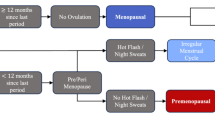
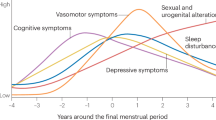
Abstract
Menopause, particularly the consequences of severe symptoms, has become the subject of intense media interest. Reducing the prevalent stigma around women’s reproductive health has allowed more women to speak openly about their menopause experiences, the effects on their lives and the barriers they face. These effects are far-reaching and illustrate the importance of improving the understanding, management and awareness of severe menopausal symptoms. The cardinal symptoms are hot flushes (or flashes) and night sweats (vasomotor symptoms), although symptoms can vary by ethnicity. For example, some Asian women report other primary symptoms (such as bone and/or joint pain). Vasomotor symptoms affect around 70% of perimenopausal and postmenopausal women and are moderate or severe in around one-third of these women. The US Food and Drug Administration considers vasomotor symptoms to be severe if they cause the person to stop their current activity. Severe vasomotor symptoms drive treatment seeking and can affect quality of life, mental health and work ability. We review the incidence, management and potential long-term health consequences of severe vasomotor symptoms, including cardiovascular disease, diabetes mellitus, cognitive dysfunction, bone health and quality of life. We discuss potential underlying mechanisms and the efficacy of available treatments. Finally, we highlight the evidence gaps in this field and directions for future research.
Key points
-
The experience of menopause varies dramatically between women, and for the same woman over time. Differences in the frequency and severity of vasomotor symptoms and uptake of treatment are evident between racial, ethnic and socio-economic groups.
-
Emerging evidence suggests that severe vasomotor symptoms are associated with long-term adverse health outcomes, including cardiovascular disease and type 2 diabetes mellitus.
-
It is not currently known whether treating severe vasomotor symptoms modifies the associations with long-term disease. Current evidence does not support use of menopause hormone therapy (MHT, also termed hormone replacement therapy) for the primary or secondary prevention of chronic disease.
-
MHT appears to be the most effective therapy for severe vasomotor symptoms. Newer agents such as neurokinin receptor antagonists are also effective in reducing the frequency and severity of hot flushes and might also improve sleep and menopause-related quality of life.
-
Future consistent implementation of validated measurement tools will better facilitate the development and evaluation of medical therapies for vasomotor symptoms through direct comparisons of treatment efficacy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Harlow, S. D. et al. Executive summary of the Stages of Reproductive Aging Workshop + 10: addressing the unfinished agenda of staging reproductive aging. J. Clin. Endocrinol. Metab. 97, 1159–1168 (2012).
Waetjen, L. E. et al. Factors associated with developing vaginal dryness symptoms in women transitioning through menopause: a longitudinal study. Menopause 25, 1094–1104 (2018).
Tepper, P. G. et al. Characterizing the trajectories of vasomotor symptoms across the menopausal transition. Menopause 23, 1067–1074 (2016).
Rance, N. E., Dacks, P. A., Mittelman-Smith, M. A., Romanovsky, A. A. & Krajewski-Hall, S. J. Modulation of body temperature and LH secretion by hypothalamic KNDy (kisspeptin, neurokinin B and dynorphin) neurons: a novel hypothesis on the mechanism of hot flushes. Front. Neuroendocrinol. 34, 211–227 (2013).
Hickey, M. et al. An empowerment model for managing menopause. Lancet 403, 947–957 (2024).
Brown, L. et al. Promoting good mental health over the menopause transition. Lancet 403, 969–983 (2024).
Mishra, G. D. et al. Optimising health after early menopause. Lancet 403, 958–968 (2024).
Zhu, D. et al. Type of menopause, age of menopause and variations in the risk of incident cardiovascular disease: pooled analysis of individual data from 10 international studies. Hum. Reprod. 35, 1933–1943 (2020).
Peate, M., Saunders, C., Cohen, P. & Hickey, M. Who is managing menopausal symptoms, sexual problems, mood and sleep disturbance after breast cancer and is it working? Findings from a large community-based survey of breast cancer survivors. Breast Cancer Res. Treat. 187, 427–435 (2021).
Zhao, D. et al. Treatment-induced menopause symptoms among women with breast cancer undergoing chemotherapy in China: a comparison to age- and menopause status-matched controls. Menopause 31, 145–153 (2024).
Hickey, M. et al. What happens after menopause? (WHAM): a prospective controlled study of vasomotor symptoms and menopause-related quality of life 12 months after premenopausal risk-reducing salpingo-oophorectomy. Gynecol. Oncol. 163, 148–154 (2021).
Toze, M. & Westwood, S. Experiences of menopause among non-binary and trans people. Int. J. Transgend. Health 26, 447–458 (2025).
Xin, M. Q. L. & Lane, R. Exploring the clinical, psychological, and social relevance of menopause for trans and gender diverse people: a qualitative study. Menopause 32, 288–294 (2025).
Thurston, R. C. & Joffe, H. Vasomotor symptoms and menopause: findings from the Study of Women’s Health across the Nation. Obst. Gynecol. Clin. North Am. 38, 489–501 (2011).
Carpenter, J. S. et al. MsFLASH participants’ priorities for alleviating menopausal symptoms. Climacteric 18, 859–866 (2015).
Lan, Q., Hickey, M., Peate, M. & Marino, J. L. Priorities for alleviating menopausal symptoms after cancer. Menopause 30, 136–142 (2023).
Lensen, S. et al. A core outcome set for vasomotor symptoms associated with menopause: the COMMA (Core Outcomes in Menopause) global initiative. Menopause 28, 852–858 (2021).
Nash, Z., Christmas, M., Gronlund, T. & Hickey, M. Top Ten menopause research priorities. Lancet 404, 2535–2536 (2025).
Santoro, N., Roeca, C., Peters, B. A. & Neal-Perry, G. The menopause transition: signs, symptoms, and management options. J. Clin. Endocrinol. Metab. 106, 1–15 (2021).
Mishra, G. D. & Kuh, D. Health symptoms during midlife in relation to menopausal transition: British prospective cohort study. BMJ 344, e402 (2012).
Avis, N. E. et al. Duration of menopausal vasomotor symptoms over the menopause transition. JAMA Intern. Med. 175, 531–539 (2015).
Anderson, D. J. et al. Obesity, smoking, and risk of vasomotor menopausal symptoms: a pooled analysis of eight cohort studies. Am. J. Obst. Gynecol. 222, 478.e1–478.e17 (2020).
US Department of Health and Human Services, Food and Drug Administration & Center for Drug Evaluation and Research. Guidance for industry: estrogen and estrogen/progestin drug products to treat vasomotor symptoms and vulvar and vaginal atrophy symptoms — recommendations for clinical evaluation. FDA https://www.fda.gov/media/71359/download (2003).
Williams, R. E. et al. Frequency and severity of vasomotor symptoms among peri- and postmenopausal women in the United States. Climacteric 11, 32–43 (2008).
Lensen, S. et al. Recommended measurement instruments for menopausal vasomotor symptoms: the COMMA (Core Outcomes in Menopause) consortium. Menopause 31, 582–590 (2024).
Rutherford, C. et al. Content validity, face validity and acceptability of three hot flash diaries for vasomotor symptoms: a cognitive interview study. Menopause https://doi.org/10.1097/GME.0000000000002696 (2025).
Iliodromiti, S. et al. Variation in menopausal vasomotor symptoms outcomes in clinical trials: a systematic review. BJOG 127, 320–333 (2019).
Gold, E. B. et al. Longitudinal analysis of the association between vasomotor symptoms and race/ethnicity across the menopausal transition: Study of Women’s Health Across the Nation. Am. J. Public Health 96, 1226–1235 (2006).
Nappi, R. E. et al. Global cross-sectional survey of women with vasomotor symptoms associated with menopause: prevalence and quality of life burden. Menopause 28, 875–882 (2021).
Kochersberger, A. et al. The association of race, ethnicity, and socioeconomic status on the severity of menopause symptoms: a study of 68,864 women. Menopause 31, 476–483 (2024).
Kim, C. et al. Prospective early adulthood risk factors for vasomotor symptoms in the Coronary Artery Risk Development in Young Adults study. Menopause 31, 108–115 (2024).
Avis, N. E., Brambilla, D., McKinlay, S. M. & Vass, K. A longitudinal analysis of the association between menopause and depression. Results from the Massachusetts Women’s Health Study. Ann. Epidemiol. 4, 214–220 (1994).
Hickey, M., Schoenaker, D. A., Joffe, H. & Mishra, G. D. Depressive symptoms across the menopause transition: findings from a large population-based cohort study. Menopause 23, 1287–1293 (2016).
Paramsothy, P. et al. Duration of the menopausal transition is longer in women with young age at onset: the multiethnic Study of Women’s Health Across the Nation. Menopause 24, 142–149 (2017).
Herber-Gast, G. C. & Mishra, G. D. Fruit, Mediterranean-style, and high-fat and -sugar diets are associated with the risk of night sweats and hot flushes in midlife: results from a prospective cohort study. Am. J. Clin. Nutr. 97, 1092–1099 (2013).
Di Donato, P. et al. Factors associated with climacteric symptoms in women around menopause attending menopause clinics in Italy. Maturitas 52, 181–189 (2005).
Mishra, G. D. & Dobson, A. J. Using longitudinal profiles to characterize women’s symptoms through midlife: results from a large prospective study. Menopause 19, 549–555 (2012).
Ruth, K. S. et al. Genetic insights into biological mechanisms governing human ovarian ageing. Nature 596, 393–397 (2021). This article provides vital insights into the biological mechanisms underlying ovarian ageing and how targeted therapeutics could be explored for future disease prevention.
Ruth, K. S. et al. Insights into the genetics of menopausal vasomotor symptoms: genome-wide analyses of routinely-collected primary care health records. BMC Med. Genom. 16, 231 (2023).
Avis, N. E. et al. Change in health-related quality of life over the menopausal transition in a multiethnic cohort of middle-aged women: Study of Women’s Health Across the Nation. Menopause 16, 860–869 (2009).
Kumari, M., Stafford, M. & Marmot, M. The menopausal transition was associated in a prospective study with decreased health functioning in women who report menopausal symptoms. J. Clin. Epidemiol. 58, 719–727 (2005).
Blumel, J. E. et al. A multinational study of sleep disorders during female mid-life. Maturitas 72, 359–366 (2012).
Jack, G. et al. Menopause in the workplace: what employers should be doing. Maturitas 85, 88–95 (2016).
Rodrigo, C. H., Sebire, E., Bhattacharya, S., Paranjothy, S. & Black, M. Effectiveness of workplace-based interventions to promote wellbeing among menopausal women: a systematic review. Post. Reprod. Health 29, 99–108 (2023).
Faubion, S. S. et al. Impact of menopause symptoms on women in the workplace. Mayo Clinic Proc. 98, 833–845 (2023).
Gartoulla, P., Worsley, R., Bell, R. J. & Davis, S. R. Moderate to severe vasomotor and sexual symptoms remain problematic for women aged 60 to 65 years. Menopause 22, 694–701 (2015).
Chung, H. F. et al. The role of sleep difficulties in the vasomotor menopausal symptoms and depressed mood relationships: an international pooled analysis of eight studies in the InterLACE consortium. Psychol. Med. 48, 2550–2561 (2018).
Worsley, R., Bell, R. J., Gartoulla, P., Robinson, P. J. & Davis, S. R. Moderate-severe vasomotor symptoms are associated with moderate-severe depressive symptoms. J. Womens Health 26, 712–718 (2017).
Maki, P. M. & Jaff, N. G. Brain fog in menopause: a health-care professional’s guide for decision-making and counseling on cognition. Climacteric 25, 570–578 (2022).
Maki, P. M. & Weber, M. T. A research primer for studies of cognitive changes across the menopause transition. Climacteric 24, 382–388 (2021).
Islam, R. M. et al. Prevalence and severity of symptoms across the menopause transition: cross-sectional findings from the Australian Women’s Midlife Years (AMY) Study. Lancet Diabetes Endocrinol. 13, 765–776 (2025).
Nash, Z. et al. Protocol for a global menopause priority setting partnership. BMJ Open 15, e096401 (2025). This article acknowledges the importance of those with lived experience in determining the future priorities in menopause research. There is much potential benefit in developing a global research agenda that is relevant and valuable.
Hanton, T. et al. Women’s health research priorities in the UK: a consensus statement from RCOG. Lancet Obst. Gynaecol. Womens Health 1, E249–E250 (2025).
Greendale, G. A., Karlamangla, A. S. & Maki, P. M. The menopause transition and cognition. JAMA 323, 1495–1496 (2020).
Furey, R. T., Thomas, E. H. X., Kulkarni, J. & Gurvich, C. Subjective versus objective cognition during menopause: a systematic review and meta-analysis. J. Int. Neuropsychol. Soc. 31, 459–477 (2025). This systematic review highlights the difficulties of understanding the cognitive symptoms increasingly reported by perimenopausal and postmenopausal women. It demonstrates the challenges in classifying these cognitive changes, showing the need for high-quality research in this important area.
Zhu, C. et al. Factors associated with subjective cognitive symptoms during the menopause transition. Menopause 33, 340–348 (2025).
Maki, P. M. et al. Hot flashes are associated with altered brain function during a memory task. Menopause 27, 269–277 (2020).
Thurston, R. C. et al. Menopausal vasomotor symptoms and white matter hyperintensities in midlife women. Neurology 100, e133–e141 (2023). This article demonstrates an important understanding of the effects of vasomotor symptoms on white matter in the brain. It provides vital foundational evidence for future research to explore whether vasomotor symptoms can serve as a female-specific midlife marker of brain health.
Williams, M. & Maki, P. M. A review of cognitive, sleep, and mood changes in the menopausal transition: beyond vasomotor symptoms. Obst. Gynecol. 146, 350–359 (2025).
Drogos, L. L. et al. Objective cognitive performance is related to subjective memory complaints in midlife women with moderate to severe vasomotor symptoms. Menopause 20, 1236–1242 (2013).
Peterson, A., Gottesman, R. T., Miller, E. C. & Tom, S. E. The association of vasomotor symptoms during the menopausal transition and cognition in later life. Menopause 29, 832–839 (2022).
Hayashi, K. et al. Complaints of reduced cognitive functioning during perimenopause: a cross-sectional analysis of the Japan Nurses’ Health Study. Womens Midlife Health 8, 6 (2022).
Greendale, G. A. et al. Menopause-associated symptoms and cognitive performance: results from the Study of Women’s Health Across the Nation. Am. J. Epidemiol. 171, 1214–1224 (2010).
Andy, C. et al. Systematic review and meta-analysis of the effects of menopause hormone therapy on cognition. Front. Endocrinol. 15, 1350318 (2024).
Gleason, C. E. et al. Long-term cognitive effects of menopausal hormone therapy: findings from the KEEPS continuation study. PLoS Med. 21, e1004435 (2024). This study demonstrates the lack of empirical support for preventative use of menopause hormone therapy (MHT) for cognitive function, an increasingly common challenge for perimenopausal and postmenopausal women and prescribers of MHT.
Thurston, R. C. Vasomotor symptoms and cardiovascular health: findings from the SWAN and the MsHeart/MsBrain studies. Climacteric 27, 75–80 (2024).
Carson, M. Y. & Thurston, R. C. Vasomotor symptoms and their links to cardiovascular disease risk. Curr. Opin. Endocr. Metab. Res. 30, 100448 (2023).
Thurston, R. C., Sutton-Tyrrell, K., Everson-Rose, S. A., Hess, R. & Matthews, K. A. Hot flashes and subclinical cardiovascular disease: findings from the Study of Women’s Health Across the Nation Heart Study. Circulation 118, 1234–1240 (2008).
Lambrinoudaki, I. et al. Menopausal symptoms are associated with subclinical atherosclerosis in healthy recently postmenopausal women. Climacteric 15, 350–357 (2012).
Thurston, R. C. et al. Hot flashes and carotid intima media thickness among midlife women. Menopause 18, 352–358 (2011).
Thurston, R. C. et al. Trajectories of vasomotor symptoms and carotid intima media thickness in the Study of Women’s Health Across the Nation. Stroke 47, 12–17 (2016).
Ryu, K. J. et al. Vasomotor symptoms and carotid artery intima-media thickness among Korean midlife women. Maturitas 159, 1–6 (2022).
Thurston, R. C. et al. Menopausal vasomotor symptoms and risk of incident cardiovascular disease events in SWAN. J. Am. Heart Assoc. 10, e017416 (2021).
Herber-Gast, G., Brown, W. J. & Mishra, G. D. Hot flushes and night sweats are associated with coronary heart disease risk in midlife: a longitudinal study. BJOG 122, 1560–1567 (2015).
Dam, V., Dobson, A. J., Onland-Moret, N. C., van der Schouw, Y. T. & Mishra, G. D. Vasomotor menopausal symptoms and cardiovascular disease risk in midlife: a longitudinal study. Maturitas 133, 32–41 (2020).
Jackson, E. A. et al. Hot flash frequency and blood pressure: data from the Study of Women’s Health Across the Nation. J. Womens Health 25, 1204–1209 (2016).
Thurston, R. C. et al. Vasomotor symptoms and accelerated epigenetic aging in the Women’s Health Initiative (WHI). J. Clin. Endocrinol. Metab. 105, 1221–1227 (2020).
Carpenter, J. S. et al. Palpitations across the menopause transition in SWAN: trajectories, characteristics, and associations with subclinical cardiovascular disease. Menopause 30, 18–27 (2023).
Zhu, D. et al. Vasomotor menopausal symptoms and risk of cardiovascular disease: a pooled analysis of six prospective studies. Am. J. Obst. Gynecol. 223, 898.e1–898.e16 (2020). This study pooled results from more than 23,300 women and demonstrated that women with severe vasomotor symptoms had more than double the risk of cardiovascular disease than those without vasomotor symptoms. It shows that severity of vasomotor symptoms is an important potential marker for those with underlying cardiovascular vulnerability.
Armeni, A. et al. Vasomotor symptoms and risk of cardiovascular disease in peri- and postmenopausal women: a systematic review and meta-analysis. Maturitas 171, 13–20 (2023).
Gray, K. E. et al. Vasomotor symptom characteristics: are they risk factors for incident diabetes? Menopause 25, 520–530 (2018).
Herber-Gast, G. C. & Mishra, G. D. Early severe vasomotor menopausal symptoms are associated with diabetes. Menopause 21, 855–860 (2014).
Thurston, R. C. et al. Vasomotor symptoms and insulin resistance in the Study of Women’s Health Across the Nation. J. Clin. Endocrinol. Metab. 97, 3487–3494 (2012).
Tural, A., Yoldemir, T. & Erenus, M. Assessment of bone mineral density should be considered earlier in perimenopausal women with vasomotor symptoms. Int. J. Gynaecol. Obst. 107, 114–116 (2009).
Anagnostis, P. et al. The association of vasomotor symptoms with fracture risk and bone mineral density in postmenopausal women: a systematic review and meta-analysis of observational studies. Osteoporos. Int. 35, 1329–1336 (2024). This timely systematic review demonstrates that although vasomotor symptoms are associated with lower bone mineral density, they do not appear to confer an increased fracture risk. Severity of symptoms was also not associated with fracture risk. These findings demonstrate the lack of empirical support for preventative use of MHT in postmenopausal women of average age where not otherwise indicated for symptoms.
Gast, G. C. et al. Vasomotor symptoms are associated with a lower bone mineral density. Menopause 16, 231–238 (2009).
Crandall, C. J. et al. Associations of menopausal vasomotor symptoms with fracture incidence. J. Clin. Endocrinol. Metab. 100, 524–534 (2015).
Kapoor, E. et al. Association of adverse childhood experiences with menopausal symptoms: results from the Data Registry on Experiences of Aging, Menopause and Sexuality (DREAMS). Maturitas 143, 209–215 (2021).
Thurston, R. C. et al. Childhood abuse or neglect is associated with increased vasomotor symptom reporting among midlife women. Menopause 15, 16–22 (2008).
Carson, M. Y. & Thurston, R. C. Childhood abuse and vasomotor symptoms among midlife women. Menopause 26, 1093–1099 (2019).
Chung, H. F. et al. Age at menarche and risk of vasomotor menopausal symptoms: a pooled analysis of six studies. BJOG 128, 603–613 (2021).
Yang, Y. et al. Premenstrual disorders, timing of menopause, and severity of vasomotor symptoms. JAMA Netw. Open 6, e2334545 (2023).
Faubion, S. S. et al. Hypertensive disorders of pregnancy and menopausal symptoms: a cross-sectional study from the data registry on experiences of aging, menopause, and sexuality. Menopause 28, 25–31 (2020).
Kingsberg, S. A., Larkin, L. C. & Liu, J. H. Clinical effects of early or surgical menopause. Obst. Gynecol. 135, 853–868 (2020).
Hickey, M. et al. What happens after menopause? (WHAM): a prospective controlled study of vasomotor symptoms and menopause-related quality of life 24 months after premenopausal risk-reducing salpingo-oophorectomy (RRSO). Gynecol. Oncol. 191, 201–211 (2024).
Hickey, M. et al. Managing menopause after cancer. Lancet 403, 984–996 (2024).
Marino, J. L. et al. How does adjuvant chemotherapy affect menopausal symptoms, sexual function, and quality of life after breast cancer? Menopause 23, 1000–1008 (2016).
Gold, E. B. et al. Vasomotor symptoms in midlife women with incident breast cancer: pink SWAN. Breast Cancer Res. Treat. 191, 125–135 (2021).
Bauld, R. & Brown, R. F. Stress, psychological distress, psychosocial factors, menopause symptoms and physical health in women. Maturitas 62, 160–165 (2009).
Sood, R. et al. Association of mindfulness and stress with menopausal symptoms in midlife women. Climacteric 22, 377–382 (2019).
Gibson, C. J. et al. Associations of intimate partner violence, sexual assault, and posttraumatic stress disorder with menopause symptoms among midlife and older women. JAMA Intern. Med. 179, 80–87 (2019).
Cao, V., Clark, A. & Aggarwal, B. Obesity and severity of menopausal symptoms: a contemporary review. Curr. Diabetes Rep. 23, 361–370 (2023).
Shobeiri, F., Jenabi, E., Poorolajal, J. & Hazavehei, S. M. The association between body mass index and hot flash in midlife women: a meta-analysis. J. Menopausal Med. 22, 14–19 (2016).
Gold, E. B. et al. Longitudinal analysis of changes in weight and waist circumference in relation to incident vasomotor symptoms: the Study of Women’s Health Across the Nation (SWAN). Menopause 24, 9–26 (2017).
Kroenke, C. H. et al. Effects of a dietary intervention and weight change on vasomotor symptoms in the Women’s Health Initiative. Menopause 19, 980–988 (2012).
Jenabi, E. & Poorolajal, J. The association between hot flushes and smoking in midlife women: a meta-analysis. Climacteric 18, 797–801 (2015).
Witkowski, S., Evard, R., Rickson, J. J., White, Q. & Sievert, L. L. Physical activity and exercise for hot flashes: trigger or treatment? Menopause 30, 218–224 (2023).
Liu, T., Chen, S., Mielke, G. I., McCarthy, A. L. & Bailey, T. G. Effects of exercise on vasomotor symptoms in menopausal women: a systematic review and meta-analysis. Climacteric 25, 552–561 (2022).
Dunneram, Y. et al. Soy intake and vasomotor menopausal symptoms among midlife women: a pooled analysis of five studies from the InterLACE consortium. Eur. J. Clin. Nutr. 73, 1501–1511 (2019).
Beezhold, B., Radnitz, C., McGrath, R. E. & Feldman, A. Vegans report less bothersome vasomotor and physical menopausal symptoms than omnivores. Maturitas 112, 12–17 (2018).
Hunter, M. S., O’Dea, I. & Britten, N. Decision-making and hormone replacement therapy: a qualitative analysis. Soc. Sci. Med. 45, 1541–1548 (1997).
Sarri, G., Pedder, H., Dias, S., Guo, Y. & Lumsden, M. A. Vasomotor symptoms resulting from natural menopause: a systematic review and network meta-analysis of treatment effects from the National Institute for Health and Care Excellence guideline on menopause. BJOG 124, 1514–1523 (2017).
Hillman, S., Shantikumar, S., Ridha, A., Todkill, D. & Dale, J. Socioeconomic status and HRT prescribing: a study of practice-level data in England. Br. J. Gen. Pract. 70, e772–e777 (2020).
Kaunitz, A. M. Oral contraceptive use in perimenopause. Am. J. Obst. Gynecol. 185, S32–S37 (2001).
Shargil, A. A. Hormone replacement therapy in perimenopausal women with a triphasic contraceptive compound: a three-year prospective study. Int. J. Fertil. 30, 18–28 (1985).
Casper, R. F., Dodin, S. & Reid, R. L. The effect of 20 µg ethinyl estradiol/1 mg norethindrone acetate (Minestrin™), a low-dose oral contraceptive, on vaginal bleeding patterns, hot flashes, and quality of life in symptomatic perimenopausal women. Menopause 4, 139–147 (1997).
Corbelli, J., Shaikh, N., Wessel, C. & Hess, R. Low-dose transdermal estradiol for vasomotor symptoms: a systematic review. Menopause 22, 114–121 (2015).
Gaudard, A. M. et al. Bioidentical hormones for women with vasomotor symptoms. Cochrane Database Syst. Rev. 2016, CD010407 (2016).
Constantine, G. D. et al. TX-001HR is associated with a clinically meaningful effect on severity of moderate to severe vasomotor symptoms in the REPLENISH trial. Menopause 27, 1236–1241 (2020).
Furness, S., Roberts, H., Marjoribanks, J. & Lethaby, A. Hormone therapy in postmenopausal women and risk of endometrial hyperplasia. Cochrane Database Syst. Rev. 8, CD000402 (2012).
Lemaitre, R. N. et al. Hormone replacement therapy and associated risk of stroke in postmenopausal women. Arch. Intern. Med. 162, 1954–1960 (2002).
Canonico, M., Plu-Bureau, G., Lowe, G. D. & Scarabin, P. Y. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ 336, 1227–1231 (2008).
Rovinski, D., Ramos, R. B., Fighera, T. M., Casanova, G. K. & Spritzer, P. M. Risk of venous thromboembolism events in postmenopausal women using oral versus non-oral hormone therapy: a systematic review and meta-analysis. Thromb. Res. 168, 83–95 (2018).
LaCroix, A. & Ensrud, K. The flash dance of midlife: the Skylight 1 trial. Nat. Med. 29, 1324–1325 (2023).
Maki, P. M. et al. Guidelines for the evaluation and treatment of perimenopausal depression: summary and recommendations. J. Womens Health 28, 117–134 (2019).
Mangione, C. M. et al. Hormone therapy for the primary prevention of chronic conditions in postmenopausal persons: US Preventive Services Task Force Recommendation Statement. JAMA 328, 1740–1746 (2022).
Ockene, J. K. et al. Symptom experience after discontinuing use of estrogen plus progestin. JAMA 294, 183–193 (2005).
Brunner, R. L. et al. Menopausal symptom experience before and after stopping estrogen therapy in the Women’s Health Initiative randomized, placebo-controlled trial. Menopause 17, 946–954 (2010).
Melville, M. et al. Menopause hormone therapy and risk of mild cognitive impairment or dementia: a systematic review and meta-analysis. Lancet Healthy Longev. 6, 100803 (2025).
Gupta, P. et al. Menopausal symptoms in women treated for breast cancer: the prevalence and severity of symptoms and their perceived effects on quality of life. Climacteric 9, 49–58 (2006).
Marino, J. L. et al. Nature and severity of menopausal symptoms and their impact on quality of life and sexual function in cancer survivors compared with women without a cancer history. Menopause 21, 267–274 (2014).
Bernhard, J. et al. Patient-reported outcomes with adjuvant exemestane versus tamoxifen in premenopausal women with early breast cancer undergoing ovarian suppression (TEXT and SOFT): a combined analysis of two phase 3 randomised trials. Lancet Oncol. 16, 848–858 (2015).
The 2023 nonhormone therapy position statement of The North American Menopause Society. Menopause 30, 573–590 (2023).
Johnson, K. et al. A phase 3, randomized, placebo-controlled, 12-week, double-blind study, plus a non-controlled extension treatment period, to assess efficacy and safety of fezolinetant, a neurokin-3 receptor antagonist, in women with moderate-to-severe vasomotor symptoms associated with menopause. Menopause 28, 1450 (2021).
Ruan, X. et al. Efficacy and safety of fezolinetant for moderate to severe vasomotor symptoms associated with menopause among women in East Asia: a phase 3 randomized study (MOONLIGHT I). J. Int. Med. Res. 52, 3000605241247684 (2024).
Pinkerton, J. V. et al. Elinzanetant for the treatment of vasomotor symptoms associated with menopause: OASIS 1 and 2 randomized clinical trials. JAMA 332, 1343–1354 (2024).
Panay, N. et al. Elinzanetant for the treatment of vasomotor symptoms associated with menopause: a phase 3 randomized clinical trial. JAMA Intern. Med. 185, 1319–1327 (2025).
US Food and Drug Administration. FDA adds warning about rare occurrence of serious liver injury with use of Veozah (fezolinetant) for hot flashes due to menopause. FDAhttps://www.fda.gov/drugs/drug-safety-and-availability/fda-adds-warning-about-rare-occurrence-serious-liver-injury-use-veozah-fezolinetant-hot-flashes-due (2024).
Hunter, M. S. Cognitive behavioral therapy for menopausal symptoms. Climacteric 24, 51–56 (2021).
Elkins, G. R., Fisher, W. I., Johnson, A. K., Carpenter, J. S. & Keith, T. Z. Clinical hypnosis in the treatment of postmenopausal hot flashes: a randomized controlled trial. Menopause 20, 291–298 (2013).
Elkins, G. et al. Self-administered hypnosis vs sham hypnosis for hot flashes: a randomized clinical trial. JAMA Netw. Open 8, e2542537 (2025).
Christmas, M. M. et al. Variation in outcome reporting and measurement tools in clinical trials of treatments for genitourinary symptoms in peri- and postmenopausal women: a systematic review. Menopause 27, 1070–1080 (2020).
Lensen, S. et al. A core outcome set for genitourinary symptoms associated with menopause: the COMMA (Core Outcomes in Menopause) global initiative. Menopause 28, 859–866 (2021).
Paramanandam, V. S. et al. Recommended measurement instruments for genitourinary symptoms associated with menopause: the COMMA (Core Outcomes in Menopause) consortium. Menopause 31, 591–599 (2024).
Burgin, J., Pyne, Y. & Hickey, M. Helping women prepare for menopause. BMJ 386, q1512 (2024).
Williams, R. E. et al. Healthcare seeking and treatment for menopausal symptoms in the United States. Maturitas 58, 348–358 (2007).
Santoro, N. Perimenopause: from research to practice. J. Womens Health 25, 332–339 (2016).
Davis, S. R. et al. The 2023 practitioner’s toolkit for managing menopause. Climacteric 26, 517–536 (2023).
Manson, J. E. et al. The Women’s Health Initiative randomized trials and clinical practice: a review. JAMA 331, 1748–1760 (2024).
Peate, M., Johnson, T. L., Avis, N. E. & Hickey, M. Addressing sociodemographic, socioeconomic, and gendered disparities for equity in menopause care. Cell Rep. Med. 5, 101616 (2024).
Carpenter, J. S. et al. Validity, cut-points, and minimally important differences for two hot flash-related daily interference scales. Menopause 24, 877–885 (2017).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
M.H. was topic expert for the 2024 NICE UK menopause guidelines and is an unpaid board member of BreastScreen Victoria, and editor for the Cochrane Collaboration. All other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Joann V. Pinkerton and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
We searched PubMed for all articles published up to 19 February 2026. The search terms included the following keywords in the title and/or abstract: (“vasomotor symptoms” or “VMS” or “hot flashes/flushes” or “night/cold sweats”) AND (“severity/severe” or “frequency/frequent” or “intensity/intense” or “duration” or “bothersome”) AND (“cardiovascular disease” or “diabetes” or “metabolic syndrome” or “osteoporosis” or “quality of life” or “chronic health” or “non-communicable disease” or “breast cancer” or “cancer” or “genitourinary symptoms” or “risk factors” or “demographic” or “socioeconomic” or “race” or “ethnicity” or “stress” or “physiological” or “lifestyle” or “reproductive” or “obesity” or “body mass index” or “weight gain” or “smoking” or “diet” or “exercise” or “physical activity” or “sedentary behaviour” or “type of menopause” or “age at menopause” or “surgical menopause” or “oophorectomy” or “hysterectomy”). This returned 2,011 results. Titles and abstracts were screened, and 298 articles were included for full-text review. We summarized evidence from systematic reviews, meta-analyses and large population-based studies (n > 500) where possible.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hickey, M., Huguenin, A., Chung, HF. et al. Risk factors, management and consequences of severe menopausal vasomotor symptoms. Nat Rev Endocrinol (2026). https://doi.org/10.1038/s41574-026-01245-w
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41574-026-01245-w