Abstract
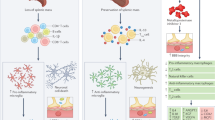
B cells represent a relatively minor cell population within both the healthy and diseased central nervous system (CNS), yet they can have profound effects. This is emphasized in multiple sclerosis, in which B cell-depleting therapies are arguably the most efficacious treatment for the condition. In this Review, we discuss how B cells enter and persist in the CNS and how, in many neurological conditions, B cells concentrate within CNS barriers but are rarely found in the parenchyma. We highlight how B cells can contribute to CNS pathology through antibody secretion, antigen presentation and secretion of neurotoxic molecules, using examples from CNS tumours, CNS infections and autoimmune conditions such as neuromyelitis optica and, in particular, multiple sclerosis. Overall, understanding common and divergent principles of B cell accumulation and their effects within the CNS could offer new insights into treating these devastating neurological conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sabatino, J. J. Jr., Probstel, A. K. & Zamvil, S. S. B cells in autoimmune and neurodegenerative central nervous system diseases. Nat. Rev. Neurosci. 20, 728–745 (2019).
Gilden, D. H. Infectious causes of multiple sclerosis. Lancet Neurol. 4, 195–202 (2005).
Li, R., Patterson, K. R. & Bar-Or, A. Reassessing B cell contributions in multiple sclerosis. Nat. Immunol. 19, 696–707 (2018).
Akkaya, M., Kwak, K. & Pierce, S. K. B cell memory: building two walls of protection against pathogens. Nat. Rev. Immunol. 20, 229–238 (2020).
Kang, T. H. & Jung, S. T. Boosting therapeutic potency of antibodies by taming Fc domain functions. Exp. Mol. Med. 51, 1–9 (2019).
Korn, T. & Kallies, A. T cell responses in the central nervous system. Nat. Rev. Immunol. 17, 179–194 (2017).
Rua, R. & McGavern, D. B. Advances in meningeal immunity. Trends Mol. Med. 24, 542–559 (2018).
Agrawal, S. M. et al. Extracellular matrix metalloproteinase inducer shows active perivascular cuffs in multiple sclerosis. Brain 136, 1760–1777 (2013).
Korin, B. et al. High-dimensional, single-cell characterization of the brain’s immune compartment. Nat. Neurosci. 20, 1300–1309 (2017). This is one of the few studies to precisely quantify the number of immune cells in the healthy CNS across the parenchyma, meninges and choroid plexus.
Brioschi, S. et al. Heterogeneity of meningeal B cells reveals a lymphopoietic niche at the CNS borders. Science 373, eabf9277 (2021).
Schafflick, D. et al. Single-cell profiling of CNS border compartment leukocytes reveals that B cells and their progenitors reside in non-diseased meninges. Nat. Neurosci. 24, 1225–1234 (2021).
Rubtsova, K., Rubtsov, A. V., Cancro, M. P. & Marrack, P. Age-associated B cells: a T-bet-dependent effector with roles in protective and pathogenic immunity. J. Immunol. 195, 1933–1937 (2015).
Fitzpatrick, Z. et al. Gut-educated IgA plasma cells defend the meningeal venous sinuses. Nature 587, 472–476 (2020). This publication highlights how B and plasma cells accumulate in the dura mater in health and disease and identified the gut as a source of IgA plasma cells.
Frischer, J. M. et al. The relation between inflammation and neurodegeneration in multiple sclerosis brains. Brain 132, 1175–1189 (2009).
Machado-Santos, J. et al. The compartmentalized inflammatory response in the multiple sclerosis brain is composed of tissue-resident CD8+ T lymphocytes and B cells. Brain 141, 2066–2082 (2018). This paper has the most thorough quantification of B and plasma cells in MS white matter lesions.
Howell, O. W. et al. Meningeal inflammation is widespread and linked to cortical pathology in multiple sclerosis. Brain 134, 2755–2771 (2011).
Prasad, R. Immunoglobulin levels in serum and cerebrospinal fluid in certain viral infections of the central nervous system. J. Infect. Dis. 148, 607 (1983).
Probstel, A. K. et al. Gut microbiota-specific IgA+ B cells traffic to the CNS in active multiple sclerosis. Sci. Immunol. 5, eabc7191 (2020).
Korin, B. et al. Short-term sleep deprivation in mice induces B cell migration to the brain compartment. Sleep 43, zsz222 (2020).
Kmiecik, J. et al. Elevated CD3+ and CD8+ tumor-infiltrating immune cells correlate with prolonged survival in glioblastoma patients despite integrated immunosuppressive mechanisms in the tumor microenvironment and at the systemic level. J. Neuroimmunol. 264, 71–83 (2013).
Domingues, P. H. et al. Immunophenotypic identification and characterization of tumor cells and infiltrating cell populations in meningiomas. Am. J. Pathol. 181, 1749–1761 (2012).
Kuhlmann, T., Lassmann, H. & Bruck, W. Diagnosis of inflammatory demyelination in biopsy specimens: a practical approach. Acta Neuropathol. 115, 275–287 (2008).
Han, S. et al. Glioma cell-derived placental growth factor induces regulatory B cells. Int. J. Biochem. Cell Biol. 57, 63–68 (2014).
Lee-Chang, C. et al. Activation of 4-1BBL+ B cells with CD40 agonism and IFNgamma elicits potent immunity against glioblastoma. J. Exp. Med. 218, e20200913 (2021).
Matschke, J. et al. Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. Lancet Neurol. 19, 919–929 (2020).
Heming, M. et al. Neurological manifestations of COVID-19 feature T cell exhaustion and dedifferentiated monocytes in cerebrospinal fluid. Immunity 54, 164–175.e6 (2021).
Song, E. et al. Divergent and self-reactive immune responses in the CNS of COVID-19 patients with neurological symptoms. Cell Rep. Med. 2, 100288 (2021).
Khateb, M., Bosak, N. & Muqary, M. Coronaviruses and central nervous system manifestations. Front. Neurol. 11, 715 (2020).
Phares, T. W., DiSano, K. D., Stohlman, S. A. & Bergmann, C. C. Progression from IgD+ IgM+ to isotype-switched B cells is site specific during coronavirus-induced encephalomyelitis. J. Virol. 88, 8853–8867 (2014).
Tschen, S. I. et al. CNS viral infection diverts homing of antibody-secreting cells from lymphoid organs to the CNS. Eur. J. Immunol. 36, 603–612 (2006).
DiSano, K. D., Stohlman, S. A. & Bergmann, C. C. Activated GL7+ B cells are maintained within the inflamed CNS in the absence of follicle formation during viral encephalomyelitis. Brain Behav. Immun. 60, 71–83 (2017).
Cepok, S. et al. Short-lived plasma blasts are the main B cell effector subset during the course of multiple sclerosis. Brain 128, 1667–1676 (2005).
Kowarik, M. C. et al. CXCL13 is the major determinant for B cell recruitment to the CSF during neuroinflammation. J. Neuroinflammation 9, 93 (2012).
Stock, A. D. et al. Tertiary lymphoid structures in the choroid plexus in neuropsychiatric lupus. JCI Insight 4, e124203 (2019).
Mok, M. Y., Chan, E. Y., Wong, W. S. & Lau, C. S. Intrathecal immunoglobulin production in patients with systemic lupus erythematosus with neuropsychiatric manifestations. Ann. Rheum. Dis. 66, 846–847 (2007).
Kowarik, M. C. et al. The cerebrospinal fluid immunoglobulin transcriptome and proteome in neuromyelitis optica reveals central nervous system-specific B cell populations. J. Neuroinflammation 12, 19 (2015).
Malviya, M. et al. NMDAR encephalitis: passive transfer from man to mouse by a recombinant antibody. Ann. Clin. Transl. Neurol. 4, 768–783 (2017).
Jelcic, I. et al. Memory B cells activate brain-homing, autoreactive CD4+ T cells in multiple sclerosis. Cell 175, 85–100.e23 (2018). In this study, memory B cells from MS patients are shown to induce CD4+ T cell proliferation and induce these cells to enter the CNS.
Kuhlmann, T. et al. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 133, 13–24 (2017).
Bell, L., Lenhart, A., Rosenwald, A., Monoranu, C. M. & Berberich-Siebelt, F. Lymphoid aggregates in the CNS of progressive multiple sclerosis patients lack regulatory T cells. Front. Immunol. 10, 3090 (2019).
Choi, S. R. et al. Meningeal inflammation plays a role in the pathology of primary progressive multiple sclerosis. Brain 135, 2925–2937 (2012).
Reali, C. et al. B cell rich meningeal inflammation associates with increased spinal cord pathology in multiple sclerosis. Brain Pathol. 30, 779–793 (2020).
Monaco, S., Nicholas, R., Reynolds, R. & Magliozzi, R. Intrathecal inflammation in progressive multiple sclerosis. Int. J. Mol. Sci. 21, 8217 (2020).
Corcione, A. et al. Recapitulation of B cell differentiation in the central nervous system of patients with multiple sclerosis. Proc. Natl Acad. Sci. USA 101, 11064–11069 (2004).
Schafflick, D. et al. Integrated single cell analysis of blood and cerebrospinal fluid leukocytes in multiple sclerosis. Nat. Commun. 11, 247 (2020).
Ramesh, A. et al. A pathogenic and clonally expanded B cell transcriptome in active multiple sclerosis. Proc. Natl Acad. Sci. USA 117, 22932–22943 (2020).
Di Pauli, F. et al. Features of intrathecal immunoglobulins in patients with multiple sclerosis. J. Neurol. Sci. 288, 147–150 (2010).
Eggers, E. L. et al. Clonal relationships of CSF B cells in treatment-naive multiple sclerosis patients. JCI Insight 2, e92724 (2017).
Munoz, U. et al. Main role of antibodies in demyelination and axonal damage in multiple sclerosis. Cell Mol. Neurobiol. https://doi.org/10.1007/s10571-021-01059-6 (2021).
Fransen, N. L. et al. Absence of B cells in brainstem and white matter lesions associates with less severe disease and absence of oligoclonal bands in MS. Neurol. Neuroimmunol. Neuroinflamm. 8, e955 (2021).
Absinta, M. et al. Gadolinium-based MRI characterization of leptomeningeal inflammation in multiple sclerosis. Neurology 85, 18–28 (2015).
Magliozzi, R. et al. Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology. Brain 130, 1089–1104 (2007).
Cepok, S. et al. Patterns of cerebrospinal fluid pathology correlate with disease progression in multiple sclerosis. Brain 124, 2169–2176 (2001).
Magliozzi, R. et al. A Gradient of neuronal loss and meningeal inflammation in multiple sclerosis. Ann. Neurol. 68, 477–493 (2010). This publication provides strong evidence that follicle-like structures in the meninges are associated with severe cortical pathology not seen in other types of MS lesions.
Howell, O. W. et al. Extensive grey matter pathology in the cerebellum in multiple sclerosis is linked to inflammation in the subarachnoid space. Neuropathol. Appl. Neurobiol. 41, 798–813 (2015).
Lovato, L. et al. Related B cell clones populate the meninges and parenchyma of patients with multiple sclerosis. Brain 134, 534–541 (2011).
Lucchinetti, C. F. et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 365, 2188–2197 (2011).
Peterson, J. W., Bo, L., Mork, S., Chang, A. & Trapp, B. D. Transected neurites, apoptotic neurons, and reduced inflammation in cortical multiple sclerosis lesions. Ann. Neurol. 50, 389–400 (2001).
Magliozzi, R. et al. Inflammatory intrathecal profiles and cortical damage in multiple sclerosis. Ann. Neurol. 83, 739–755 (2018).
Mitsdorffer, M. et al. Formation and immunomodulatory function of meningeal B-cell aggregates in progressive CNS autoimmunity. Brain 144, 1697–1710 (2021).
Pikor, N. B. et al. Integration of Th17- and lymphotoxin-derived signals initiates meningeal-resident stromal cell remodeling to propagate neuroinflammation. Immunity 43, 1160–1173 (2015).
Grewal, I. S. et al. CD62L is required on effector cells for local interactions in the CNS to cause myelin damage in experimental allergic encephalomyelitis. Immunity 14, 291–302 (2001).
Dang, A. K., Tesfagiorgis, Y., Jain, R. W., Craig, H. C. & Kerfoot, S. M. Meningeal infiltration of the spinal cord by non-classically activated B cells is associated with chronic disease course in a spontaneous B cell-dependent model of CNS autoimmune disease. Front. Immunol. 6, 470 (2015).
Serafini, B., Rosicarelli, B., Magliozzi, R., Stigliano, E. & Aloisi, F. Detection of ectopic B-cell follicles with germinal centers in the meninges of patients with secondary progressive multiple sclerosis. Brain Pathol. 14, 164–174 (2004).
Michel, L. et al. Activated leukocyte cell adhesion molecule regulates B lymphocyte migration across central nervous system barriers. Sci. Transl. Med. 11, eaaw0475 (2019). Using a combination of mouse models and live imaging, this publication identifies ALCAM as an important mediator of B cell recruitment into the CNS and quantifies other recruitment molecules on B cells.
Alter, A. et al. Determinants of human B cell migration across brain endothelial cells. J. Immunol. 170, 4497–4505 (2003).
Haas, J. et al. The choroid plexus is permissive for a preactivated antigen-experienced memory B-cell subset in multiple sclerosis. Front. Immunol. 11, 618544 (2020).
van Langelaar, J. et al. Induction of brain-infiltrating T-bet-expressing B cells in multiple sclerosis. Ann. Neurol. 86, 264–278 (2019).
Hussain, R. Z. et al. alpha4-integrin deficiency in B cells does not affect disease in a T-cell-mediated EAE disease model. Neurol. Neuroimmunol. Neuroinflamm. 6, e563 (2019).
Lehmann-Horn, K., Sagan, S. A., Bernard, C. C., Sobel, R. A. & Zamvil, S. S. B-cell very late antigen-4 deficiency reduces leukocyte recruitment and susceptibility to central nervous system autoimmunity. Ann. Neurol. 77, 902–908 (2015).
Hausler, D. et al. CNS inflammation after natalizumab therapy for multiple sclerosis: A retrospective histopathological and CSF cohort study. Brain Pathol. 31, e12969 (2021).
Tesfagiorgis, Y., Zhu, S. L., Jain, R. & Kerfoot, S. M. Activated B cells participating in the anti-myelin response are excluded from the inflamed central nervous system in a model of autoimmunity that allows for B cell recognition of autoantigen. J. Immunol. 199, 449–457 (2017).
Pollok, K. et al. The chronically inflamed central nervous system provides niches for long-lived plasma cells. Acta Neuropathol. Commun. 5, 88 (2017).
Rojas, O. L. et al. Recirculating Intestinal IgA-producing cells regulate neuroinflammation via IL-10. Cell 176, 610–624.e18 (2019). This study identified that IgA+ plasma cells from the gut move into the CNS during neuroinflammation and suppress EAE using IL-10.
Nguyen, D. C. et al. Factors of the bone marrow microniche that support human plasma cell survival and immunoglobulin secretion. Nat. Commun. 9, 3698 (2018).
Baker, D., Pryce, G., James, L. K., Schmierer, K. & Giovannoni, G. Failed B cell survival factor trials support the importance of memory B cells in multiple sclerosis. Eur. J. Neurol. 27, 221–228 (2020).
Touil, H. et al. Human central nervous system astrocytes support survival and activation of B cells: implications for MS pathogenesis. J. Neuroinflammation 15, 114 (2018).
Chan, T. D. et al. Antigen affinity controls rapid T-dependent antibody production by driving the expansion rather than the differentiation or extrafollicular migration of early plasmablasts. J. Immunol. 183, 3139–3149 (2009).
Quinn, J. L., Kumar, G., Agasing, A., Ko, R. M. & Axtell, R. C. Role of TFH cells in promoting T helper 17-induced neuroinflammation. Front. Immunol. 9, 382 (2018).
Chihara, N. et al. Plasmablasts as migratory IgG-producing cells in the pathogenesis of neuromyelitis optica. PLoS One 8, e83036 (2013).
O’Connor, T. et al. Age-related gliosis promotes central nervous system lymphoma through CCL19-mediated tumor cell retention. Cancer Cell 36, 250–267.e9 (2019).
Williams, M. T. et al. Interleukin-15 enhances cellular proliferation and upregulates CNS homing molecules in pre-B acute lymphoblastic leukemia. Blood 123, 3116–3127 (2014).
Krumbholz, M. et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J. Exp. Med. 201, 195–200 (2005).
DiSano, K. D., Royce, D. B., Gilli, F. & Pachner, A. R. Central nervous system inflammatory aggregates in the Theiler’s virus model of progressive multiple sclerosis. Front. Immunol. 10, 1821 (2019).
Hohlfeld, R., Dornmair, K., Meinl, E. & Wekerle, H. The search for the target antigens of multiple sclerosis, part 2: CD8+ T cells, B cells, and antibodies in the focus of reverse-translational research. Lancet Neurol. 15, 317–331 (2016).
Lucchinetti, C. et al. Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann. Neurol. 47, 707–717 (2000).
Sadaba, M. C. et al. Axonal and oligodendrocyte-localized IgM and IgG deposits in MS lesions. J. Neuroimmunol. 247, 86–94 (2012).
Genain, C. P., Cannella, B., Hauser, S. L. & Raine, C. S. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat. Med. 5, 170–175 (1999).
Zhang, Y. et al. Clonal expansion of IgA-positive plasma cells and axon-reactive antibodies in MS lesions. J. Neuroimmunol. 167, 120–130 (2005).
Keegan, M. et al. Relation between humoral pathological changes in multiple sclerosis and response to therapeutic plasma exchange. Lancet 366, 579–582 (2005).
Silber, E., Semra, Y. K., Gregson, N. A. & Sharief, M. K. Patients with progressive multiple sclerosis have elevated antibodies to neurofilament subunit. Neurology 58, 1372–1381 (2002).
Blauth, K. et al. Antibodies produced by clonally expanded plasma cells in multiple sclerosis cerebrospinal fluid cause demyelination of spinal cord explants. Acta Neuropathol. 130, 765–781 (2015).
Brandle, S. M. et al. Distinct oligoclonal band antibodies in multiple sclerosis recognize ubiquitous self-proteins. Proc. Natl Acad. Sci. USA 113, 7864–7869 (2016).
Willis, S. N. et al. Investigating the antigen specificity of multiple sclerosis central nervous system-derived immunoglobulins. Front. Immunol. 6, 600 (2015).
Benjamins, J. A. et al. Exosome-enriched fractions from MS B cells induce oligodendrocyte death. Neurol. Neuroimmunol. Neuroinflamm. 6, e550 (2019). This paper identified that exosomes produced from B cells can be toxic to oligodendrocytes, opening an entire new field of study.
Lisak, R. P. et al. B cells from patients with multiple sclerosis induce cell death via apoptosis in neurons in vitro. J. Neuroimmunol. 309, 88–99 (2017).
Bettelli, E., Baeten, D., Jager, A., Sobel, R. A. & Kuchroo, V. K. Myelin oligodendrocyte glycoprotein-specific T and B cells cooperate to induce a Devic-like disease in mice. J. Clin. Invest. 116, 2393–2402 (2006).
Kuerten, S. et al. Tertiary lymphoid organ development coincides with determinant spreading of the myelin-specific T cell response. Acta Neuropathol. 124, 861–873 (2012).
Parker Harp, C. R. et al. B cells are capable of independently eliciting rapid reactivation of encephalitogenic CD4 T cells in a murine model of multiple sclerosis. PLoS One 13, e0199694 (2018).
Pierson, E. R., Stromnes, I. M. & Goverman, J. M. B cells promote induction of experimental autoimmune encephalomyelitis by facilitating reactivation of T cells in the central nervous system. J. Immunol. 192, 929–939 (2014).
Claes, N. et al. Age-associated B cells with proinflammatory characteristics are expanded in a proportion of multiple sclerosis patients. J. Immunol. 197, 4576–4583 (2016).
Li, R. et al. Proinflammatory GM-CSF-producing B cells in multiple sclerosis and B cell depletion therapy. Sci. Transl. Med. 7, 310ra166 (2015). In this study, GM-CSF-producing B cells are shown to induce inflammatory polarization of macrophages, creating a link between B cell and macrophage biology in MS.
McWilliam, O., Sellebjerg, F., Marquart, H. V. & von Essen, M. R. B cells from patients with multiple sclerosis have a pathogenic phenotype and increased LTα and TGFβ1 response. J. Neuroimmunol. 324, 157–164 (2018).
Stein, J. et al. Intrathecal B cells in ms have significantly greater lymphangiogenic potential compared to B cells derived from non-MS subjects. Front. Neurol. 9, 554 (2018).
Gardner, C. et al. Cortical grey matter demyelination can be induced by elevated pro-inflammatory cytokines in the subarachnoid space of MOG-immunized rats. Brain 136, 3596–3608 (2013).
Magliozzi, R. et al. Meningeal inflammation changes the balance of TNF signalling in cortical grey matter in multiple sclerosis. J. Neuroinflammation 16, 259 (2019).
James, R. E. et al. Persistent elevation of intrathecal pro-inflammatory cytokines leads to multiple sclerosis-like cortical demyelination and neurodegeneration. Acta Neuropathol. Commun. 8, 66 (2020).
van Olst, L. et al. Meningeal inflammation in multiple sclerosis induces phenotypic changes in cortical microglia that differentially associate with neurodegeneration. Acta Neuropathol. 141, 881–899 (2021).
Fehres, C. M. et al. APRIL induces a novel subset of IgA+ regulatory B cells that suppress inflammation via expression of IL-10 and PD-L1. Front. Immunol. 10, 1368 (2019).
Fillatreau, S., Sweenie, C. H., McGeachy, M. J., Gray, D. & Anderton, S. M. B cells regulate autoimmunity by provision of IL-10. Nat. Immunol. 3, 944–950 (2002).
Pennati, A. et al. Regulatory B cells induce formation of IL-10-expressing T cells in mice with autoimmune neuroinflammation. J. Neurosci. 36, 12598–12610 (2016).
Bjarnadottir, K. et al. B cell-derived transforming growth factor-beta1 expression limits the induction phase of autoimmune neuroinflammation. Sci. Rep. 6, 34594 (2016).
Piancone, F. et al. B lymphocytes in multiple sclerosis: Bregs and BTLA/CD272 expressing-CD19+ lymphocytes modulate disease severity. Sci. Rep. 6, 29699 (2016).
Shen, P. et al. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature 507, 366–370 (2014).
Khan, A. R. et al. PD-L1hi B cells are critical regulators of humoral immunity. Nat. Commun. 6, 5997 (2015).
Lehmann-Horn, K. et al. CNS accumulation of regulatory B cells is VLA-4-dependent. Neurol. Neuroimmunol. Neuroinflamm. 3, e212 (2016).
Matsumoto, M. et al. Interleukin-10-producing plasmablasts exert regulatory function in autoimmune inflammation. Immunity 41, 1040–1051 (2014). This study demonstrates that plasmablasts are a major regulatory population in EAE induced through immunization that inhibits T cell responses in the lymph nodes where T cells are differentiating.
Wang, A., Rojas, O., Lee, D. & Gommerman, J. L. Regulation of neuroinflammation by B cells and plasma cells. Immunol. Rev. 299, 45–60 (2021).
Stebegg, M. et al. Regulation of the germinal center response. Front. Immunol. 9, 2469 (2018).
Roco, J. A. et al. Class-switch recombination occurs infrequently in germinal centers. Immunity 51, 337–350.e7 (2019).
Lepennetier, G. et al. Cytokine and immune cell profiling in the cerebrospinal fluid of patients with neuro-inflammatory diseases. J. Neuroinflammation 16, 219 (2019).
Flach, A. C. et al. Autoantibody-boosted T-cell reactivation in the target organ triggers manifestation of autoimmune CNS disease. Proc. Natl Acad. Sci. USA 113, 3323–3328 (2016).
Romanelli, E. et al. Myelinosome formation represents an early stage of oligodendrocyte damage in multiple sclerosis and its animal model. Nat. Commun. 7, 13275 (2016).
Jegou, J. F. et al. C3d binding to the myelin oligodendrocyte glycoprotein results in an exacerbated experimental autoimmune encephalomyelitis. J. Immunol. 178, 3323–3331 (2007).
Molnarfi, N. et al. MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies. J. Exp. Med. 210, 2921–2937 (2013).
Murakami, M., Kamimura, D. & Hirano, T. Pleiotropy and specificity: insights from the interleukin 6 family of cytokines. Immunity 50, 812–831 (2019).
Pennati, A., Nylen, E. A., Duncan, I. D. & Galipeau, J. Regulatory B cells normalize CNS myeloid cell content in a mouse model of multiple sclerosis and promote oligodendrogenesis and remyelination. J. Neurosci. 40, 5105–5115 (2020).
Acknowledgements
R.W.J. acknowledges postdoctoral fellowship funding from the University of Calgary Eyes High program, the Multiple Sclerosis of Canada and Roche unrestricted educational fellowship. V.W.Y. acknowledges salary support from the Canada Research Chair Tier 1 program and operating grant support from the Canadian Institutes of Health Research and the Multiple Sclerosis Society of Canada.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Immunology thanks S. Pillai and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- B1 B cells
-
A subset of mature B cells with limited B cell receptor diversity predominantly found within the peritoneal and pleural cavities that typically differentiates directly into plasma cells, forming few memory cells.
- Follicular B cells
-
A subset of mature B cells, also known as B2 B cells, that represents the majority of B cells in the body and are commonly found within all lymphoid organs.
- Marginal zone B cells
-
A subset of mature B cells with limited B cell receptor diversity that is predominantly found in the marginal zone of the spleen and which typically differentiates directly into plasma cells but can also generate memory cells.
- Germinal centre B cells
-
This subset of activated B cells is an intermediate stage of differentiation and is the precursor of higher-affinity memory B cells and plasma cells.
- Memory B cells
-
A subset of B cells that is antigen experienced and has reacquired a quiescent phenotype and can participate in secondary immune responses, or sometimes refers to activated B cells that retain an activated phenotype.
- T-bet+ memory B cells
-
A subset of memory B cells that expresses the transcription factor T-bet whose numbers tend to increase with age as well as in autoimmune disorders and viral infections.
- Plasma cells
-
Terminally differentiated B cells that produce large amounts of antibodies and are short-lived unless they find a survival niche allowing long-term maintenance.
- Regulatory B (Breg) cells
-
This subset of B cells suppresses inflammation through the secretion of anti-inflammatory proteins or through physical interactions.
- Plasmablasts
-
The precursor to plasma cells that has begun to produce antibodies but also retains some features of B cells, such as surface B cell receptor and MHC class II expression, and is still proliferating.
- T helper 17 (TH17) cells
-
A subset of CD4+ T cells characterized by the expression of the transcription factor RORγT and the expression of IL-17A that is commonly associated with immunity against extracellular pathogens.
- T follicular helper cells
-
A subset of CD4+ T cells characterized by the expression of the transcriptional repressor Bcl6 and the chemokine receptor CXCR5 allowing these cells to co-localize with B cells and promote their differentiation through physical interactions.
- T helper 1 (TH1) cells
-
A subset of CD4+ T cells characterized by the expression of the transcription factor T-bet and the expression of IFNγ that is commonly associated with cellular immunity.
Rights and permissions
About this article
Cite this article
Jain, R.W., Yong, V.W. B cells in central nervous system disease: diversity, locations and pathophysiology. Nat Rev Immunol 22, 513–524 (2022). https://doi.org/10.1038/s41577-021-00652-6
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41577-021-00652-6
This article is cited by
-
Spilling the T: T cells in tauopathy mechanisms, disease progression, and therapeutic horizons
Molecular Neurodegeneration Advances (2026)
-
Secreted IgM deficiency alters the retinal landscape enhancing neurodegeneration associated with aging
Immunity & Ageing (2025)
-
Breaking boundaries: role of the brain barriers in metastatic process
Fluids and Barriers of the CNS (2025)
-
B cells are not drivers of stromal cell activation during acute CNS infection
Journal of Neuroinflammation (2025)
-
EBV induces CNS homing of B cells attracting inflammatory T cells
Nature (2025)