Abstract
This Review discusses the current state of knowledge on the contribution of the gut microbiome as a potential key actor in defining how we age. The gut microbiome is a complex ecosystem that establishes lifelong dynamic interactions with the host at multiple levels (several gut–organ axes), differently influencing ageing patterns and age-related disease onset and progression across populations. Accordingly, the definition of a ‘normative’ gut microbiome remains elusive, depending largely on the interaction with the external environment. In this complex scenario, the causal role of the gut microbiome in defining the ageing trajectory and its precise contribution to various organ-specific age-related diseases is still uncertain in clinical terms and could be context specific. Multiparametric and uniqueness indexes within a given population have shown a certain capacity for predicting disability and mortality. However, the gut microbiome is shaped over time by exposure to different intrinsic and environmental factors, resulting in a high degree of inter-individual variability, a key phenomenon that should be considered to develop novel personalized strategies to counteract age-related disease and frailty.
Key points
-
Ageing is associated with a reconfiguration of the gut microbial communities towards dysbiosis. This process is influenced by life-long environmental exposures and is highly individualized, resulting in elevated inter-individual microbiome variability.
-
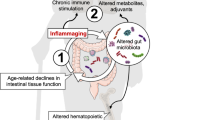
Gut microbiome reconfiguration during ageing is influenced by gastrointestinal cell senescence and provides a great amount of antigenic stimulation to the senescent immune system, fuelling inflammageing and favouring trajectories towards the frailty syndrome.
-
The gut microbiome of centenarians shows an exceptional capacity of adaptation to the pathophysiological processes leading to inflammageing, with persistence of a set of bacterial taxa with anti-inflammatory properties.
-
The dynamic interaction among lifestyle, gut microbiome and cell and organ senescence could influence the adaptation of each individual to the biological mechanisms of ageing, resulting in different clinical trajectories towards health or disease.
-
Modulation of the gut microbiome with diet, exercise and nutritional supplements containing probiotics, prebiotics or food bioactives could represent an effective strategy to improve the ageing trajectory in older individuals.
-
Microbiome-centred interventions should be personalized and take into account the inter-individual variability of microbial responses. Microbiome profiling and metabotyping should be increased to enable more efficient translation of microbiome science into clinical practice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Berg, B. et al. Microbiome definition re-visited: old concepts and new challenges. Microbiome 8, 103 (2020).
de Vos, W. M., Tilg, H., Van Hul, M. & Cani, P. D. Gut microbiome and health: mechanistic insights. Gut 71, 1020–1032 (2022).
Joos, R. et al. Examining the healthy human microbiome concept. Nat. Rev. Microbiol. 23, 192–205 (2024).
Posteraro, B., De Maio, F. & Gasbarrini, A. Profiling the gastrointestinal microbiota. Methods Mol. Biol. 2283, 83–92 (2021).
Joseph, T. A. & Pe’er, I. An introduction to whole-metagenome shotgun sequencing studies. Methods Mol. Biol. 2243, 107–122 (2021).
Jurburg, S. D. et al. The community ecology perspective of omics data. Microbiome 10, 225 (2022).
Xiao, L. & Zhao, F. Microbial transmission, colonisation and succession: from pregnancy to infancy. Gut 72, 772–786 (2023).
Dominguez-Bello, M. G. et al. Partial restoration of the microbiota of caesarean-born infants via vaginal microbial transfer. Nat. Med. 22, 250–253 (2016).
Bogaert, D. et al. Mother-to-infant microbiota transmission and infant microbiota development across multiple body sites. Cell Host Microbe 31, 447–460.e6 (2023).
Stewart, C. J. et al. Temporal development of the gut microbiome in early childhood from the TEDDY study. Nature 562, 583–588 (2018).
Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 (2018).
Wilde, J., Slack, E. & Foster, K. R. Host control of the microbiome: mechanisms, evolution, and disease. Science 385, eadi3338 (2024).
Bäcked, F. et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 17, 690–703 (2015).
Gacesa, R. et al. Environmental factors shaping the gut microbiome in a Dutch population. Nature 604, 732–739 (2022).
Zhernakova, A. et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 352, 565–569 (2016).
Mancabelli, L. et al. Taxonomic and metabolic development of the human gut microbiome across life stages: a worldwide metagenomic investigation. mSystems 9, e0129423 (2024).
Kurilshikov, A. et al. Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat. Genet. 53, 156–165 (2021).
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
Asnicar, F. et al. Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals. Nat. Med. 27, 321–332 (2021).
Lynch, S. V. & Pedersen, O. The human intestinal microbiome in health and disease. N. Engl. J. Med. 365, 2369–2379 (2016).
Sommer, F., Moltzau Anderson, J. M., Bharti, R., Raes, J. & Rosenstiel, P. The resilience of the intestinal microbiota influences health and disease. Nat. Rev. Microbiol. 15, 630–638 (2017).
Schwartz, D. J., Langdon, A. E. & Dantas, G. Understanding the impact of antibiotic perturbation on the human microbiome. Genome Med. 12, 82 (2020).
Fragiadakis, G. K. et al. Long-term dietary intervention reveals resilience of the gut microbiota despite changes in diet and weight. Am. J. Clin. Nutr. 111, 1127–1136 (2020).
Ross, F. C. et al. The interplay between diet and the gut microbiome: implications for health and disease. Nat. Rev. Microbiol. 22, 671–686 (2024).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Bhattarai, S. K. et al. Commensal antimicrobial resistance mediates microbiome resilience to antibiotic disruption. Sci. Transl. Med. 16, eadi9711 (2024).
Tiffany, C. R. & Bäumler, A. J. Dysbiosis: from fiction to function. Am. J. Physiol. Gastrointest. Liver Physiol. 317, G602–G608 (2019).
Ling, Z. et al. Gut microbiota and aging. Crit. Rev. Food Sci. Nutr. 62, 3509–3534 (2022).
Kroemer, G. et al. From geroscience to precision geromedicine: understanding and managing aging. Cell 188, 2043–2062 (2025).
Haran, J. P. & McCormick, B. A. Aging, frailty, and the microbiome — how dysbiosis influences human aging and disease. Gastroenterology 160, 507–523 (2021).
Bradley, E. & Haran, J. P. The human gut microbiome and aging. Gut Microbes 16, 2359677 (2024).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Jackson, M. A. et al. Gut microbiota associations with common diseases and prescription medications in a population-based cohort. Nat. Commun. 9, 2655 (2018).
Schmidt, T. S. B., Raes, J. & Bork, P. The human gut microbiome: from association to modulation. Cell 172, 1198–1215 (2018).
Tilg, H., Adolph, T. E. & Trauner, M. Gut–liver axis: pathophysiological concepts and clinical implications. Cell Metab. 34, 1700–1718 (2022).
Cryan, J. F. et al. The microbiota–gut–brain axis. Physiol. Rev. 99, 1877–2013 (2019).
Stavropoulou, E. et al. Focus on the gut–kidney axis in health and disease. Front. Med. 7, 620102 (2021).
Ticinesi, A. et al. Aging gut microbiota at the cross-road between nutrition, physical frailty, and sarcopenia: is there a gut–muscle axis? Nutrients 9, 1303 (2017).
Ticinesi, A., Siniscalchi, C., Meschi, T. & Nouvenne, A. Gut microbiome and bone health: update on mechanisms, clinical correlations and possible treatment strategies. Osteoporos. Int. 36, 167–191 (2025).
Wang, L. et al. The bidirectional gut–lung axis in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 207, 1145–1160 (2023).
Blaak, E. E. et al. Short chain fatty acids in human gut and metabolic health. Benef. Microbes 11, 411–455 (2020).
Belcheva, A. et al. Gut microbial metabolism drives transformation of Msh2-deficient colon epithelial cells. Cell 158, 288–299 (2014).
Singh, V. et al. Dysregulated microbial fermentation of soluble fibers induces cholestatic liver cancer. Cell 175, 679–694 (2018).
Okumura, S. et al. Gut bacteria identified in colorectal cancer patients promote tumourigenesis via butyrate secretion. Nat. Commun. 12, 5674 (2021).
Strasser, B., Wolters, M., Weyh, C., Krüger, K. & Ticinesi, A. The effects of lifestyle and diet on gut microbiota composition, inflammation and muscle performance in our aging society. Nutrients 13, 2045 (2021).
Conway, J. & Duggal, N. A. Ageing of the gut microbiome: potential influences on immune senescence and inflammageing. Ageing Res. Rev. 68, 101323 (2021).
Maynard, C. & Weinkove, D. The gut microbiota and ageing. Subcell. Biochem. 90, 351–371 (2018).
O’Toole, P. W. & Shiels, P. G. The role of the microbiota in sedentary lifestyle disorders and ageing: lessons from the animal kingdom. J. Intern. Med. 287, 271–282 (2020).
Touvier, M. Health effects of ultra-processed food: uncovering causal mechanisms. Nat. Rev. Endocrinol. 22, 66–67 (2025).
Minnis, H. et al. The bio-exposome: intracellular processes, stress physiology and the environment. Nat. Ment. Health 2, 132–140 (2024).
Kontele, I. et al. Socio-demographic determinants of Mediterranean diet adherence: results of the EU-National Health Interview Survey (EHIS3). J. Hum. Nutr. Diet. 38, e70023 (2025).
Maugeri, A. et al. Adherence to the Mediterranean diet partially mediates socioeconomic differences in leukocyte LINE-1 methylation: evidence from a cross-sectional study in Italian women. Sci. Rep. 10, 14360 (2020).
Shanahan, F., Ghosh, T. S. & O’Toole, P. W. The healthy microbiome — what is the definition of a healthy gut microbiome? Gastroenterology 160, 483–494 (2021).
Van Hul, M. et al. What defines a healthy gut microbiome? Gut 73, 1893–1908 (2024).
Najmanová, L., Vídeňská, P. & Cahová, M. Healthy microbiome — a mere idea or a sound concept? Physiol. Res. 71, 719–738 (2022).
Wilmanski, T., Rappaport, N., Diener, C., Gibbons, S. M. & Price, N. D. From taxonomy to metabolic output: what factors define gut microbiome health? Gut Microbes 13, e1907270 (2021).
De Siqueira, M. K. et al. Infection-elicited microbiota promotes host adaptation to nutrient restriction. Proc. Natl Acad. Sci. USA 120, e2214484120 (2023).
Carter, M. M. et al. Ultra-deep sequencing of Hadza hunter-gatherers recovers vanishing gut microbes. Cell 186, 3111–3124.e13 (2023).
Vandeputte, D. et al. Temporal variability in quantitative human gut microbiome profiles and implications for clinical research. Nat. Commun. 12, 6740 (2021).
Nishijima, S. et al. Fecal microbial load is a major determinant of gut microbiome variation and a confounder for disease associations. Cell 188, 222–236 (2025).
Gupta, V. K. et al. A predictive index for health status using species-level gut microbiome profiling. Nat. Commun. 11, 4635 (2020).
Salosensaari, A. et al. Taxonomic signatures of cause-specific mortality risk in human gut microbiome. Nat. Commun. 12, 2671 (2021).
Wilmanski, T. et al. Gut microbiome pattern reflects healthy ageing and predicts survival in humans. Nat. Metab. 3, 274–286 (2021).
Si, J. et al. Long-term life history predicts current gut microbiome in a population-based cohort study. Nat. Aging 2, 885–895 (2022).
Ghosh, T. S., Das, M., Jeffery, I. B. & O’Toole, P. W. Adjusting for age improves identification of gut microbiome alterations in multiple diseases. eLife 9, e50240 (2020).
Claesson, M. J. et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 488, 178–184 (2012).
Rahayu, E. S. et al. Gut microbiota profile in healthy Indonesians. World J. Gastroenterol. 25, 1478–1491 (2019).
Xu, Y. et al. Altered fecal microbiota composition in older adults with frailty. Front. Cell. Infect. Microbiol. 11, 696186 (2021).
Haran, J. P. et al. The nursing home older adult gut microbiome composition shows time-dependent dysbiosis and is influenced by medication exposures, age, environment, and frailty. J. Gerontol. A Biol. Sci. Med. Sci. 76, 1930–1938 (2021).
Ghosh, T. S., Shanahan, F. & O’Toole, P. W. Toward an improved definition of a healthy microbiome for healthy aging. Nat. Aging 2, 1054–1069 (2022).
Ferrucci, L. et al. Measuring biological aging in humans: a quest. Aging Cell 19, e13080 (2020).
Fong, S. et al. Principal component-based clinical aging clocks identify signatures of healthy aging and targets for clinical intervention. Nat. Aging 4, 1137–1152 (2024).
Drewelies, J. et al. Using blood test parameters to define biological age among older adults: association with morbidity and mortality independent of chronological age validated in two separate birth cohorts. Geroscience 44, 2685–2699 (2022).
Kwon, D. & Belsky, D. W. A toolkit for quantification of biological age from blood chemistry and organ function test data: bioage. Geroscience 43, 2795–2808 (2021).
Gems, D., Virk, R. S. & de Magalhaes, J. P. Epigenetic clocks and programmatic aging. Ageing Res. Rev. 101, 102546 (2024).
Prattichizzo, F. et al. Organ-specific biological clocks: ageotyping for personalized anti-aging medicine. Ageing Res. Rev. 96, 102253 (2024).
Apsley, A. T. et al. Cross-tissue comparison of epigenetic aging clocks in humans. Aging Cell 24, e14451 (2025).
Huang, S. et al. Human skin, oral, and gut microbiomes predict chronological age. mSystems 5, e00630-19 (2020).
Galkin, F. et al. Human gut microbiome aging clock based on taxonomic profiling and deep learning. iScience 23, 101199 (2020).
Gopu, S. et al. An accurate aging clock developed from large-scale gut microbiome and human gene expression data. iScience 27, 108538 (2023).
Wang, H. et al. A gut aging clock using microbiome multi-view profiles is associated with health and frail risk. Gut Microbes 16, 2297852 (2024).
Wang, T. et al. Divergent age-associated and metabolism-associated gut microbiome signatures modulate cardiovascular disease risk. Nat. Med. 30, 1722–1731 (2024).
Sayed, N. et al. An inflammatory aging clock (iAge) based on deep learning tracks multimorbidity, immunosenescence, frailty and cardiovascular aging. Nat. Aging 1, 598–615 (2021).
Kim, D. H. & Rockwood, K. Frailty in older adults. N. Engl. J. Med. 391, 538–548 (2024).
Picca, A., Coelho-Junior, H. J., Calvani, R., Marzetti, E. & Vetrano, D. L. Biomarkers shared by frailty and sarcopenia in older adults: a systematic review and meta-analysis. Ageing Res. Rev. 73, 101530 (2022).
Boehme, M. et al. Microbiota from young mice counteracts selective age-associated behavioral deficits. Nat. Aging 1, 666–676 (2021).
Mo, X. et al. Faecal microbiota transplantation from young rats attenuates age-related sarcopenia revealed by multiomics analysis. J. Cachexia Sarcopenia Muscle 14, 2168–2183 (2023).
Zhu, M. et al. Fecal microbiota transplantation attenuates frailty via gut–muscle axis in old mice. Aging Dis. 16, 1180–1198 (2024).
Fielding, R. A. et al. Muscle strength is increased in mice that are colonized with microbiota from high-functioning older adults. Exp. Gerontol. 127, 110722 (2019).
Farsijani, S. et al. Associations between walking speed and gut microbiome composition in older men from the MrOS Study. J. Gerontol. A Biol. Sci. Med. Sci. 79, glae030 (2024).
Guo, Y. et al. Serum metabolome and gut microbiome alterations are associated with low handgrip strength in older adults. Aging 16, 2368–2656 (2024).
Lapauw, L. et al. Associations between gut microbiota and sarcopenia or its defining parameters in older adults: a systematic review. J. Cachexia Sarcopenia Muscle 15, 2190–2207 (2024).
Almeida, H. M., Sardeli, A. V., Conway, J., Duggal, N. D. & Cavaglieri, C. R. Comparison between frail and non-frail older adults’ gut microbiota: a systematic review and meta-analysis. Ageing Res. Rev. 82, 101773 (2022).
Jackson, M. A. et al. Signatures of early frailty in the gut microbiota. Genome Med. 8, 8 (2016).
Verdi, S. et al. An investigation into physical frailty as a link between the gut microbiome and cognitive health. Front. Aging Neurosci. 10, 398 (2018).
Lim, M. Y., Hong, S., Kim, J. H. & Nam, Y. D. Association between gut microbiome and frailty in the older adult population in Korea. J. Gerontol. A Biol. Sci. Med. Sci. 76, 1362–1368 (2021).
Pu, Y. et al. Gut microbial features and circulating metabolomic signatures of frailty in older adults. Nat. Aging 4, 1249–1262 (2024).
Fulop, T. et al. Immunology of aging: the birth of inflammaging. Clin. Rev. Allergy Immunol. 64, 109–122 (2023).
Ferrucci, L. & Fabbri, E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 15, 505–522 (2018).
Fulop, T. et al. Immunosenescence is both functional/adaptive and dysfunctional/maladaptive. Semin. Immunopathol. 42, 521–536 (2020).
Franceschi, C., Olivieri, F., Moskalev, A., Ivanchenko, M. & Santoro, A. Towards precision interventions and metrics of inflammaging. Nat. Aging 5, 1441–1454 (2025).
Franck, M. et al. Nonuniversality of inflammaging across human populations. Nat. Aging 5, 1481–1490 (2025).
Olivieri, F. et al. Antifragility and antiinflammaging: can they play a role for a healthy longevity? Ageing Res. Rev. 84, 101836 (2023).
Gensous, N. et al. Targeted epigenetic clock for the prediction of biological age. Cells 11, 4044 (2022).
Franceschi, C. et al. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Franceschi, C. et al. Immunobiography and the heterogeneity of immune responses in the elderly: a focus on inflammaging and trained immunity. Front. Immunol. 8, 982 (2017).
Untersmayr, E. et al. The intestinal barrier dysfunction as driving factor of inflammaging. Nutrients 14, 949 (2022).
Witkowski, J. M. et al. Immunosenescence and COVID-19. Mech. Ageing Dev. 204, 111672 (2022).
Domínguez-Andrés, J. et al. Trained immunity: adaptation within innate immune mechanisms. Physiol. Rev. 103, 313–343 (2023).
Blacher, E. et al. Aging disrupts circadian age regulation and function in macrophages. Nat. Immun. 23, 229–236 (2022).
Carrasco, E. et al. The role of T cells in age-related diseases. Nat. Rev. Immunol. 22, 97–111 (2022).
Guedj, A. et al. Gut microbiota shape ‘inflamm-ageing’ cytokines and account for age-dependent decline in DNA damage repair. Gut 69, 1064–1075 (2020).
Bashir, H. et al. Age-mediated gut microbiota dysbiosis promotes the loss of dendritic cells tolerance. Aging Cell 22, e13838 (2023).
Koeth, R. A. et al. L-carnitine in omnivorous diets induces an atherogenic gut microbial pathway in humans. J. Clin. Invest. 129, 373–387 (2019).
Song, Y. et al. Gut microbiota-dependent phyenylacetylglutamine in cardiovascular disease: current knowledge and new insights. Front. Med. 18, 31–45 (2024).
Zhang, L., Yu, F. & Xia, J. Trimethylamine-N-oxide: role in cell senescence and age-related diseases. Eur. J. Nutr. 62, 525–541 (2023).
Yang, H. et al. Gut microbial-derived phenylacetylglutamine accelerates host cellular senescence. Nat. Aging 5, 401–418 (2025).
Liu, Y. et al. Dietary polyphenols as anti-aging agents: targeting the hallmarks of aging. Nutrients 16, 3305 (2024).
Zumerle, S. et al. Targeting senescence induced by age or chemotherapy with a polyphenol-rich natural extract improves longevity and healthspan in mice. Nat. Aging 4, 1231–1248 (2024).
Santoro, A. et al. Gut microbiota changes in the extreme decades of human life: a focus on centenarians. Cell. Mol. Life Sci. 75, 129–148 (2018).
Wang, J. et al. The landscape in the gut microbiome of long-lived families reveals new insights on longevity and aging — relevant neural and immune function. Gut Microbes 14, e2107288 (2022).
Li, C. et al. Deep insights into the gut microbial community of extreme longevity in south Chinese centenarians by ultra-deep metagenomics and large-scale culturomics. npj Biofilms Microbiomes 8, 28 (2022).
Johansen, J. et al. Centenarians have a diverse gut virome with the potential to modulate metabolism and promote healthy lifespan. Nat. Microbiol. 8, 1064–1078 (2023).
Pu, L. et al. The gut mycobiome signatures in long-lived populations. iScience 27, 110412 (2024).
Chen, S. et al. Consistent signatures in the human gut microbiome of longevous populations. Gut Microbes 16, 2393756 (2024).
Pang, S. et al. Longevity of centenarians is reflected by the gut microbiome with youth-associated signatures. Nat. Aging 3, 436–449 (2023).
Rampelli, S. et al. Shotgun metagenomics of gut microbiota in humans with up to extreme longevity and the increasing role of xenobiotic degradation. mSystems 5, e00124-20 (2020).
Sato, Y. et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 599, 458–464 (2021).
Tavella, T., Turroni, S., Brigidi, P., Candela, M. & Rampelli, S. The human gut resistome up to extreme longevity. mSphere 6, e00691–21 (2021).
Lu, C. et al. Antimicrobial peptides from the gut microbiome of the centenarians: diversification of biosynthesis and youthful development of resistance genes. J. Gerontol. A Biol. Sci. Med. Sci. 79, glae218 (2024).
Santos-Pujol, E. et al. The multiomics blueprint of the individual with the most extreme lifespan. Cell Rep. Med. 6, 102368 (2025).
Jang, D. H. et al. The connection between aging, cellular senescence and gut microbiome alterations: a comprehensive review. Aging Cell 23, e14315 (2024).
Kawamoto, S. & Hara, E. Crosstalk between gut microbiota and cellular senescence: a vicious cycle leading to aging gut. Trends Cell Biol. 34, 626–634 (2024).
Kawamoto, S. et al. Bacterial induction of B cell senescence promotes age-related changes in the gut microbiota. Nat. Cell Biol. 25, 865–876 (2023).
Di Vincenzo, F., Del Gaudio, A., Petito, V., Lopetuso, L. R. & Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern. Emerg. Med. 19, 275–293 (2024).
Zhang, D. et al. Neutrophil ageing is regulated by the microbiome. Nature 525, 528–532 (2015).
Shoshkes-Carmel, M. Telocytes in the luminal GI tract. Cell. Mol. Gastroenterol. Hepatol. 17, 697–701 (2024).
Ahmad Fadzuli, N. I. et al. Faecal intestinal permeability and intestinal inflammatory markers in older adults with age-related disorders: a systematic review and meta-analysis. Ageing Res. Rev. 101, 102506 (2024).
Boschetti, E. et al. Serum zonulin levels are increased in Alzheimer’s disease but not in vascular dementia. Aging Clin. Exp. Res. 35, 1835–1843 (2023).
Palermo, F. et al. Investigating gut alterations in Alzheimer’s disease: in-depth analysis with micro- and nano-3D X-ray phase contrast tomography. Sci. Adv. 11, eadr8511 (2025).
Carloni, S. et al. Identification of a choroid plexus vascular barrier closing during intestinal inflammation. Science 374, 439–448 (2021).
Carloni, S. & Rescigno, M. The gut–brain vascular axis in neuroinflammation. Semin. Immunol. 69, 101802 (2023).
Mustafa, M. A. et al. The gut–brain axis in Alzheimer’s disease: exploring microbial influences and therapeutic strategies. Mol. Neurobiol. 63, 151 (2025).
Ahmed, H. et al. Microbiota-derived metabolites as drivers of gut–brain communication. Gut Microbes 14, 2102878 (2022).
Borbolis, F., Mytilinaiou, E. & Palikaras, K. The crosstalk between microbiome and mitochondrial homeostasis in neurodegeneration. Cells 12, 429 (2023).
Miller, B. C., Mathai, M., Yadav, H. & Jain, S. Geroprotective potential of microbiome modulators in the Caenorhabditis elegans model. Geroscience 46, 129–151 (2024).
Liu, C. et al. Understanding the gut microbiota and sarcopenia: a systematic review. J. Cachexia Sarcopenia Muscle 12, 1393–1407 (2021).
de Rijke, T. J. et al. A systematic review on the effects of different types of probiotics in animal Alzheimer’s disease studies. Front. Psychiatry 13, 879491 (2022).
Herisson, F. M. et al. Targeting the gut–heart axis improves cardiac remodeling in a clinical scale model of cardiometabolic syndrome. JACC Basic Transl. Sci. 10, 1–15 (2025).
Lazou-Ahrén, I. et al. Probiotic-reduced inflammaging in older adults : a randomized double-blind, placebo-controlled trial. Probiot. Antimicrob. Prot. 17, 3429–3439 (2025).
Castro-Herrera, V. M. et al. Combination of the probiotics Lacticaseibacillus rhamnosus GG and Bifidobacterium animalis subsp. lactis, BB-12 has limited effect on biomarkers of immunity and inflammation in older people resident in care homes: results from the Probiotics to Reduce Infections iN CarE home reSidentS randomized controlled trial. Front. Immunol. 12, 643321 (2021).
Besora-Moreno, M., Llauradò, E., Valls, R. M., Pedret, R. & Solà, R. Effects of probiotics, prebiotics, and synbiotics on sarcopenia parameters in older adults: a systematic review and meta-analysis of randomized controlled trials. Nutr. Rev. 83, e1693–e1708 (2025).
Lapauw, L. et al. Effect of host and gut microbiota-altering interventions on sarcopenia or its defining parameters: a systematic review and meta-analysis of nutrition-based intervention studies. Aging Clin. Exp. Res. 38, 17 (2025).
Karim, A. et al. A multistrain probiotic reduces sarcopenia by modulating Wnt signaling biomarkers in patients with chronic heart failure. J. Cardiol. 80, 449–455 (2022).
Karim, A. et al. A multistrain probiotic improves handgrip strength and functional capacity in patients with COPD: a randomized controlled trial. Arch. Gerontol. Geriatr. 102, 140721 (2022).
Nistor-Cseppento, C. et al. The contribution of diet therapy and probiotics in the treatment of sarcopenia induced by prolonged immobilization caused by the COVID-19 pandemic. Nutrients 14, 4701 (2021).
Rondanelli, M. et al. Effect of a novel food composed of leucine, omega-3 fatty acids and probiotic Lactobacillus paracasei PS23 for the treatment of sarcopenia in elderly subjects: a 2-month randomized double-blind placebo-controlled trial. Nutrients 14, 4566 (2022).
Qaisar, R. et al. Probiotics supplements improve the sarcopenia-related quality of life in older adults with age-related muscle decline. Calcif. Tissue Int. 114, 583–591 (2024).
Zhang, Z. et al. Bifidobacterium animalis Probio-M8 improves sarcopenia physical performance by mitigating creatine restrictions imposed by microbial metabolites. npj Biofilms Microbiomes 10, 144 (2024).
Kang, C. H. et al. Pasteurized Akkermansia muciniphila HB05 (HB05P) improves muscle strength and function: a 12-week, randomized, double-blind, placebo-controlled clinical trial. Nutrients 16, 4037 (2024).
Román, E. et al. Home exercise, branched-chain amino acids, and probiotics improve frailty in cirrhosis: a randomized clinical trial. Hepatol. Commun. 8, e0443 (2024).
Buigues, C. et al. Effect of a prebiotic formulation on frailty syndrome: a randomized, double-blind clinical trial. Int. J. Mol. Sci. 17, 932 (2016).
Theou, O. et al. Can a prebiotic formulation reduce frailty levels in older people? J. Frailty Aging 8, 48–52 (2019).
Tominaga, K. et al. Increase in muscle mass associated with the prebiotic effects of 1-kestose in super-elderly patients with sarcopenia. Biosci. Microbiota Food Health 40, 150–155 (2021).
Yang, J. et al. Probiotics improve frailty status in community-dwelling older individuals in a double-blind, randomized, controlled trial. J. Clin. Invest. 134, e176507 (2024).
Ni Lochlainn, M. et al. Effect of gut microbiome modulation on muscle function and cognition: the PROMOTe randomised controlled trial. Nat. Commun. 15, 1859 (2024).
Liu, X., Ning, L., Fan, W., Jia, C. & Ge, L. Probiotics and cognitive-related health outcomes: an umbrella review of systematic reviews and meta-analyses of randomized controlled trials. Nutr. Rev. 83, 2144–2158 (2025).
Guo, H. Q. et al. Effects of probiotics on cognitive function across the human lifespan: a meta-analysis. Eur. J. Clin. Nutr. 80, 46–61 (2026).
Calzada-Gonzales, N. et al. Efficacy and safety of probiotic supplements on cognitive function: a systematic review and meta-analysis of randomized clinical trials. BMC Complement. Med. Ther. 25, 432 (2025).
Guarente, L., Sinclair, D. A. & Kroemer, G. Human trials exploring anti-aging medicines. Cell Metab. 36, 354–376 (2024).
Rolland, Y., Ticinesi, A., Sokol, H. & De Souto Barreto, P. Therapeutic perspectives of pre-, pro-, post-biotics in the treatment of sarcopenia. J. Nutr. Health Aging 28, 100298 (2024).
Tran, T. T. T. et al. Prebiotic supplementation in frail older people affects specific gut microbiota taxa but not global diversity. Microbiome 7, 39 (2019).
Salvesi, C. et al. Impact of a probiotic diet on well-being of healthy senior: THE PROBIOSENIOR PROJECT. J. Appl. Microbiol. 133, 2941–2953 (2022).
Di Giosia, P. et al. The role of nutrition in inflammaging. Ageing Res. Rev. 77, 101596 (2022).
Ticinesi, A., Nouvenne, A., Cerundolo, N., Parise, A., Mena, P. & Meschi, T. The interaction between Mediterranean diet and intestinal microbiome: relevance for preventive strategies against frailty in older individuals. Aging Clin. Exp. Res. 36, 58 (2024).
Ghosh, T. S. et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut 69, 1218–1228 (2020).
Muralidharan, J. et al. Effect on gut microbiota of a 1-y lifestyle intervention with Mediterranean diet compared with energy-reduced Mediterranean diet and physical activity promotion: PREDIMED-Plus Study. Am. J. Clin. Nutr. 114, 1148–1158 (2021).
Sbierski-Kind, J. et al. Effects of caloric restriction on the gut microbiome are linked with immune senescence. Microbiome 10, 57 (2022).
Gensous, N. et al. One-year Mediterranean diet promotes epigenetic rejuvenation with country- and sex-specific effects: a pilot study from the NU-AGE project. Geroscience 42, 687–701 (2020).
Waziry, R. et al. Effect of long-term caloric restriction on DNA methylation measures of biological aging in healthy adults from the CALERIE trial. Nat. Aging 3, 248–257 (2023).
Nagpal, R. et al. Modified Mediterranean-ketogenic diet modulates the gut microbiome and short-chain fatty acids in association with Alzheimer’s disease markers in subjects with mild cognitive impairment: a pilot study. eBioMedicine 59, 102950 (2019).
Dilmore, A. H. et al. Effect of a ketogenic and low-fat diet on the human metabolome, microbiome, and foodome in adults at risk for Alzheimer’s disease. Alzheimers Dement. 19, 4805–4816 (2023).
Ford, A. L. et al. Microbiota stability and gastrointestinal tolerance in response to a high-protein diet with and without a prebiotic, probiotic, and synbiotic: a randomized, double-blind, placebo-controlled trial in older women. J. Acad. Nutr. Diet. 120, 500–516 (2020).
Allen, J. M. et al. Exercise alters gut microbiota composition and function in lean and obese humans. Med. Sci. Sports Exerc. 50, 747–757 (2018).
Grosicki, G. J. et al. Gut check: unveiling the influence of acute exercise on the gut microbiota. Exp. Physiol. 108, 1466–1480 (2023).
Qi, X., Kong, H., Li, J. & Chen, C. The relationship between insomnia and multidimensional frailty in community-dwelling older adults: a systematic review. Arch. Gerontol. Geriatr. 129, 105661 (2025).
Estaki, M. et al. Association of subjective and objective measures of sleep with gut microbiota composition and diversity in older men: the Osteoporotic Fractures in Men Study. J. Gerontol. A Biol. Sci. Med. Sci. 78, 1925–1932 (2023).
Palmnäs, M. et al. Perspective: metabotyping-a potential personalized nutrition strategy for precision prevention of cardiometabolic disease. Adv. Nutr. 11, 524–532 (2020).
Calderón-Pérez, L. et al. A single-blinded, randomized, parallel intervention to evaluate genetics and omics-based personalized nutrition in general population via an e-commerce tool: the PREVENTOMICS e-commerce study. Am. J. Clin. Nutr. 120, 129–144 (2024).
Quinn-Bohmann, N. et al. Microbial community-scale metabolic modelling predicts personalized short-chain fatty acid production profiles in the human gut. Nat. Microbiol. 9, 1700–1712 (2024).
Cortés-Martín, A. et al. Where to look into the puzzle of polyphenols and health? The postbiotics and gut microbiota associated with human metabotypes. Mol. Nutr. Food Res. 64, 1900952 (2020).
Favari, C. et al. Factors driving the inter-individual variability in the metabolism and bioavailability of (poly)phenolic metabolites: a systematic review of human studies. Redox Biol. 71, 103095 (2024).
Tosi, N. et al. Unravelling phenolic metabotypes in the frame of the COMBAT study, a randomized, controlled trial with cranberry supplementation. Food Res. Int. 172, 113187 (2023).
Cortés-Martín, A. et al. The gut microbiota urolithin metabotypes revisited: the human metabolism of ellagic acid is mainly determined by aging. Food Funct. 9, 4100–4106 (2018).
Meroño, T. et al. The relevance of urolithins-based metabotyping for assessing the effects of a polyphenol-rich dietary intervention on intestinal permeability: a post-hoc analysis of the MaPLE trial. Food Res. Int. 159, 11632 (2022).
Ratiner, K., Ciocan, D., Abdeen, S. K. & Elinav, E. Utilization of the microbiome in personalized medicine. Nat. Rev. Microbiol. 22, 291–308 (2024).
Tian, S. et al. A designed synthetic microbiota provides insight to community function in Clostridioides difficile resistance. Cell Host Microbe 33, 1–15 (2025).
Author information
Authors and Affiliations
Contributions
A.T. and C.F. researched data for the article. S.M., G.Z., A.N. and C.F. contributed substantially to discussion of the content. A.T. wrote the article. S.M., G.Z., A.N. and C.F. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Cristina Menni, who co-reviewed with Ruth Bowyer, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ticinesi, A., Maggi, S., Nouvenne, A. et al. The gut microbiome and ageing trajectories: mechanisms and clinical implications. Nat Rev Endocrinol (2026). https://doi.org/10.1038/s41574-026-01236-x
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41574-026-01236-x