Abstract
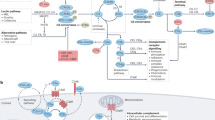
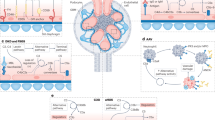
The complement system is a recognized pillar of host defence against infection and noxious self-derived antigens. Complement is traditionally known as a serum-effective system, whereby the liver expresses and secretes most complement components, which participate in the detection of bloodborne pathogens and drive an inflammatory reaction to safely remove the microbial or antigenic threat. However, perturbations in normal complement function can cause severe disease and, for reasons that are currently not fully understood, the kidney is particularly vulnerable to dysregulated complement activity. Novel insights into complement biology have identified cell-autonomous and intracellularly active complement — the complosome — as an unexpected central orchestrator of normal cell physiology. For example, the complosome controls mitochondrial activity, glycolysis, oxidative phosphorylation, cell survival and gene regulation in innate and adaptive immune cells, and in non-immune cells, such as fibroblasts and endothelial and epithelial cells. These unanticipated complosome contributions to basic cell physiological pathways make it a novel and central player in the control of cell homeostasis and effector responses. This discovery, together with the realization that an increasing number of human diseases involve complement perturbations, has renewed interest in the complement system and its therapeutic targeting. Here, we summarize the current knowledge about the complosome across healthy cells and tissues, highlight contributions from dysregulated complosome activities to human disease and discuss potential therapeutic implications.
Key points
-
Complement function is compartmentalized and operates systemically, locally in the extracellular space, and intracellularly within sub-cellular compartments and organelles.
-
Intracellular complement — the complosome — serves non-classical roles as a novel central regulator of basic cell physiological processes including mitochondrial respiration, glycolysis, autophagy and gene transcription.
-
The complosome functions across immune and non-immune cells and tissues where it controls normal cell turnover, the responses to infectious and non-infectious stimuli and the return to homeostasis.
-
Perturbations in complosome activities contribute to human disease, including infections and infection-related pathological conditions, arthritic disease, atherosclerosis, cancer and kidney disease.
-
Targeting the complosome, possibly in combination with the extracellularly active complement, might be therapeutically beneficial in complement-mediated pathological conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bordet, J. G. O. Sur l’existence de substances sensibilisatrices dans la plupart des serums antimicrobiens. Ann. De. l’Inst. Pasteur 15, 289–303 (1901).
Merle, N. S., Church, S. E., Fremeaux-Bacchi, V. & Roumenina, L. T. Complement system part I — molecular mechanisms of activation and regulation. Front. Immunol. 6, 262 (2015).
Merle, N. S., Noe, R., Halbwachs-Mecarelli, L., Fremeaux-Bacchi, V. & Roumenina, L. T. Complement system part II: role in immunity. Front. Immunol. 6, 257 (2015).
Pepys, M. B. Role of complement in induction of the allergic response. Nat. N. Biol. 237, 157–159 (1972).
Killick, J., Morisse, G., Sieger, D. & Astier, A. L. Complement as a regulator of adaptive immunity. Semin. Immunopathol. 40, 37–48 (2018).
Carroll, M. C. & Isenman, D. E. Regulation of humoral immunity by complement. Immunity 37, 199–207 (2012).
Kolev, M., Le Friec, G. & Kemper, C. Complement — tapping into new sites and effector systems. Nat. Rev. Immunol. 14, 811–820 (2014).
Liszewski, M. K. et al. Intracellular complement activation sustains T cell homeostasis and mediates effector differentiation. Immunity 39, 1143–1157 (2013).
Kolev, M. et al. Complement regulates nutrient influx and metabolic reprogramming during Th1 cell responses. Immunity 42, 1033–1047 (2015).
Kremlitzka, M. et al. Interaction of serum-derived and internalized C3 with DNA in human B cells — a potential involvement in regulation of gene transcription. Front. Immunol. 10, 493 (2019).
King, B. C., Renstrom, E. & Blom, A. M. Intracellular cytosolic complement component C3 regulates cytoprotective autophagy in pancreatic β cells by interaction with ATG16L1. Autophagy 15, 919–921 (2019).
West, E. E., Kolev, M. & Kemper, C. Complement and the regulation of T cell responses. Annu. Rev. Immunol. 36, 309–338 (2018).
Kolev, M. et al. Inside-out of complement in cancer. Front. Immunol. 13, 931273 (2022).
Freiwald, T. & Afzali, B. Renal diseases and the role of complement: linking complement to immune effector pathways and therapeutics. Adv. Immunol. 152, 1–81 (2021).
Hajishengallis, G., Reis, E. S., Mastellos, D. C., Ricklin, D. & Lambris, J. D. Novel mechanisms and functions of complement. Nat. Immunol. 18, 1288–1298 (2017).
Reis, E. S., Mastellos, D. C., Hajishengallis, G. & Lambris, J. D. New insights into the immune functions of complement. Nat. Rev. Immunol. 19, 503–516 (2019).
Laumonnier, Y., Karsten, C. M. & Kohl, J. Novel insights into the expression pattern of anaphylatoxin receptors in mice and men. Mol. Immunol. 89, 44–58 (2017).
Bosmann, M. & Ward, P. A. Role of C3, C5 and anaphylatoxin receptors in acute lung injury and in sepsis. Adv. Exp. Med. Biol. 946, 147–159 (2012).
Mathern, D. R. & Heeger, P. S. Molecules great and small: the complement system. Clin. J. Am. Soc. Nephrol. 10, 1636–1650 (2015).
West, E. E. & Kemper, C. in Paul’s Fundamental Immunology, 8th edn (eds Flajnik, M. F., Singh, N. J. & Holland, S. M.) 1–39 (Wolters Kluwer, 2022).
Fernandez-Sola, J. et al. Persistent low C3 levels associated with meningococcal meningitis and membranoproliferative glomerulonephritis. Am. J. Nephrol. 10, 426–430 (1990).
Defendi, F., Thielens, N. M., Clavarino, G., Cesbron, J. Y. & Dumestre-Perard, C. The immunopathology of complement proteins and innate immunity in autoimmune disease. Clin. Rev. Allergy Immunol. 58, 229–251 (2020).
Conigliaro, P. et al. Complement, infection, and autoimmunity. Curr. Opin. Rheumatol. 31, 532–541 (2019).
Pouw, R. B. & Ricklin, D. Tipping the balance: intricate roles of the complement system in disease and therapy. Semin. Immunopathol. 43, 757–771 (2021).
Liszewski, M. K. & Atkinson, J. P. Complement regulators in human disease: lessons from modern genetics. J. Intern. Med. 277, 294–305 (2015).
Cserhalmi, M., Papp, A., Brandus, B., Uzonyi, B. & Jozsi, M. Regulation of regulators: role of the complement factor H-related proteins. Semin. Immunol. 45, 101341 (2019).
Matsumoto, A. K. et al. Intersection of the complement and immune systems: a signal transduction complex of the B lymphocyte-containing complement receptor type 2 and CD19. J. Exp. Med. 173, 55–64 (1991).
Cumpelik, A. et al. Dynamic regulation of B cell complement signaling is integral to germinal center responses. Nat. Immunol. 22, 757–768 (2021).
Astier, A., Trescol-Biemont, M. C., Azocar, O., Lamouille, B. & Rabourdin-Combe, C. Cutting edge: CD46, a new costimulatory molecule for T cells, that induces p120CBL and LAT phosphorylation. J. Immunol. 164, 6091–6095 (2000).
Zaffran, Y. et al. CD46/CD3 costimulation induces morphological changes of human T cells and activation of Vav, Rac, and extracellular signal-regulated kinase mitogen-activated protein kinase. J. Immunol. 167, 6780–6785 (2001).
Le Friec, G. et al. The CD46-Jagged1 interaction is critical for human TH1 immunity. Nat. Immunol. 13, 1213–1221 (2012).
Heesterbeek, D. A. C., Angelier, M. L., Harrison, R. A. & Rooijakkers, S. H. M. Complement and bacterial infections: from molecular mechanisms to therapeutic applications. J. Innate Immun. 10, 455–464 (2018).
Botto, M. et al. Homozygous C1q deficiency causes glomerulonephritis associated with multiple apoptotic bodies. Nat. Genet. 19, 56–59 (1998).
Strainic, M. G. et al. Locally produced complement fragments C5a and C3a provide both costimulatory and survival signals to naive CD4+ T cells. Immunity 28, 425–435 (2008).
Lalli, P. N. et al. Locally produced C5a binds to T cell-expressed C5aR to enhance effector T-cell expansion by limiting antigen-induced apoptosis. Blood 112, 1759–1766 (2008).
Lalli, P. N., Strainic, M. G., Lin, F., Medof, M. E. & Heeger, P. S. Decay accelerating factor can control T cell differentiation into IFN-γ-producing effector cells via regulating local C5a-induced IL-12 production. J. Immunol. 179, 5793–5802 (2007).
Fang, C., Miwa, T. & Song, W. C. Decay-accelerating factor regulates T-cell immunity in the context of inflammation by influencing costimulatory molecule expression on antigen-presenting cells. Blood 118, 1008–1014 (2011).
Liu, J. et al. IFN-γ and IL-17 production in experimental autoimmune encephalomyelitis depends on local APC-T cell complement production. J. Immunol. 180, 5882–5889 (2008).
Cravedi, P. et al. Immune cell-derived C3a and C5a costimulate human T cell alloimmunity. Am. J. Transpl. 13, 2530–2539 (2013).
Pratt, J. R., Basheer, S. A. & Sacks, S. H. Local synthesis of complement component C3 regulates acute renal transplant rejection. Nat. Med. 8, 582–587 (2002).
Angeletti, A. et al. Loss of decay-accelerating factor triggers podocyte injury and glomerulosclerosis. J. Exp. Med. https://doi.org/10.1084/jem.20191699 (2020).
Arbore, G. et al. T helper 1 immunity requires complement-driven NLRP3 inflammasome activity in CD4+ T cells. Science 352, aad1210 (2016).
Niyonzima, N. et al. Mitochondrial C5aR1 activity in macrophages controls IL-1β production underlying sterile inflammation. Sci. Immunol. 6, eabf2489 (2021).
Sorbara, M. T. et al. Complement C3 drives autophagy-dependent restriction of cyto-invasive bacteria. Cell Host Microbe 23, 644–652 e645 (2018).
Zeng, J. et al. CD46 splice variant enhances translation of specific mRNAs linked to an aggressive tumor cell phenotype in bladder cancer. Mol. Ther. Nucleic Acids 24, 140–153 (2021).
Ding, P. et al. Intracellular complement C5a/C5aR1 stabilizes β-catenin to promote colorectal tumorigenesis. Cell Rep. 39, 110851 (2022).
Ishii, M., Beeson, G., Beeson, C. & Rohrer, B. Mitochondrial C3a receptor activation in oxidatively stressed epithelial cells reduces mitochondrial respiration and metabolism. Front. Immunol. 12, 628062 (2021).
Karsten, C. M., Laumonnier, Y. & Kohl, J. Functional analysis of C5a effector responses in vitro and in vivo. Methods Mol. Biol. 1100, 291–304 (2014).
Hess, C. & Kemper, C. Complement-mediated regulation of metabolism and basic cellular processes. Immunity 45, 240–254 (2016).
Kolev, M. & Kemper, C. Keeping it all going — complement meets metabolism. Front. Immunol. 8, 1 (2017).
Liszewski, M. K., Elvington, M., Kulkarni, H. S. & Atkinson, J. P. Complement’s hidden arsenal: new insights and novel functions inside the cell. Mol. Immunol. 84, 2–9 (2017).
King, B. C. & Blom, A. M. Complement in metabolic disease: metaflammation and a two-edged sword. Semin. Immunopathol. 43, 829–841 (2021).
Wong, Y. C., Kim, S., Peng, W. & Krainc, D. Regulation and function of mitochondria-lysosome membrane contact sites in cellular homeostasis. Trends Cell Biol. 29, 500–513 (2019).
Settembre, C. et al. A lysosome-to-nucleus signalling mechanism senses and regulates the lysosome via mTOR and TFEB. EMBO J. 31, 1095–1108 (2012).
Condon, K. J. & Sabatini, D. M. Nutrient regulation of mTORC1 at a glance. J. Cell Sci. https://doi.org/10.1242/jcs.222570 (2019).
Perucha, E. et al. The cholesterol biosynthesis pathway regulates IL-10 expression in human Th1 cells. Nat. Commun. 10, 498 (2019).
Xu, K. et al. Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science 371, 405–410 (2021).
Arbore, G. et al. Complement receptor CD46 co-stimulates optimal human CD8+ T cell effector function via fatty acid metabolism. Nat. Commun. 9, 4186 (2018).
Rahman, J., Singh, P., Merle, N. S., Niyonzima, N. & Kemper, C. Complement’s favourite organelle — mitochondria? Br. J. Pharmacol. 178, 2771–2785 (2021).
Kim, K. H. & Lee, M. S. Autophagy — a key player in cellular and body metabolism. Nat. Rev. Endocrinol. 10, 322–337 (2014).
Viret, C. et al. Regulation of anti-microbial autophagy by factors of the complement system. Microb. Cell 7, 93–105 (2020).
Zhang, S. et al. The regulation, function, and role of lipophagy, a form of selective autophagy, in metabolic disorders. Cell Death Dis. 13, 132 (2022).
Li, Y. et al. Intracellular C3 prevents hepatic steatosis by promoting autophagy and very-low-density lipoprotein secretion. FASEB J. 35, e22037 (2021).
King, B. C. & Blom, A. M. Intracellular complement: evidence, definitions, controversies, and solutions. Immunol. Rev. https://doi.org/10.1111/imr.13135 (2022).
Ni Choileain, S. et al. The dynamic processing of CD46 intracellular domains provides a molecular rheostat for T cell activation. PLoS ONE 6, e16287 (2011).
Ni Choileain, S. & Astier, A. L. CD46 processing: a means of expression. Immunobiology 217, 169–175 (2012).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013).
Cardone, J. et al. Complement regulator CD46 temporally regulates cytokine production by conventional and unconventional T cells. Nat. Immunol. 11, 862–871 (2010).
Ni Choileain, S. et al. TCR-stimulated changes in cell surface CD46 expression generate type 1 regulatory T cells. Sci. Signal. https://doi.org/10.1126/scisignal.aah6163 (2017).
Arbore, G., Kemper, C. & Kolev, M. Intracellular complement — the complosome — in immune cell regulation. Mol. Immunol. 89, 2–9 (2017).
Arbore, G. & Kemper, C. A novel “complement-metabolism-inflammasome axis” as a key regulator of immune cell effector function. Eur. J. Immunol. 46, 1563–1573 (2016).
Ling, G. S. et al. C1q restrains autoimmunity and viral infection by regulating CD8+ T cell metabolism. Science 360, 558–563 (2018).
Paiano, J. et al. Follicular B2 cell activation and class switch recombination depend on autocrine C3ar1/C5ar1 signaling in B2 cells. J. Immunol. 203, 379–388 (2019).
Jimenez-Reinoso, A. et al. Human plasma C3 is essential for the development of memory B, but not T, lymphocytes. J. Allergy Clin. Immunol. 141, 1151–1154 e1114 (2018).
Kolev, M. et al. Diapedesis-induced integrin signaling via LFA-1 facilitates tissue immunity by inducing intrinsic complement C3 expression in immune cells. Immunity 52, 513–527.e8 (2020).
Liew, P. X. & Kubes, P. The neutrophil’s role during health and disease. Physiol. Rev. 99, 1223–1248 (2019).
Bashant, K. R. et al. Proteomic, biomechanical and functional analyses define neutrophil heterogeneity in systemic lupus erythematosus. Ann. Rheum. Dis. 80, 209–218 (2021).
Benoit, M. E., Clarke, E. V., Morgado, P., Fraser, D. A. & Tenner, A. J. Complement protein C1q directs macrophage polarization and limits inflammasome activity during the uptake of apoptotic cells. J. Immunol. 188, 5682–5693 (2012).
Baudino, L. et al. C3 opsonization regulates endocytic handling of apoptotic cells resulting in enhanced T-cell responses to cargo-derived antigens. Proc. Natl Acad. Sci. USA 111, 1503–1508 (2014).
Martin, M. et al. Factor H uptake regulates intracellular C3 activation during apoptosis and decreases the inflammatory potential of nucleosomes. Cell Death Differ. 23, 903–911 (2016).
Tam, J. C., Bidgood, S. R., McEwan, W. A. & James, L. C. Intracellular sensing of complement C3 activates cell autonomous immunity. Science 345, 1256070 (2014).
Wang, Y., Tong, X., Zhang, J. & Ye, X. The complement C1qA enhances retinoic acid-inducible gene-I-mediated immune signalling. Immunology 136, 78–85 (2012).
Liszewski, M. K. & Atkinson, J. P. Membrane cofactor protein (MCP; CD46): deficiency states and pathogen connections. Curr. Opin. Immunol. 72, 126–134 (2021).
Schafer, N. et al. Complement factor H-related 3 enhanced inflammation and complement activation in human RPE cells. Front. Immunol. 12, 769242 (2021).
Kulkarni, H. S. et al. Intracellular C3 protects human airway epithelial cells from stress-associated cell death. Am. J. Respir. Cell Mol. Biol. 60, 144–157 (2019).
Chaudhary, N., Jayaraman, A., Reinhardt, C., Campbell, J. D. & Bosmann, M. A single-cell lung atlas of complement genes identifies the mesothelium and epithelium as prominent sources of extrahepatic complement proteins. Mucosal Immunol. 15, 927–939 (2022).
Sunderhauf, A. et al. GC1qR cleavage by caspase-1 drives aerobic glycolysis in tumor cells. Front. Oncol. 10, 575854 (2020).
Mahajan, S. et al. Local complement factor H protects kidney endothelial cell structure and function. Kidney Int. 100, 824–836 (2021).
Correa-Gallegos, D., Jiang, D. & Rinkevich, Y. Fibroblasts as confederates of the immune system. Immunol. Rev. 302, 147–162 (2021).
Friscic, J. et al. The complement system drives local inflammatory tissue priming by metabolic reprogramming of synovial fibroblasts. Immunity 54, 1002–1021 e1010 (2021).
Terri, M. et al. Mechanisms of peritoneal fibrosis: focus on immune cells-peritoneal stroma interactions. Front. Immunol. 12, 607204 (2021).
Shidham, V. B. The panorama of different faces of mesothelial cells. Cytojournal 18, 31 (2021).
Liu, Y. et al. Transition of mesothelial cell to fibroblast in peritoneal dialysis: EMT, stem cell or bystander? Perit. Dial. Int. 35, 14–25 (2015).
Lubbers, R., van Essen, M. F., van Kooten, C. & Trouw, L. A. Production of complement components by cells of the immune system. Clin. Exp. Immunol. 188, 183–194 (2017).
Markiewski, M. M. et al. C3a and C3b activation products of the third component of complement (C3) are critical for normal liver recovery after toxic injury. J. Immunol. 173, 747–754 (2004).
Yilmaz, M. et al. Overexpression of schizophrenia susceptibility factor human complement C4A promotes excessive synaptic loss and behavioral changes in mice. Nat. Neurosci. 24, 214–224 (2021).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
Stevens, B. et al. The classical complement cascade mediates CNS synapse elimination. Cell 131, 1164–1178 (2007).
Wang, C. et al. Microglia mediate forgetting via complement-dependent synaptic elimination. Science 367, 688–694 (2020).
Kunz, N. & Kemper, C. Complement has brains — do intracellular complement and immunometabolism cooperate in tissue homeostasis and behavior? Front. Immunol. 12, 629986 (2021).
Tenner, A. J. Complement-mediated events in Alzheimer’s disease: mechanisms and potential therapeutic targets. J. Immunol. 204, 306–315 (2020).
Hong, S. et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 352, 712–716 (2016).
Brennan, F. H., Lee, J. D., Ruitenberg, M. J. & Woodruff, T. M. Therapeutic targeting of complement to modify disease course and improve outcomes in neurological conditions. Semin. Immunol. 28, 292–308 (2016).
Sekar, A. et al. Schizophrenia risk from complex variation of complement component 4. Nature 530, 177–183 (2016).
Jacob, A. & Alexander, J. J. Complement and blood–brain barrier integrity. Mol. Immunol. 61, 149–152 (2014).
Veerhuis, R., Nielsen, H. M. & Tenner, A. J. Complement in the brain. Mol. Immunol. 48, 1592–1603 (2011).
Datta, D. et al. Classical complement cascade initiating C1q protein within neurons in the aged rhesus macaque dorsolateral prefrontal cortex. J. Neuroinflammation 17, 8 (2020).
Ten, V. S. et al. Complement component c1q mediates mitochondria-driven oxidative stress in neonatal hypoxic-ischemic brain injury. J. Neurosci. 30, 2077–2087 (2010).
Meri, S. et al. Human protectin (CD59), an 18,000-20,000 MW complement lysis restricting factor, inhibits C5b-8 catalysed insertion of C9 into lipid bilayers. Immunology 71, 1–9 (1990).
Golec, E. et al. Alternative splicing encodes functional intracellular CD59 isoforms that mediate insulin secretion and are down-regulated in diabetic islets. Proc. Natl Acad. Sci. USA 119, e2120083119 (2022).
Golec, E. et al. A cryptic non-GPI-anchored cytosolic isoform of CD59 controls insulin exocytosis in pancreatic β-cells by interaction with SNARE proteins. FASEB J. 33, 12425–12434 (2019).
Krus, U. et al. The complement inhibitor CD59 regulates insulin secretion by modulating exocytotic events. Cell Metab. 19, 883–890 (2014).
King, B. C. et al. Complement component C3 is highly expressed in human pancreatic islets and prevents β cell death via ATG16L1 interaction and autophagy regulation. Cell Metab. 29, 202–210 e206 (2019).
Zelek, W. M., Xie, L., Morgan, B. P. & Harris, C. L. Compendium of current complement therapeutics. Mol. Immunol. 114, 341–352 (2019).
Schartz, N. D. & Tenner, A. J. The good, the bad, and the opportunities of the complement system in neurodegenerative disease. J. Neuroinflammation 17, 354 (2020).
Dijkstra, D. J., Joeloemsingh, J. V., Bajema, I. M. & Trouw, L. A. Complement activation and regulation in rheumatic disease. Semin. Immunol. 45, 101339 (2019).
Ghannam, A., Fauquert, J. L., Thomas, C., Kemper, C. & Drouet, C. Human complement C3 deficiency: Th1 induction requires T cell-derived complement C3a and CD46 activation. Mol. Immunol. 58, 98–107 (2014).
Ghannam, A. et al. Human C3 deficiency associated with impairments in dendritic cell differentiation, memory B cells, and regulatory T cells. J. Immunol. 181, 5158–5166 (2008).
Yan, B. et al. SARS-CoV-2 drives JAK1/2-dependent local complement hyperactivation. Sci. Immunol. https://doi.org/10.1126/sciimmunol.abg0833 (2021).
Afzali, B., Noris, M., Lambrecht, B. N. & Kemper, C. The state of complement in COVID-19. Nat. Rev. Immunol. 22, 77–84 (2022).
Bosmann, M. Complement control for COVID-19. Sci. Immunol. https://doi.org/10.1126/sciimmunol.abj1014 (2021).
Ma, L. et al. Increased complement activation is a distinctive feature of severe SARS-CoV-2 infection. Sci. Immunol. https://doi.org/10.1126/sciimmunol.abh2259 (2021).
Banda, N. K. et al. Analysis of complement gene expression, clinical associations, and biodistribution of complement proteins in the synovium of early rheumatoid arthritis patients reveals unique pathophysiologic features. J. Immunol. 208, 2482–2496 (2022).
Ellinghaus, U. et al. Dysregulated CD46 shedding interferes with Th1-contraction in systemic lupus erythematosus. Eur. J. Immunol. 47, 1200–1210 (2017).
Aragam, K. G. et al. Discovery and systematic characterization of risk variants and genes for coronary artery disease in over a million participants. Nat. Genet. 54, 1803–1815 (2022).
Sunderhauf, A. et al. Regulation of epithelial cell expressed C3 in the intestine — relevance for the pathophysiology of inflammatory bowel disease? Mol. Immunol. 90, 227–238 (2017).
Satyam, A. et al. Intracellular activation of complement 3 is responsible for intestinal tissue damage during mesenteric ischemia. J. Immunol. 198, 788–797 (2017).
Roumenina, L. T., Daugan, M. V., Petitprez, F., Sautes-Fridman, C. & Fridman, W. H. Context-dependent roles of complement in cancer. Nat. Rev. Cancer 19, 698–715 (2019).
Reis, E. S., Mastellos, D. C., Ricklin, D., Mantovani, A. & Lambris, J. D. Complement in cancer: untangling an intricate relationship. Nat. Rev. Immunol. 18, 5–18 (2018).
Daugan, M. V. et al. Intracellular factor H drives tumor progression independently of the complement cascade. Cancer Immunol. Res. 9, 909–925 (2021).
Daugan, M. V. et al. Complement C1s and C4d as prognostic biomarkers in renal cancer: emergence of noncanonical functions of C1s. Cancer Immunol. Res. 9, 891–908 (2021).
Thurman, J. M. Complement and the kidney: an overview. Adv. Chronic Kidney Dis. 27, 86–94 (2020).
Richards, A. et al. Mutations in human complement regulator, membrane cofactor protein (CD46), predispose to development of familial hemolytic uremic syndrome. Proc. Natl Acad. Sci. USA 100, 12966–12971 (2003).
Sheerin, N. S. et al. Synthesis of complement protein C3 in the kidney is an important mediator of local tissue injury. FASEB J. 22, 1065–1072 (2008).
Brown, K. M. et al. Influence of donor C3 allotype on late renal-transplantation outcome. N. Engl. J. Med. 354, 2014–2023 (2006).
Portilla, D. & Xavier, S. Role of intracellular complement activation in kidney fibrosis. Br. J. Pharmacol. 178, 2880–2891 (2021).
Tziastoudi, M. et al. Key genetic components of fibrosis in diabetic nephropathy: an updated systematic review and meta-analysis. Int. J. Mol. Sci. https://doi.org/10.3390/ijms232315331 (2022).
Heurich, M. et al. Common polymorphisms in C3, factor B, and factor H collaborate to determine systemic complement activity and disease risk. Proc. Natl Acad. Sci. USA 108, 8761–8766 (2011).
Samstad, E. O. et al. Cholesterol crystals induce complement-dependent inflammasome activation and cytokine release. J. Immunol. 192, 2837–2845 (2014).
Zimmer, S. et al. Cyclodextrin promotes atherosclerosis regression via macrophage reprogramming. Sci. Transl. Med. 8, 333ra350 (2016).
Bakke, S. S. et al. Cyclodextrin reduces cholesterol crystal-induced inflammation by modulating complement activation. J. Immunol. 199, 2910–2920 (2017).
Gerard, A., Cope, A. P., Kemper, C., Alon, R. & Kochl, R. LFA-1 in T cell priming, differentiation, and effector functions. Trends Immunol. 42, 706–722 (2021).
Seya, T. et al. CD46 (membrane cofactor protein of complement, measles virus receptor): structural and functional divergence among species (review). Int. J. Mol. Med. 1, 809–816 (1998).
West, E. E., Kunz, N. & Kemper, C. Complement and human T cell metabolism: location, location, location. Immunol. Rev. 295, 68–81 (2020).
Kremlitzka, M. et al. Alternative translation and retrotranslocation of cytosolic C3 that detects cytoinvasive bacteria. Cell Mol. Life Sci. 79, 291 (2022).
Russkamp, N. F. et al. Experimental design of complement component 5a-induced acute lung injury (C5a-ALI): a role of CC-chemokine receptor type 5 during immune activation by anaphylatoxin. FASEB J. 29, 3762–3772 (2015).
Tenner, A. J., Stevens, B. & Woodruff, T. M. New tricks for an ancient system: physiological and pathological roles of complement in the CNS. Mol. Immunol. 102, 3–13 (2018).
Ratajczak, M. Z. & Kucia, M. Hematopoiesis and innate immunity: an inseparable couple for good and bad times, bound together by an hormetic relationship. Leukemia 36, 23–32 (2022).
Elvington, M., Liszewski, M. K., Bertram, P., Kulkarni, H. S. & Atkinson, J. P. A C3(H20) recycling pathway is a component of the intracellular complement system. J. Clin. Invest. 127, 970–981 (2017).
West, E. E. & Kemper, C. Complement and T cell metabolism: food for thought. Immunometabolism 1, e190006 (2019).
Song, W. C. Crosstalk between complement and toll-like receptors. Toxicol. Pathol. 40, 174–182 (2012).
Barratt-Due, A., Pischke, S. E., Nilsson, P. H., Espevik, T. & Mollnes, T. E. Dual inhibition of complement and Toll-like receptors as a novel approach to treat inflammatory diseases — C3 or C5 emerge together with CD14 as promising targets. J. Leukoc. Biol. 101, 193–204 (2017).
Asgari, E. et al. C3a modulates IL-1β secretion in human monocytes by regulating ATP efflux and subsequent NLRP3 inflammasome activation. Blood 122, 3473–3481 (2013).
Niyonzima, N. et al. Cholesterol crystals use complement to increase NLRP3 signaling pathways in coronary and carotid atherosclerosis. EBioMedicine 60, 102985 (2020).
Triantafilou, K., Hughes, T. R., Triantafilou, M. & Morgan, B. P. The complement membrane attack complex triggers intracellular Ca2+ fluxes leading to NLRP3 inflammasome activation. J. Cell Sci. 126, 2903–2913 (2013).
Kumar, V. The complement system, toll-like receptors and inflammasomes in host defense: three musketeers’ one target. Int. Rev. Immunol. 38, 131–156 (2019).
Liu, H. et al. Mannan binding lectin attenuates double-stranded RNA-mediated TLR3 activation and innate immunity. FEBS Lett. 588, 866–872 (2014).
Acknowledgements
Work in the Kemper laboratory is supported in part by the Intramural Research Program of the National Institutes of Health, National Heart, Lung, and Blood Institute (ZIA/hl006223 to C.K.).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made substantial contributions to discussions of the content and wrote, reviewed or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Nephrology thanks M. Bosmann, P. Cravedi, A. Jayaraman, L. Roumenina and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
West, E.E., Kemper, C. Complosome — the intracellular complement system. Nat Rev Nephrol 19, 426–439 (2023). https://doi.org/10.1038/s41581-023-00704-1
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41581-023-00704-1
This article is cited by
-
Herpes simplex virus-1 induces complement-mediated microglial phagocytosis of synapses in murine primary brain cultures and tissues
Cell Communication and Signaling (2026)
-
Complement system in tumor growth and metastases
British Journal of Cancer (2026)
-
Complement C3a Suppresses Spinal Cord Neural Stem Cell Activation by Inhibiting UCHL1 via the NF-κB p65/Nrf2 Pathway
Neuroscience Bulletin (2026)
-
Complosome Activation after Myocardial Infarction in a Murine Model of Permanent LAD Ligation
Stem Cell Reviews and Reports (2026)
-
Targeting the complement-mTOR-autophagy axis: the role of apolipoprotein E in depression
BMC Biology (2025)