. et al. Frequency of interruptions to sitting time: benefits for postprandial metabolism in type 2 diabetes. Diabetes Care 44, 1254–1263 (2021).
Schwingshackl, L., Chaimani, A., Hoffmann, G., Schwedhelm, C. & Boeing, H. A network meta-analysis on the comparative efficacy of different dietary approaches on glycaemic control in patients with type 2 diabetes mellitus. Eur. J. Epidemiol. 33, 157–170 (2018).
Lv, M. et al. Effects of vegetarian or vegan diets on glycemic and cardiometabolic health in type 2 diabetes: a systematic review and meta-analysis. Nutr. Rev. 83, 1438–1449 (2025).
Ajala, O., English, P. & Pinkney, J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am. J. Clin. Nutr. 97, 505–516 (2013).
Churuangsuk, C. et al. Diets for weight management in adults with type 2 diabetes: an umbrella review of published meta-analyses and systematic review of trials of diets for diabetes remission. Diabetologia 65, 14–36 (2022).
Liu, G. et al. Adherence to a healthy lifestyle in association with microvascular complications among adults with type 2 diabetes. JAMA Netw. Open 6, e2252239 (2023).
Gidudu, M. et al. Factors affecting lifestyle modification among adults with type II diabetes mellitus attending care at Mbale regional referral hospital in Mbale City, Eastern Uganda: a mixed methods study. J. Public Health Res. 14, 22799036251395268 (2025).
Davies, M. J. et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE Diabetes randomized clinical trial. JAMA 314, 687–699 (2015).
Davies, M. et al. Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet 397, 971–984 (2021).
Garvey, W. T. et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 402, 613–626 (2023). This trial shows that tirzepatide significantly improves weight loss and glycaemic outcomes in T2DM with obesity.
Lee, M. M. Y. et al. Cardiovascular and kidney outcomes and mortality with long-acting injectable and oral glucagon-like peptide 1 receptor agonists in individuals with type 2 diabetes: a systematic review and meta-analysis of randomized trials. Diabetes Care 48, 846–859 (2025).
Hammad, B. F. et al. Exploring the multifaceted roles of GLP-1 receptor agonists; a comprehensive review. Front. Clin. Diabetes Healthc. 6, 1590530 (2025).
Sanyal, A. J. et al. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N. Engl. J. Med. 392, 2089–2099 (2025).
Srinivasan, N. M., Farokhnia, M., Farinelli, L. A., Ferrulli, A. & Leggio, L. GLP-1 therapeutics and their emerging role in alcohol and substance use disorders: an endocrinology primer. J. Endocr. Soc. 9, bvaf141 (2025).
Wharton, S. et al. Managing the gastrointestinal side effects of GLP-1 receptor agonists in obesity: recommendations for clinical practice. Postgrad. Med. 134, 14–19 (2022).
Azizi, Z., Rodriguez, F. & Assimes, T. L. Digital footprints of obesity treatment: GLP-1 receptor agonists and the health equity divide. Circulation 150, 171–173 (2024).
Waldrop, S. W., Johnson, V. R. & Stanford, F. C. Inequalities in the provision of GLP-1 receptor agonists for the treatment of obesity. Nat. Med. 30, 22–25 (2024).
Hussein, H. et al. Efficacy and tolerability of sodium-glucose co-transporter-2 inhibitors and glucagon-like peptide-1 receptor agonists: a systematic review and network meta-analysis. Diabetes Obes. Metab. 22, 1035–1046 (2020).
Kleinaki, Z., Kapnisi, S., Theodorelou-Charitou, S.-A., Nikas, I. P. & Paschou, S. A. Type 2 diabetes mellitus management in patients with chronic kidney disease: an update. Hormones 19, 467–476 (2020).
Patel, S. M. et al. Sodium-glucose cotransporter-2 inhibitors and major adverse cardiovascular outcomes: a SMART-C collaborative meta-analysis. Circulation 149, 1789–1801 (2024).
Banerjee, M., Pal, R., Nair, K. & Mukhopadhyay, S. SGLT2 inhibitors and cardiovascular outcomes in heart failure with mildly reduced and preserved ejection fraction: a systematic review and meta-analysis. Indian Heart J. 75, 122–127 (2023).
Zannad, F. et al. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-reduced and DAPA-HF trials. Lancet 396, 819–829 (2020).
Mavrakanas, T. A., Tsoukas, M. A., Brophy, J. M., Sharma, A. & Gariani, K. SGLT-2 inhibitors improve cardiovascular and renal outcomes in patients with CKD: a systematic review and meta-analysis. Sci. Rep. 13, 15922 (2023).
Ferrannini, E. Sodium-glucose co-transporters and their inhibition: clinical physiology. Cell Metab. 26, 27–38 (2017).
Santos-Gallego, C. G. et al. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J. Am. Coll. Cardiol. 73, 1931–1944 (2019).
Vallon, V. & Verma, S. Effects of SGLT2 inhibitors on kidney and cardiovascular function. Annu. Rev. Physiol. 83, 503–528 (2021).
Roberts, D. J. & Miyamoto, S. Hexokinase II integrates energy metabolism and cellular protection: akting on mitochondria and TORCing to autophagy. Cell Death Differ. 22, 248–257 (2015).
Kanbay, M. et al. Effect of sodium-glucose cotransporter 2 inhibitors on hemoglobin and hematocrit levels in type 2 diabetes: a systematic review and meta-analysis. Int. Urol. Nephrol. 54, 827–841 (2022).
Han, J. H. et al. The beneficial effects of empagliflozin, an SGLT2 inhibitor, on atherosclerosis in ApoE -/- mice fed a western diet. Diabetologia 60, 364–376 (2017).
Dihoum, A., Brown, A. J., McCrimmon, R. J., Lang, C. C. & Mordi, I. R. Dapagliflozin, inflammation and left ventricular remodelling in patients with type 2 diabetes and left ventricular hypertrophy. BMC Cardiovasc. Disord. 24, 356 (2024).
Forrester, E. A. et al. Crucial role for sensory nerves and Na/H exchanger inhibition in dapagliflozin- and empagliflozin-induced arterial relaxation. Cardiovasc. Res. 120, 1811–1824 (2024).
Kraus, B. J. et al. Characterization and implications of the initial estimated glomerular filtration rate ‘dip’ upon sodium-glucose cotransporter-2 inhibition with empagliflozin in the EMPA-REG OUTCOME trial. Kidney Int. 99, 750–762 (2021).
Jongs, N. et al. Correlates and consequences of an acute change in eGFR in response to the SGLT2 inhibitor dapagliflozin in patients with CKD. J. Am. Soc. Nephrol. 33, 2094–2107 (2022).
UK Prospective Diabetes Study Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 352, 854–865 (1998).
Monami, M. et al. Effect of metformin on all-cause mortality and major adverse cardiovascular events: an updated meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 31, 699–704 (2021).
Charles, M. A. & Leslie, R. D. Diabetes: concepts of β-cell organ dysfunction and failure would lead to earlier diagnoses and prevention. Diabetes 70, 2444–2456 (2021).
Zhu, M. et al. β Cell aging and age-related diabetes. Aging 13, 7691–7706 (2021).
American Diabetes Association Professional Practice Committee. 9. Pharmacologic approaches to glycemic treatment: standards of care in diabetes — 2025. Diabetes Care 48, S181–S206 (2024).
Maiorino, M. I. et al. Insulin and glucagon-like peptide 1 receptor agonist combination therapy in type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Care 40, 614–624 (2017).
Yang, R., Yang, Z., Chi, J. & Zhu, Y. Insulin delivery devices in diabetes management: applications and advancements. Intell. Pharm. 3, 235–242 (2025).
Wysham, C. et al. Effect of insulin degludec vs insulin glargine u100 on hypoglycemia in patients with type 2 diabetes: the SWITCH 2 randomized clinical trial. JAMA 318, 45–56 (2017).
Philis-Tsimikas, A. et al. Rationale and design of the phase 3a development programme (ONWARDS 1-6 trials) investigating once-weekly insulin icodec in diabetes. Diabetes Obes. Metab. 25, 331–341 (2023).
Bergenstal, R. M. et al. Once-weekly insulin efsitora alfa: design and rationale for the QWINT phase 3 clinical development programme. Diabetes Obes. Metab. 26, 3020–3030 (2024).
Shetty, S. & Suvarna, R. Efficacy and safety of once-weekly insulin icodec in type 2 diabetes: a meta-analysis of ONWARDS phase 3 randomized controlled trials. Diabetes Obes. Metab. 26, 1069–1081 (2024).
Bajaj, H. S. et al. Once-weekly insulin icodec compared with daily basal insulin analogues in type 2 diabetes: participant-level meta-analysis of the ONWARDS 1-5 trials. Diabetes Obes. Metab. 26, 3810–3820 (2024).
Goldman, J., Triplitt, C. & Isaacs, D. Icodec: a novel once-weekly basal insulin for diabetes management. Ann. Pharmacother. 59, 554–569 (2025).
Rosenstock, J. et al. Weekly fixed-dose insulin efsitora in type 2 diabetes without previous insulin therapy. N. Engl. J. Med. 393, 325–335 (2025).
Wysham, C. et al. Insulin efsitora versus degludec in type 2 diabetes without previous insulin treatment. N. Engl. J. Med. 391, 2201–2211 (2024).
Trevisan, R., Conti, M. & Ciardullo, S. Once-weekly insulins: a promising approach to reduce the treatment burden in people with diabetes. Diabetologia 67, 1480–1492 (2024).
Bajaj, H. S. et al. Continuous glucose monitoring-based metrics and hypoglycemia duration in insulin-experienced individuals with long-standing type 2 diabetes switched from a daily basal insulin to once-weekly insulin icodec: post hoc analysis of ONWARDS 2 and ONWARDS 4. Diabetes Care 47, 729–738 (2024).
Philis-Tsimikas, A. et al. Once-weekly insulin efsitora alfa versus once-daily insulin degludec in adults with type 2 diabetes currently treated with basal insulin (QWINT-3): a phase 3, randomised, non-inferiority trial. Lancet 405, 2279–2289 (2025).
Blevins, T. et al. Once-weekly insulin efsitora alfa versus once-daily insulin glargine U100 in adults with type 2 diabetes treated with basal and prandial insulin (QWINT-4): a phase 3, randomised, non-inferiority trial. Lancet 405, 2290–2301 (2025).
Liu, Y. et al. Recent progress in glucose-responsive insulin. Diabetes 73, 1377–1388 (2024).
Tomlinson, B., Patil, N. G., Fok, M., Chan, P. & Lam, C. W. K. The role of sulfonylureas in the treatment of type 2 diabetes. Expert Opin. Pharmacother. 23, 387–403 (2022).
Dormandy, J. A. et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 366, 1279–1289 (2005).
Kernan, W. N. et al. Pioglitazone after ischemic stroke or transient ischemic attack. N. Engl. J. Med. 374, 1321–1331 (2016).
Li, C., Luo, J., Jiang, M. & Wang, K. The efficacy and safety of the combination therapy with GLP-1 receptor agonists and SGLT-2 inhibitors in type 2 diabetes mellitus: a systematic review and meta-analysis. Front. Pharmacol. 13, 838277 (2022).
DeFronzo, R. A. Combination therapy with GLP-1 receptor agonist and SGLT2 inhibitor. Diabetes Obes. Metab. 19, 1353–1362 (2017).
Chen, B., Tao, L., Tian, M. & Ji, Z. Efficacy and safety of combination of semaglutide and basal insulin in patients with of type 2 diabetes mellitus: a systematic review and meta-analysis. Clin. Nutr. ESPEN 66, 564–572 (2025).
Amiel, S. A. The consequences of hypoglycaemia. Diabetologia 64, 963–970 (2021).
Htoo, P. T. et al. Risk of severe hypoglycemia with newer second-line glucose-lowering medications in older adults with type 2 diabetes stratified by known indicators of hypoglycemia risk. J. Gerontol. A 78, 2426–2434 (2023).
Zaccardi, F., Ling, S., Lawson, C., Davies, M. J. & Khunti, K. Severe hypoglycaemia and absolute risk of cause-specific mortality in individuals with type 2 diabetes: a UK primary care observational study. Diabetologia 63, 2129–2139 (2020).
Zoungas, S. et al. Severe hypoglycemia and risks of vascular events and death. N. Engl. J. Med. 363, 1410–1418 (2010).
Freckmann, G. et al. A comparative analysis of glycemic metrics derived from three continuous glucose monitoring systems. Diabetes Care 48, 1213–1217 (2025).
Battelino, T. et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care 42, 1593–1603 (2019).
Barchiesi, M. A. et al. Continuous glucose monitoring in type 2 diabetes: a systematic review of barriers and opportunities for care improvement. Int. J. Qual. Health Care 37, mzaf046 (2025).
Espinoza, J., Xu, N. Y., Nguyen, K. T. & Klonoff, D. C. The need for data standards and implementation policies to integrate CGM data into the electronic health record. J. Diabetes Sci. Technol. 17, 495–502 (2023).
Novo Nordisk. Novo Nordisk A/S: Wegovy approved in the US for the treatment of MASH. Novo Nordisk https://www.novonordisk.com/news-and-media/news-and-ir-materials/news-details.html?id=916416&s=09# (15 August 2025).
Belfort, R. et al. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N. Engl. J. Med. 355, 2297–2307 (2006).
Cusi, K. et al. Long-term pioglitazone treatment for patients with nonalcoholic steatohepatitis and prediabetes or type 2 diabetes mellitus: a randomized trial. Ann. Intern. Med. 165, 305–315 (2016).
Wei, Q., Xu, X., Guo, L., Li, J. & Li, L. Effect of SGLT2 inhibitors on type 2 diabetes mellitus with non-alcoholic fatty liver disease: a meta-analysis of randomized controlled trials. Front. Endocrinol. 12, 635556 (2021).
Lv, X. et al. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) for the management of nonalcoholic fatty liver disease (NAFLD): a systematic review. Endocrinol. Diabetes Metab. 3, e00163 (2020).
Rosenstock, J. et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet 398, 143–155 (2021).
Packer, M. et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N. Engl. J. Med. 392, 427–437 (2025).
Malhotra, A. et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N. Engl. J. Med. 391, 1193–1205 (2024).
Loomba, R. et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N. Engl. J. Med. 391, 299–310 (2024).
Nicholls, S. J. et al. Cardiovascular outcomes with tirzepatide versus dulaglutide in type 2 diabetes. N. Engl. J. Med. 393, 2409–2420 (2025).
Phillips, L. S. et al. Clinical inertia. Ann. Intern. Med. 135, 825–834 (2001).
Almigbal, T. H. et al. Clinical inertia in the management of type 2 diabetes mellitus: a systematic review. Medicina 59, 182 (2023).
Khunti, K. & Davies, M. J. Clinical inertia — time to reappraise the terminology? Prim. Care Diabetes 11, 105–106 (2017).
Khunti, K., Wolden, M. L., Thorsted, B. L., Andersen, M. & Davies, M. J. Clinical inertia in people with type 2 diabetes: a retrospective cohort study of more than 80,000 people. Diabetes Care 36, 3411–3417 (2013).
Khunti, S., Khunti, K. & Seidu, S. Therapeutic inertia in type 2 diabetes: prevalence, causes, consequences and methods to overcome inertia. Ther. Adv. Endocrinol. Metab. 10, 2042018819844694 (2019). This review highlights the prevalence, causes and consequences of therapeutic inertia in T2DM and strategies to address it.
Mauricio, D. et al. Change in insulin dose and HbA1c by geographical region — results from the diabetes unmet need with basal insulin evaluation (DUNE) study. Diabetes 67, 1037-P (2018).
Shalaeva, E. V. et al. Impact of persistent medication adherence and compliance with lifestyle recommendations on major cardiovascular events and one-year mortality in patients with type 2 diabetes and advanced stages of atherosclerosis: results from a prospective cohort study. Glob. Heart 18, 61 (2023).
Evans, M. et al. Adherence to and persistence with antidiabetic medications and associations with clinical and economic outcomes in people with type 2 diabetes mellitus: a systematic literature review. Diabetes Obes. Metab. 24, 377–390 (2022).
Polonsky, W. H. et al. Assessment of diabetes-related distress. Diabetes Care 18, 754–760 (1995).
Konstantinou, P. et al. Barriers, facilitators, and interventions for medication adherence across chronic conditions with the highest non-adherence rates: a scoping review with recommendations for intervention development. Transl. Behav. Med. 10, 1390–1398 (2020).
Wulandari, N., Lamuri, A., van Hasselt, F., Feenstra, T. & Taxis, K. The burden of depression among patients with type 2 diabetes: an umbrella review of systematic reviews. J. Diabetes Complications 39, 109004 (2025).
Ascher-Svanum, H. et al. Associations between glycemic control, depressed mood, clinical depression, and diabetes distress before and after insulin initiation: an exploratory, post hoc analysis. Diabetes Ther. 6, 303–316 (2015).
Lamptey, R., Berry, E., Hermanns, N. & Snoek, F. Editorial: highlights in diabetes self-management 2021/22. Front. Clin. Diabetes Healthc. 4, 1116879 (2023).
Speight, J. et al. Bringing an end to diabetes stigma and discrimination: an international consensus statement on evidence and recommendations. Lancet Diabetes Endocrinol. 12, 61–82 (2024). This consensus statement provides evidence-based recommendations to address diabetes-related stigma and discrimination.
Speight, J., Conn, J., Dunning, T. & Skinner, T. C.; Diabetes Australia. Diabetes Australia position statement. A new language for diabetes: improving communications with and about people with diabetes. Diabetes Res. Clin. Pract. 97, 425–431 (2012).
Bhattarai, M. et al. Association of sodium-glucose cotransporter 2 inhibitors with cardiovascular outcomes in patients with type 2 diabetes and other risk factors for cardiovascular disease: a meta-analysis. JAMA Netw. Open 5, e2142078 (2022).
Undiagnosed type 2 diabetes: an invisible risk factor. Lancet Diabetes Endocrinol. 12, 215 (2024).
Owolabi, M. O. et al. Gaps in guidelines for the management of diabetes in low- and middle-income versus high-income countries — a systematic review. Diabetes Care 41, 1097–1105 (2018).
Sargeant, J. A. et al. Adults with early-onset type 2 diabetes (aged 18–39 years) are severely underrepresented in diabetes clinical research trials. Diabetologia 63, 1516–1520 (2020).
Wang, M. C., Shah, N. S., Carnethon, M. R., O’Brien, M. J. & Khan, S. S. Age at diagnosis of diabetes by race and ethnicity in the United States from 2011 to 2018. JAMA Intern. Med. 181, 1537–1539 (2021).
Paul, S. K. et al. Comparison of body mass index at diagnosis of diabetes in a multi-ethnic population: a case-control study with matched non-diabetic controls. Diabetes Obes. Metab. 19, 1014–1023 (2017).
Xie, J. et al. Global burden of type 2 diabetes in adolescents and young adults, 1990-2019: systematic analysis of the Global Burden of Disease Study 2019. BMJ 379, e072385 (2022).
Nanayakkara, N. et al. Impact of age at type 2 diabetes mellitus diagnosis on mortality and vascular complications: systematic review and meta-analyses. Diabetologia 64, 275–287 (2021).
Goldney, J. et al. Age at onset of type 2 diabetes and prevalence of vascular disease and heart failure: systematic review and dose-response meta-analysis. J. Diabetes Complications 38, 108849 (2024).
Barker, M. M. et al. Age at diagnosis of type 2 diabetes and depressive symptoms, diabetes-specific distress, and self-compassion. Diabetes Care 46, 579–586 (2023).
Misra, S. et al. Managing early-onset type 2 diabetes in the individual and at the population level. Lancet 405, 2341–2354 (2025).
Lim, L. L. et al. Aspects of multicomponent integrated care promote sustained improvement in surrogate clinical outcomes: a systematic review and meta-analysis. Diabetes Care 41, 1312–1320 (2018).
Wagner, E. H. et al. Finding common ground: patient-centeredness and evidence-based chronic illness care. J. Altern. Complement. Med. 11, S7–S15 (2005).
Amgen. Amgen announces robust weight loss with maritide in people living with obesity or overweight at 52 weeks in a phase 2 study. Amgen https://go.nature.com/40esdb4 (2024).
Zhang, B. et al. Efficacy and safety of mazdutide in Chinese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled phase 2 trial. Diabetes Care 47, 160–168 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04153929 (2022).
Rosenstock, J. et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet 402, 529–544 (2023). This trial demonstrates potent metabolic effects of retatrutide, a triple GIP–GLP1–glucagon agonist, in T2DM.
Romero-Gómez, M. et al. A phase IIa active-comparator-controlled study to evaluate the efficacy and safety of efinopegdutide in patients with non-alcoholic fatty liver disease. J. Hepatol. 79, 888–897 (2023).
Sanyal, A. J. et al. A phase 2 randomized trial of survodutide in MASH and fibrosis. N. Engl. J. Med. 391, 311–319 (2024).
Sanyal, A. J. et al. Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nat. Med. 30, 2037–2048 (2024). This trial evaluates retatrutide in steatotic liver disease, highlighting potential benefits beyond glucose control.
Heymsfield, S. B. et al. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA Netw. Open 4, e2033457 (2021).
McGowan, B. M. et al. Efficacy and safety of once-weekly semaglutide 2.4 mg versus placebo in people with obesity and prediabetes (STEP 10): a randomised, double-blind, placebo-controlled, multicentre phase 3 trial. Lancet Diabetes Endocrinol. 12, 631–642 (2024).
Eli Lilly and Company. Tirzepatide reduced the risk of developing type 2 diabetes by 94% in adults with pre-diabetes and obesity or overweight. Eli Lilly https://go.nature.com/4b9JU0n (2024).
Misra, S. et al. Precision subclassification of type 2 diabetes: a systematic review. Commun. Med. 3, 138 (2023).
RADIANT Study Group. The rare and atypical diabetes network (RADIANT) study: design and early results. Diabetes Care 46, 1265–1270 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06603571 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06373146 (2026).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06565611 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT06564753 (2025).
Siminerio, L. M. The role of technology and the chronic care model. J. Diabetes Sci. Technol. 4, 470–475 (2010).
NHS England. Quality and outcomes framework guidance for 2024/25. NHS England https://www.england.nhs.uk/publication/quality-and-outcomes-framework-guidance-for-2024-25/ (2024).
Emonena, H. & Ojo, O. The efficacy of tele-monitoring in maintaining glycated haemoglobin levels in patients with type 2 diabetes mellitus: a systematic review. Int. J. Env. Res. Public Health 19, 16722 (2022). This systematic review finds telemonitoring to be effective in maintaining glycaemic control in T2DM.
Ni, K., Tampe, C. A., Sol, K., Cervantes, L. & Pereira, R. I. Continuous glucose monitor: reclaiming type 2 diabetes self-efficacy and mitigating disparities. J. Endocr. Soc. 8, bvae125 (2024).
Ferreira, R. O. M. et al. Continuous glucose monitoring systems in noninsulin-treated people with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Technol. Ther. 26, 252–262 (2024).
Eeg-Olofsson, K. et al. Real-world study of flash glucose monitoring among adults with type 2 diabetes within the Swedish National Diabetes Register. Diabetes Vasc. Dis. Res. 20, 14791641211067418 (2023).
Edelman, S., Cheatham, W. W., Norton, A. & Close, K. L. Patient perspectives on the benefits and challenges of diabetes and digital technology. Clin. Diabetes 42, 243–256 (2024).
Cornelius, J., Doran, F., Jefford, E. & Salehi, N. Patient decision aids in clinical practice for people with diabetes: a scoping review. Diabetol. Int. 11, 344–359 (2020).
Daly, A. & Hovorka, R. Technology in the management of type 2 diabetes: present status and future prospects. Diabetes Obes. Metab. 23, 1722–1732 (2021).
Acknowledgements
The authors acknowledge M. Savage (Research Associate, Diabetes Research Centre, Leicester, UK), who provided considerable support with collating sections and formatting the final manuscript. The authors also acknowledge C. Franklin (Senior Creative, Diabetes Research Centre, Leicester, UK), who provided considerable support in drafting and amending the original figure concepts.
Author information
Authors and Affiliations
Contributions
M.J.D. and S.L. are co-first authors. Introduction (all authors); Epidemiology (A.P.-T.); Mechanisms/pathophysiology (S.L., J.G. and T.S.); Diagnosis, screening and prevention (T.T.); Management (I.L., S.L., J.G., T.Y., D.R.F., A.P.-T. and M.J.D.); Quality of Life (R.L., J.G. and T.S.); Outlook (D.R.F., J.G. and R.L.); overview of Primer (M.J.D.).
Corresponding authors
Ethics declarations
Competing interests
M.J.D. has acted as a consultant/adviser and speaker for Eli Lilly and Company, Novo Nordisk and Sanofi, has attended advisory boards for AbbVie, Amgen, AstraZeneca, Biomea Fusion, Carmot/Roche, Daewoong Pharmaceutical, Sanofi, Zealand Pharma, Regeneron, GSK and EktaH and as a speaker for AstraZeneca, Boehringer Ingelheim and Zuellig Pharma. She has received grants from AstraZeneca, Boehringer Ingelheim and Novo Nordisk. S.L. is an advisory board member for Novo Nordisk and AstraZeneca, and has served on the speakers’ bureau of Novo Nordisk, Sanofi, Boehringer Ingelheim and AstraZeneca. He has received research funding from Chong Kun Dang and Daewoong Pharma. A.P.-T. performs research and serves as an adviser on behalf of their employer for Abbvie, Corcept, Dexcom, Eli Lilly and Company, Genentech, Medtronic, Novo Nordisk, Regeneron and Roche; there has been no direct or indirect transfer of funds. D.R.F. has served as an advisor to Eli Lilly and Company, Novo Nordisk, Abbott, Medtronic, AstraZeneca and Embecta, has received research support from Eli Lilly and Company and Novo Nordisk, and has received fees for speaking from AstraZeneca, Eli Lilly and Company, Abbott, Medtronic and Novo Nordisk. T.Y. has received investigator-initiated funding from AstraZeneca, contracted research funding from the Reinsurance Group of America and has acted as a consultant for Regeneron. I.L. received research funding (paid to institution) and/or product from Novo Nordisk, Boehringer Ingelheim, Dexcom, Roche, Pfizer and Eli Lilly and Company. I.L. received research-related consulting fees (paid to institution) from Novo Nordisk, advisory/consulting fees and/or other support from Aadvarak Therapeutics, Abbvie, Altimmune, Alveus Therapeutics, Amgen, Antag Therapeutics, AstraZeneca, Bain Capital, Bayer, Betagenon AB, Bioio, Biomea, Boehringer Ingelheim, Boston Scientific, Carmot, Corxel, Cytoki Pharma, Eli Lilly and Company, Genentech, Intercept, Janssen/J&J, Juvena, Keros Therapeutic, Mediflix, Merck, Metsera, Neurocrine, Novo Nordisk, Pfizer, Regeneron, Roche, Sanofi, Shionogi, Skye Bio, Source Bio, Structure Therapeutics, TERNS Pharma, The Comm Group, Verdiva Bio, WebMD and Zealand Pharma. T.T., R.L., T.S. and J.G. declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks P. Froguel, S. Cromer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Davies, M.J., Lim, S., Slater, T. et al. Type 2 diabetes mellitus. Nat Rev Dis Primers 12, 13 (2026). https://doi.org/10.1038/s41572-026-00687-w
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41572-026-00687-w