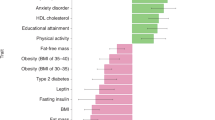
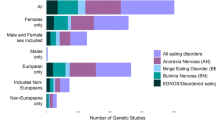
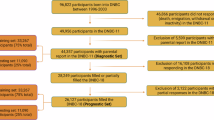
Abstract
Despite their prevalence, eating disorders (EDs) are under-researched and often misunderstood. A recent focus of research on the biological underpinnings of EDs has helped to reframe our understanding of their origins, but there remain a lack of effective treatment options, high rates of relapse and, unfortunately, high mortality and morbidity. In this Review, we highlight the many facets of normal and pathological feeding behaviour and body weight regulation and suggest that these provide a framework with which to develop integrative methods to study, and ultimately treat, EDs. We propose that a better understanding of the biological causes of ED, and their crucial interactions with psychological and environmental factors, is necessary to progress the field. This can be achieved through a combination of preclinical and clinical investigations, which provide complementary information on these highly complex disorders. In the era of individualized medicine and with the advent of artificial intelligence tools that allow the amalgamation of multimodal data, we hope that a better understanding of the biology of EDs may hold the answer to effectively overcoming the debilitating effects of these conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galmiche, M., Déchelotte, P., Lambert, G. & Tavolacci, M. P. Prevalence of eating disorders over the 2000–2018 period: a systematic literature review. Am. J. Clin. Nutr. 109, 1402–1413 (2019).
Watson, H. J. et al. Genome-wide association study identifies eight risk loci and implicates metabo-psychiatric origins for anorexia nervosa. Nat. Genet. 51, 1207–1214 (2019). This seminal study identified genetic loci associated with anorexia nervosa, revealing significant correlations with both psychiatric and metabolic traits, thereby redefining the disorder as a metabo-psychiatric condition and highlighting the need for integrated treatment approaches.
Burstein, D. et al. Genome-wide analysis of a model-derived binge eating disorder phenotype identifies risk loci and implicates iron metabolism. Nat. Genet. 55, 1462–1470 (2023).
Hay, P. et al. Epidemiology of eating disorders: population, prevalence, disease burden and quality of life informing public policy in Australia — a rapid review. J. Eat. Disord. 11, 23 (2023).
Corsica, J. A. & Hood, M. M. Eating disorders in an obesogenic environment. J. Am. Diet. Assoc. 111, 996–1000 (2011).
Tsofliou, F., Casey, C. & Hughes, C. Eating Disorders (eds Patel, V. & Preedy, V.) 1–23 (Springer International Publishing, 2021).
Hardaway, J. A., Crowley, N. A., Bulik, C. M. & Kash, T. L. Integrated circuits and molecular components for stress and feeding: implications for eating disorders. Genes Brain Behav. 14, 85–97 (2015).
Bulik, C. M., Carroll, I. M. & Mehler, P. Reframing anorexia nervosa as a metabo-psychiatric disorder. Trends Endocrinol. Metab. 32, 752–761 (2021).
Hübel, C. et al. One size does not fit all. Genomics differentiates among anorexia nervosa, bulimia nervosa, and binge-eating disorder. Int. J. Eat. Disord. 54, 785–793 (2021).
Treasure, J., Stein, D. & Maguire, S. Has the time come for a staging model to map the course of eating disorders from high risk to severe enduring illness? An examination of the evidence. Early Interv. Psychiatry 9, 173–184 (2015).
Hambleton, A. et al. Psychiatric and medical comorbidities of eating disorders: findings from a rapid review of the literature. J. Eat. Disord. 10, 132 (2022).
Kass, A. E., Kolko, R. P. & Wilfley, D. E. Psychological treatments for eating disorders. Curr. Opin. Psychiatry 26, 549–555 (2013).
Sadybekov, A. V. & Katritch, V. Computational approaches streamlining drug discovery. Nature 616, 673–685 (2023).
Raison, C. L. et al. Single-dose psilocybin treatment for major depressive disorder: a randomized clinical trial. JAMA 330, 843–853 (2023).
Himmerich, H. & Treasure, J. Psychopharmacological advances in eating disorders. Expert Rev. Clin. Pharmacol. 11, 95–108 (2018).
Steward, T. Endocrinology-informed neuroimaging in eating disorders: GLP1, orexins, and psilocybin. Trends Mol. Med. 30, 321–323 (2024).
Bulik, C. M. et al. Genetics and neurobiology of eating disorders. Nat. Neurosci. 25, 543–554 (2022).
Frank, G. K. W., Shott, M. E. & DeGuzman, M. C. The neurobiology of eating disorders. Child Adolesc. Psychiatr. Clin. N. Am. 28, 629–640 (2019).
Huckins, L. M. et al. What next for eating disorder genetics? Replacing myths with facts to sharpen our understanding. Mol. Psychiatry 27, 3929–3938 (2022).
Hamamoto, Y. et al. Neural mechanisms of perceptual and affective body-image disturbance during own-body and ideal-body estimation. Behav. Brain Res. 444, 114349 (2023).
Bailer, U. F. et al. Altered 5-HT(2A) receptor binding after recovery from bulimia-type anorexia nervosa: relationships to harm avoidance and drive for thinness. Neuropsychopharmacology 29, 1143–1155 (2004).
Campos, A., Port, J. D. & Acosta, A. Integrative hedonic and homeostatic food intake regulation by the central nervous system: insights from neuroimaging. Brain Sci. 12, 431 (2022).
Higgs, S. Social norms and their influence on eating behaviours. Appetite 86, 38–44 (2015).
Sanzari, C. M. et al. The impact of social media use on body image and disordered eating behaviors: content matters more than duration of exposure. Eat. Behav. 49, 101722 (2023).
Akhlaghi, M. & Kohanmoo, A. Sleep deprivation in development of obesity, effects on appetite regulation, energy metabolism, and dietary choices. Nutr. Res. Rev. 38, 4–24 (2025).
Kuckuck, S. et al. Glucocorticoids, stress and eating: the mediating role of appetite-regulating hormones. Obes. Rev. 24, e13539 (2023).
Berner, L. A. et al. Neuroendocrinology of reward in anorexia nervosa and bulimia nervosa: beyond leptin and ghrelin. Mol. Cell. Endocrinol. 497, 110320 (2019). This paper emphasizes how neuroendocrine disruptions in reward pathways, beyond leptin and ghrelin, contribute to anorexia and bulimia nervosa, pointing to novel hormonal treatment targets.
Alcantara, I. C., Tapia, A. P. M., Aponte, Y. & Krashes, M. J. Acts of appetite: neural circuits governing the appetitive, consummatory, and terminating phases of feeding. Nat. Metab. 4, 836–847 (2022).
Stuber, G. D. & Wise, R. A. Lateral hypothalamic circuits for feeding and reward. Nat. Neurosci. 19, 198–205 (2016).
Tellez, L. A. et al. Separate circuitries encode the hedonic and nutritional values of sugar. Nat. Neurosci. 19, 465–470 (2016).
Avena, N. M., Rada, P. & Hoebel, B. G. Evidence for sugar addiction: behavioral and neurochemical effects of intermittent, excessive sugar intake. Neurosci. Biobehav. Rev. 32, 20–39 (2008).
Thomas, J. M. et al. Satiation attenuates BOLD activity in brain regions involved in reward and increases activity in dorsolateral prefrontal cortex: an fMRI study in healthy volunteers. Am. J. Clin. Nutr. 101, 701–708 (2015).
Higgs, S. et al. Interactions between metabolic, reward and cognitive processes in appetite control: implications for novel weight management therapies. J. Psychopharmacol. 31, 1460–1474 (2017).
Mehr, J. B., Mitchison, D., Bowrey, H. E. & James, M. H. Sleep dysregulation in binge eating disorder and ‘food addiction’: the orexin (hypocretin) system as a potential neurobiological link. Neuropsychopharmacology 46, 2051–2061 (2021).
O’Hara, C. B., Campbell, I. C. & Schmidt, U. A reward-centred model of anorexia nervosa: a focussed narrative review of the neurological and psychophysiological literature. Neurosci. Biobehav. Rev. 52, 131–152 (2015).
Bronleigh, M., Baumann, O. & Stapleton, P. Neural correlates associated with processing food stimuli in anorexia nervosa and bulimia nervosa: an activation likelihood estimation meta-analysis of fMRI studies. Eat. Weight. Disord. 27, 2309–2320 (2022).
Kaye, W. H. et al. Neural insensitivity to the effects of hunger in women remitted from anorexia nervosa. Am. J. Psychiatry 177, 601–610 (2020).
Eddy, K. T. et al. Neural activation of regions involved in food reward and cognitive control in young females with anorexia nervosa and atypical anorexia nervosa versus healthy controls. Transl. Psychiatry 13, 220 (2023).
Santel, S., Baving, L., Krauel, K., Münte, T. F. & Rotte, M. Hunger and satiety in anorexia nervosa: fMRI during cognitive processing of food pictures. Brain Res. 1114, 138–148 (2006).
Wierenga, C. E. et al. Hunger does not motivate reward in women remitted from anorexia nervosa. Biol. Psychiatry 77, 642–652 (2015).
Lindstedt, K. et al. How do your genes feel? Subjective experience of anorexia nervosa in patients with high vs. low polygenic risk. Psychatr. Genet. https://doi.org/10.1097/YPG.0000000000000395 (2025).
Simon, J. J. et al. Neuroimaging of hypothalamic mechanisms related to glucose metabolism in anorexia nervosa and obesity. J. Clin. Invest. 130, 4094–4103 (2020). This elegantly designed study revealed blunted hypothalamic responses to glucose and disrupted connectivity with reward-related brain regions in anorexia nervosa, suggesting that impaired central glucose sensing may contribute to the pathophysiology of the disorder.
Khalsa, S. S. et al. Altered interoceptive awareness in anorexia nervosa: effects of meal anticipation, consumption and bodily arousal. Int. J. Eat. Disord. 48, 889–897 (2015).
Walton, E. et al. Brain structure in acutely underweight and partially weight-restored individuals with anorexia nervosa: a coordinated analysis by the ENIGMA eating disorders working group. Biol. Psychiatry 92, 730–738 (2022).
Blanke, O., Slater, M. & Serino, A. Behavioral, neural, and computational principles of bodily self-consciousness. Neuron 88, 145–166 (2015).
Corbetta, M. & Shulman, G. L. Control of goal-directed and stimulus-driven attention in the brain. Nat. Rev. Neurosci. 3, 201–215 (2002).
Balodis, I. M. et al. Divergent neural substrates of inhibitory control in binge eating disorder relative to other manifestations of obesity. Obesity 21, 367–377 (2013).
Romei, A., Voigt, K. & Verdejo-Garcia, A. A perspective on candidate neural underpinnings of binge eating disorder: reward and homeostatic systems. Curr. Pharm. Des. 26, 2327–2333 (2020).
Schienle, A., Schäfer, A., Hermann, A. & Vaitl, D. Binge-eating disorder: reward sensitivity and brain activation to images of food. Biol. Psychiatry 65, 654–661 (2009).
Weygandt, M., Schaefer, A., Schienle, A. & Haynes, J. D. Diagnosing different binge‐eating disorders based on reward‐related brain activation patterns. Hum. Brain Mapp. 33, 2135–2146 (2012).
Giel, K. E., Teufel, M., Junne, F., Zipfel, S. & Schag, K. Food-related impulsivity in obesity and binge eating disorder — a systematic update of the evidence. Nutrients 9, 1170 (2017).
Leehr, E. J. et al. Emotion regulation model in binge eating disorder and obesity — a systematic review. Neurosci. Biobehav. Rev. 49, 125–134 (2015).
Leenaerts, N., Jongen, D., Ceccarini, J., Van Oudenhove, L. & Vrieze, E. The neurobiological reward system and binge eating: a critical systematic review of neuroimaging studies. Int. J. Eat. Disord. 55, 1421–1458 (2022).
Schag, K. et al. Impulsivity in binge eating disorder: food cues elicit increased reward responses and disinhibition. PLoS ONE 8, e76542 (2013).
Hartogsveld, B., Quaedflieg, C. W., van Ruitenbeek, P. & Smeets, T. Volume and connectivity differences in brain networks associated with cognitive constructs of binge eating. eNeuro 9, ENEURO.0080-21.2021 (2022).
Chen, X., Li, W., Liu, Y., Xiao, M. & Chen, H. Altered effective connectivity between reward and inhibitory control networks in people with binge eating episodes: a spectral dynamic causal modeling study. Appetite 188, 106763 (2023).
Monteleone, P. et al. Circulating ghrelin is decreased in non-obese and obese women with binge eating disorder as well as in obese non-binge eating women, but not in patients with bulimia nervosa. Psychoneuroendocrinology 30, 243–250 (2005).
Troisi, A. et al. Plasma ghrelin in anorexia, bulimia, and binge-eating disorder: relations with eating patterns and circulating concentrations of cortisol and thyroid hormones. Neuroendocrinology 81, 259–266 (2005).
Hernandez, D., Mehta, N. & Geliebter, A. Meal‐related acyl and des‐acyl ghrelin and other appetite‐related hormones in people with obesity and binge eating. Obesity 27, 629–635 (2019).
Geliebter, A., Hashim, S. A. & Gluck, M. E. Appetite-related gut peptides, ghrelin, PYY, and GLP-1 in obese women with and without binge eating disorder (BED). Physiol. Behav. 94, 696–699 (2008).
Schalla, M. A. & Stengel, A. The role of ghrelin in anorexia nervosa. Int. J. Mol. Sci. 19, 2117 (2018).
Mancuso, C. et al. Changes in appetite-regulating hormones following food intake are associated with changes in reported appetite and a measure of hedonic eating in girls and young women with anorexia nervosa. Psychoneuroendocrinology 113, 104556 (2020).
Seidel, M. et al. A systematic review and meta-analysis finds increased blood levels of all forms of ghrelin in both restricting and binge-eating/purging subtypes of anorexia nervosa. Nutrients 13, 709 (2021).
Muhammed, M. et al. Gut-derived appetite regulating hormones across the anorexia nervosa spectrum. Psychoneuroendocrinology 172, 107257 (2025).
Berridge, K. C. ‘Liking’ and ‘wanting’ food rewards: brain substrates and roles in eating disorders. Physiol. Behav. 97, 537–550 (2009).
Davis, C. et al. Binge eating disorder and the dopamine D2 receptor: genotypes and sub-phenotypes. Prog. Neuro-psychopharmacol. Biol. Psychiatry 38, 328–335 (2012).
Kontis, D. & Theochari, E. Dopamine in anorexia nervosa: a systematic review. Behav. Pharmacol. 23, 496–515 (2012).
Steward, T. et al. Reduced plasma orexin — a concentrations are associated with cognitive deficits in anorexia nervosa. Sci. Rep. 9, 7910 (2019).
Bryant, E., Aouad, P., Hambleton, A., Touyz, S. & Maguire, S. ‘In an otherwise limitless world, I was sure of my limit’. Experiencing anorexia nervosa: a phenomenological metasynthesis. Front. Psychiatry 13, 894178 (2022).
Hill, M. N. & Lee, F. S. Endocannabinoids and stress resilience: is deficiency sufficient to promote vulnerability? Biol. Psychiatry 79, 792–793 (2016).
Wenzel, J. M. & Cheer, J. F. Endocannabinoid regulation of reward and reinforcement through interaction with dopamine and endogenous opioid signaling. Neuropsychopharmacology 43, 103–115 (2018).
Kleiman, S. C. et al. The intestinal microbiota in acute anorexia nervosa and during renourishment: relationship to depression, anxiety, and eating disorder psychopathology. Psychosom. Med. 77, 969–981 (2015).
Leyrolle, Q. et al. Specific gut microbial, biological, and psychiatric profiling related to binge eating disorders: a cross-sectional study in obese patients. Clin. Nutr. 40, 2035–2044 (2021).
Fan, Y. et al. The gut microbiota contributes to the pathogenesis of anorexia nervosa in humans and mice. Nat. Microbiol. 8, 787–802 (2023). This paper establishes a causal role for altered gut microbiota in anorexia nervosa pathogenesis via human-to-mouse microbiome transfers, highlighting the microbiome as a novel therapeutic target.
Seitz, J., Trinh, S. & Herpertz-Dahlmann, B. The microbiome and eating disorders. Psychiatr. Clin. 42, 93–103 (2019).
Terry, S. M., Barnett, J. A. & Gibson, D. L. A critical analysis of eating disorders and the gut microbiome. J. Eat. Disord. 10, 154 (2022).
Saunders, J. & Smith, T. Malnutrition: causes and consequences. Clin. Med. 10, 624 (2010).
Gauthier, C. et al. Symptoms of depression and anxiety in anorexia nervosa: links with plasma tryptophan and serotonin metabolism. Psychoneuroendocrinology 39, 170–178 (2014).
Morales, F., Montserrat-de la Paz, S., Leon, M. J. & Rivero-Pino, F. Effects of malnutrition on the immune system and infection and the role of nutritional strategies regarding Improvements in Children’s Health Status: a literature review. Nutrients 16, 1 (2023).
Seitz, J., Herpertz-Dahlmann, B. & Konrad, K. Brain morphological changes in adolescent and adult patients with anorexia nervosa. J. Neural Transm. 123, 949–959 (2016).
Wronski, M.-L. et al. Dynamic amygdala nuclei alterations in relation to weight status in anorexia nervosa are mediated by leptin. J. Am. Acad. Child Adolesc. Psychiatry 63, 624–639 (2024).
Hanachi, M. et al. Micronutrients deficiencies in 374 severely malnourished anorexia nervosa inpatients. Nutrients 11, 792 (2019).
Astrup, A. & Bügel, S. Overfed but undernourished: recognizing nutritional inadequacies/deficiencies in patients with overweight or obesity. Int. J. Obes. 43, 219–232 (2019).
Arold, D. et al. Predicting long-term outcome in anorexia nervosa: a machine learning analysis of brain structure at different stages of weight recovery. Psychol. Med. 53, 7827–7836 (2023).
Mack, I. et al. Weight gain in anorexia nervosa does not ameliorate the faecal microbiota, branched chain fatty acid profiles and gastrointestinal complaints. Sci. Rep. 6, 26752 (2016).
Leinninger, G. M. et al. Leptin acts via leptin receptor-expressing lateral hypothalamic neurons to modulate the mesolimbic dopamine system and suppress feeding. Cell Metab. 10, 89–98 (2009).
Cassioli, E. et al. Reward and psychopathological correlates of eating disorders: the explanatory role of leptin. Psychiatry Res. 290, 113071 (2020).
McCuen-Wurst, C., Ruggieri, M. & Allison, K. C. Disordered eating and obesity: associations between binge-eating disorder, night-eating syndrome, and weight-related comorbidities. Ann. N. Y. Acad. Sci. 1411, 96–105 (2018).
Lowe, C. J., Morton, J. B. & Reichelt, A. C. Adolescent obesity and dietary decision making — a brain-health perspective. Lancet Child Adolesc. Health 4, 388–396 (2020).
Huerta-Canseco, C., Caba, M. & Camacho-Morales, A. Obesity-mediated lipoinflammation modulates food reward responses. Neuroscience 529, 37–53 (2023).
Ehrlich, S. et al. The role of leptin and cortisol in hyperactivity in patients with acute and weight-recovered anorexia nervosa. Prog. Neuro-psychopharmacol. Biol. Psychiatry 33, 658–662 (2009).
Steiger, H. & Thaler, L. Eating disorders, gene–environment interactions and the epigenome: roles of stress exposures and nutritional status. Physiol. Behav. 162, 181–185 (2016).
Swinbourne, J. et al. The comorbidity between eating disorders and anxiety disorders: prevalence in an eating disorder sample and anxiety disorder sample. Aust. N. Zeal. J. Psychiatry 46, 118–131 (2012).
Fiksdal, A. et al. Associations between symptoms of depression and anxiety and cortisol responses to and recovery from acute stress. Psychoneuroendocrinology 102, 44–52 (2019).
Robert, M. et al. Resilience is associated with less eating disorder symptoms in the NutriNet-Santé cohort study. Int. J. Environ. Res. Public Health 19, 1471 (2022).
Hill, D. C., Moss, R. H., Sykes-Muskett, B., Conner, M. & O’Connor, D. B. Stress and eating behaviors in children and adolescents: systematic review and meta-analysis. Appetite 123, 14–22 (2018).
Henderson, K. A. et al. Coping in adolescents: a mediator between stress and disordered eating. Eat. Behav. 47, 101626 (2022).
Leal, A. & Moreira, A. C. Food and the circadian activity of the hypothalamic–pituitary–adrenal axis. Braz. J. Med. Biol. Res. 30, 1391–1405 (1997).
O’Connor, E. C. et al. Accumbal D1R neurons projecting to lateral hypothalamus authorize feeding. Neuron 88, 553–564 (2015).
Ip, C. K. et al. Critical role of lateral habenula circuits in the control of stress-induced palatable food consumption. Neuron 111, 2583–2600.e6 (2023). This study identifies the lateral habenula as a key brain region regulating stress-induced overeating of palatable foods, revealing a novel circuit-level mechanism linking stress and reward-driven eating.
Rosenberg, N. et al. Cortisol response and desire to binge following psychological stress: comparison between obese subjects with and without binge eating disorder. Psychiatry Res. 208, 156–161 (2013).
Gluck, M. E., Geliebter, A., Hung, J. & Yahav, E. Cortisol, hunger, and desire to binge eat following a cold stress test in obese women with binge eating disorder. Psychosom. Med. 66, 876–881 (2004).
Smith, K. E. et al. Dynamic stress responses and real-time symptoms in binge-eating disorder. Ann. Behav. Med. 55, 758–768 (2021).
Williamson, G., Cotter, E. W., Cao, L., Guidinger, C. & Kelly, N. R. Ecological momentary assessment of state affect prior to and following loss of control eating in young men. Eat. Behav. 50, 101782 (2023).
Tamres, L. K., Janicki, D. & Helgeson, V. S. Sex differences in coping behavior: a meta-analytic review and an examination of relative coping. Pers. Soc. Psychol. Rev. 6, 2–30 (2002).
Lo Sauro, C., Ravaldi, C., Cabras, P. L., Faravelli, C. & Ricca, V. Stress, hypothalamic–pituitary–adrenal axis and eating disorders. Neuropsychobiology 57, 95–115 (2008).
Schmalbach, I. et al. Cortisol reactivity in patients with anorexia nervosa after stress induction. Transl. Psychiatry 10, 275 (2020).
Seed, J. A., McCue, P. M., Wesnes, K. A., Dahabra, S. & Young, A. H. Basal activity of the HPA axis and cognitive function in anorexia nervosa. Int. J. Neuropsychopharmacol. 5, 17–25 (2002).
Monteleone, A. M. et al. Deranged emotional and cortisol responses to a psychosocial stressor in anorexia nervosa women with childhood trauma exposure: evidence for a ‘maltreated ecophenotype’? J. Psychiatr. Res. 104, 39–45 (2018).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5 Vol. 5 (American Psychiatric Association, 2013).
Pike, K. M., Claudino, D. A. & Claudino, A. M. in Eating Disorders: An International Comprehensive View (eds Robinson, P. et al.) 19–39 (Springer, 2024).
Livney, J., Pehlivan, M., Martin, N. G. & Maguire, S. Diagnostic integrity of DSM categorized eating disorders: exploration of alternative methods of classification and the implications for genetic research. Twin Res. Hum. Genet. 28, 28–33 (2025). This Review discusses how the DSM-5’s rigid categorical classifications fail to capture the fluidity of eating disorder presentations and advocates for alternative dimensional models to enhance the validity and power of genetic research into their aetiology.
Castellini, G. et al. Diagnostic crossover and outcome predictors in eating disorders according to DSM-IV and DSM-V proposed criteria: a 6-year follow-up study. Psychosom. Med. 73, 270–279 (2011).
Udo, T. & Grilo, C. M. Psychiatric and medical correlates of DSM-5 eating disorders in a nationally representative sample of adults in the United States. Int. J. Eat. Disord. 52, 42–50 (2019).
Salbach-Andrae, H. et al. Psychiatric comorbidities among female adolescents with anorexia nervosa. Child Psychiatry Hum. Dev. 39, 261–272 (2008).
Seymour, K. E., Reinblatt, S. P., Benson, L. & Carnell, S. Overlapping neurobehavioral circuits in ADHD, obesity, and binge eating: evidence from neuroimaging research. CNS Spectr. 20, 401–411 (2015).
Kotov, R. et al. The Hierarchical Taxonomy of Psychopathology (HiTOP): a dimensional alternative to traditional nosologies. J. Abnorm. Psychol. 126, 454 (2017).
Cuthbert, B. N. The RDoC framework: facilitating transition from ICD/DSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry 13, 28–35 (2014).
Bernardoni, F. et al. Altered medial frontal feedback learning signals in anorexia nervosa. Biol. Psychiatry 83, 235–243 (2018).
Foerde, K. & Steinglass, J. E. Decreased feedback learning in anorexia nervosa persists after weight restoration. Int. J. Eat. Disord. 50, 415–423 (2017).
Hildebrandt, T. et al. Evidence of prefrontal hyperactivation to food-cue reversal learning in adolescents with anorexia nervosa. Behav. Res. Ther. 111, 36–43 (2018).
Pike, A. C. et al. Adaptive learning from outcome contingencies in eating-disorder risk groups. Transl. Psychiatry 13, 340 (2023).
Schuman, I., Wang, J., Ballard, I. C. & Lapate, R. C. Willing to wait: anorexia nervosa symptomatology is associated with higher future orientation and reduced intertemporal discounting. Sci. Rep. 15, 4508 (2025).
Pietrzak, M. et al. Ghrelin decreases sensitivity to negative feedback and increases prediction-error related caudate activity in humans, a randomized controlled trial. Neuropsychopharmacology 49, 1042–1049 (2024).
Bernardoni, F. et al. Metabolic state and value-based decision-making in acute and recovered female patients with anorexia nervosa. J. Psychiatry Neurosci. 45, 253–261 (2020).
Voon, V. et al. Disorders of compulsivity: a common bias towards learning habits. Mol. Psychiatry 20, 345–352 (2015).
Waltmann, M. et al. Neurocomputational mechanisms underlying differential reinforcement learning from wins and losses in obesity with and without binge eating. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 9, 1281–1290 (2024).
Steward, T. et al. Delay discounting of reward and impulsivity in eating disorders: from anorexia nervosa to binge eating disorder. Eur. Eat. Disord. Rev. 25, 601–606 (2017).
Ferracci, S. et al. The role of impulsivity and binge eating in outpatients with overweight or obesity: an EEG temporal discounting study. J. Eat. Disord. 12, 130 (2024).
Morris, L. S. et al. Biases in the explore–exploit tradeoff in addictions: the role of avoidance of uncertainty. Neuropsychopharmacology 41, 940–948 (2016).
Lee, P. H. et al. Genomic relationships, novel loci, and pleiotropic mechanisms across eight psychiatric disorders. Cell 179, 1469–1482.e11 (2019).
Brassard, S. L. & Balodis, I. M. A review of effort-based decision-making in eating and weight disorders. Prog. Neuro-psychopharmacol. Biol. Psychiatry 110, 110333 (2021).
Colton, E., Wilson, K.-E., Chong, T. T. J. & Verdejo-Garcia, A. Dysfunctional decision-making in binge-eating disorder: a meta-analysis and systematic review. Neurosci. Biobehav. Rev. 152, 105250 (2023).
Garcia-Burgos, D., Wilhelm, P., Vögele, C. & Munsch, S. Food restriction in anorexia nervosa in the light of modern learning theory: a narrative review. Behav. Sci. 13, 96 (2023).
Steding, J. et al. Goal-directed vs. habitual instrumental behavior during reward processing in anorexia nervosa: an fMRI study. Sci. Rep. 9, 13529 (2019). This study demonstrates that acutely underweight people with anorexia nervosa segregate into goal-directed versus habit-driven subgroups, distinguished by differential medial orbitofrontal cortex activation during reward anticipation.
Daw, N. D. & Doya, K. The computational neurobiology of learning and reward. Curr. Opin. Neurobiol. 16, 199–204 (2006).
Walsh, B. T. The enigmatic persistence of anorexia nervosa. Am. J. Psychiatry 170, 477–484 (2013).
Schwabe, L. & Wolf, O. T. Stress prompts habit behavior in humans. J. Neurosci. 29, 7191–7198 (2009).
Di Lodovico, L. et al. Is decision-making impairment an endophenotype of anorexia nervosa? Eur. Psychiatry 65, e68 (2022).
Reilly, E. E., Wierenga, C. E. & Le Grange, D. Testing the role of associative learning in evidence‐based treatments for anorexia nervosa. Int. J. Eat. Disord. 57, 1088–1095 (2024). This study underscores the potential of individual difference in associative learning to explain varied responses to anorexia nervosa treatments and advocates for research into these mechanisms.
Levinson, C. A. et al. d-cycloserine facilitation of exposure therapy improves weight regain in patients with anorexia nervosa: a pilot randomized controlled trial. J. Clin. Psychiatry 76, 4033 (2015).
Carhart-Harris, R. L. & Friston, K. J. REBUS and the anarchic brain: toward a unified model of the brain action of psychedelics. Pharmacol. Rev. 71, 316–344 (2019).
Peck, S. K. et al. Psilocybin therapy for females with anorexia nervosa: a phase 1, open-label feasibility study. Nat. Med. 29, 1947–1953 (2023). This landmark phase I study demonstrates the safety and feasibility of psilocybin-assisted therapy in females with anorexia nervosa, providing foundational clinical evidence for psychedelic treatment in eating disorders.
Balleine, B. W. & O’doherty, J. P. Human and rodent homologies in action control: corticostriatal determinants of goal-directed and habitual action. Neuropsychopharmacology 35, 48–69 (2010). This influential review outlines the shared corticostriatal circuits in humans and rodents that govern goal-directed versus habitual behaviour, providing a critical framework for understanding maladaptive action control in disorders such as anorexia and addiction.
Berntson, G. G. & Khalsa, S. S. Neural circuits of interoception. Trends Neurosci. 44, 17–28 (2021).
Bliss-Moreau, E. & Rudebeck, P. H. Animal models of human mood. Neurosci. Biobehav. Rev. 120, 574–582 (2021).
Horn, C. C. et al. Why can’t rodents vomit? A comparative behavioral, anatomical, and physiological study. PLoS ONE 8, e60537 (2013).
Shemesh, Y. & Chen, A. A paradigm shift in translational psychiatry through rodent neuroethology. Mol. Psychiatry 28, 993–1003 (2023).
Mirabella, P. N. & Fenselau, H. Advanced neurobiological tools to interrogate metabolism. Nat. Rev. Endocrinol. 19, 639–654 (2023). This review highlights cutting-edge neurobiological tools — such as optogenetics, chemogenetics and imaging — that enable precise dissection of brain–metabolism interactions, advancing our ability to study and treat metabolic aspects of eating disorders.
Formozov, A., Dieter, A. & Wiegert, J. S. A flexible and versatile system for multi-color fiber photometry and optogenetic manipulation. Cell Rep. Methods 3, 100418 (2023).
Monosov, I. E., Zimmermann, J., Frank, M. J., Mathis, M. W. & Baker, J. T. Ethological computational psychiatry: challenges and opportunities. Curr. Opin. Neurobiol. 86, 102881 (2024).
Loos, R. J. F. & Yeo, G. S. H. The genetics of obesity: from discovery to biology. Nat. Rev. Genet. 23, 120–133 (2022).
Huckins, L. M., Brennand, K. & Bulik, C. M. Dissecting the biology of feeding and eating disorders. Trends Mol. Med. 30, 380–391 (2024).
Gutierrez, E. A rat in the labyrinth of anorexia nervosa: contributions of the activity-based anorexia rodent model to the understanding of anorexia nervosa. Int. J. Eat. Disord. 46, 289–301 (2013).
Huang, K. et al. Rapid, automated, and experimenter-free touchscreen testing reveals reciprocal interactions between cognitive flexibility and activity-based anorexia in female rats. eLife 12, e84961 (2023).
Milton, L. K. et al. Suppression of corticostriatal circuit activity improves cognitive flexibility and prevents body weight loss in activity-based anorexia in rats. Biol. Psychiatry 90, 819–828 (2021). This study shows that targeted suppression of corticostriatal activity enhances cognitive flexibility and prevents weight loss in a rodent model of anorexia, implicating this circuit as a potential therapeutic target for core behavioural features of the disorder.
François, M. & Zeltser, L. M. Rethinking the approach to preclinical models of anorexia nervosa. Curr. Psychiatry Rep. 24, 71–76 (2022).
Foldi, C. J. Taking better advantage of the activity-based anorexia model. Trends Mol. Med. 30, 330–338 (2024).
Milton, L. K., Oldfield, B. J. & Foldi, C. J. Evaluating anhedonia in the activity-based anorexia (ABA) rat model. Physiol. Behav. 194, 324–332 (2018).
Beeler, J. A. et al. Vulnerable and resilient phenotypes in a mouse model of anorexia nervosa. Biol. Psychiatry 90, 829–842 (2021).
Gurfein, B. T. et al. The calm mouse: an animal model of stress reduction. Mol. Med. 18, 606–617 (2012).
Svensson, M. et al. Forced treadmill exercise can induce stress and increase neuronal damage in a mouse model of global cerebral ischemia. Neurobiol. Stress 5, 8–18 (2016).
Walle, R. et al. Nucleus accumbens D1- and D2-expressing neurons control the balance between feeding and activity-mediated energy expenditure. Nat. Commun. 15, 2543 (2024). This study reveals that D1-expressing and D2-expressing neurons in the nucleus accumbens distinctly regulate feeding and energy expenditure through activity, uncovering a neural mechanism for balancing energy homeostasis relevant to eating disorder pathophysiology.
Wable, G. S., Chen, Y. W., Rashid, S. & Aoki, C. Exogenous progesterone exacerbates running response of adolescent female mice to repeated food restriction stress by changing α4-GABAA receptor activity of hippocampal pyramidal cells. Neuroscience 310, 322–341 (2015).
Lee, T. J. & Kinzig, K. P. Repeated adolescent activity-based anorexia influences central estrogen signaling and adulthood anxiety-like behaviors in rats. Physiol. Behav. 171, 199–206 (2017).
Foldi, C. J., Milton, L. K. & Oldfield, B. J. The role of mesolimbic reward neurocircuitry in prevention and rescue of the activity-based anorexia (ABA) phenotype in rats. Neuropsychopharmacology 42, 2292–2300 (2017).
Kucukdereli, H. et al. Repeated stress triggers seeking of a starvation-like state in anxiety-prone female mice. Neuron 112, 2130–2141.e7 (2024). This study demonstrates that repeated stress induces a drive towards a starvation-like state in anxiety-prone female mice, offering a compelling neurobiological model linking stress, sex differences and vulnerability to anorexia nervosa.
Sutton Hickey, A. K. et al. AgRP neurons coordinate the mitigation of activity-based anorexia. Mol. Psychiatry 28, 1622–1635 (2023).
Anversa, R. G. et al. A model of emotional stress-induced binge eating in female mice with no history of food restriction. Genes Brain Behav. 19, e12613 (2020).
Anversa, R. G. et al. A paraventricular thalamus to insular cortex glutamatergic projection gates ‘emotional’ stress-induced binge eating in females. Neuropsychopharmacology 48, 1931–1940 (2023). This study identifies a glutamatergic circuit from the paraventricular thalamus to the insular cortex that drives stress-induced binge eating in females, revealing a sex-specific neural mechanism underlying emotional eating.
Rehn, S., Raymond, J. S., Boakes, R. A. & Leenaars, C. H. C. A systematic review and meta-analysis of animal models of binge eating — part 1: definitions and food/drink intake outcomes. Neurosci. Biobehav. Rev. 132, 1137–1156 (2022).
Notaras, M., Hill, R. & van den Buuse, M. The BDNF gene Val66Met polymorphism as a modifier of psychiatric disorder susceptibility: progress and controversy. Mol. Psychiatry 20, 916–930 (2015).
Madra, M. & Zeltser, L. M. BDNF-Val66Met variant and adolescent stress interact to promote susceptibility to anorexic behavior in mice. Transl. Psychiatry 6, e776 (2016).
Doss, M. K. et al. Psilocybin therapy increases cognitive and neural flexibility in patients with major depressive disorder. Transl. Psychiatry 11, 574 (2021).
Rutherford, S. et al. The normative modeling framework for computational psychiatry. Nat. Protoc. 17, 1711–1734 (2022).
Lv, J. et al. Individual deviations from normative models of brain structure in a large cross-sectional schizophrenia cohort. Mol. Psychiatry 26, 3512–3523 (2021).
Levinson, C. A., Cusack, C., Brown, M. L. & Smith, A. R. A network approach can improve eating disorder conceptualization and treatment. Nat. Rev. Psychol. 1, 419–430 (2022).
Hebebrand, J., Hinney, A. & Antel, J. Could leptin substitution therapy potentially terminate entrapment in anorexia nervosa? Nat. Rev. Endocrinol. 19, 435–436 (2023).
Tozzi, L. et al. Personalized brain circuit scores identify clinically distinct biotypes in depression and anxiety. Nat. Med. 30, 2076–2087 (2024). This study introduces personalized brain circuit scores that distinguish clinically meaningful biotypes in depression and anxiety, advancing precision psychiatry and offering a model for circuit-based classification potentially applicable to eating disorders.
Baydili, İ., Tasci, B. & Tasci, G. Artificial intelligence in psychiatry: a review of biological and behavioral data analyses. Diagnostics 15, 434 (2025).
Haynos, A. F. et al. Machine learning enhances prediction of illness course: a longitudinal study in eating disorders. Psychol. Med. 51, 1392–1402 (2021). This longitudinal study demonstrates that machine-learning models significantly improve prediction of illness trajectory in eating disorders, paving the way for more personalized and proactive treatment approaches.
Monaco, F. et al. An advanced artificial intelligence platform for a personalised treatment of eating disorders. Front. Psychiatry 15, 1414439 (2024).
Rodebaugh, T. L., Frumkin, M. R. & Piccirillo, M. L. The long road from person-specific models to personalized mental health treatment. BMC Med. 18, 365 (2020).
Brown, R. M. & James, M. H. Binge eating, overeating and food addiction: approaches for examining food overconsumption in laboratory rodents. Prog. Neuropsychopharmacol. Biol. Psychiatry 123, 110717 (2023).
Jian-min, C. et al. Assessment of lisdexamfetamine on executive function in rats: a translational cognitive research. Exp. Neurol. 374, 114718 (2024).
Aday, J. S., Davis, A. K., Mitzkovitz, C. M., Bloesch, E. K. & Davoli, C. C. Predicting reactions to psychedelic drugs: a systematic review of states and traits related to acute drug effects. ACS Pharmacol. Transl. Sci. 4, 424–435 (2021).
Griffiths, K. R. et al. Impulsivity and its relationship with lisdexamfetamine dimesylate treatment in binge eating disorder. Front. Psychol. 12, 716010 (2021). This study explores how impulsivity traits influence response to lisdexamfetamine treatment in binge eating disorder, highlighting the importance of targeting impulsivity to optimize therapeutic outcomes.
Frank, G. K. W. From desire to dread — a neurocircuitry based model for food avoidance in anorexia nervosa. J. Clin. Med. 10, 2228 (2021).
Frank, G. K. W., Shott, M. E., Stoddard, J., Swindle, S. & Pryor, T. L. Association of brain reward response with body mass index and ventral striatal-hypothalamic circuitry among young women with eating disorders. JAMA Psychiatry 78, 1123–1133 (2021).
Fladung, A. K., Schulze, U. M., Schöll, F., Bauer, K. & Grön, G. Role of the ventral striatum in developing anorexia nervosa. Transl. Psychiatry 3, e315 (2013).
Seeger, G., Braus, D. F., Ruf, M., Goldberger, U. & Schmidt, M. H. Body image distortion reveals amygdala activation in patients with anorexia nervosa — a functional magnetic resonance imaging study. Neurosci. Lett. 326, 25–28 (2002).
King, J. A. et al. Exaggerated frontoparietal control over cognitive effort-based decision-making in young women with anorexia nervosa. Mol. Psychiatry 30, 861–869 (2025).
Yilmaz, Z. et al. Examination of the shared genetic basis of anorexia nervosa and obsessive–compulsive disorder. Mol. Psychiatry 25, 2036–2046 (2020).
Xue, A. M. et al. Neural representations of food-related attributes in the human orbitofrontal cortex during choice deliberation in anorexia nervosa. J. Neurosci. 42, 109–120 (2022).
Beucke, J. C. et al. Abnormally high degree connectivity of the orbitofrontal cortex in obsessive-compulsive disorder. JAMA Psychiatry 70, 619–629 (2013).
Vocks, S. et al. Effects of body image therapy on the activation of the extrastriate body area in anorexia nervosa: an fMRI study. Psychiatry Res. Neuroimaging 183, 114–118 (2010).
Steuber, E. R. & McGuire, J. F. A meta-analysis of transcranial magnetic stimulation in obsessive-compulsive disorder. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 8, 1145–1155 (2023).
McElroy, S. L. et al. Lisdexamfetamine dimesylate effects on binge eating behaviour and obsessive–compulsive and impulsive features in adults with binge eating disorder. Eur. Eat. Disord. Rev. 24, 223–231 (2016).
Siegel, J. S. et al. Psilocybin desynchronizes the human brain. Nature 632, 131–138 (2024).
Renelli, M. et al. An exploratory study of experiences with conventional eating disorder treatment and ceremonial ayahuasca for the healing of eating disorders. Eat. Weight Disord. 25, 437–444 (2020).
Hayashi, D. et al. What is food noise? A conceptual model of food cue reactivity. Nutrients 15, 4809 (2023).
O’Neil, P. M. et al. Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: a randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. Lancet 392, 637–649 (2018).
Quddos, F. et al. Semaglutide and tirzepatide reduce alcohol consumption in individuals with obesity. Sci. Rep. 13, 20998 (2023).
Richards, J. et al. Successful treatment of binge eating disorder with the GLP-1 agonist semaglutide: a retrospective cohort study. Obes. Pillars 7, 100080 (2023).
Aoun, L. et al. GLP-1 receptor agonists: a novel pharmacotherapy for binge eating (binge eating disorder and bulimia nervosa)? A systematic review. J. Clin. Transl. Endocrinol. 35, 100333 (2024).
Bartel, S., McElroy, S. L., Levangie, D. & Keshen, A. Use of glucagon-like peptide-1 receptor agonists in eating disorder populations. Int. J. Eat. Disord. 57, 286–293 (2024).
Kenny, T. E., Van Wijk, M., Singleton, C. & Carter, J. C. An examination of the relationship between binge eating disorder and insomnia symptoms. Eur. Eat. Disord. Rev. 26, 186–196 (2018).
Carpi, M. et al. Clinical usefulness of dual orexin receptor antagonism beyond insomnia: neurological and psychiatric comorbidities. Neuropharmacology 245, 109815 (2023).
Biederman, J. et al. Lisdexamfetamine dimesylate and mixed amphetamine salts extended-release in children with ADHD: a double-blind, placebo-controlled, crossover analog classroom study. Biol. Psychiatry 62, 970–976 (2007).
Carhart-Harris, R. L. et al. Psilocybin with psychological support for treatment-resistant depression: six-month follow-up. Psychopharmacology 235, 399–408 (2018).
Hinnen, D. Glucagon-like peptide 1 receptor agonists for type 2 diabetes. Diabetes Spectr. 30, 202–210 (2017).
Acknowledgements
C.J.F. received financial support from the National Health and Medical Research Council (NHMRC) of Australia (GTN 2001722 and GTN 2011334).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neuroscience thanks Stefan Ehrlich; Jamie Feusner, who co-reviewed with Hayden Peel; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Adiposity
-
The extent of body fat accumulation, typically quantified as the mass or proportion of adipose tissue relative to total body mass, serving as an index of energy storage and metabolic health.
- Anorexigenic hormones
-
Hormones that suppress appetite and reduce food intake by signalling satiety to the brain.
- Case–control studies
-
Observational studies that compare individuals with a specific condition or disease (cases) with those without it (controls) to identify factors associated with the outcome.
- Cognitive control
-
The mental ability to regulate thoughts and actions in accordance with internal goals, including processes such as attention, inhibition and working memory.
- Epigenetic mechanisms
-
Molecular processes — such as DNA methylation, histone modifications and non-coding RNA activity — that stably alter gene expression without changing the underlying DNA sequence.
- Face validity
-
The extent to which a test or measure appears, on the surface, to assess what it is intended to measure.
- Genome-wide association studies
-
(GWASs). Studies that scan the entire genome to identify genetic variants associated with specific traits or diseases in a population.
- Gut–brain axis
-
The bidirectional communication network linking the gastrointestinal tract and the central nervous system, involving neural, hormonal and immune pathways.
- Neuroendocrine signalling
-
The process by which neurons communicate with endocrine cells to regulate physiological functions through the release of hormones into the bloodstream.
- Orexigenic hormones
-
Hormones that stimulate appetite and increase food intake by acting on brain regions involved in hunger regulation.
- Psychological therapies
-
Structured interventions aimed at improving mental health by addressing thoughts, emotions and behaviours through techniques such as cognitive behavioural therapy and counselling.
- Psychosocial factors
-
Social and psychological influences that affect an individual’s behaviour, mental health and well-being.
- Reward
-
A positive stimulus or outcome that reinforces behaviour by activating brain circuits associated with pleasure andmotivation.
- State-based factors
-
Temporary internal conditions or circumstances, such as mood or physiological status, that influence behaviour or cognition at a given moment.
- Trait-based factors
-
Stable, enduring characteristics or qualities of an individual that influence behaviour and cognition over time.
- Transdiagnostic
-
Processes or factors that apply across multiple distinct mental health disorders rather than being specific to one diagnosis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Foldi, C.J., Griffiths, K.R. Examining the biological causes of eating disorders to inform treatment strategies. Nat. Rev. Neurosci. 26, 554–570 (2025). https://doi.org/10.1038/s41583-025-00940-3
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41583-025-00940-3