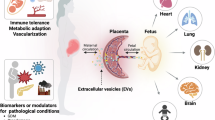
Abstract
The intricate processes of feto–placental development are regulated by both genetics and the nutritional environment. The placenta mediates nutrient transfer to the fetus and waste transfer to the maternal circulation to facilitate proper fetal cell fate specification. Disruptions in these fate-regulatory programmes can lead to maternal and fetal complications. We explore how metabolic–epigenetic mechanisms programme feto–placental development, discussing the roles of metabolic sensing mechanisms. This Review covers the roles of nutrient metabolite-dependent protein modifications, lipid signalling, steroid signalling and oxygen signalling in regulating particular phases and lineages of feto–placental development. Understanding these environmental–genetic interactions is crucial for elucidating the limits of developmental plasticity, the pathogenesis of pregnancy-related disorders and the potential therapeutic windows we can leverage to improve maternal and fetal outcomes.
Key points
-
Placental metabolism is shaped by a conflict between fetal demands and maternal constraints, with evidence from within the past 10 years confirming the existence of feto–placental genes that respond to metabolic–epigenetic interactions and fine-tune fetal development.
-
Placental trophoblast differentiation into the syncytiotrophoblast and extravillous trophoblasts ensures nutrient exchange, spiral artery remodelling and endocrine signalling critical for maternal adaptations and fetal development.
-
Metabolite sensing mechanisms and switches (such as O-GlcNAcylation, N-acylation, lactylation and methylation) have been discovered to dynamically regulate the epigenetic programming of particular lineages’ cell fates during feto–placental development.
-
Steroid-sensing nuclear hormone receptor signalling, oxygen-sensing hypoxia-inducible factor 1α (HIF1α) signalling and redox signalling also regulate tissue progenitor multipotency or lineage differentiation according to different levels of steroids and oxygen.
-
Metabolic–epigenetic disruptions (such as aberrant tricarboxylic acid levels, histone acetylation, DNA methylation or RNA methylation) impair feto–placental development, triggering recurrent miscarriage, pre-eclampsia or fetal growth restriction.
-
Feto–placental metabolic stress induces epigenetic reprogramming (such as 5-methylcytosine and N6-methyladenosine modifications), linking tissue progenitors to adult insulin resistance and developmental origins of adult-onset metabolic syndrome.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wooding, P. & Burton, G. Comparative Placentation: Structures, Functions and Evolution (Springer, 2008).
Burton, G. J. & Fowden, A. L. Review: the placenta and developmental programming: balancing fetal nutrient demands with maternal resource allocation. Placenta 33 (Suppl), S23–S27 (2012).
Kotler, J. Encyclopedia of Evolutionary Psychological Science (Springer, 2016).
Fowden, A. L. & Moore, T. Maternal–fetal resource allocation: co-operation and conflict. Placenta 33 (Suppl. 2), e11–e15 (2012).
Díaz, P., Powell, T. L. & Jansson, T. The role of placental nutrient sensing in maternal–fetal resource allocation. Biol. Reprod. 91, 82 (2014).
Lazo-de-la-Vega-Monroy, M. L. et al. Association of placental nutrient sensing pathways with birth weight. Reproduction 160, 455–468 (2020).
Hofmeister, R. J. et al. Parent-of-origin effects on complex traits in up to 236,781 individuals. Nature 646, 647–656 (2025).
Laundon, D., Gostling, N. J., Sengers, B. G., Chavatte-Palmer, P. & Lewis, R. M. Placental evolution from a three-dimensional and multiscale structural perspective. Evolution 78, 13–25 (2023).
Stadtmauer, D. J. et al. Cell type and cell signalling innovations underlying mammalian pregnancy. Nat. Ecol. Evol. 9, 1469–1486 (2025).
Afzal, J. et al. Evidence for coopetition at the maternal–fetal interface shaping placental invasion. Proc. Natl Acad. Sci. USA 122, e2323038122 (2025).
Godfrey, K. M., Gluckman, P. D. & Hanson, M. A. Developmental origins of metabolic disease: life course and intergenerational perspectives. Trends Endocrinol. Metab. 21, 199–205 (2010).
Renfree, M. B. & Shaw, G. Diapause. Annu. Rev. Physiol. 62, 353–375 (2000).
Shao, X. et al. The mystery of the life tree: the placentas†. Biol. Reprod. 107, 301–316 (2022).
Turco, M. Y. & Moffett, A. Development of the human placenta. Development 146, dev163428 (2019).
Wang, F. et al. Single-cell immune landscape of human recurrent miscarriage. Genom. Proteom. Bioinform. 19, 208–222 (2021).
Pijnenborg, R., Vercruysse, L. & Hanssens, M. The uterine spiral arteries in human pregnancy: facts and controversies. Placenta 27, 939–958 (2006).
Perazzolo, S., Lewis, R. M. & Sengers, B. G. Modelling nutrient transfer based on 3D imaging of the human placental microstructure. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2016, 5953–5956 (2016).
Barbour, L. A. et al. Human placental growth hormone increases expression of the p85 regulatory unit of phosphatidylinositol 3-kinase and triggers severe insulin resistance in skeletal muscle. Endocrinology 145, 1144–1150 (2004).
Li, Y. et al. Expression of P450 aromatase and 17β-hydroxysteroid dehydrogenase type 1 at fetal–maternal interface during tubal pregnancy. J. Steroid Biochem. Mol. Biol. 87, 241–246 (2003).
Shao, X. et al. Orchestrated feedback regulation between melatonin and sex hormones involving GPER1-PKA-CREB signaling in the placenta. J. Pineal Res. 75, e12913 (2023).
Shao, X. et al. Testosterone represses estrogen signaling by upregulating miR-22. Hypertension 69, 721–730 (2017).
Yu, D. et al. A multi-tissue metabolome atlas of primate pregnancy. Cell 187, 764–781 (2024).
Lopez-Tello, J. et al. Fetal manipulation of maternal metabolism is a critical function of the imprinted Igf2 gene. Cell Metab. 35, 1195–1208 (2023).
Sferruzzi-Perri, A. N., López-Tello, J., Fowden, A. L. & Constancia, M. Maternal and fetal genomes interplay through phosphoinositol 3-kinase(PI3K)-p110α signaling to modify placental resource allocation. Proc. Natl Acad. Sci. USA 113, 11255–11260 (2016).
López-Tello, J. et al. Fetal and trophoblast PI3K p110α have distinct roles in regulating resource supply to the growing fetus in mice. eLife 8, e45282 (2019).
Shao, X. et al. Placental trophoblast syncytialization potentiates macropinocytosis via mTOR signaling to adapt to reduced amino acid supply. Proc. Natl Acad. Sci. USA 118, e2017092118 (2021).
Burton, G. J. & Jones, C. J. Syncytial knots, sprouts, apoptosis, and trophoblast deportation from the human placenta. Taiwan. J. Obstet. Gynecol. 48, 28–37 (2009).
Li, Z., Wang, S. & Li, L. Advanced oxidative protein products drive trophoblast cells into senescence by inhibiting the autophagy: the potential implication of preeclampsia. Front. Cell Dev. Biol. 10, 810282 (2022).
Dimitriadis, E. et al. Pre-eclampsia. Nat. Rev. Dis. Primers 9, 8 (2023).
Schneider, H. Placental dysfunction as a key element in the pathogenesis of preeclampsia. Dev. Period. Med. 21, 309–316 (2017).
Hoffman, D. J., Powell, T. L., Barrett, E. S. & Hardy, D. B. Developmental origins of metabolic diseases. Physiol. Rev. 101, 739–795 (2021).
Mayhew, T. M. A stereological perspective on placental morphology in normal and complicated pregnancies. J. Anat. 215, 77–90 (2009).
Lager, S. & Powell, T. L. Regulation of nutrient transport across the placenta. J. Pregnancy 2012, 179827 (2012).
Brett, K. E., Ferraro, Z. M., Yockell-Lelievre, J., Gruslin, A. & Adamo, K. B. Maternal–fetal nutrient transport in pregnancy pathologies: the role of the placenta. Int. J. Mol. Sci. 15, 16153–16185 (2014).
Sferruzzi-Perri, A. N., Lopez-Tello, J. & Salazar-Petres, E. Placental adaptations supporting fetal growth during normal and adverse gestational environments. Exp. Physiol. 108, 371–397 (2023).
Zaugg, J. et al. Obesogenic diet in pregnancy disrupts placental iron handling and ferroptosis and stress signalling in association with fetal growth alterations. Cell Mol. Life Sci. 81, 151 (2024).
Sferruzzi-Perri, A. N., Higgins, J. S., Vaughan, O. R., Murray, A. J. & Fowden, A. L. Placental mitochondria adapt developmentally and in response to hypoxia to support fetal growth. Proc. Natl Acad. Sci. USA 116, 1621–1626 (2019).
Chi, F., Sharpley, M. S., Nagaraj, R., Roy, S. S. & Banerjee, U. Glycolysis-independent glucose metabolism distinguishes TE from ICM fate during mammalian embryogenesis. Dev. Cell 53, 9–26 (2020).
Liu, J. et al. Quantitative chemoproteomics reveals O-GlcNAcylation of cystathionine γ-lyase (CSE) represses trophoblast syncytialization. Cell Chem. Biol. 28, 788–801 (2021).
Hao, Y. et al. Chemoproteomic and transcriptomic analysis reveals that O-GlcNAc regulates mouse embryonic stem cell fate through the pluripotency network. Angew. Chem. Int. Ed. 62, e202300500 (2023).
Cao, J. et al. Oxidative phosphorylation safeguards pluripotency via UDP-N-acetylglucosamine. Protein Cell 14, 376–381 (2023).
Liu, Q. et al. O-GlcNAcase regulates pluripotency states of human embryonic stem cells. Stem Cell Rep. 19, 993–1009 (2024).
Andres, L. M. et al. Chemical modulation of protein O-glcNAcylation via OGT inhibition promotes human neural cell differentiation. ACS Chem. Biol. 12, 2030–2039 (2017).
Cao, D. et al. Selective utilization of glucose metabolism guides mammalian gastrulation. Nature 634, 919–928 (2024).
Villaronga-Luque, A. et al. Integrated molecular-phenotypic profiling reveals metabolic control of morphological variation in a stem-cell-based embryo model. Cell Stem Cell 32, 759–777 (2025).
Stapornwongkul, K. S. et al. Glycolytic activity instructs germ layer proportions through regulation of nodal and Wnt signaling. Cell Stem Cell 32, 744–758 (2025).
Miyazawa, H. et al. Glycolytic flux-signaling controls mouse embryo mesoderm development. eLife 11, e83299 (2022).
Dingare, C., Cao, D., Yang, J. J., Sozen, B. & Steventon, B. Mannose controls mesoderm specification and symmetry breaking in mouse gastruloids. Dev. Cell 59, 1523–1537 (2024).
Oginuma, M. et al. A gradient of glycolytic activity coordinates FGF and Wnt signaling during elongation of the body axis in amniote embryos. Dev. Cell 40, 342–353 (2017).
Oginuma, M. et al. Intracellular pH controls WNT downstream of glycolysis in amniote embryos. Nature 584, 98–101 (2020).
Michaut, A. et al. Extracellular volume expansion drives vertebrate axis elongation. Curr. Biol. 35, 843–853 (2025).
Bulusu, V. et al. Spatiotemporal analysis of a glycolytic activity gradient linked to mouse embryo mesoderm development. Dev. Cell 40, 331–341 (2017).
Diaz-Cuadros, M. et al. Metabolic regulation of species-specific developmental rates. Nature 613, 550–557 (2023).
Corrales, P., Vidal-Puig, A. & Medina-Gómez, G. Obesity and pregnancy, the perfect metabolic storm. Eur. J. Clin. Nutr. 75, 1723–1734 (2021).
Marchlewicz, E. H. et al. Lipid metabolism is associated with developmental epigenetic programming. Sci. Rep. 6, 34857 (2016).
Şanlı, E. & Kabaran, S. Maternal obesity, maternal overnutrition and fetal programming: effects of epigenetic mechanisms on the development of metabolic disorders. Curr. Genom. 20, 419–427 (2019).
Cai, S. et al. Nutritional status impacts epigenetic regulation in early embryo development: a scoping review. Adv. Nutr. 12, 1877–1892 (2021).
McDonnell, E. et al. Lipids reprogram metabolism to become a major carbon source for histone acetylation. Cell Rep. 17, 1463–1472 (2016).
Moussaieff, A. et al. Glycolysis-mediated changes in acetyl-CoA and histone acetylation control the early differentiation of embryonic stem cells. Cell Metab. 21, 392–402 (2015).
Shyh-Chang, N. & Daley, G. Q. Metabolic switches linked to pluripotency and embryonic stem cell differentiation. Cell Metab. 21, 349–350 (2015).
Yu, X. et al. Acetyl-CoA metabolism maintains histone acetylation for syncytialization of human placental trophoblast stem cells. Cell Stem Cell 31, 1280–1297 (2024).
Aye, I. et al. Placental sex-dependent spermine synthesis regulates trophoblast gene expression through acetyl-coA metabolism and histone acetylation. Commun. Biol. 5, 586 (2022).
Festa, J. & Trefely, S. Acetate to the rescue: acetyl-CoA facilitates placental development. Cell Stem Cell 31, 1241–1243 (2024).
Eddy, A. C., Chapman, H. & George, E. M. Acute hypoxia and chronic ischemia induce differential total changes in placental epigenetic modifications. Reprod. Sci. 26, 766–773 (2019).
Yang, D. et al. Histone lactylation is involved in mouse oocyte maturation and embryo development. Int. J. Mol. Sci. 25, 4821 (2024).
Merkuri, F., Rothstein, M. & Simoes-Costa, M. Histone lactylation couples cellular metabolism with developmental gene regulatory networks. Nat. Commun. 15, 90 (2024).
Li, J. et al. Lactate regulates major zygotic genome activation by H3K18 lactylation in mammals. Natl Sci. Rev. 11, nwad295 (2023).
Zhao, Y. et al. Lactate modulates zygotic genome activation through H3K18 lactylation rather than H3K27 acetylation. Cell Mol. Life Sci. 81, 298 (2024).
Yan, K. et al. Deficient histone H3 propionylation by BRPF1-KAT6 complexes in neurodevelopmental disorders and cancer. Sci. Adv. 6, eaax0021 (2020).
Fang, Y. et al. Histone crotonylation promotes mesoendodermal commitment of human embryonic stem cells. Cell Stem Cell 28, 748–763 (2021).
Gao, D. et al. P300 regulates histone crotonylation and preimplantation embryo development. Nat. Commun. 15, 6418 (2024).
Huang, B. et al. Inhibition of HDAC activity directly reprograms murine embryonic stem cells to trophoblast stem cells. Dev. Cell 59, 2101–2117 (2024).
Ye, Q. et al. Butyrate drives the acetylation of histone H3K9 to activate steroidogenesis through PPARγ and PGC1α pathways in ovarian granulosa cells. FASEB J. 35, e21316 (2021).
Lipinska, P., Pawlak, P. & Warzych, E. Species and embryo genome origin affect lipid droplets in preimplantation embryos. Front. Cell Dev. Biol. 11, 1187832 (2023).
Pawlak, P., Malyszka, N., Szczerbal, I. & Kolodziejski, P. Fatty acid induced lipolysis influences embryo development, gene expression and lipid droplet formation in the porcine cumulus cellsdagger. Biol. Reprod. 103, 36–48 (2020).
Yamada, M. et al. A medium-chain fatty acid as an alternative energy source in mouse preimplantation development. Sci. Rep. 2, 930 (2012).
Dunning, K. R., Akison, L. K., Russell, D. L., Norman, R. J. & Robker, R. L. Increased beta-oxidation and improved oocyte developmental competence in response to l-carnitine during ovarian in vitro follicle development in mice. Biol. Reprod. 85, 548–555 (2011).
Shafiei, G. et al. l-carnitine reduces the adverse effects of ROS and up-regulates the expression of implantation related genes in in vitro developed mouse embryos. Theriogenology 145, 59–66 (2020).
Logsdon, D. M. et al. Fatty acids present in commercial albumin preparations differentially affect development of murine embryos before and during implantation. F S Sci. 2, 50–58 (2021).
Zander-Fox, D., Villarosa, L. & McPherson, N. O. Albumin used in human IVF contain different levels of lipids and modify embryo and fetal growth in a mouse model. J. Assist. Reprod. Genet. 38, 2371–2381 (2021).
Berger, P. S. & Wood, P. A. Disrupted blastocoele formation reveals a critical developmental role for long-chain acyl-CoA dehydrogenase. Mol. Genet. Metab. 82, 266–272 (2004).
Huang, J. C. et al. Stimulation of embryo hatching and implantation by prostacyclin and peroxisome proliferator-activated receptor delta activation: implication in IVF. Hum. Reprod. 22, 807–814 (2007).
Guo, J. et al. Peroxisome proliferator-activated receptor δ improves porcine blastocyst hatching via the regulation of fatty acid oxidation. Theriogenology 90, 266–275 (2017).
Mumford, S. L. et al. Dietary fat intake and reproductive hormone concentrations and ovulation in regularly menstruating women. Am. J. Clin. Nutr. 103, 868–877 (2016).
Abolghasemi, A., Dirandeh, E., Ansari Pirsaraei, Z. & Shohreh, B. Dietary conjugated linoleic acid supplementation alters the expression of genes involved in the endocannabinoid system in the bovine endometrium and increases plasma progesterone concentrations. Theriogenology 86, 1453–1459 (2016).
Ye, Q. et al. Maternal short and medium chain fatty acids supply during early pregnancy improves embryo survival through enhancing progesterone synthesis in rats. J. Nutr. Biochem. 69, 98–107 (2019).
Luo, L. et al. Muscle injuries induce a prostacyclin-PPARgamma/PGC1a-FAO spike that boosts regeneration. Adv. Sci. 10, e2301519 (2023).
Aye, I. L., Waddell, B. J., Mark, P. J. & Keelan, J. A. Oxysterols inhibit differentiation and fusion of term primary trophoblasts by activating liver X receptors. Placenta 32, 183–191 (2011).
Aye, I. L., Gao, X., Weintraub, S. T., Jansson, T. & Powell, T. L. Adiponectin inhibits insulin function in primary trophoblasts by PPARα-mediated ceramide synthesis. Mol. Endocrinol. 28, 512–524 (2014).
Yang, Q., Wang, W., Liu, C., Wang, Y. & Sun, K. Compartmentalized localization of 11β-HSD 1 and 2 at the feto-maternal interface in the first trimester of human pregnancy. Placenta 46, 63–71 (2016).
Solmonson, A. et al. Compartmentalized metabolism supports midgestation mammalian development. Nature 604, 349–353 (2022).
Van Winkle, L. J. & Ryznar, R. One-carbon metabolism regulates embryonic stem cell fate through epigenetic DNA and histone modifications: implications for transgenerational metabolic disorders in adults. Front. Cell Dev. Biol. 7, 300 (2019).
Shyh-Chang, N. et al. Influence of threonine metabolism on S-adenosylmethionine and histone methylation. Science 339, 222–226 (2013).
Mirzadeh Azad, F. et al. Spic regulates one-carbon metabolism and histone methylation in ground-state pluripotency. Sci. Adv. 9, eadg7997 (2023).
Tang, S. et al. Methionine metabolism is essential for SIRT1-regulated mouse embryonic stem cell maintenance and embryonic development. EMBO J. 36, 3175–3193 (2017).
Baksh, S. C. & Finley, L. W. S. Metabolic coordination of cell fate by α-ketoglutarate-dependent dioxygenases. Trends Cell Biol. 31, 24–36 (2021).
Betto, R. M. et al. Metabolic control of DNA methylation in naive pluripotent cells. Nat. Genet. 53, 215–229 (2021).
TeSlaa, T. et al. α-Ketoglutarate accelerates the initial differentiation of primed human pluripotent stem cells. Cell Metab. 24, 485–493 (2016).
Liu, T. M. et al. Ascorbate and iron are required for the specification and long-term self-renewal of human skeletal mesenchymal stromal cells. Stem Cell Rep. 14, 210–225 (2020).
Zhao, J. et al. Metabolic remodelling during early mouse embryo development. Nat. Metab. 3, 1372–1384 (2021).
Ispada, J. et al. Tricarboxylic acid cycle metabolites as mediators of DNA methylation reprogramming in bovine preimplantation embryos. Int. J. Mol. Sci. 21, 6868 (2020).
Wang, X. H. et al. Low chorionic villous succinate accumulation associates with recurrent spontaneous abortion risk. Nat. Commun. 12, 3428 (2021).
Pastor, W. A. & Kwon, S. Y. Distinctive aspects of the placental epigenome and theories as to how they arise. Cell Mol. Life Sci. 79, 569 (2022).
Rahnama, F., Shafiei, F., Gluckman, P. D., Mitchell, M. D. & Lobie, P. E. Epigenetic regulation of human trophoblastic cell migration and invasion. Endocrinology 147, 5275–5283 (2006).
Andrews, S. et al. Mechanisms and function of de novo DNA methylation in placental development reveals an essential role for DNMT3B. Nat. Commun. 14, 371 (2023).
Clark, S. J. et al. Single-cell multi-omics profiling links dynamic DNA methylation to cell fate decisions during mouse early organogenesis. Genome Biol. 23, 202 (2022).
Wilson, R. L. et al. Characterization of 5-methylcytosine and 5-hydroxymethylcytosine in human placenta cell types across gestation. Epigenetics 14, 660–671 (2019).
He, Z. et al. The promoter methylomes of monochorionic twin placentas reveal intrauterine growth restriction-specific variations in the methylation patterns. Sci. Rep. 6, 20181 (2016).
Zhang, Y., Zheng, D., Fang, Q. & Zhong, M. Aberrant hydroxymethylation of ANGPTL4 is associated with selective intrauterine growth restriction in monochorionic twin pregnancies. Epigenetics 15, 887–899 (2020).
Li, X. et al. Ten-eleven translocation 2 demethylates the MMP9 promoter, and its down-regulation in preeclampsia impairs trophoblast migration and invasion. J. Biol. Chem. 293, 10059–10070 (2018).
Ma, M., Zhou, Q. J., Xiong, Y., Li, B. & Li, X. T. Preeclampsia is associated with hypermethylation of IGF-1 promoter mediated by DNMT1. Am. J. Transl. Res. 10, 16–39 (2018).
Wu, A. H. et al. Expression of TET and 5-HmC in trophoblast villi of women with normal pregnancy and with early pregnancy loss. Curr. Med. Sci. 38, 505–512 (2018).
Song, D. & Shyh-Chang, N. An RNA methylation code to regulate protein translation and cell fate. Cell Prolif. 55, e13224 (2022).
Ning, J., Yan, J. & Yang, H. Exploring the role of m6A modification in the great obstetrical syndromes. J. Matern. Fetal Neonatal Med. 36, 2234541 (2023).
Gu, Y., Chu, X., Morgan, J. A., Lewis, D. F. & Wang, Y. Upregulation of METTL3 expression and m6A RNA methylation in placental trophoblasts in preeclampsia. Placenta 103, 43–49 (2021).
Ning, J. et al. Demethylase FTO-mediated m6A modification of SIK1 modulates placental cytotrophoblast syncytialization in type 2 diabetes mellitus. iScience 27, 109900 (2024).
Andraweera, P. H. et al. The obesity associated FTO gene variant and the risk of adverse pregnancy outcomes: evidence from the SCOPE study. Obesity 24, 2600–2607 (2016).
Guang, L. et al. An obesogenic FTO allele causes accelerated development, growth and insulin resistance in human skeletal muscle cells. Nat. Commun. 16, 1645 (2025).
Zhu, H. et al. Lin28a transgenic mice manifest size and puberty phenotypes identified in human genetic association studies. Nat. Genet. 42, 626–630 (2010).
Zhu, H. et al. The Lin28/let-7 axis regulates glucose metabolism. Cell 147, 81–94 (2011).
Nguyen, L. H. et al. Lin28b is sufficient to drive liver cancer and necessary for its maintenance in murine models. Cancer Cell 26, 248–261 (2014).
Wang, P. et al. Adult progenitor rejuvenation with embryonic factors. Cell Prolif. 56, e13459 (2023).
Song, D., Chen, Y., Wang, P., Cheng, Y. & Shyh-Chang, N. Lin28a forms an RNA-binding complex with Igf2bp3 to regulate m6A-modified stress response genes in stress granules of muscle stem cells. Cell Prolif. 57, e13707 (2024).
Zhang, J. et al. LIN28 regulates stem cell metabolism and conversion to primed pluripotency. Cell Stem Cell 19, 66–80 (2016).
Seabrook, J. L. et al. Role of LIN28A in mouse and human trophoblast cell differentiation. Biol. Reprod. 89, 95 (2013).
Wang, P. et al. Lin28a maintains a subset of adult muscle stem cells in an embryonic-like state. Cell Res. 33, 712–726 (2023).
Shinoda, G. et al. Fetal deficiency of Lin28 programs life-long aberrations in growth and glucose metabolism. Stem Cell 31, 1563–1573 (2013).
Zhao, H., Wong, R. J. & Stevenson, D. K. The impact of hypoxia in early pregnancy on placental cells. Int. J. Mol. Sci. 22, 9675 (2021).
Morin, S. J. Oxygen tension in embryo culture: does a shift to 2% O2 in extended culture represent the most physiologic system? J. Assist. Reprod. Genet. 34, 309–314 (2017).
Burton, G. J., Cindrova-Davies, T., Yung, H. W. & Jauniaux, E. Hypoxia and reproductive health: oxygen and development of the human placenta. Reproduction 161, F53–f65 (2021).
Rodesch, F., Simon, P., Donner, C. & Jauniaux, E. Oxygen measurements in endometrial and trophoblastic tissues during early pregnancy. Obstet. Gynecol. 80, 283–285 (1992).
Fischer, B. & Bavister, B. D. Oxygen tension in the oviduct and uterus of rhesus monkeys, hamsters and rabbits. J. Reprod. Fertil. 99, 673–679 (1993).
Soares, M. J., Iqbal, K. & Kozai, K. Hypoxia and Placental Development. Birth Defects Res. 109, 1309–1329 (2017).
Prater, M. et al. RNA-seq reveals changes in human placental metabolism, transport and endocrinology across the first-second trimester transition. Biol. Open 10, bio058222 (2021).
Jauniaux, E., Hempstock, J., Greenwold, N. & Burton, G. J. Trophoblastic oxidative stress in relation to temporal and regional differences in maternal placental blood flow in normal and abnormal early pregnancies. Am. J. Pathol. 162, 115–125 (2003).
Jauniaux, E. et al. Onset of maternal arterial blood flow and placental oxidative stress: a possible factor in human early pregnancy failure. Am. J. Pathol. 157, 2111–2122 (2000).
Burton, G. J. Oxygen, the janus gas; its effects on human placental development and function. J. Anat. 215, 27–35 (2009).
Chang, C. W., Wakeland, A. K. & Parast, M. M. Trophoblast lineage specification, differentiation and their regulation by oxygen tension. J. Endocrinol. 236, R43–R56 (2018).
Yang, Y. et al. Hypoxic stress forces irreversible differentiation of a majority of mouse trophoblast stem cells despite FGF41. Biol. Reprod. 95, 110 (2016).
Red-Horse, K. et al. Trophoblast differentiation during embryo implantation and formation of the maternal–fetal interface. J. Clin. Invest. 114, 744–754 (2004).
Wakeland, A. K. et al. Hypoxia directs human extravillous trophoblast differentiation in a hypoxia-inducible factor-dependent manner. Am. J. Pathol. 187, 767–780 (2017).
Adelman, D. M., Gertsenstein, M., Nagy, A., Simon, M. C. & Maltepe, E. Placental cell fates are regulated in vivo by HIF-mediated hypoxia responses. Genes Dev. 14, 3191–3203 (2000).
Cowden Dahl, K. D. et al. Hypoxia-inducible factors 1α and 2α regulate trophoblast differentiation. Mol. Cell Biol. 25, 10479–10491 (2005).
Maltepe, E. et al. Hypoxia-inducible factor-dependent histone deacetylase activity determines stem cell fate in the placenta. Development 132, 3393–3403 (2005).
Spaans, F., de Vos, P., Bakker, W. W., van Goor, H. & Faas, M. M. Danger signals from ATP and adenosine in pregnancy and preeclampsia. Hypertension 63, 1154–1160 (2014).
Sitkovsky, M. & Lukashev, D. Regulation of immune cells by local-tissue oxygen tension: HIF1α and adenosine receptors. Nat. Rev. Immunol. 5, 712–721 (2005).
Ohta, A. et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc. Natl Acad. Sci. USA 103, 13132–13137 (2006).
Sayama, S. et al. Maternal erythrocyte ENT1-mediated AMPK activation counteracts placental hypoxia and supports fetal growth. JCI Insight 5, e130205 (2020).
Huang, A. et al. Elevated adenosine induces placental DNA hypomethylation independent of A2B receptor signaling in preeclampsia. Hypertension 70, 209–218 (2017).
Salmon, J. E. & Girardi, G. Antiphospholipid antibodies and pregnancy loss: a disorder of inflammation. J. Reprod. Immunol. 77, 51–56 (2008).
Lyu, C. et al. Insufficient GDF15 expression predisposes women to unexplained recurrent pregnancy loss by impairing extravillous trophoblast invasion. Cell Prolif. 56, e13514 (2023).
Romero, R., Espinoza, J. & Mazor, M. Can endometrial infection/inflammation explain implantation failure, spontaneous abortion, and preterm birth after in vitro fertilization? Fertil. Steril. 82, 799–804 (2004).
Zhang, Y. et al. The autocrine role of placental extracellular vesicles from missed miscarriage in causing senescence: possible pathogenesis of missed miscarriage. Cells 11, 3873 (2022).
Chen, E. X. et al. Suppression of GATA3 promotes epithelial-mesenchymal transition and simultaneous cellular senescence in human extravillous trophoblasts. Biochim. Biophys. Acta Mol. Cell Res. 1871, 119768 (2024).
Ives, C. W., Sinkey, R., Rajapreyar, I., Tita, A. T. N. & Oparil, S. Preeclampsia-pathophysiology and clinical presentations: JACC state-of-the-art review. J. Am. Coll. Cardiol. 76, 1690–1702 (2020).
Magee, L. A. et al. The 2021 international society for the study of hypertension in pregnancy classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 27, 148–169 (2022).
Sircar, M., Thadhani, R. & Karumanchi, S. A. Pathogenesis of preeclampsia. Curr. Opin. Nephrol. Hypertens. 24, 131–138 (2015).
Thilaganathan, B. & Kalafat, E. Cardiovascular system in preeclampsia and beyond. Hypertension 73, 522–531 (2019).
Yao, Z., Chen, Y., Cao, W. & Shyh-Chang, N. Chromatin-modifying drugs and metabolites in cell fate control. Cell Prolif. 53, e12898 (2020).
Liu, H. et al. 14-3-3 tau (YWHAQ) gene promoter hypermethylation in human placenta of preeclampsia. Placenta 35, 981–988 (2014).
Gao, Y. et al. Increased expression and altered methylation of HERVWE1 in the human placentas of smaller fetuses from monozygotic, dichorionic, discordant twins. PLoS ONE 7, e33503 (2012).
Ma, X., Li, J., Brost, B., Cheng, W. & Jiang, S. W. Decreased expression and DNA methylation levels of GATAD1 in preeclamptic placentas. Cell Signal. 26, 959–967 (2014).
Tang, Y. et al. Hypermethylation of the HLA-G promoter is associated with preeclampsia. Mol. Hum. Reprod. 21, 736–744 (2015).
Zhu, L. et al. Genome-wide mapping of 5mC and 5hmC identified differentially modified genomic regions in late-onset severe preeclampsia: a pilot study. PLoS ONE 10, e0134119 (2015).
Cuffe, J. S. M., Holland, O., Salomon, C., Rice, G. E. & Perkins, A. V. Review: placental derived biomarkers of pregnancy disorders. Placenta 54, 104–110 (2017).
Barker, D. J. et al. Fetal nutrition and cardiovascular disease in adult life. Lancet 341, 938–941 (1993).
Barker, D. J. & Osmond, C. Infant mortality, childhood nutrition, and ischaemic heart disease in England and Wales. Lancet 1, 1077–1081 (1986).
Naruse, K., Tsunemi, T., Kawahara, N. & Kobayashi, H. Preliminary evidence of a paternal-maternal genetic conflict on the placenta: Link between imprinting disorder and multi-generational hypertensive disorders. Placenta 84, 69–73 (2019).
Berti, C. et al. Early-life nutritional exposures and lifelong health: immediate and long-lasting impacts of probiotics, vitamin D, and breastfeeding. Nutr. Rev. 75, 83–97 (2017).
Boney, C. M., Verma, A., Tucker, R. & Vohr, B. R. Metabolic syndrome in childhood: association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 115, e290–e296 (2005).
Oken, E., Taveras, E. M., Kleinman, K. P., Rich-Edwards, J. W. & Gillman, M. W. Gestational weight gain and child adiposity at age 3 years. Am. J. Obstet. Gynecol. 196, 322 (2007).
Pettitt, D. J. et al. Congenital susceptibility to NIDDM. Role of intrauterine environment. Diabetes 37, 622–628 (1988).
Li, S. et al. Childhood cardiovascular risk factors and carotid vascular changes in adulthood: the Bogalusa Heart Study. JAMA 290, 2271–2276 (2003).
Wei, J. et al. FTO mediates LINE1 m6A demethylation and chromatin regulation in mESCs and mouse development. Science 376, 968–973 (2022).
Li, C. W. et al. Pathogenesis of sarcopenia and the relationship with fat mass: descriptive review. J. Cachexia Sarcopenia Muscle 13, 781–794 (2022).
Ma, S. & Shyh-Chang, N. The metabaging cycle. Cell Prolif. 55, e13197 (2022).
Cheng, Y. et al. The metabaging cycle promotes non-metabolic chronic diseases of ageing. Cell Prolif. 57, e13712 (2024).
Yu, D., Luo, L., Wang, H. & Shyh-Chang, N. Pregnancy-induced metabolic reprogramming and regenerative responses to pro-aging stresses. Trends Endocrinol. Metab. 36, 482–494 (2025).
Akhaphong, B. et al. Placental mTOR complex 1 regulates fetal programming of obesity and insulin resistance in mice. JCI Insight 6, e149271 (2021).
Zhang, Q. et al. Adrenomedullin has a pivotal role in trophoblast differentiation: A promising nanotechnology-based therapeutic target for early-onset preeclampsia. Sci. Adv. 9, eadi4777 (2023).
Peng, B. et al. Red blood cell extracellular vesicles deliver therapeutic siRNAs to skeletal muscles for treatment of cancer cachexia. Mol. Ther. 31, 1418–1436 (2023).
Lees, C. C. et al. Clinical opinion: The diagnosis and management of suspected fetal growth restriction: an evidence-based approach. Am. J. Obstet. Gynecol. 226, 366–378 (2022).
Dimitriadis, E., Menkhorst, E., Saito, S., Kutteh, W. H. & Brosens, J. J. Recurrent pregnancy loss. Nat. Rev. Dis. Primers 6, 98 (2020).
Girardi, G. & Bremer, A. A. The intersection of maternal metabolic syndrome, adverse pregnancy outcomes, and future metabolic health for the mother and offspring. Metab. Syndr. Relat. Disord. 20, 251–254 (2022).
Acknowledgements
The authors acknowledge the support of grants from the National Key Research and Development Program of China (grant no. 2022YFC2702400) and the National Natural Science Foundation of China (grant nos. 82421003, 82192872, 82495190, 32400655, U21A20396 and 2024YFA1802200).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article. N.S.-C., H.W. and Y.-L.W. contributed substantially to discussion of the content. N.S.-C., H.W., Y.-L.W., Y.G, X.Y. and Y.W. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Trond Michelsen, Ye Yuan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, Y., Yu, X., Wang, Y. et al. Metabolic control of feto–placental development and pregnancy outcomes. Nat Rev Endocrinol 22, 153–165 (2026). https://doi.org/10.1038/s41574-025-01206-9
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41574-025-01206-9