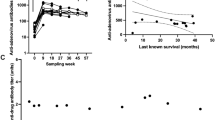
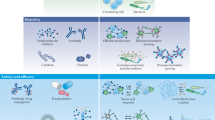
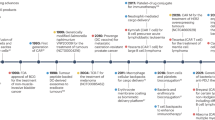
Abstract
Engineered cell therapies are transforming precision medicine by enabling real-time, context-responsive interventions that act upon disease-specific cues. Inspired by the success of CAR-T cells in oncology, next-generation platforms are being developed using diverse immune cells and stem cells to address a broader spectrum of diseases. These living therapeutics harness synthetic gene circuits to induce targeted cytotoxicity, to modulate the secretion of effector proteins or to coordinate both functions in response to endogenous signals or externally delivered molecular and physical triggers. Ex vivo engineering of autologous cells remains the norm, but challenges in scalability, cost and accessibility are fuelling efforts towards allogeneic products and in vivo reprogramming. Advances in targeted delivery — using viral vectors, mRNA-loaded nanoparticles and virus-like particles — are expanding the toolkit for direct programming of cells within the body. This Review discusses emerging strategies for engineering human cells with therapeutic functions, highlighting modular control systems, delivery innovations and the translational hurdles that lie ahead.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
Majzner, R. G. & Mackall, C. L. Clinical lessons learned from the first leg of the CAR T cell journey. Nat. Med. 25, 1341–1355 (2019).
Jorgensen, L. V., Christensen, E. B., Barnkob, M. B. & Barington, T. The clinical landscape of CAR NK cells. Exp. Hematol. Oncol. 14, 46 (2025).
Sloas, C., Gill, S. & Klichinsky, M. Engineered CAR-macrophages as adoptive immunotherapies for solid tumors. Front. Immunol. 12, 783305 (2021).
Mackensen, A. et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat. Med. 28, 2124–2132 (2022).
Oh, S. et al. Precision targeting of autoantigen-specific B cells in muscle-specific tyrosine kinase myasthenia gravis with chimeric autoantibody receptor T cells. Nat. Biotechnol. 41, 1229–1238 (2023).
Rurik, J. G. et al. CAR T cells produced in vivo to treat cardiac injury. Science 375, 91–96 (2022).
Galvan, S., Xie, Y. Q., Teixeira, A. P. & Fussenegger, M. Synthetic closed-loop gene circuit for phenylalanine regulation. Nucleic Acids Res. 53, gkaf1151 (2025).
Amor, C. et al. Prophylactic and long-lasting efficacy of senolytic CAR T cells against age-related metabolic dysfunction. Nat. Aging 4, 336–349 (2024).
Arias, J., Yu, J., Varshney, M., Inzunza, J. & Nalvarte, I. Hematopoietic stem cell- and induced pluripotent stem cell-derived CAR-NK cells as reliable cell-based therapy solutions. Stem Cell Transl. Med. 10, 987–995 (2021).
Iriguchi, S. et al. A clinically applicable and scalable method to regenerate T-cells from iPSCs for off-the-shelf T-cell immunotherapy. Nat. Commun. 12, 430 (2021).
Ueda, T. et al. Optimization of the proliferation and persistency of CAR T cells derived from human induced pluripotent stem cells. Nat. Biomed. Eng. 7, 24–37 (2023).
Kosti, P. et al. Hypoxia-sensing CAR T cells provide safety and efficacy in treating solid tumors. Cell Rep. Med. 2, 100227 (2021).
Li, H. S. et al. Multidimensional control of therapeutic human cell function with synthetic gene circuits. Science 378, 1227–1234 (2022).
Nicolai, C. J. et al. In vivo CAR T-cell generation in nonhuman primates using lentiviral vectors displaying a multidomain fusion ligand. Blood 144, 977–987 (2024).
Tabebordbar, M. et al. Directed evolution of a family of AAV capsid variants enabling potent muscle-directed gene delivery across species. Cell 184, 4919–4938.e4922 (2021).
Parayath, N. N., Stephan, S. B., Koehne, A. L., Nelson, P. S. & Stephan, M. T. In vitro-transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo. Nat. Commun. 11, 6080 (2020).
An, M. et al. Engineered virus-like particles for transient delivery of prime editor ribonucleoprotein complexes in vivo. Nat. Biotechnol. 42, 1526–1537 (2024).
Banskota, S. et al. Engineered virus-like particles for efficient in vivo delivery of therapeutic proteins. Cell 185, 250–265.e216 (2022).
Bulcha, J. T., Wang, Y., Ma, H., Tai, P. W. L. & Gao, G. Viral vector platforms within the gene therapy landscape. Signal. Transduct. Target. Ther. 6, 53 (2021).
Dunbar, C. E. et al. Gene therapy comes of age. Science 359, eaan4672 (2018).
Halle, S., Halle, O. & Forster, R. Mechanisms and dynamics of T cell-mediated cytotoxicity in vivo. Trends Immunol. 38, 432–443 (2017).
Benmebarek, M. R. et al. Killing mechanisms of chimeric antigen receptor (CAR) T cells. Int. J. Mol. Sci. 20, 1283 (2019).
Goff, S. L. et al. Pilot trial of adoptive transfer of chimeric antigen receptor-transduced T cells targeting EGFRvIII in patients with glioblastoma. J. Immunother. 42, 126–135 (2019).
O’Rourke, D. M. et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aaa0984 (2017).
Raje, N. et al. Anti-BCMA CAR T-cell therapy bb2121 in relapsed or refractory multiple myeloma. N. Engl. J. Med. 380, 1726–1737 (2019).
Du, B. et al. CAR-T therapy in solid tumors. Cancer Cell 43, 665–679 (2025).
Qi, C. et al. Claudin-18 isoform 2-specific CAR T-cell therapy (satri-cel) versus treatment of physician’s choice for previously treated advanced gastric or gastro-oesophageal junction cancer (CT041-ST-01): a randomised, open-label, phase 2 trial. Lancet 405, 2049–2060 (2025).
Uslu, U. & June, C. H. Beyond the blood: expanding CAR T cell therapy to solid tumors. Nat. Biotechnol. 43, 506–515 (2025).
Doan, A. E. et al. FOXO1 is a master regulator of memory programming in CAR T cells. Nature 629, 211–218 (2024).
Seo, H. et al. BATF and IRF4 cooperate to counter exhaustion in tumor-infiltrating CAR T cells. Nat. Immunol. 22, 983–995 (2021).
Feng, B. et al. The type 2 cytokine Fc-IL-4 revitalizes exhausted CD8(+) T cells against cancer. Nature 634, 712–720 (2024).
Steffin, D. et al. Interleukin-15-armoured GPC3 CAR T cells for patients with solid cancers. Nature 637, 940–946 (2025).
Zhao, Y. et al. IL-10-expressing CAR T cells resist dysfunction and mediate durable clearance of solid tumors and metastases. Nat. Biotechnol. 42, 1693–1704 (2024).
Svoboda, J. et al. Enhanced CAR T-cell therapy for lymphoma after previous failure. N. Engl. J. Med. 392, 1824–1835 (2025).
Chen, A. X. Y. et al. Rewiring endogenous genes in CAR T cells for tumour-restricted payload delivery. Nature 644, 241–251 (2025).
Muller, F. et al. CD19-targeted CAR T cells in refractory antisynthetase syndrome. Lancet 401, 815–818 (2023).
Muller, F. et al. CD19 CAR T-cell therapy in autoimmune disease — a case series with follow-up. N. Engl. J. Med. 390, 687–700 (2024).
Mao, Y. et al. Efficacy and safety of novel multifunctional M10 CAR-T cells in HIV-1-infected patients: a phase I, multicenter, single-arm, open-label study. Cell Discov. 10, 49 (2024).
Yashaswini, C. N. et al. In vivo anti-FAP CAR T therapy reduces fibrosis and restores liver homeostasis in metabolic dysfunction-associated steatohepatitis. Preprint at bioRxiv https://doi.org/10.1101/2025.02.25.640143 (2025).
Amor, C. et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583, 127–132 (2020).
Yang, D. et al. NKG2D-CAR T cells eliminate senescent cells in aged mice and nonhuman primates. Sci. Transl. Med. 15, eadd1951 (2023).
Liu, E. et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N. Engl. J. Med. 382, 545–553 (2020).
Marin, D. et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19(+) B cell tumors: a phase 1/2 trial. Nat. Med. 30, 772–784 (2024).
Van den Eynde, A. et al. IL-15-secreting CAR natural killer cells directed toward the pan-cancer target CD70 eliminate both cancer cells and cancer-associated fibroblasts. J. Hematol. Oncol. 17, 8 (2024).
Askanase, A. et al. A phase 1 study of NKX019, an allogeneic chimeric antigen receptor natural killer (CAR-NK) cell therapy in patients with systemic lupus erythematosus. Blood 144, 4846.4841–4846.4841 (2024).
Maia, A., Tarannum, M. & Romee, R. Genetic manipulation approaches to enhance the clinical application of NK cell-based immunotherapy. Stem Cell Transl. Med. 13, 230–242 (2024).
Dong, H. et al. Memory-like NK cells armed with a neoepitope-specific CAR exhibit potent activity against NPM1 mutated acute myeloid leukemia. Proc. Natl Acad. Sci. USA 119, e2122379119 (2022).
Li, Y. et al. KIR-based inhibitory CARs overcome CAR-NK cell trogocytosis-mediated fratricide and tumor escape. Nat. Med. 28, 2133–2144 (2022).
Ghobadi, A. et al. Induced pluripotent stem-cell-derived CD19-directed chimeric antigen receptor natural killer cells in B-cell lymphoma: a phase 1, first-in-human trial. Lancet 405, 127–136 (2025).
Zhang, W. et al. Chimeric antigen receptor macrophage therapy for breast tumours mediated by targeting the tumour extracellular matrix. Br. J. Cancer 121, 837–845 (2019).
Klichinsky, M. et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat. Biotechnol. 38, 947–953 (2020).
Pierini, S. et al. Chimeric antigen receptor macrophages (CAR-M) sensitize HER2+ solid tumors to PD1 blockade in pre-clinical models. Nat. Commun. 16, 706 (2025).
Lei, A. et al. A second-generation M1-polarized CAR macrophage with antitumor efficacy. Nat. Immunol. 25, 102–116 (2024).
Reiss, K. A. et al. CAR-macrophage therapy for HER2-overexpressing advanced solid tumors: a phase 1 trial. Nat. Med. 31, 1171–1182 (2025).
Zhang, J. et al. Generation of anti-GD2 CAR macrophages from human pluripotent stem cells for cancer immunotherapies. Stem Cell Rep. 18, 585–596 (2023).
Shah, Z. et al. Human anti-PSCA CAR macrophages possess potent antitumor activity against pancreatic cancer. Cell Stem Cell 31, 803–817.e806 (2024).
Nishimoto, K. P. et al. Allogeneic CD20-targeted ɣδ T cells exhibit innate and adaptive antitumor activities in preclinical B-cell lymphoma models. Clin. Transl. Immunol 11, e1373 (2022).
Lu, B. et al. Effects of IL-2 and IL-15 on the proliferative and antitumor capacities of allogeneic CD20 CAR-engineered γδ T cells in a 3D B cell lymphoma spheroid assay. J. ImmunoTher. Cancer https://doi.org/10.1136/jitc-2020-SITC2020.0107 (2021).
Boonchalermvichian, C. et al. invariant natural killer T cell therapy as a novel therapeutic approach in hematological malignancies. Front. Transpl. 3, 1353803 (2024).
Zhou, X. et al. CAR-redirected natural killer T cells demonstrate superior antitumor activity to CAR-T cells through multimodal CD1d-dependent mechanisms. Nat. Cancer 5, 1607–1621 (2024).
Heczey, A. et al. Anti-GD2 CAR-NKT cells in relapsed or refractory neuroblastoma: updated phase 1 trial interim results. Nat. Med. 29, 1379–1388 (2023).
Ghasemi, A. et al. Cytokine-armed dendritic cell progenitors for antigen-agnostic cancer immunotherapy. Nat. Cancer 5, 240–261 (2024).
Demaria, O. et al. A tetraspecific engager armed with a non-alpha IL-2 variant harnesses natural killer cells against B cell non-Hodgkin lymphoma. Sci. Immunol. 9, eadp3720 (2024).
Gauthier, L. et al. Multifunctional natural killer cell engagers targeting NKp46 trigger protective tumor immunity. Cell 177, 1701–1713.e1716 (2019).
Reddy, N. R. et al. Engineering synthetic suppressor T cells that execute locally targeted immunoprotective programs. Science 386, eadl4793 (2024).
Simic, M. S. et al. Programming tissue-sensing T cells that deliver therapies to the brain. Science 386, eadl4237 (2024).
Choi, Y. R. et al. A genome-engineered bioartificial implant for autoregulated anticytokine drug delivery. Sci. Adv. 7, eabj1414 (2021).
Bai, P. et al. A synthetic biology-based device prevents liver injury in mice. J. Hepatol. 65, 84–94 (2016).
Kemmer, C. et al. Self-sufficient control of urate homeostasis in mice by a synthetic circuit. Nat. Biotechnol. 28, 355–360 (2010).
Schukur, L., Geering, B., Charpin-El Hamri, G. & Fussenegger, M. Implantable synthetic cytokine converter cells with AND-gate logic treat experimental psoriasis. Sci. Transl. Med. 7, 318ra201 (2015).
Smole, A., Lainscek, D., Bezeljak, U., Horvat, S. & Jerala, R. A synthetic mammalian therapeutic gene circuit for sensing and suppressing inflammation. Mol. Ther. 25, 102–119 (2017).
Unal, G., Xie, Y. Q. & Fussenegger, M. A closed-loop cholesterol shunt controlling experimental dyslipidemia. Cell Metab. 37, 2066–2075.e2063 (2025).
Franko, N. et al. Regulation of therapeutic protein release in response to circadian biomarkers. Nat. Commun. 16, 9812 (2025).
Levy, O. et al. Shattering barriers toward clinically meaningful MSC therapies. Sci. Adv. 6, eaba6884 (2020).
Liu, L. et al. Mechanoresponsive stem cells to target cancer metastases through biophysical cues. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aan2966 (2017).
Pollock, K. et al. Human mesenchymal stem cells genetically engineered to overexpress brain-derived neurotrophic factor improve outcomes in Huntington’s disease mouse models. Mol. Ther. 24, 965–977 (2016).
Kleinboehl, E. W. et al. Engineering memory T cells as a platform for long-term enzyme replacement therapy in lysosomal storage disorders. Mol. Ther. 32, 3865–3878 (2024).
Cheng, R. Y. et al. Ex vivo engineered human plasma cells exhibit robust protein secretion and long-term engraftment in vivo. Nat. Commun. 13, 6110 (2022).
Liu, H. et al. A precision gene-engineered B cell medicine producing sustained levels of active factor IX for hemophilia B therapy. Mol. Ther. 34, 266–280 (2026).
Moffett, H. F. et al. B cells engineered to express pathogen-specific antibodies protect against infection. Sci. Immunol. 4, eaax0644 (2019).
Hill, T. F. et al. Human plasma cells engineered to secrete bispecifics drive effective in vivo leukemia killing. Mol. Ther. 32, 2676–2691 (2024).
Teixeira, A. P. & Fussenegger, M. Synthetic macromolecular switches for precision control of therapeutic cell functions. Nat. Rev. Bioeng. 2, 1005–1022 (2024).
Bertschi, A. et al. Controlling therapeutic protein expression via inhalation of a butter flavor molecule. Nucleic Acids Res. 51, e28 (2023).
Galvan, S., Madderson, O., Xue, S., Teixeira, A. P. & Fussenegger, M. Regulation of transgene expression by the natural sweetener xylose. Adv. Sci. 9, e2203193 (2022).
Wang, Y. et al. A versatile genetic control system in mammalian cells and mice responsive to clinically licensed sodium ferulate. Sci. Adv. 6, eabb9484 (2020).
Bai, P. et al. A fully human transgene switch to regulate therapeutic protein production by cooling sensation. Nat. Med. 25, 1266–1273 (2019).
Mahameed, M., Wang, P., Xue, S. & Fussenegger, M. Engineering receptors in the secretory pathway for orthogonal signalling control. Nat. Commun. 13, 7350 (2022).
Scheller, L., Strittmatter, T., Fuchs, D., Bojar, D. & Fussenegger, M. Generalized extracellular molecule sensor platform for programming cellular behavior. Nat. Chem. Biol. 14, 723–729 (2018).
Zhu, I. et al. Modular design of synthetic receptors for programmed gene regulation in cell therapies. Cell 185, 1431–1443.e1416 (2022).
Bertschi, A., Wang, P., Galvan, S., Teixeira, A. P. & Fussenegger, M. Combinatorial protein dimerization enables precise multi-input synthetic computations. Nat. Chem. Biol. 19, 767–777 (2023).
Franko, N., Teixeira, A. P., Xue, S., Charpin-El Hamri, G. & Fussenegger, M. Design of modular autoproteolytic gene switches responsive to anti-coronavirus drug candidates. Nat. Commun. 12, 6786 (2021).
Nihongaki, Y., Otabe, T., Ueda, Y. & Sato, M. A split CRISPR–Cpf1 platform for inducible genome editing and gene activation. Nat. Chem. Biol. 15, 882–888 (2019).
Franko, N. et al. Integrated compact regulators of protein activity enable control of signaling pathways and genome-editing in vivo. Cell Discov. 10, 9 (2024).
Nihongaki, Y., Yamamoto, S., Kawano, F., Suzuki, H. & Sato, M. CRISPR–Cas9-based photoactivatable transcription system. Chem. Biol. 22, 169–174 (2015).
Tycko, J. et al. Development of compact transcriptional effectors using high-throughput measurements in diverse contexts. Nat. Biotechnol. 43, 1525–1538 (2025).
Tycko, J. et al. High-throughput discovery and characterization of human transcriptional effectors. Cell 183, 2020–2035.e2016 (2020).
Liu, R. et al. Optogenetic control of RNA function and metabolism using engineered light-switchable RNA-binding proteins. Nat. Biotechnol. 40, 779–786 (2022).
Saito, H. et al. Synthetic translational regulation by an L7Ae-kink-turn RNP switch. Nat. Chem. Biol. 6, 71–78 (2010).
Monteys, A. M. et al. Regulated control of gene therapies by drug-induced splicing. Nature 596, 291–295 (2021).
Xiang, J. S. et al. Massively parallel RNA device engineering in mammalian cells with RNA-Seq. Nat. Commun. 10, 4327 (2019).
Nakahara, E., Mullapudi, V., Collier, G. E., Joachimiak, L. A. & Hulleman, J. D. Development of a new DHFR-based destabilizing domain with enhanced basal turnover and applicability in mammalian systems. ACS Chem. Biol. 17, 2877–2889 (2022).
Cho, J. H. et al. Engineering advanced logic and distributed computing in human CAR immune cells. Nat. Commun. 12, 792 (2021).
Minutolo, N. G. et al. Quantitative control of gene-engineered T-Cell activity through the covalent attachment of targeting ligands to a universal immune receptor. J. Am. Chem. Soc. 142, 6554–6568 (2020).
Stepanov, A. V. et al. Control of the antitumour activity and specificity of CAR T cells via organic adapters covalently tethering the CAR to tumour cells. Nat. Biomed. Eng. 8, 529–543 (2024).
Labanieh, L. et al. Enhanced safety and efficacy of protease-regulated CAR-T cell receptors. Cell 185, 1745–1763.e1722 (2022).
Li, H. S. et al. High-performance multiplex drug-gated CAR circuits. Cancer Cell 40, 1294–1305.e1294 (2022).
Giordano Attianese, G. M. P. et al. Dual ON/OFF-switch chimeric antigen receptor controlled by two clinically approved drugs. Proc. Natl Acad. Sci. USA 121, e2405085121 (2024).
Giordano-Attianese, G. et al. A computationally designed chimeric antigen receptor provides a small-molecule safety switch for T-cell therapy. Nat. Biotechnol. 38, 426–432 (2020).
Weber, E. W. et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science 372, eaba1786 (2021).
Piraner, D. I. et al. Engineered receptors for soluble cellular communication and disease sensing. Nature 638, 805–813 (2025).
Choe, J. H. et al. SynNotch-CAR T cells overcome challenges of specificity, heterogeneity, and persistence in treating glioblastoma. Sci. Transl. Med. 13, eabe7378 (2021).
Hernandez-Lopez, R. A. et al. T cell circuits that sense antigen density with an ultrasensitive threshold. Science 371, 1166–1171 (2021).
Hyrenius-Wittsten, A. et al. SynNotch CAR circuits enhance solid tumor recognition and promote persistent antitumor activity in mouse models. Sci. Transl. Med. 13, eabd8836 (2021).
Lajoie, M. J. et al. Designed protein logic to target cells with precise combinations of surface antigens. Science 369, 1637–1643 (2020).
Tousley, A. M. et al. Co-opting signalling molecules enables logic-gated control of CAR T cells. Nature 615, 507–516 (2023).
Bojar, D., Scheller, L., Hamri, G. C., Xie, M. & Fussenegger, M. Caffeine-inducible gene switches controlling experimental diabetes. Nat. Commun. 9, 2318 (2018).
Huang, J. et al. Aspirin-responsive gene switch regulating therapeutic protein expression. Nat. Commun. 16, 2028 (2025).
Wang, H., Xie, M., Charpin-El Hamri, G., Ye, H. & Fussenegger, M. Treatment of chronic pain by designer cells controlled by spearmint aromatherapy. Nat. Biomed. Eng. 2, 114–123 (2018).
Zhao, H., Xue, S., Hussherr, M. D., Teixeira, A. P. & Fussenegger, M. Autonomous push button-controlled rapid insulin release from a piezoelectrically activated subcutaneous cell implant. Sci. Adv. 8, eabm4389 (2022).
Zhou, Y. et al. A small and highly sensitive red/far-red optogenetic switch for applications in mammals. Nat. Biotechnol. 40, 262–272 (2022).
Mansouri, M. et al. Smart-watch-programmed green-light-operated percutaneous control of therapeutic transgenes. Nat. Commun. 12, 3388 (2021).
Huang, J., Xue, S., Teixeira, A. P. & Fussenegger, M. A mediator-free sonogenetic switch for therapeutic protein expression in mammalian cells. Nucleic Acids Res. 53, gkaf191 (2025).
Lin, Z., Guha Ray, P., Huang, J., Buchmann, P. & Fussenegger, M. Electromagnetic wireless remote control of mammalian transgene expression. Nat. Nanotechnol. 20, 1071–1078 (2025).
Huang, J., Xue, S., Buchmann, P., Teixeira, A. P. & Fussenegger, M. An electrogenetic interface to program mammalian gene expression by direct current. Nat. Metab. 5, 1395–1407 (2023).
Krawczyk, K. et al. Electrogenetic cellular insulin release for real-time glycemic control in type 1 diabetic mice. Science 368, 993–1001 (2020).
Wang, T. et al. Repurposing salicylic acid as a versatile inducer of proximity. Nat. Chem. Biol. 21, 1444–1456 (2025).
Teixeira, A. P., Xue, S., Huang, J. & Fussenegger, M. Evolution of molecular switches for regulation of transgene expression by clinically licensed gluconate. Nucleic Acids Res. 51, e85 (2023).
De Carluccio, G., Fusco, V. & di Bernardo, D. Engineering a synthetic gene circuit for high-performance inducible expression in mammalian systems. Nat. Commun. 15, 3311 (2024).
Allan, D. S. J. et al. Systematic improvements in lentiviral transduction of primary human natural killer cells undergoing ex vivo expansion. Mol. Ther. Methods Clin. Dev. 20, 559–571 (2021).
Colamartino, A. B. L. et al. Efficient and robust NK-Cell transduction with baboon envelope pseudotyped lentivector. Front. Immunol. 10, 2873 (2019).
Abdou, Y. et al. Abstract P2-12-17: a phase 1, first-in-human study of autologous monocytes engineered to express an anti-HER2 chimeric antigen receptor (CAR) in participants with HER2 overexpressing solid tumors. Clin. Cancer Res. https://doi.org/10.1158/1557-3265.SABCS24-P2-12-17 (2025).
Fiumara, M. et al. Genotoxic effects of base and prime editing in human hematopoietic stem cells. Nat. Biotechnol. 42, 877–891 (2024).
Holstein, M. et al. Efficient non-viral gene delivery into human hematopoietic stem cells by minicircle Sleeping Beauty transposon vectors. Mol. Ther. 26, 1137–1153 (2018).
Eyquem, J. et al. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543, 113–117 (2017).
Suchy, F. P. et al. Genome engineering with Cas9 and AAV repair templates generates frequent concatemeric insertions of viral vectors. Nat. Biotechnol. 43, 204–213 (2025).
Frangoul, H. et al. CRISPR–Cas9 gene editing for sickle cell disease and β-thalassemia. N. Engl. J. Med. 384, 252–260 (2021).
Wong, C. UK first to approve CRISPR treatment for diseases: what you need to know. Nature 623, 676–677 (2023).
Ramos, C. A. et al. Allogeneic NKT cells expressing a CD19-specific CAR in patients with relapsed or refractory B-cell malignancies: an interim analysis. Blood 138, 2819 (2021).
Martin, K. E., Hammer, Q., Perica, K., Sadelain, M. & Malmberg, K. J. Engineering immune-evasive allogeneic cellular immunotherapies. Nat. Rev. Immunol. 24, 680–693 (2024).
Mo, F. et al. Engineered off-the-shelf therapeutic T cells resist host immune rejection. Nat. Biotechnol. 39, 56–63 (2021).
Benjamin, R. et al. UCART19, a first-in-class allogeneic anti-CD19 chimeric antigen receptor T-cell therapy for adults with relapsed or refractory B-cell acute lymphoblastic leukaemia (CALM): a phase 1, dose-escalation trial. Lancet Haematol. 9, e833–e843 (2022).
Liu, D. D. et al. Umbilical cord blood: a promising source for allogeneic CAR-T cells. Front. Oncol. 12, 944248 (2022).
Wang, X. et al. Allogeneic CD19-targeted CAR-T therapy in patients with severe myositis and systemic sclerosis. Cell 187, 4890–4904.e4899 (2024).
Hu, X. et al. Hypoimmune anti-CD19 chimeric antigen receptor T cells provide lasting tumor control in fully immunocompetent allogeneic humanized mice. Nat. Commun. 14, 2020 (2023).
Jo, S. et al. Endowing universal CAR T-cell with immune-evasive properties using TALEN-gene editing. Nat. Commun. 13, 3453 (2022).
Hu, X. et al. Hypoimmune induced pluripotent stem cells survive long term in fully immunocompetent, allogeneic rhesus macaques. Nat. Biotechnol. 42, 413–423 (2024).
Hu, X. et al. Hypoimmune islets achieve insulin independence after allogeneic transplantation in a fully immunocompetent non-human primate. Cell Stem Cell 31, 334–340.e335 (2024).
Mehta, A. et al. Interim phase I clinical data of FT819-101, a study of the first-ever, off-the-shelf, iPSC-derived TCR-less CD19 CAR T-cell therapy for patients with relapsed/refractory B-cell malignancies. Blood 140, 4577–4578 (2022).
Paasch, D. et al. Ex vivo generation of CAR macrophages from hematopoietic stem and progenitor cells for use in cancer therapy. Cells 11, 994 (2022).
Jing, R. et al. EZH1 repression generates mature iPSC-derived CAR T cells with enhanced antitumor activity. Cell Stem Cell 29, 1181–1196.e1186 (2022).
Li, Y., Hermanson, D. L., Moriarity, B. S. & Kaufman, D. S. Human iPSC-derived natural killer cells engineered with chimeric antigen receptors enhance anti-tumor activity. Cell Stem Cell 23, 181–192.e185 (2018).
Shen, J. et al. Activating innate immune responses repolarizes hPSC-derived CAR macrophages to improve anti-tumor activity. Cell Stem Cell 31, 1003–1019.e1009 (2024).
Wang, Z. et al. 3D-organoid culture supports differentiation of human CAR+ iPSCs into highly functional CAR T cells. Cell Stem Cell 29, 515–527 (2022).
Wang, B. et al. Generation of hypoimmunogenic T cells from genetically engineered allogeneic human induced pluripotent stem cells. Nat. Biomed. Eng. 5, 429–440 (2021).
Cabrera, A. et al. The sound of silence: transgene silencing in mammalian cell engineering. Cell Syst. 13, 950–973 (2022).
Jing, R. et al. Maturation and persistence of CAR T cells derived from human pluripotent stem cells via chemical inhibition of G9a/GLP. Cell Stem Cell 32, 71–85 (2025).
van der Stegen, S. J. C. et al. Generation of T-cell-receptor-negative CD8αβ-positive CAR T cells from T-cell-derived induced pluripotent stem cells. Nat. Biomed. Eng. 6, 1284–1297 (2022).
Kim, D. Y. et al. Efficient CRISPR editing with a hypercompact Cas12f1 and engineered guide RNAs delivered by adeno-associated virus. Nat. Biotechnol. 40, 94–102 (2022).
Tornabene, P. et al. Intein-mediated protein trans-splicing expands adeno-associated virus transfer capacity in the retina. Sci. Transl. Med. 11, eaav4523 (2019).
Demircan, M. B. et al. T-cell specific in vivo gene delivery with DART-AAVs targeted to CD8. Mol. Ther. 32, 3470–3484 (2024).
Hanlon, K. S. et al. Selection of an efficient AAV vector for robust CNS transgene expression. Mol. Ther. Methods Clin. Dev. 15, 320–332 (2019).
Cripe, T. P. et al. Leveraging gene therapy to achieve long-term continuous or controllable expression of biotherapeutics. Sci. Adv. 8, eabm1890 (2022).
Andorko, J. I. et al. Targeted in vivo delivery of genetic medicines utilizing an engineered lentiviral vector platform results in CAR T and NK cell generation. Mol. Ther. 33, 4937–4952 (2025).
Milani, M. et al. Genome editing for scalable production of alloantigen-free lentiviral vectors for in vivo gene therapy. EMBO Mol. Med. 9, 1558–1573 (2017).
Chiocca, E. A. et al. Combined immunotherapy with controlled interleukin-12 gene therapy and immune checkpoint blockade in recurrent glioblastoma: an open-label, multi-institutional phase I trial. Neuro Oncol. 24, 951–963 (2022).
Chiocca, E. A. et al. Regulatable interleukin-12 gene therapy in patients with recurrent high-grade glioma: results of a phase 1 trial. Sci. Transl. Med. 11, eaaw5680 (2019).
Chen, H. et al. Branched chemically modified poly(A) tails enhance the translation capacity of mRNA. Nat. Biotechnol. 43, 194–203 (2025).
Bloom, K., van den Berg, F. & Arbuthnot, P. Self-amplifying RNA vaccines for infectious diseases. Gene Ther. 28, 117–129 (2019).
Vo, J. N. et al. The landscape of circular RNA in cancer. Cell 176, 869–881.e813 (2019).
Rothgangl, T. et al. In vivo adenine base editing of PCSK9 in macaques reduces LDL cholesterol levels. Nat. Biotechnol. 39, 949–957 (2021).
Villiger, L. et al. In vivo cytidine base editing of hepatocytes without detectable off-target mutations in RNA and DNA. Nat. Biomed. Eng. 5, 179–189 (2021).
Hunter, T. L. et al. In vivo CAR T cell generation to treat cancer and autoimmune disease. Science 388, 1311–1317 (2025).
Zou, Y. et al. Blood–brain barrier-penetrating single CRISPR–Cas9 nanocapsules for effective and safe glioblastoma gene therapy. Sci. Adv. 8, eabm8011 (2022).
Granit, V. et al. Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomised phase 1b/2a study. Lancet Neurol. 22, 578–590 (2023).
Raguram, A., An, M., Chen, P. Z. & Liu, D. R. Directed evolution of engineered virus-like particles with improved production and transduction efficiencies. Nat. Biotechnol. 43, 1635–1647 (2025).
Geilenkeuser, J. et al. Engineered nucleocytosolic vehicles for loading of programmable editors. Cell 188, 2637–2655.e2631 (2025).
Hamilton, J. R. et al. In vivo human T cell engineering with enveloped delivery vehicles. Nat. Biotechnol. 42, 1684–1692 (2024).
Schambach, A., Morgan, M. & Fehse, B. Two cases of T cell lymphoma following Piggybac-mediated CAR T cell therapy. Mol. Ther. 29, 2631–2633 (2021).
Musunuru, K. et al. Patient-specific in vivo gene editing to treat a rare genetic disease. N. Engl. J. Med. 392, 2235–2243 (2025).
Ji, R. J. et al. Epitope prime editing shields hematopoietic cells from CD123 immunotherapy for acute myeloid leukemia. Cell Stem Cell 31, 1650–1666 e1658 (2024).
Witte, I. P. et al. Programmable gene insertion in human cells with a laboratory-evolved CRISPR-associated transposase. Science 388, eadt5199 (2025).
Cole, A. J. et al. A chimeric viral platform for directed evolution in mammalian cells. Nat. Commun. 16, 4250 (2025).
Ma, L. & Lin, Y. Orthogonal RNA replication enables directed evolution and Darwinian adaptation in mammalian cells. Nat. Chem. Biol. 21, 451–463 (2025).
Ruffolo, J. A. et al. Design of highly functional genome editors by modeling the universe of CRISPR–Cas sequences. Preprint at BioRxiv https://doi.org/10.1101/2024.04.22.590591 (2024).
Acknowledgements
We thank A. Fussenegger-Aubel for advice on the figure design.
Author information
Authors and Affiliations
Contributions
All authors contributed to all aspects of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Genetics thanks Melita Irving, Graham Lord, who co-reviewed with Isabella Barbutti, and Katayoun Rezvani, who co-reviewed with Rafet Basar, for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Allogeneic cell therapies
-
Therapies using cells from a healthy donor and administered to a different recipient.
- Autologous cell therapies
-
Therapies using the patient’s own cells, modified ex vivo and re-infused.
- Encapsulation
-
The physical confinement of cells within semi-permeable materials that protect them from immune attack while allowing diffusion of nutrients, oxygen and therapeutic molecules.
- Envelope protein
-
Viral surface protein embedded in the lipid envelope that mediates host-cell binding and membrane fusion, determining tropism and entry efficiency.
- Immune reset
-
A transient therapeutic reprogramming of the immune system, with ablation of pathogenic cells, enabling immune reconstitution towards a healthier state.
- Kill switches
-
Engineered safety mechanisms that trigger selective elimination of therapeutic cells to prevent adverse effects.
- Logic gates
-
Engineered genetic circuits implementing Boolean rules (such as AND, OR, NOT, IF–THEN) so that effector functions occur only under specific combinations of molecular or environmental cues.
- Priming
-
Transient ex vivo conditioning of cells to enhance therapeutic function without permanent genetic modifications.
- Safe-harbour locus
-
Genomic site that supports stable transgene expression without disrupting endogenous gene function.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Teixeira, A.P., Aubel, D. & Fussenegger, M. Next-generation programmable cell therapies for precision medicine. Nat Rev Genet (2026). https://doi.org/10.1038/s41576-026-00945-3
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41576-026-00945-3