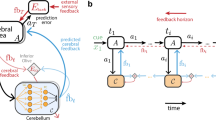
Abstract
The rise of the deep neural network as the workhorse of artificial intelligence has brought increased attention to how network architectures serve specialized functions. The cerebellum, with its largely shallow, feedforward architecture, provides a curious example of such a specialized network. Within the cerebellum, tiny supernumerary granule cells project to a monolayer of giant Purkinje neurons that reweight synaptic inputs under the instructive influence of a unitary synaptic input from climbing fibres. What might this predominantly feedforward organization confer computationally? Here we review evidence for and against the hypothesis that the cerebellum learns basic associative feedforward control policies to speed up motor control and learning. We contrast and link this feedforward control framework with another prominent set of theories proposing that the cerebellum computes internal models. Ultimately, we suggest that the cerebellum may implement control through mechanisms that resemble internal models but involve model-free implicit mappings of high-dimensional sensorimotor contexts to motor output.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Eccles, J. C., Ito, M. & Szentágothai, J. The Cerebellum as a Neuronal Machine (Springer, 1967).
Cisek, P. Evolution of behavioural control from chordates to primates. Philos. Trans. R. Soc. Lond. B Biol. Sci. 377, 20200522 (2022). This review discusses neural function through the lens of evolutionary transitions that require novel computations.
Shadmehr, R. & Mussa-Ivaldi S. Biological Learning and Control (MIT Press, 2023). This book provides an invaluable introduction to computational approaches to studying biological motor control.
Holmes, G. The Croonian Lectures on the clinical symptoms of cerebellar disease and their interpretation. Lecture I. 1922. Cerebellum 6, 142–147 (2007). This work presents a fascinating and thorough description of motor deficits that emerged in World War I soldiers who suffered gunshot wounds to the cerebellum.
Shadmehr, R. Population coding in the cerebellum: a machine learning perspective. J. Neurophysiol. 124, 2022–2051 (2020).
Spong, M. W. & Fujita, M. in The Impact of Control Technology: Overview, Success Stories, and Research Challenges (eds Samad, T. and Annaswamy, A.) 49–56 (IEEE Control Systems Society, 2011).
Wolpert, D. M. & Miall, R. C. Forward models for physiological motor control. Neural Netw. 9, 1265–1279 (1996). This paper beautifully describes how forward models can be used to solve many motor control problems common in biological systems.
Wolpert, D. M., Miall, R. C. & Kawato, M. Internal models in the cerebellum. Trends Cogn. Sci. 2, 338–347 (1998).
Shadmehr, R., Smith, M. A. & Krakauer, J. W. Error correction, sensory prediction, and adaptation in motor control. Annu. Rev. Neurosci. 33, 89–108 (2010).
Cullen, K. E. Internal models of self-motion: neural computations by the vestibular cerebellum. Trends Neurosci. 46, 986–1002 (2023).
Ohyama, T., Nores, W. L., Murphy, M. & Mauk, M. D. What the cerebellum computes. Trends Neurosci. 26, 222–227 (2003). This perspective article proposes that feedforward control principles underlying DEC could be extended to other motor control domains.
Raymond, J. L., Lisberger, S. G. & Mauk, M. D. The cerebellum: a neuronal learning machine? Science 272, 1126–1131 (1996).
Bastian, A. J. Learning to predict the future: the cerebellum adapts feedforward movement control. Curr. Opin. Neurobiol. 16, 645–649 (2006).
McCormick, D. A. & Thompson, R. F. Cerebellum: essential involvement in the classically conditioned eyelid response. Science 223, 296–299 (1984).
Moyer, J. R., Deyo, R. A. & Disterhoft, J. F. Hippocampectomy disrupts trace eye-blink conditioning in rabbits. Behav. Neurosci. 129, 523–532 (2015).
Kalmbach, B. E., Ohyama, T., Kreider, J. C., Riusech, F. & Mauk, M. D. Interactions between prefrontal cortex and cerebellum revealed by trace eyelid conditioning. Learn. Mem. 16, 86–95 (2009).
Medina, J. F., Nores, W. L., Ohyama, T. & Mauk, M. D. Mechanisms of cerebellar learning suggested by eyelid conditioning. Curr. Opin. Neurobiol. 10, 717–724 (2000).
Jirenhed, D.-A., Bengtsson, F. & Hesslow, G. Acquisition, extinction, and reacquisition of a cerebellar cortical memory trace. J. Neurosci. 27, 2493–2502 (2007).
Jirenhed, D.-A. & Hesslow, G. Are Purkinje cell pauses drivers of classically conditioned blink responses? Cerebellum 15, 526–534 (2016).
Halverson, H. E., Khilkevich, A. & Mauk, M. D. Relating cerebellar Purkinje cell activity to the timing and amplitude of conditioned eyelid responses. J. Neurosci. 35, 7813–7832 (2015).
Ten Brinke, M. M. et al. Dynamic modulation of activity in cerebellar nuclei neurons during Pavlovian eyeblink conditioning in mice. eLife 6, e28132 (2017).
Mauk, M. D. & Buonomano, D. V. The neural basis of temporal processing. Annu. Rev. Neurosci. 27, 307–340 (2004).
Rasmussen, A., Jirenhed, D.-A., Wetmore, D. Z. & Hesslow, G. Changes in complex spike activity during classical conditioning. Front. Neural Circuits 8, 90 (2014).
Ohmae, S. & Medina, J. F. Climbing fibers encode a temporal-difference prediction error during cerebellar learning in mice. Nat. Neurosci. 18, 1798–1803 (2015).
ten Brinke, M. M. et al. Evolving models of Pavlovian conditioning: cerebellar cortical dynamics in awake behaving mice. Cell Rep. 13, 1977–1988 (2015).
Boele, H.-J. et al. Impact of parallel fiber to Purkinje cell long-term depression is unmasked in absence of inhibitory input. Sci. Adv. 4, eaas9426 (2018).
Vilis, T. & Hore, J. in Progress in Brain Research Vol. 64 (eds. Freund, H.-J., Büttner, U., Cohen, B. & Noth, J.) 207–215 (Elsevier, 1986).
Flament, D., Hore, J. & Vilis, T. Braking of fast and accurate elbow flexions in the monkey. J. Physiol. 349, 195–202 (1984).
Vilis, T. & Hore, J. Effects of changes in mechanical state of limb on cerebellar intention tremor. J. Neurophysiol. 40, 1214–1224 (1977).
Pasalar, S., Roitman, A. V., Durfee, W. K. & Ebner, T. J. Force field effects on cerebellar Purkinje cell discharge with implications for internal models. Nat. Neurosci. 9, 1404–1411 (2006).
Robinson, D. A. The use of control systems analysis in the neurophysiology of eye movements. Annu. Rev. Neurosci. 4, 463–503 (1981).
Robinson, F. R. & Fuchs, A. F. The role of the cerebellum in voluntary eye movements. Annu. Rev. Neurosci. 24, 981–1004 (2001).
Smith, M. A., Brandt, J. & Shadmehr, R. Motor disorder in Huntington’s disease begins as a dysfunction in error feedback control. Nature 403, 544–549 (2000).
Kawato, M. & Gomi, H. A computational model of four regions of the cerebellum based on feedback-error learning. Biol. Cybern. 68, 95–103 (1992).
Flash, T. & Sejnowski, T. J. Computational approaches to motor control. Curr. Opin. Neurobiol. 11, 655–662 (2001).
Dean, P. & Porrill, J. Adaptive-filter models of the cerebellum: computational analysis. Cerebellum 7, 567–571 (2008).
Heiney, S. A., Wojaczynski, G. J. & Medina, J. F. Action-based organization of a cerebellar module specialized for predictive control of multiple body parts. Neuron 109, 2981–2994.e5 (2021).
Albus, J. S. A theory of cerebellar function. Math. Biosci. 10, 25–61 (1971). This impressive work presents a sweeping hypothesis of cerebellar control of movements that proves a useful framework more than 50 years later.
Safo, P. & Regehr, W. G. Timing dependence of the induction of cerebellar LTD. Neuropharmacology 54, 213–218 (2008).
Medina, J. F. & Mauk, M. D. Computer simulation of cerebellar information processing. Nat. Neurosci. 3, 1205–1211 (2000).
Becker, M. I. & Person, A. L. Cerebellar control of reach kinematics for endpoint precision. Neuron 103, 335–348.e5 (2019).
Guo, J.-Z. et al. Disrupting cortico-cerebellar communication impairs dexterity. eLife 10, e65906 (2021).
Gao, Z. et al. Excitatory cerebellar nucleocortical circuit provides internal amplification during associative conditioning. Neuron 89, 645–657 (2016).
Wang, X., Yu, S.-Y., Ren, Z., De Zeeuw, C. I. & Gao, Z. A FN–MdV pathway and its role in cerebellar multimodular control of sensorimotor behavior. Nat. Commun. 11, 6050 (2020).
Noda, H. et al. Saccadic eye movements evoked by microstimulation of the fastigial nucleus of macaque monkeys. J. Neurophysiol. 60, 1036–1052 (1988).
Sun, Z., Junker, M., Dicke, P. W. & Thier, P. Individual neurons in the caudal fastigial oculomotor region convey information on both macro- and microsaccades. Eur. J. Neurosci. 44, 2531–2542 (2016).
Özcan, O. O. et al. Differential coding strategies in glutamatergic and gabaergic neurons in the medial cerebellar nucleus. J. Neurosci. 40, 159–170 (2020).
Burton, J. E. & Onoda, N. Interpositus neuron discharge in relation to a voluntary movement. Brain Res. 121, 167–172 (1977).
Fortier, P. A., Kalaska, J. F. & Smith, A. M. Cerebellar neuronal activity related to whole-arm reaching movements in the monkey. J. Neurophysiol. 62, 198–211 (1989).
Zhai, P. et al. Whisker kinematics in the cerebellum. J. Physiol. 602, 153–181 (2024).
Brooks, J. X., Carriot, J. & Cullen, K. E. Learning to expect the unexpected: rapid updating in primate cerebellum during voluntary self-motion. Nat. Neurosci. 18, 1310–1317 (2015). This study provides a pristine example of learning and reafference cancellation within the primate cerebellum.
Fuchs, A. F., Robinson, F. R. & Straube, A. Role of the caudal fastigial nucleus in saccade generation. I. Neuronal discharge pattern. J. Neurophysiol. 70, 1723–1740 (1993).
Low, A. Y. T. et al. Precision of discrete and rhythmic forelimb movements requires a distinct neuronal subpopulation in the interposed anterior nucleus. Cell Rep. 22, 2322–2333 (2018).
Thanawalla, A. R., Chen, A. I. & Azim, E. The cerebellar nuclei and dexterous limb movements. Neuroscience 450, 168–183 (2020).
Dacre, J. et al. A cerebellar–thalamocortical pathway drives behavioral context-dependent movement initiation. Neuron 109, 2326–2338.e8 (2021).
Nashef, A., Cohen, O., Harel, R., Israel, Z. & Prut, Y. Reversible block of cerebellar outflow reveals cortical circuitry for motor coordination. Cell Rep. 27, 2608–2619.e4 (2019).
Rispal-Padel, L., Cicirata, F. & Pons, C. Cerebellar nuclear topography of simple and synergistic movements in the alert baboon (Papio papio). Exp. Brain Res. 47, 365–380 (1982).
Rispal-Padel, L., Cicirata, F. & Pons, C. Contribution of the dentato-thalamo-cortical system to control of motor synergy. Neurosci. Lett. 22, 137–144 (1981).
Thach, W. T. Cerebellar output: properties, synthesis and uses. Brain Res. 40, 89–97 (1972).
Apps, R. & Garwicz, M. Anatomical and physiological foundations of cerebellar information processing. Nat. Rev. Neurosci. 6, 297–311 (2005).
Brown, S. T. & Raman, I. M. Sensorimotor integration and amplification of reflexive whisking by well-timed spiking in the cerebellar corticonuclear circuit. Neuron 99, 564–575.e2 (2018). This study discovers conditions under which cerebellar output is coupled or decoupled from cerebellar cortical signalling, providing a precedent for considering how sensory and motor feedback are handled differently by the cerebellum.
Proville, R. D. et al. Cerebellum involvement in cortical sensorimotor circuits for the control of voluntary movements. Nat. Neurosci. 17, 1233–1239 (2014).
Chen, S., Augustine, G. J. & Chadderton, P. Serial processing of kinematic signals by cerebellar circuitry during voluntary whisking. Nat. Commun. 8, 232 (2017).
Gaffield, M. A. & Christie, J. M. Movement rate is encoded and influenced by widespread, coherent activity of cerebellar molecular layer interneurons. J. Neurosci. 37, 4751–4765 (2017).
Gao, Z. et al. A cortico-cerebellar loop for motor planning. Nature 563, 113–116 (2018).
Chabrol, F. P., Blot, A. & Mrsic-Flogel, T. D. Cerebellar contribution to preparatory activity in motor neocortex. Neuron 103, 506–519.e4 (2019).
Jung, S. J. et al. Novel cerebello-amygdala connections provide missing link between cerebellum and limbic system. Front. Syst. Neurosci. 16, 879634 (2022).
Stoodley, C. J. & Tsai, P. T. Adaptive prediction for social contexts: the cerebellar contribution to typical and atypical social behaviors. Annu. Rev. Neurosci. 44, 475–493 (2021).
Fallahnezhad, M. et al. Cerebellar control of a unitary head direction sense. Proc. Natl Acad. Sci. USA 120, e2214539120 (2023).
Chen, C. H. et al. A Purkinje cell to parabrachial nucleus pathway enables broad cerebellar influence over the forebrain. Nat. Neurosci. 26, 1929–1941 (2023).
Frontera, J. L. et al. Bidirectional control of fear memories by cerebellar neurons projecting to the ventrolateral periaqueductal grey. Nat. Commun. 11, 5207 (2020).
Schmahmann, J. D. Disorders of the cerebellum: ataxia, dysmetria of thought, and the cerebellar cognitive affective syndrome. J. Neuropsychiatry Clin. Neurosci. 16, 367–378 (2004).
Uusisaari, M., Obata, K. & Knöpfel, T. Morphological and electrophysiological properties of GABAergic and non-GABAergic cells in the deep cerebellar nuclei. J. Neurophysiol. 97, 901–911 (2007).
Heiney, S. A., Kim, J., Augustine, G. J. & Medina, J. F. Precise control of movement kinematics by optogenetic inhibition of Purkinje cell activity. J. Neurosci. 34, 2321–2330 (2014).
Garcia, K. S. & Mauk, M. D. Pharmacological analysis of cerebellar contributions to the timing and expression of conditioned eyelid responses. Neuropharmacology 37, 471–480 (1998).
Attwell, P. J. E., Ivarsson, M., Millar, L. & Yeo, C. H. Cerebellar mechanisms in eyeblink conditioning. Ann. N. Y. Acad. Sci. 978, 79–92 (2002).
Ebner, T. J. A role for the cerebellum in the control of limb movement velocity. Curr. Opin. Neurobiol. 8, 762–769 (1998).
Fu, Q. G., Suarez, J. I. & Ebner, T. J. Neuronal specification of direction and distance during reaching movements in the superior precentral premotor area and primary motor cortex of monkeys. J. Neurophysiol. 70, 2097–2116 (1993).
Hewitt, A. L., Popa, L. S., Pasalar, S., Hendrix, C. M. & Ebner, T. J. Representation of limb kinematics in Purkinje cell simple spike discharge is conserved across multiple tasks. J. Neurophysiol. 106, 2232–2247 (2011).
Herzfeld, D. J., Kojima, Y., Soetedjo, R. & Shadmehr, R. Encoding of action by the Purkinje cells of the cerebellum. Nature 526, 439–442 (2015). This study invigorates the field of cerebellar cortical coding by identifying a population coding principle in which complex-spike tuning defines populations of Purkinje neurons that, despite heterogeneous individual tuning, as a group are exquisitely tuned to behaviour.
Herzfeld, D. J., Kojima, Y., Soetedjo, R. & Shadmehr, R. Encoding of error and learning to correct that error by the Purkinje cells of the cerebellum. Nat. Neurosci. 21, 736–743 (2018).
Zobeiri, O. A. & Cullen, K. E. Cerebellar Purkinje cells in male macaques combine sensory and motor information to predict the sensory consequences of active self-motion. Nat. Commun. 15, 4003 (2024).
Raghavan, R. T. & Lisberger, S. G. Responses of Purkinje cells in the oculomotor vermis of monkeys during smooth pursuit eye movements and saccades: comparison with floccular complex. J. Neurophysiol. 118, 986–1001 (2017).
Calame, D. J., Becker, M. I. & Person, A. L. Cerebellar associative learning underlies skilled reach adaptation. Nat. Neurosci. 26, 1068–1079 (2023).
Thier, P., Dicke, P. W., Haas, R. & Barash, S. Encoding of movement time by populations of cerebellar Purkinje cells. Nature 405, 72–76 (2000).
Sedaghat-Nejad, E., Pi, J. S., Hage, P., Fakharian, M. A. & Shadmehr, R. Synchronous spiking of cerebellar Purkinje cells during control of movements. Proc. Natl Acad. Sci. USA 119, e2118954119 (2022).
Fakharian, M. A., Shoup, A. M., Hage, P., Elseweifi, H. Y. & Shadmehr, R. A vector calculus for neural computation in the cerebellum. Science 388, 869–875 (2025). This study discovers an organizational logic within cerebellar cortical population coding that implements output null and output potent signalling, suggesting that not all cerebellar activity exerts control.
Hage, P. et al. Control of tongue movements by the Purkinje cells of the cerebellum. PLoS Biol. 23, e3003110 (2025).
Sawtell, N. B. Neural mechanisms for predicting the sensory consequences of behavior: insights from electrosensory systems. Annu. Rev. Physiol. 79, 381–399 (2017).
Wang, X., Novello, M., Gao, Z., Ruigrok, T. J. H. & De Zeeuw, C. I. Input and output organization of the mesodiencephalic junction for cerebro-cerebellar communication. J. Neurosci. Res. 100, 620–637 (2022).
Person, A. L. & Raman, I. M. Purkinje neuron synchrony elicits time-locked spiking in the cerebellar nuclei. Nature 481, 502–505 (2011).
Witter, L., Canto, C. B., Hoogland, T. M., de Gruijl, J. R. & De Zeeuw, C. I. Strength and timing of motor responses mediated by rebound firing in the cerebellar nuclei after Purkinje cell activation. Front. Neural Circuits 7, 133 (2013).
De Zeeuw, C. I. Bidirectional learning in upbound and downbound microzones of the cerebellum. Nat. Rev. Neurosci. 22, 92–110 (2021).
Blot, F. G. C. et al. Purkinje cell microzones mediate distinct kinematics of a single movement. Nat. Commun. 14, 4358 (2023).
Ohmae, S., Ohmae, K., Heiney, S., Subramanian, D. & Medina, J. A recurrent circuit links antagonistic cerebellar modules during associative motor learning. Preprint at bioRxiv https://doi.org/10.1101/2021.11.16.468438 (2021).
Khilkevich, A., Zambrano, J., Richards, M.-M. & Mauk, M. D. Cerebellar implementation of movement sequences through feedback. eLife 7, e37443 (2018).
Geminiani, A. et al. Mesoscale simulations predict the role of synergistic cerebellar plasticity during classical eyeblink conditioning. PLoS Comput. Biol. 20, e1011277 (2024).
De Zeeuw, C. I., Koppen, J., Bregman, G. G., Runge, M. & Narain, D. Heterogeneous encoding of temporal stimuli in the cerebellar cortex. Nat. Commun. 14, 7581 (2023).
Raymond, J. L. & Medina, J. F. Computational principles of supervised learning in the cerebellum. Annu. Rev. Neurosci. 41, 233–253 (2018).
Kozareva, V. et al. A transcriptomic atlas of mouse cerebellar cortex comprehensively defines cell types. Nature 598, 214–219 (2021).
Tang, T. et al. Heterogeneity of Purkinje cell simple spike–complex spike interactions: zebrin- and non-zebrin-related variations. J. Physiol. 595, 5341–5357 (2017).
Mauk, M. D., Steinmetz, J. E. & Thompson, R. F. Classical conditioning using stimulation of the inferior olive as the unconditioned stimulus. Proc. Natl Acad. Sci. USA 83, 5349–5353 (1986).
Steinmetz, J. E., Lavond, D. G., Thompson, R. F. Classical conditioning in rabbits using pontine nucleus stimulation as a conditioned stimulus and inferior olive stimulation as an unconditioned stimulus. Synapse 3, 225–233 (1989).
Silva, N. T., Ramírez-Buriticá, J., Pritchett, D. L. & Carey, M. R. Climbing fibers provide essential instructive signals for associative learning. Nat. Neurosci. 27, 940–951 (2024).
Ito, M. Error detection and representation in the olivo-cerebellar system. Front. Neural Circuits 7, 1 (2013).
Yang, Y. & Lisberger, S. G. Purkinje-cell plasticity and cerebellar motor learning are graded by complex-spike duration. Nature 510, 529–532 (2014).
Medina, J. F. & Lisberger, S. G. Links from complex spikes to local plasticity and motor learning in the cerebellum of awake-behaving monkeys. Nat. Neurosci. 11, 1185–1192 (2008).
Muller, S. Z. et al. Complex spikes perturb movements and reveal the sensorimotor map of Purkinje cells. Curr. Biol. 33, 4869–4879.e3 (2023).
Pi, J. S. et al. The olivary input to the cerebellum dissociates sensory events from movement plans. Proc. Natl Acad. Sci. USA 121, e2318849121 (2024).
Popa, L. S., Streng, M. L., Hewitt, A. L. & Ebner, T. J. The errors of our ways: understanding error representations in cerebellar-dependent motor learning. Cerebellum 15, 93–103 (2016).
Hull, C. Prediction signals in the cerebellum: beyond supervised motor learning. eLife 9, e54073 (2020).
Kitazawa, S., Kimura, T. & Yin, P. B. Cerebellar complex spikes encode both destinations and errors in arm movements. Nature 392, 494–497 (1998).
Streng, M. L., Popa, L. S. & Ebner, T. J. Modulation of sensory prediction error in Purkinje cells during visual feedback manipulations. Nat. Commun. 9, 1099 (2018).
Popa, L. S., Streng, M. L. & Ebner, T. J. Purkinje cell representations of behavior: diary of a busy neuron. Neuroscientist 25, 241–257 (2019). This work presents a fantastic review of the complex field of Purkinje neuron tuning.
Bouvier, G. et al. Cerebellar learning using perturbations. eLife 7, e31599 (2018).
Heffley, W. et al. Coordinated cerebellar climbing fiber activity signals learned sensorimotor predictions. Nat. Neurosci. 21, 1431–1441 (2018).
Bina, L., Romano, V., Hoogland, T. M., Bosman, L. W. J. & De Zeeuw, C. I. Purkinje cells translate subjective salience into readiness to act and choice performance. Cell Rep. 37, 110116 (2021).
Wagner, M. J. et al. A neural circuit state change underlying skilled movements. Cell 184, 3731–3747.e21 (2021).
Kostadinov, D., Beau, M., Pozo, M. B. & Häusser, M. Predictive and reactive reward signals conveyed by climbing fiber inputs to cerebellar Purkinje cells. Nat. Neurosci. 22, 950–962 (2019).
Imamizu, H. & Kawato, M. Neural correlates of predictive and postdictive switching mechanisms for internal models. J. Neurosci. 28, 10751–10765 (2008).
Streng, M. L. et al. Mesoscale Ca2+ imaging reveals networks of Purkinje cell dendritic and somatic modulation, with divergent roles of activity versus correlation during behavior. Preprint at bioRxiv https://doi.org/10.1101/2023.10.05.561090 (2023).
Nguyen, V. & Stell, B. M. Rapid motor adaptation via population-level modulation of cerebellar error signals. Preprint at bioRxiv https://doi.org/10.1101/2024.01.03.574031 (2024).
Ojakangas, C. L. & Ebner, T. J. Purkinje cell complex spike activity during voluntary motor learning: relationship to kinematics. J. Neurophysiol. 72, 2617–2630 (1994).
Mann-Metzer, P. & Yarom, Y. Electrotonic coupling interacts with intrinsic properties to generate synchronized activity in cerebellar networks of inhibitory interneurons. J. Neurosci. 19, 3298–3306 (1999).
Lefler, Y., Yarom, Y. & Uusisaari, M. Y. Cerebellar inhibitory input to the inferior olive decreases electrical coupling and blocks subthreshold oscillations. Neuron 81, 1389–1400 (2014).
Kim, O. A., Ohmae, S. & Medina, J. F. A cerebello-olivary signal for negative prediction error is sufficient to cause extinction of associative motor learning. Nat. Neurosci. 23, 1550–1554 (2020).
Hoang, H. et al. Electrical coupling controls dimensionality and chaotic firing of inferior olive neurons. PLoS Comput. Biol. 16, e1008075 (2020).
Hoang, H. et al. Dynamic organization of cerebellar climbing fiber response and synchrony in multiple functional components reduces dimensions for reinforcement learning. eLife 12, e86340 (2023).
Van Der Giessen, R. S. et al. Role of olivary electrical coupling in cerebellar motor learning. Neuron 58, 599–612 (2008).
Marr, D. A theory of cerebellar cortex. J. Physiol. 202, 437–470 (1969).
Ito, M. Cerebellar control of the vestibulo-ocular reflex—around the flocculus hypothesis. Annu. Rev. Neurosci. 5, 275–296 (1982).
Nguyen-Vu, T. D. B. et al. Cerebellar Purkinje cell activity drives motor learning. Nat. Neurosci. 16, 1734–1736 (2013).
Kimpo, R. R., Rinaldi, J. M., Kim, C. K., Payne, H. L. & Raymond, J. L. Gating of neural error signals during motor learning. eLife 3, e02076 (2014).
Rowan, M. J. M. et al. Graded control of climbing-fiber-mediated plasticity and learning by inhibition in the cerebellum. Neuron 99, 999–1015.e6 (2018).
Jörntell, H. & Hansel, C. Synaptic memories upside down: bidirectional plasticity at cerebellar parallel fiber–Purkinje cell synapses. Neuron 52, 227–238 (2006).
Sejnowski, T. J. Storing covariance with nonlinearly interacting neurons. J. Math. Biol. 4, 303–321 (1977).
Suvrathan, A., Payne, H. L. & Raymond, J. L. Timing rules for synaptic plasticity matched to behavioral function. Neuron 92, 959–967 (2016).
Jayabal, S. et al. Experience alters the timing rules governing synaptic plasticity and learning. Preprint at bioRxiv https://doi.org/10.1101/2022.11.28.518128 (2024).
Lang, E. J., Sugihara, I., Welsh, J. P. & Llinás, R. Patterns of spontaneous Purkinje cell complex spike activity in the awake rat. J. Neurosci. 19, 2728–2739 (1999).
Medina, J. F., Nores, W. L. & Mauk, M. D. Inhibition of climbing fibres is a signal for the extinction of conditioned eyelid responses. Nature 416, 330–333 (2002).
Mauk, M. D. & Donegan, N. H. A model of Pavlovian eyelid conditioning based on the synaptic organization of the cerebellum. Learn. Mem. 4, 130–158 (1997).
Zhang, K. et al. Molecular layer disinhibition unlocks climbing-fiber-instructed motor learning in the cerebellum. Preprint at bioRxiv https://doi.org/10.1101/2023.08.04.552059 (2023).
Bonnan, A., Zhang, K., Gaffield, M. A. & Christie, J. M. Expression of a form of cerebellar motor memory requires learned alterations to the activity of inhibitory molecular layer interneurons. J. Neurosci. 43, 601–612 (2023).
Gaffield, M. A., Rowan, M. J. M., Amat, S. B., Hirai, H. & Christie, J. M. Inhibition gates supralinear Ca2+ signaling in Purkinje cell dendrites during practiced movements. eLife 7, e36246 (2018).
Lackey, E. P. et al. Specialized connectivity of molecular layer interneuron subtypes leads to disinhibition and synchronous inhibition of cerebellar Purkinje cells. Neuron 112, 2333–2348.e6 (2024).
Blot, A. & Barbour, B. Ultra-rapid axon–axon ephaptic inhibition of cerebellar Purkinje cells by the pinceau. Nat. Neurosci. 17, 289–295 (2014).
Mittmann, W., Koch, U. & Häusser, M. Feed-forward inhibition shapes the spike output of cerebellar Purkinje cells. J. Physiol. 563, 369–378 (2005).
Kennedy, A. et al. A temporal basis for predicting the sensory consequences of motor commands in an electric fish. Nat. Neurosci. 17, 416–422 (2014).
Narain, D., Remington, E. D., Zeeuw, C. I. D. & Jazayeri, M. A cerebellar mechanism for learning prior distributions of time intervals. Nat. Commun. 9, 469 (2018).
Wagner, M. J., Kim, T. H., Savall, J., Schnitzer, M. J. & Luo, L. Cerebellar granule cells encode the expectation of reward. Nature 544, 96–100 (2017).
Giovannucci, A. et al. Cerebellar granule cells acquire a widespread predictive feedback signal during motor learning. Nat. Neurosci. 20, 727–734 (2017).
Knogler, L. D., Markov, D. A., Dragomir, E. I., Štih, V. & Portugues, R. Sensorimotor representations in cerebellar granule cells in larval zebrafish are dense, spatially organized, and non-temporally patterned. Curr. Biol. 27, 1288–1302 (2017).
Cayco-Gajic, N. A., Clopath, C. & Silver, R. A. Sparse synaptic connectivity is required for decorrelation and pattern separation in feedforward networks. Nat. Commun. 8, 1116 (2017).
Litwin-Kumar, A., Harris, K. D., Axel, R., Sompolinsky, H. & Abbott, L. F. Optimal degrees of synaptic connectivity. Neuron 93, 1153–1164.e7 (2017).
Lanore, F., Cayco-Gajic, N. A., Gurnani, H., Coyle, D. & Silver, R. A. Cerebellar granule cell axons support high-dimensional representations. Nat. Neurosci. 24, 1142–1150 (2021).
Garcia-Garcia, M. G. et al. A cerebellar granule cell-climbing fiber computation to learn to track long time intervals. Neuron 112, 2749–2764.e7 (2024).
Fleming, E. A., Field, G. D., Tadross, M. R. & Hull, C. Local synaptic inhibition mediates cerebellar granule cell pattern separation and enables learned sensorimotor associations. Nat. Neurosci. 27, 689–701 (2024).
Thoroughman, K. A. & Shadmehr, R. Learning of action through adaptive combination of motor primitives. Nature 407, 742–747 (2000).
Huang, C.-C. et al. Convergence of pontine and proprioceptive streams onto multimodal cerebellar granule cells. eLife 2, e00400 (2013).
Nguyen, T. M. et al. Structured cerebellar connectivity supports resilient pattern separation. Nature 613, 543–549 (2023).
Bengtsson, F. & Jörntell, H. Sensory transmission in cerebellar granule cells relies on similarly coded mossy fiber inputs. Proc. Natl Acad. Sci. USA 106, 2389–2394 (2009).
Jörntell, H. & Ekerot, C.-F. Properties of somatosensory synaptic integration in cerebellar granule cells in vivo. J. Neurosci. 26, 11786–11797 (2006).
Rancz, E. A. et al. High-fidelity transmission of sensory information by single cerebellar mossy fibre boutons. Nature 450, 1245–1248 (2007).
Billings, G., Piasini, E., Lőrincz, A., Nusser, Z. & Silver, R. A. Network structure within the cerebellar input layer enables lossless sparse encoding. Neuron 83, 960–974 (2014).
Chabrol, F. P., Arenz, A., Wiechert, M. T., Margrie, T. W. & DiGregorio, D. A. Synaptic diversity enables temporal coding of coincident multisensory inputs in single neurons. Nat. Neurosci. 18, 718–727 (2015).
Fujita, M. Adaptive filter model of the cerebellum. Biol. Cybern. 45, 195–206 (1982).
Gilmer, J. I. et al. An emergent temporal basis set robustly supports cerebellar time-series learning. J. Neurophysiol. 129, 159–176 (2023).
Buonomano, D. V. & Maass, W. State-dependent computations: spatiotemporal processing in cortical networks. Nat. Rev. Neurosci. 10, 113–125 (2009).
Barri, A., Wiechert, M. T., Jazayeri, M. & DiGregorio, D. A. Synaptic basis of a sub-second representation of time in a neural circuit model. Nat. Commun. 13, 7902 (2022).
Straub, I. et al. Gradients in the mammalian cerebellar cortex enable fourier-like transformation and improve storing capacity. eLife 9, e51771 (2020).
Huson, V. & Regehr, W. G. Realistic mossy fiber input patterns to unipolar brush cells evoke a continuum of temporal responses comprised of components mediated by different glutamate receptors. eLife 13, RP102618 (2025).
Herzfeld, D. J. et al. Contributions of the cerebellum and the motor cortex to acquisition and retention of motor memories. NeuroImage 98, 147–158 (2014).
Xu-Wilson, M., Chen-Harris, H., Zee, D. S. & Shadmehr, R. Cerebellar contributions to adaptive control of saccades in humans. J. Neurosci. 29, 12930–12939 (2009).
Miall, R. C., Weir, D. J., Wolpert, D. M. & Stein, J. F. Is the cerebellum a Smith predictor? J. Mot. Behav. 25, 203–216 (1993).
Miall, R. C., Christensen, L. O. D., Cain, O. & Stanley, J. Disruption of state estimation in the human lateral cerebellum. PLoS Biol. 5, e316 (2007).
Pisella, L. et al. An ‘automatic pilot’ for the hand in human posterior parietal cortex: toward reinterpreting optic ataxia. Nat. Neurosci. 3, 729–736 (2000).
Desmurget, M. et al. Role of the posterior parietal cortex in updating reaching movements to a visual target. Nat. Neurosci. 2, 563–567 (1999).
Vilis, T., Hore, J., Meyer-Lohmann, J. & Brooks, V. B. Dual nature of the precentral responses to limb perturbations revealed by cerebellar cooling. Brain Res. 117, 336–340 (1976).
Bollu, T. et al. Cortex-dependent corrections as the tongue reaches for and misses targets. Nature 594, 82–87 (2021).
Bollu, T. et al. Motor cortical inactivation impairs corrective submovements in mice performing a hold-still center-out reach task. J. Neurophysiol. 132, 829–848 (2024).
Pruszynski, J. A. et al. Primary motor cortex underlies multi-joint integration for fast feedback control. Nature 478, 387–390 (2011).
Desmurget, M. & Grafton, S. Forward modeling allows feedback control for fast reaching movements. Trends Cogn. Sci. 4, 423–431 (2000).
Schneider, D. M., Sundararajan, J. & Mooney, R. A cortical filter that learns to suppress the acoustic consequences of movement. Nature 561, 391–395 (2018).
Holey, B. E. & Schneider, D. M. Sensation and expectation are embedded in mouse motor cortical activity. Cell Rep. 43, 114396 (2024).
Mathis, M. W., Mathis, A. & Uchida, N. Somatosensory cortex plays an essential role in forelimb motor adaptation in mice. Neuron 93, 1493–1503.e6 (2017).
Zimmet, A. M., Cao, D., Bastian, A. J. & Cowan, N. J. Cerebellar patients have intact feedback control that can be leveraged to improve reaching. eLife 9, e53246 (2020).
Wagner, M. J. et al. Shared cortex–cerebellum dynamics in the execution and learning of a motor task. Cell 177, 669–682.e24 (2019).
Diedrichsen, J., Hashambhoy, Y., Rane, T. & Shadmehr, R. Neural correlates of reach errors. J. Neurosci. 25, 9919–9931 (2005).
Todorov, E. & Jordan, M. I. Optimal feedback control as a theory of motor coordination. Nat. Neurosci. 5, 1226–1235 (2002).
Sakaguchi, Y., Tanaka, M. & Inoue, Y. Adaptive intermittent control: a computational model explaining motor intermittency observed in human behavior. Neural Netw. 67, 92–109 (2015).
Soetedjo, R. & Horwitz, G. D. Closed-loop optogenetic perturbation of macaque oculomotor cerebellum: evidence for an internal saccade model. J. Neurosci. 44, e1317232023 (2024).
Low, A. Y. T. et al. Reverse-translational identification of a cerebellar satiation network. Nature 600, 269–273 (2021).
National Aeronautics and Space Administration. Mars exploration rovers: Spirit and Opportunity. NASA Science https://science.nasa.gov/mission/mars-exploration-rovers-spirit-and-opportunity/ (2024).
Azim, E. & Alstermark, B. Skilled forelimb movements and internal copy motor circuits. Curr. Opin. Neurobiol. 33, 16–24 (2015).
Acknowledgements
The authors thank members of the Person laboratory and our three reviewers for their thoughtful and constructive suggestions. We also thank A. Haith for insightful conversations about motor policy learning. The authors acknowledge support to K.P.N. (NS134561) and A.L.P. (NS114430 and NS131839).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neuroscience thanks Michael Mauk and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Adaptive filter
-
A neural circuit computation that adapts its outputs to changing inputs, achieving flexible goals.
- Associative learning
-
A learning process that links together two stimuli. This term is often used in context of Pavlovian processes in which one stimulus predicts the occurrence of a second.
- Cerebellar ataxia
-
A suite of motor discoordination profiles characteristic of cerebellar dysfunction.
- Cerebellum proper
-
The cerebellum as distinct from cerebellar-like structures such as the dorsal cochlear nucleus or electrosensory lobule of the electric fish. The cerebellum proper is unique in that it possess climbing fibre inputs.
- Classical conditioning
-
A Pavlovian learning paradigm in which a neutral ‘conditioning’ stimulus (CS) is repeatedly paired with a reflex-inducing ‘unconditioned’ stimulus (US). Animals learn to associate the CS with the US through the process of associative learning.
- Complex spike
-
A burst of action potentials generated by Purkinje cells (PCs) in response to climbing fibre inputs. We use the term here to include dendritic Ca2+ events driven by climbing fibre inputs to PCs, even though these signals are mechanistically distinct from the spikelets emitted by the soma.
- Control theory
-
A field of engineering that formalizes processes for generating a control variable to behave in a desired way by using inputs in various ways. These inputs can be feedback or copies of output commands, relevant examples for biological versions of controllers.
- Efference copy
-
A copy of a motor command being sent to the periphery.
- Eligibility trace
-
A hypothesized molecular mechanism that renders a synapse eligible for synaptic plasticity. Eligibility traces are thought to be labile in time and can be specific to individual synapses.
- Internal models
-
Hypothesized frameworks in which the brain generates world-based and body-based models used for functions as diverse as motor control and social cognition.
- Long-term depression
-
Use-dependent weakening of synaptic strength.
- Long-term potentiation
-
Use-dependent strengthening of synaptic strength.
- Lookup table
-
An engineering term depicting a discrete mapping of an input onto an output, as in a table indexed by rows and columns.
- Microzone
-
A small cerebellar module defined by interconnected olivary climbing fibre projections to a subset of Purkinje cells (PCs), their convergent targets in the cerebellar nuclei and their projections back to the same olivary region.
- Population coding
-
The principle that neurons encode information as ensembles with temporally evolving dynamics rather than individually.
- Saccades
-
Brief, nearly ballistic eye movements from one target to another.
- Simple spikes
-
An action potential type in Purkinje cells (PCs). Simple spikes are the typical type of sodium spike that propagates down the axon. They are in contrast to complex spikes, which have distinct characteristics.
- Supervised learning
-
A machine learning perspective in which networks are trained through labelled instructive signals.
- Temporal difference learning
-
A reinforcement learning algorithm that estimates future outcomes based on the difference between predicted and actual outcomes using intermediate cues. In the case of cerebellar learning, this refers to a condition stimulus becoming instructive through learned representation in the climbing fibre pathway.
- Trace eyelid conditioning
-
A variation on a classical conditioning paradigm in which the end of the conditioned stimulus (CS) is separated in time from the unconditioned stimulus (US). By contrast, in delay eyelid conditioning (DEC) the CS and US co-occur.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nguyen, K.P., Person, A.L. Cerebellar circuit computations for predictive motor control. Nat. Rev. Neurosci. 26, 538–553 (2025). https://doi.org/10.1038/s41583-025-00936-z
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41583-025-00936-z
This article is cited by
-
Consensus Paper: Models of Cerebellar Functions
The Cerebellum (2026)
-
Stage-dependent cerebrocerebellar communication during sensorimotor processing
Nature Communications (2025)
-
Commentary on the Flocculonodular Lobe as a Cerebellar Resilience Zone: Could Exercise Really be the Best Medicine?
The Cerebellum (2025)