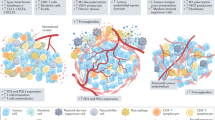
Abstract
Immune evasion is a hallmark of cancer progression but the role of steroid hormones in this evasion has long been underrated. This oversight is particularly notable for glucocorticoids given that exogenous glucocorticoids remain a cornerstone therapy in various oncological treatment regimens, supportive care and treatment of immune-related adverse events caused by immune-checkpoint inhibitors. Cortisol, the main endogenous glucocorticoid in humans, is secreted by the adrenal cortex in response to stress. Additionally, cortisol and its inactive metabolite cortisone can be interconverted to further modulate tissue-dependent glucocorticoid action. In the past 5 years, intratumoural production of glucocorticoids, by both immune and tumour cells, has been shown to support tumour immune evasion. Here, we summarize current progress at the crossroads of endocrinology and immuno-oncology. We outline the known effects of steroid hormones on different immune cell types with a focus on glucocorticoids and androgens. We conclude with options for pharmaceutical intervention, including the engineering of cell-based therapies that resist the immunosuppressive action of steroid hormones. Overall, local steroid production and metabolism are emerging elements of tumour immune suppression that are potentially amenable to therapeutic intervention. Targeting steroid hormones to enhance anticancer therapies could increase their efficacy but will require expertise in endocrine care.
Key points
-
High-dose or long-term glucocorticoid use can compromise immunotherapy efficacy.
-
Both immune cells and tumour cells potentially possess the capability to engage in steroid hormone synthesis.
-
Identifying a dependable biomarker could help pinpoint tumours that are likely to respond well to glucocorticoid-modulating therapies.
-
Therapeutic strategies could involve targeting steroid hormone pathways through various approaches, including inhibition of cortisol and androgen synthesis (via CYP11B1 and CYP17A1 inhibitors), blocking cortisol activation (with HSD11B1 inhibitors), or antagonizing glucocorticoid and androgen receptors.
-
When using high-dose systemic inhibitors of steroid hormone synthesis, glucocorticoid replacement is necessary, but this replacement, along with potential reactivation by HSD11B1, might inadvertently lead to unwanted glucocorticoid effects in the tumour microenvironment.
-
Cell-targeted drug delivery and cell-based therapies will enable precise treatment by selectively blocking steroid hormone signalling in effector cells, avoiding the risks of systemic glucocorticoid deficiency.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fujiwara, Y., Mittra, A., Naqash, A. R. & Takebe, N. A review of mechanisms of resistance to immune checkpoint inhibitors and potential strategies for therapy. Cancer Drug Resist. 3, 252–275 (2020).
Acharya, N. et al. Endogenous glucocorticoid signaling regulates CD8+ T cell differentiation and development of dysfunction in the tumor microenvironment. Immunity 53, 658–671.e6 (2020).
Martins Nascentes Melo, L. et al. Glucocorticoid activation by HSD11B1 limits T cell-driven interferon signaling and response to PD-1 blockade in melanoma. J. Immunother. Cancer 11, e004150 (2023).
Taves, M. D. et al. Tumors produce glucocorticoids by metabolite recycling, not synthesis, and activate Tregs to promote growth. J. Clin. Invest. 133, e164599 (2023).
Ikuta, K., Ejima, A., Abe, S. & Shimba, A. Control of immunity and allergy by steroid hormones. Allergol. Int. 71, 432–436 (2022).
Anderson, A. C. & Acharya, N. Steroid hormone regulation of immune responses in cancer. Immunometabolism 4, e00012 (2022).
Zhang, X. et al. Androgen signaling contributes to sex differences in cancer by inhibiting NF-κB activation in T cells and suppressing antitumor immunity. Cancer Res. 83, 906–921 (2023).
Xing, Y., Parker, C. R., Edwards, M. & Rainey, W. E. ACTH is a potent regulator of gene expression in human adrenal cells. J. Mol. Endocrinol. 45, 59–68 (2010).
Antoniou-Tsigkos A. et al. Adrenal androgens. Endotext (MDText.com, 2019).
Zobniw, C. M., Causebrook, A. & Fong, M. K. Clinical use of abiraterone in the treatment of metastatic castration-resistant prostate cancer. Res. Rep. Urol. 6, 97–105 (2014).
Pivonello, R., Simeoli, C., Di Paola, N. & Colao, A. Cushing’s disease: adrenal steroidogenesis inhibitors. Pituitary 25, 726–732 (2022).
Chapman, K., Holmes, M. & Seckl, J. 11β-Hydroxysteroid dehydrogenases: intracellular gate-keepers of tissue glucocorticoid action. Physiol. Rev. 93, 1139–1206 (2013).
Okamoto, N. et al. A membrane transporter is required for steroid hormone uptake in Drosophila. Dev. Cell 47, 294–305.e7 (2018).
Noddings, C. M., Johnson, J. L. & Agard, D. A. Cryo-EM reveals how Hsp90 and FKBP immunophilins co-regulate the glucocorticoid receptor. Nat. Struct. Mol. Biol. 30, 1867–1877 (2023).
Zannas, A. S., Wiechmann, T., Gassen, N. C. & Binder, E. B. Gene–stress–epigenetic regulation of FKBP5: clinical and translational implications. Neuropsychopharmacology 41, 261–274 (2016).
Timmermans, S., Souffriau, J. & Libert, C. A general introduction to glucocorticoid biology. Front. Immunol. 10, 1545 (2019).
Clarisse, D., Van Moortel, L., Van Leene, C., Gevaert, K. & De Bosscher, K. Glucocorticoid receptor signaling: intricacies and therapeutic opportunities. Trends Biochem. Sci. 49, 431–444 (2024).
Galon, J. et al. Gene profiling reveals unknown enhancing and suppressive actions of glucocorticoids on immune cells. FASEB J. 16, 61–71 (2002).
Sutanto, W. & de Kloet, E. R. Mineralocorticoid receptor ligands: biochemical, pharmacological, and clinical aspects. Med. Res. Rev. 11, 617–639 (1991).
Williams, D. M. Clinical pharmacology of corticosteroids. Respir. Care 63, 655–670 (2018).
Martin-Grace, J., Tomkins, M., O’Reilly, M. W. & Sherlock, M. Iatrogenic adrenal insufficiency in adults. Nat. Rev. Endocrinol. 20, 209–227 (2024).
Pivonello, R. et al. Complications of Cushing’s syndrome: state of the art. Lancet Diabetes Endocrinol. 4, 611–629 (2016).
Caratti, B. et al. The glucocorticoid receptor associates with RAS complexes to inhibit cell proliferation and tumor growth. Sci. Signal. 15, eabm4452 (2022).
Auger, J. P. et al. Metabolic rewiring promotes anti-inflammatory effects of glucocorticoids. Nature 629, 184–192 (2024).
Fizazi, K. et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N. Engl. J. Med. 377, 352–360 (2017).
Garg, A. D., Martin, S., Golab, J. & Agostinis, P. Danger signalling during cancer cell death: origins, plasticity and regulation. Cell Death Differ. 21, 26–38 (2014).
Yang, K., Halima, A. & Chan, T. A. Antigen presentation in cancer — mechanisms and clinical implications for immunotherapy. Nat. Rev. Clin. Oncol. 20, 604–623 (2023).
Limumpornpetch, P. et al. The effect of endogenous Cushing syndrome on all-cause and cause-specific mortality. J. Clin. Endocrinol. Metab. 107, 2377–2388 (2022).
Bouillet, P. et al. Proapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science 286, 1735–1738 (1999).
Tilly, H. et al. Diffuse large B-cell lymphoma (DLBCL): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 26, v116–125 (2015).
Cain, D. W. & Cidlowski, J. A. Immune regulation by glucocorticoids. Nat. Rev. Immunol. 17, 233–247 (2017).
Capelle, C. M. et al. Stress hormone signalling inhibits Th1 polarization in a CD4 T-cell-intrinsic manner via mTORC1 and the circadian gene PER1. Immunology 165, 428–444 (2022).
Liberman, A. C. et al. Regulatory and mechanistic actions of glucocorticoids on T and inflammatory cells. Front. Endocrinol. 9, 235 (2018).
Aston, W. J. et al. Dexamethasone differentially depletes tumour and peripheral blood lymphocytes and can impact the efficacy of chemotherapy/checkpoint blockade combination treatment. Oncoimmunology 8, e1641390 (2019).
Landwehr, L. S. et al. Interplay between glucocorticoids and tumor-infiltrating lymphocytes on the prognosis of adrenocortical carcinoma. J. Immunother. Cancer 8, e000469 (2020).
Taves, M. D. & Ashwell, J. D. Glucocorticoids in T cell development, differentiation and function. Nat. Rev. Immunol. 21, 233–243 (2021).
Elftman, M. D., Norbury, C. C., Bonneau, R. H. & Truckenmiller, M. E. Corticosterone impairs dendritic cell maturation and function. Immunology 122, 279–290 (2007).
Cohen, N. et al. GILZ expression in human dendritic cells redirects their maturation and prevents antigen-specific T lymphocyte response. Blood 107, 2037–2044 (2006).
Hamdi, H. et al. Induction of antigen-specific regulatory T lymphocytes by human dendritic cells expressing the glucocorticoid-induced leucine zipper. Blood 110, 211–219 (2007).
Kelly, P. M., Davison, R. S., Bliss, E. & McGee, J. O. Macrophages in human breast disease: a quantitative immunohistochemical study. Br. J. Cancer 57, 174–177 (1988).
Belgiovine, C., Digifico, E., Anfray, C., Ummarino, A. & Torres Andón, F. Targeting tumor-associated macrophages in anti-cancer therapies: convincing the traitors to do the right thing. J. Clin. Med. 9, 3226 (2020).
Skytthe, M. K., Graversen, J. H. & Moestrup, S. K. Targeting of CD163+ macrophages in inflammatory and malignant diseases. Int. J. Mol. Sci. 21, 5497 (2020).
Mantovani, A., Allavena, P., Marchesi, F. & Garlanda, C. Macrophages as tools and targets in cancer therapy. Nat. Rev. Drug Discov. 21, 799–820 (2022).
Zizzo, G., Hilliard, B. A., Monestier, M. & Cohen, P. L. Efficient clearance of early apoptotic cells by human macrophages requires M2c polarization and MerTK induction. J. Immunol. 189, 3508–3520 (2012).
Kleiman, A. & Tuckermann, J. P. Glucocorticoid receptor action in beneficial and side effects of steroid therapy: lessons from conditional knockout mice. Mol. Cell Endocrinol. 275, 98–108 (2007).
Stifel, U. et al. Glucocorticoids coordinate macrophage metabolism through the regulation of the tricarboxylic acid cycle. Mol. Metab. 57, 101424 (2022).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Cook, M. B. et al. Sex disparities in cancer incidence by period and age. Cancer Epidemiol. Biomarkers Prev. 18, 1174–1182 (2009).
Ainslie, R. J., Simitsidellis, I., Kirkwood, P. M. & Gibson, D. A. RISING STARS: androgens and immune cell function. J. Endocrinol. 261, e230398 (2024).
Kissick, H. T. et al. Androgens alter T-cell immunity by inhibiting T-helper 1 differentiation. Proc. Natl Acad. Sci. USA 111, 9887–9892 (2014).
Fijak, M. et al. Testosterone replacement effectively inhibits the development of experimental autoimmune orchitis in rats: evidence for a direct role of testosterone on regulatory T cell expansion. J. Immunol. 186, 5162–5172 (2011).
Walecki, M. et al. Androgen receptor modulates Foxp3 expression in CD4+CD25+Foxp3+ regulatory T-cells. Mol. Biol. Cell 26, 2845–2857 (2015).
Liva, S. M. & Voskuhl, R. R. Testosterone acts directly on CD4+ T lymphocytes to increase IL-10 production. J. Immunol. 167, 2060–2067 (2001).
Kwon, H. et al. Androgen conspires with the CD8+ T cell exhaustion program and contributes to sex bias in cancer. Sci. Immunol. 7, eabq2630 (2022).
Yang, C. et al. Androgen receptor-mediated CD8+ T cell stemness programs drive sex differences in antitumor immunity. Immunity 55, 1268–1283.e9 (2022).
Chi, L. et al. Sexual dimorphism in skin immunity is mediated by an androgen-ILC2-dendritic cell axis. Science 384, eadk6200 (2024).
D’Agostino, P. et al. Sex hormones modulate inflammatory mediators produced by macrophages. Ann. N. Y. Acad. Sci. 876, 426–429 (1999).
Cioni, B. et al. Androgen receptor signalling in macrophages promotes TREM-1-mediated prostate cancer cell line migration and invasion. Nat. Commun. 11, 4498 (2020).
Yamaguchi, M. et al. Androgens enhance the ability of intratumoral macrophages to promote breast cancer progression. Oncol. Rep. 46, 188 (2021).
Wu, L. et al. Feminizing adrenocortical carcinoma: the source of estrogen production and the role of adrenal-gonadal dedifferentiation. J. Clin. Endocrinol. Metab. 103, 3706–3713 (2018).
Nakaya, M., Tachibana, H. & Yamada, K. Effect of estrogens on the interferon-gamma producing cell population of mouse splenocytes. Biosci. Biotechnol. Biochem. 70, 47–53 (2006).
Seillet, C. et al. The TLR-mediated response of plasmacytoid dendritic cells is positively regulated by estradiol in vivo through cell-intrinsic estrogen receptor α signaling. Blood 119, 454–464 (2012).
Fox, H. S., Bond, B. L. & Parslow, T. G. Estrogen regulates the IFN-gamma promoter. J. Immunol. 146, 4362–4367 (1991).
Chakraborty, B. et al. Inhibition of estrogen signaling in myeloid cells increases tumor immunity in melanoma. J. Clin. Invest. 131, e151347 (2021).
Adurthi, S. et al. Oestrogen receptor-α binds the FOXP3 promoter and modulates regulatory T-cell function in human cervical cancer. Sci. Rep. 7, 17289 (2017).
Hao, S. et al. Modulation of 17β-estradiol on the number and cytotoxicity of NK cells in vivo related to MCM and activating receptors. Int. Immunopharmacol. 7, 1765–1775 (2007).
Kovats, S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell Immunol. 294, 63–69 (2015).
Hoffmann, J. P., Liu, J. A., Seddu, K. & Klein, S. L. Sex hormone signaling and regulation of immune function. Immunity 56, 2472–2491 (2023).
Jones, L. A. et al. Differential modulation of TLR3- and TLR4-mediated dendritic cell maturation and function by progesterone. J. Immunol. 185, 4525–4534 (2010).
Menzies, F. M., Henriquez, F. L., Alexander, J. & Roberts, C. W. Selective inhibition and augmentation of alternative macrophage activation by progesterone. Immunology 134, 281–291 (2011).
Lee, J. H., Ulrich, B., Cho, J., Park, J. & Kim, C. H. Progesterone promotes differentiation of human cord blood fetal T cells into T regulatory cells but suppresses their differentiation into Th17 cells. J. Immunol. 187, 1778–1787 (2011).
Lei, K. et al. Progesterone acts via the nuclear glucocorticoid receptor to suppress IL-1β-induced COX-2 expression in human term myometrial cells. PLoS One 7, e50167 (2012).
Duma, N. et al. Sex differences in tolerability to anti-programmed cell death protein 1 therapy in patients with metastatic melanoma and non-small cell lung cancer: are we all equal? Oncologist 24, e1148–e1155 (2019).
Pala, L. et al. Sex and cancer immunotherapy: current understanding and challenges. Cancer Cell 40, 695–700 (2022).
Orzołek, I., Sobieraj, J. & Domagała-Kulawik, J. Estrogens, cancer and immunity. Cancers 14, 2265 (2022).
Pardoll, D. M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Hong, M., Clubb, J. D. & Chen, Y. Y. Engineering CAR-T cells for next-generation cancer therapy. Cancer Cell 38, 473–488 (2020).
Klobuch, S., Seijkens, T. T. P., Schumacher, T. N. & Haanen, J. Tumour-infiltrating lymphocyte therapy for patients with advanced-stage melanoma. Nat. Rev. Clin. Oncol. 21, 173–184 (2024).
Scheetz, L. et al. Engineering patient-specific cancer immunotherapies. Nat. Biomed. Eng. 3, 768–782 (2019).
Goebeler, M. E. & Bargou, R. C. T cell-engaging therapies — BiTEs and beyond. Nat. Rev. Clin. Oncol. 17, 418–434 (2020).
Märkl, F. et al. Bispecific antibodies redirect synthetic agonistic receptor modified T cells against melanoma. J. Immunother. Cancer 11, e006436 (2023).
Postow, M. A., Sidlow, R. & Hellmann, M. D. Immune-related adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 378, 158–168 (2018).
Brahmer, J. R. et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 36, 1714–1768 (2018).
Santomasso, B. D. et al. Management of immune-related adverse events in patients treated with chimeric antigen receptor T-cell therapy: ASCO guideline. J. Clin. Oncol. 39, 3978–3992 (2021).
Haanen, J. et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 33, 1217–1238 (2022).
Shimabukuro-Vornhagen, A. et al. Cytokine release syndrome. J. Immunother. Cancer 6, 56 (2018).
Adkins, S. CAR T-cell therapy: adverse events and management. J. Adv. Pract. Oncol. 10, 21–28 (2019).
Rejeski, K., Jain, M. D., Shah, N. N., Perales, M. A. & Subklewe, M. Immune effector cell-associated haematotoxicity after CAR T-cell therapy: from mechanism to management. Lancet Haematol. 11, e459–e470 (2024).
Freeman-Keller, M. et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin. Cancer Res. 22, 886–894 (2016).
Maher, V. E. et al. Analysis of the association between adverse events and outcome in patients receiving a programmed death protein 1 or programmed death ligand 1 antibody. J. Clin. Oncol. 37, 2730–2737 (2019).
Topalian, S. L. et al. Five-year survival and correlates among patients with advanced melanoma, renal cell carcinoma, or non-small cell lung cancer treated with nivolumab. JAMA Oncol. 5, 1411–1420 (2019).
Eggermont, A. M. M. et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 6, 519–527 (2020).
Arbour, K. C. et al. Impact of baseline steroids on efficacy of programmed cell death-1 and programmed death-ligand 1 blockade in patients with non-small-cell lung cancer. J. Clin. Oncol. 36, 2872–2878 (2018).
Scott, S. C. & Pennell, N. A. Early use of systemic corticosteroids in patients with advanced NSCLC treated with nivolumab. J. Thorac. Oncol. 13, 1771–1775 (2018).
Ricciuti, B. et al. Immune checkpoint inhibitor outcomes for patients with non-small-cell lung cancer receiving baseline corticosteroids for palliative versus nonpalliative indications. J. Clin. Oncol. 37, 1927–1934 (2019).
Horvat, T. Z. et al. Immune-related adverse events, need for systemic immunosuppression, and effects on survival and time to treatment failure in patients with melanoma treated with ipilimumab at Memorial Sloan Kettering Cancer Center. J. Clin. Oncol. 33, 3193–3198 (2015).
Weber, J. S. et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J. Clin. Oncol. 35, 785–792 (2017).
Van Buren, I. et al. Survival among veterans receiving steroids for immune-related adverse events after immune checkpoint inhibitor therapy. JAMA Netw. Open 6, e2340695 (2023).
Bai, X. et al. Early use of high-dose glucocorticoid for the management of irAE is associated with poorer survival in patients with advanced melanoma treated with anti-PD-1 monotherapy. Clin. Cancer Res. 27, 5993–6000 (2021).
Iorgulescu, J. B. et al. Concurrent dexamethasone limits the clinical benefit of immune checkpoint blockade in glioblastoma. Clin. Cancer Res. 27, 276–287 (2021).
Albarrán, V. et al. Negative association of steroids with immunotherapy efficacy in a multi-tumor cohort: time and dose-dependent. Cancer Immunol. Immunother. 73, 186 (2024).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
De Philippis, C. et al. Impact of preemptive use of tocilizumab on chimeric antigen receptor T cell outcomes in non-Hodgkin lymphoma. Transpl. Cell Ther. 29, 429.e1–429.e6 (2023).
Verheijden, R. J., van Eijs, M. J. M., May, A. M., van Wijk, F. & Suijkerbuijk, K. P. M. Immunosuppression for immune-related adverse events during checkpoint inhibition: an intricate balance. NPJ Precis. Oncol. 7, 41 (2023).
Fassnacht, M. et al. European Society of Endocrinology Clinical Practice Guidelines on the management of adrenocortical carcinoma in adults, in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 179, G1–G46 (2018).
Vanbrabant, T., Fassnacht, M., Assie, G. & Dekkers, O. M. Influence of hormonal functional status on survival in adrenocortical carcinoma: systematic review and meta-analysis. Eur. J. Endocrinol. 179, 429–436 (2018).
Sada, A. et al. The effect of hormonal secretion on survival in adrenocortical carcinoma: a multi-center study. Surgery 175, 80–89 (2024).
Berruti, A. et al. Prognostic role of overt hypercortisolism in completely operated patients with adrenocortical cancer. Eur. Urol. 65, 832–838 (2014).
Georgantzoglou, N., Kokkali, S., Tsourouflis, G. & Theocharis, S. Tumor microenvironment in adrenocortical carcinoma: barrier to immunotherapy success? Cancers 13, 1798 (2021).
Landwehr, L. S. et al. Expression and prognostic relevance of PD-1, PD-L1, and CTLA-4 immune checkpoints in adrenocortical carcinoma. J. Clin. Endocrinol. Metab. 109, 2325–2334 (2024).
Ababneh, O. et al. The efficacy and safety of immune checkpoint inhibitors in adrenocortical carcinoma: a systematic review and meta-analysis. Cancers 16, 900 (2024).
Thorsson, V. et al. The immune landscape of cancer. Immunity 48, 812–830.e4 (2018).
Zheng, S. et al. Comprehensive pan-genomic characterization of adrenocortical carcinoma. Cancer Cell 29, 723–736 (2016).
Greenstein, A. E., Habra, M. A., Wadekar, S. A. & Grauer, A. Adrenal tumors provide insight into the role of cortisol in NK cell activity. Endocr. Relat. Cancer 28, 583–592 (2021).
Tierney, J. F. et al. Expression of programmed death ligand 1 and 2 in adrenocortical cancer tissues: an exploratory study. Surgery 165, 196–201 (2019).
Baechle, J. J. et al. Integrative computational immunogenomic profiling of cortisol-secreting adrenocortical carcinoma. J. Cell Mol. Med. 25, 10061–10072 (2021).
Wilmouth, J. J. Jr. et al. Sexually dimorphic activation of innate antitumor immunity prevents adrenocortical carcinoma development. Sci. Adv. 8, eadd0422 (2022).
Kanzawa, M. et al. Effects of the cortisol milieu on tumor-infiltrating immune cells in corticotroph tumors. Endocrinology 165, bqae016 (2024).
Taves, M. D., Gomez-Sanchez, C. E. & Soma, K. K. Extra-adrenal glucocorticoids and mineralocorticoids: evidence for local synthesis, regulation, and function. Am. J. Physiol. Endocrinol. Metab. 301, E11–24 (2011).
Taves, M. D., Donahue, K. M., Bian, J., Cam, M. C. & Ashwell, J. D. Aire drives steroid hormone biosynthesis by medullary thymic epithelial cells. Sci. Immunol. 8, eabo7975 (2023).
Slominski, A. & Wortsman, J. Neuroendocrinology of the skin. Endocr. Rev. 21, 457–487 (2000).
Hannen, R. F. et al. Steroid synthesis by primary human keratinocytes; implications for skin disease. Biochem. Biophys. Res. Commun. 404, 62–67 (2011).
Sidler, D. et al. Colon cancer cells produce immunoregulatory glucocorticoids. Oncogene 30, 2411–2419 (2011).
Mahata, B. et al. Single-cell RNA sequencing reveals T helper cells synthesizing steroids de novo to contribute to immune homeostasis. Cell Rep. 7, 1130–1142 (2014).
Mahata, B. et al. Tumors induce de novo steroid biosynthesis in T cells to evade immunity. Nat. Commun. 11, 3588 (2020).
Cirillo, N. et al. Characterisation of the cancer-associated glucocorticoid system: key role of 11β-hydroxysteroid dehydrogenase type 2. Br. J. Cancer 117, 984–993 (2017).
Saito, R. et al. 11β Hydroxysteroid dehydrogenase 1: a new marker for predicting response to immune-checkpoint blockade therapy in non-small-cell lung carcinoma. Br. J. Cancer 123, 61–71 (2020).
Guan, X. et al. Androgen receptor activity in T cells limits checkpoint blockade efficacy. Nature 606, 791–796 (2022).
Kalfeist, L. et al. Impact of glucocorticoid use in oncology in the immunotherapy era. Cells 11, 770 (2022).
Poinot, H. et al. Activation of endogenous glucocorticoids by HSD11B1 inhibits the antitumor immune response in renal cancer. Oncoimmunology 13, 2286820 (2024).
Othonos, N. et al. 11β-HSD1 inhibition in men mitigates prednisolone-induced adverse effects in a proof-of-concept randomised double-blind placebo-controlled trial. Nat. Commun. 14, 1025 (2023).
Schiffer, L. et al. Inhibition of the glucocorticoid-activating enzyme 11β-hydroxysteroid dehydrogenase type 1 drives concurrent 11-oxygenated androgen excess. FASEB J. 38, e23574 (2024).
Fleseriu, M. et al. Mifepristone, a glucocorticoid receptor antagonist, produces clinical and metabolic benefits in patients with Cushing’s syndrome. J. Clin. Endocrinol. Metab. 97, 2039–2049 (2012).
Desai, K. B. et al. A phase I trial of enzalutamide plus selective glucocorticoid receptor modulator relacorilant in patients with metastatic castration-resistant prostate cancer. Clin. Cancer Res. 30, 2384–2392 (2024).
Colombo, N. et al. Relacorilant + Nab-paclitaxel in patients with recurrent, platinum-resistant ovarian cancer: a three-arm, randomized, controlled, open-label phase II study. J. Clin. Oncol. 41, 4779–4789 (2023).
Denlinger, N., Bond, D. & Jaglowski, S. CAR T-cell therapy for B-cell lymphoma. Curr. Probl. Cancer 46, 100826 (2022).
Brown, C. E. et al. Off-the-shelf, steroid-resistant, IL13Rα2-specific CAR T cells for treatment of glioblastoma. Neuro-oncology 24, 1318–1330 (2022).
Kaeuferle, T. et al. CRISPR-Cas9-mediated glucocorticoid resistance in virus-specific T cells for adoptive T cell therapy posttransplantation. Mol. Ther. 28, 1965–1973 (2020).
Koukoulias, K. et al. “Cerberus” T cells: a glucocorticoid-resistant, multi-pathogen specific T cell product to fight infections in severely immunocompromised patients. Front. Immunol. 11, 608701 (2020).
Schauer, M. P. et al. ROR-1 specific CAR-T cells with CRISPR/CAS9 mediated glucocorticoid receptor-knockout exert potent antitumor efficacy in advanced adrenocortical carcinoma. Endocrine Abst. 93, https://doi.org/10.1530/endoabs.93.OC20 (2023).
Munoz, A. M. et al. Dexamethasone potentiates chimeric antigen receptor T cell persistence and function by enhancing IL-7Rα expression. Mol. Ther. 32, 527–539 (2024).
Wu, K. et al. Discovery of a glucocorticoid receptor (GR) activity signature correlates with immune cell infiltration in adrenocortical carcinoma. J. Immunother. Cancer 11, e007528 (2023).
Deng, Y. et al. Glucocorticoid receptor regulates PD-L1 and MHC-I in pancreatic cancer cells to promote immune evasion and immunotherapy resistance. Nat. Commun. 12, 7041 (2021).
Acknowledgements
This work resulted, in part, from collaborative work on a grant application on the topic that finally did not receive funding. Work in the authors’ laboratories was supported by the German Research Consortium (Deutsche Forschungsgemeinschaft) within the collaborative research centres SFB Transregio no. TRR205 (project number 314061271) to F.B. and M.K., TRR 388 (project number 452881907) to S.T. and S.K., and an individual grant No. 391587558 (to S.T.). S.K. is funded by the Bavarian Cancer Research Center (TANGO), the Deutsche Forschungsgemeinschaft (grant number: KO5055-2-1 and KO5055/3-1), the international doctoral programme ‘i-Target: immunotargeting of cancer’ (funded by the Elite Network of Bavaria), the Melanoma Research Alliance (grant number 409510), Marie Sklodowska-Curie Training Network for Optimizing Adoptive T Cell Therapy of Cancer (funded by the Horizon 2020 programme of the European Union; grant 955575), Else Kröner-Fresenius-Stiftung (IOLIN), German Cancer Aid (AvantCAR.de), the Wilhelm-Sander-Stiftung, Ernst Jung Stiftung, Institutional Strategy LMUexcellent of LMU Munich (within the framework of the German Excellence Initiative), the Go-Bio-Initiative, the m4-Award of the Bavarian Ministry for Economical Affairs, Bundesministerium für Bildung und Forschung, European Research Council (Starting Grant 756017, CoG 101124203 and PoC Grant 101100460), by the SFB-TRR 338/1 2021–Fritz-Bender Foundation (to S.K.), Deutsche José Carreras Leukämie Stiftung (to S.K.), Hector Foundation, Monika-Kutzner Foundation for Cancer Research (to S.K.), Bavarian Research Foundation (BAYCELLATOR), the Bruno and Helene Jöster Foundation (360° CAR). P.S. received funding from the Else Kröner-Fresenius-Stiftung as part of the clinician-scientist programme “RISE – Rare Important Syndromes in Endocrinology”. S.T. received research grants by the Bruno und Helene Jöster Stiftung, Köln, and the Thomas Kirch Stiftung, München. We further acknowledge structural support from the Bavarian Cancer Research Center and the German Consortium for Translational Cancer Research. G.A. received support from the ITMO Cancer AVIESAN (Alliance Nationale pour les Sciences de la Vie et de la Santé, National Alliance for Life Sciences & Health) within the framework of the Cancer Plan, the Agence Nationale de la Recherche (18-CE14-0008-01), the French Ministry of Health and the French National Cancer Institute (COMETE-CARE, PRT-K 2020-15367), the SIRIC CARPEM (CAncer Research for PErsonalized Medicine), and COST Action CA20122 Harmonisation.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article. All authors contributed substantially to discussion of the content. M.K., P.S., A.T., G.A., A.J., S.T., F.B. and S.K. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.K. has received institutional remuneration (to institution) for contract research from Corcept, Inc. S.K. has received honoraria from Cymab, Plectonic, TCR2 Inc., Novartis, BMS, Miltenyi and GSK. S.K. is an inventor of several patents in the field of immuno-oncology. S.K. received license fees from TCR2 Inc. and Carina Biotech. S.K. received research support from Tabby Therapeutics, TCR2 Inc., Plectonic GmBH, Catalym GmBH and Arcus Bioscience for work unrelated to the manuscript.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Hidenori Fukuoka and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Schwarzlmueller, P., Triebig, A., Assié, G. et al. Steroid hormones as modulators of anti-tumoural immunity. Nat Rev Endocrinol 21, 331–343 (2025). https://doi.org/10.1038/s41574-025-01102-2
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41574-025-01102-2
This article is cited by
-
Gender and sex interactions are intrinsic components of cancer phenotypes
Nature Reviews Cancer (2025)
-
Gene expression and molecular pathway analyses differentiate immunotherapy-induced myositis from spontaneous dermatomyositis
Scientific Reports (2025)
-
Surgical outcomes of lung cancer associated with autoimmune disease-related interstitial pneumonia
General Thoracic and Cardiovascular Surgery (2025)
-
Impact of steroid differentiation on tumor microenvironment revealed by single-nucleus atlas of adrenal tumors
Nature Communications (2025)