Abstract
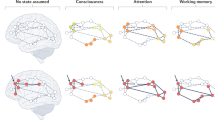
Half a century of neurophysiological recordings from single electrodes established a ‘localized’ viewpoint on function in the brain — that complex behaviour results from computations that are carried out and representations that occur across distinct brain areas, each of which has a specialized role. Data generated from new techniques for specific, high-throughput measurement of neuronal activity and behaviour in rodents have prompted an alternative viewpoint, which posits that neural encoding of behavioural variables is distributed across a wide range of areas: ‘everything, everywhere, all at once’. After briefly introducing these paradigms, we evaluate which of them better describes cognition — the manipulation of internal variables that enables flexible behaviour. Measurements of neuronal activity in both rodents and primates suggest that cognitive variables are reflected broadly but not ubiquitously across the brain, including, to a surprising degree, in regions engaged in controlling movement. We close by discussing why cognitive signals may appear in such areas, as well as the factors that affect the breadth of the brain-wide network that is recruited for cognition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Freedman, D. J. et al. Categorical representation of visual stimuli in the primate prefrontal cortex. Science 291, 312–316 (2001).
Seger, C. A. & Miller, E. K. Category learning in the brain. Annu. Rev. Neurosci. 33, 203–219 (2010).
Wallis, J. D., Anderson, K. C. & Miller, E. K. Single neurons in prefrontal cortex encode abstract rules. Nature 411, 953–956 (2001).
Stoet, G. & Snyder, L. H. Single neurons in posterior parietal cortex of monkeys encode cognitive set. Neuron 42, 1003–1012 (2004).
Mansouri, F. A., Freedman, D. J. & Buckley, M. J. Emergence of abstract rules in the primate brain. Nat. Rev. Neurosci. 21, 595–610 (2020).
Snyder, L. H., Batista, A. P. & Andersen, R. A. Coding of intention in the posterior parietal cortex. Nature 386, 167–170 (1997).
Cui, H. & Andersen, R. A. Posterior parietal cortex encodes autonomously selected motor plans. Neuron 56, 552–559 (2007).
Miller, E. K. & Cohen, J. D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 24, 167–202 (2001).
Christophel, T. B. et al. The distributed nature of working memory. Trends Cogn. Sci. 21, 111–124 (2017).
Haxby, J. V. et al. Distributed and overlapping representations of faces and objects in ventral temporal cortex. Science 293, 2425–2430 (2001).
Spiridon, M. & Kanwisher, N. How distributed is visual category information in human occipito-temporal cortex? An fMRI study. Neuron 35, 1157–1165 (2002).
Op de Beeck, H. P., Haushofer, J. & Kanwisher, N. G. Interpreting fMRI data: maps, modules and dimensions. Nat. Rev. Neurosci. 9, 123–135 (2008).
Fritsch, G. & Hitzig, E. Electric excitability of the cerebrum (Uber die elektrische Erregbarkeit des Grosshirns). Epilepsy Behav. 15, 123–130 (2009).
Ferrier, D. & Burdon-Sanderson, J. S. Experiments on the brain of monkeys. — no. I. Proc. R. Soc. Lond. 23, 409–430 (1874).
Beevor, C. E. & Horsley, V. A. H. VI A minute analysis (experiments) of the various movements produced by stimulating in the monkey different regions of the cortical centre for the upper limb, as defined by Professor Ferrier. Philos. Trans. R. Soc. Lond. B 178, 153–167 (1887).
Penfield, W. & Boldrey, E. Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation. Brain 60, 389–443 (1937).
Hubel, D. H. & Wiesel, T. N. Receptive fields, binocular interaction and functional architecture in the cat’s visual cortex. J. Physiol. 160, 106–154.2 (1962).
Hubel, D. H. & Wiesel, T. N. Ferrier lecture — functional architecture of macaque monkey visual cortex. Proc. R. Soc. Lond. B Biol. Sci. 198, 1–59 (1997).
Dubner, R. & Zeki, S. M. Response properties and receptive fields of cells in an anatomically defined region of the superior temporal sulcus in the monkey. Brain Res. 35, 528–532 (1971).
Zeki, S. M. Functional organization of a visual area in the posterior bank of the superior temporal sulcus of the rhesus monkey. J. Physiol. 236, 549–573 (1974).
Maunsell, J. H. & Van Essen, D. C. Functional properties of neurons in middle temporal visual area of the macaque monkey. I. Selectivity for stimulus direction, speed, and orientation. J. Neurophysiol. 49, 1127–1147 (1983).
Gross, C. G., Bender, D. B. & Rocha-Miranda, C. E. Visual receptive fields of neurons in inferotemporal cortex of the monkey. Science 166, 1303–1306 (1969).
Gross, C. G., Rocha-Miranda, C. E. & Bender, D. B. Visual properties of neurons in inferotemporal cortex of the macaque. J. Neurophysiol. 35, 96–111 (1972).
Desimone, R., Albright, T. D., Gross, C. G. & Bruce, C. Stimulus-selective properties of inferior temporal neurons in the macaque. J. Neurosci. 4, 2051–2062 (1984).
Tanji, J. & Evarts, E. V. Anticipatory activity of motor cortex neurons in relation to direction of an intended movement. J. Neurophysiol. 39, 1062–1068 (1976).
Georgopoulos, A. P., Kalaska, J. F., Caminiti, R. & Massey, J. T. On the relations between the direction of two-dimensional arm movements and cell discharge in primate motor cortex. J. Neurosci. 2, 1527–1537 (1982).
Bizzi, E. Discharge of frontal eye field neurons during saccadic and following eye movements in unanesthetized monkeys. Exp. Brain Res. 6, 69–80 (1968).
Bizzi, E. & Schiller, P. H. Single unit activity in the frontal eye fields of unanesthetized monkeys during eye and head movement. Exp. Brain Res. 10, 150–158 (1970).
Wurtz, R. H. & Goldberg, M. E. Superior colliculus cell responses related to eye movements in awake monkeys. Science 171, 82–84 (1971).
Juavinett, A. L., Bekheet, G. & Churchland, A. K. Chronically implanted Neuropixels probes enable high-yield recordings in freely moving mice. eLife 8, e47188 (2019).
Luo, T. Z. et al. An approach for long-term, multi-probe Neuropixels recordings in unrestrained rats. eLife 9, e59716 (2020).
Steinmetz, N. A. et al. Neuropixels 2.0: a miniaturized high-density probe for stable, long-term brain recordings. Science 372, eabf4588 (2021).
Manley, J. et al. Simultaneous, cortex-wide dynamics of up to 1 million neurons reveal unbounded scaling of dimensionality with neuron number. Neuron 112, 1694–1709.e5 (2024).
Sofroniew, N. J., Flickinger, D., King, J. & Svoboda, K. A large field of view two-photon mesoscope with subcellular resolution for in vivo imaging. eLife 5, e14472 (2016).
Zong, W. et al. Large-scale two-photon calcium imaging in freely moving mice. Cell 185, 1240–1256.e30 (2022).
Zhang, Y. et al. Fast and sensitive GCaMP calcium indicators for imaging neural populations. Nature 615, 884–891 (2023).
Machado, T. A., Kauvar, I. V. & Deisseroth, K. Multiregion neuronal activity: the forest and the trees. Nat. Rev. Neurosci. 23, 683–704 (2022).
Mathis, A. et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat. Neurosci. 21, 1281–1289 (2018).
Pereira, T. D. et al. Fast animal pose estimation using deep neural networks. Nat. Methods 16, 117–125 (2019).
Pereira, T. D. et al. SLEAP: a deep learning system for multi-animal pose tracking. Nat. Methods 19, 486–495 (2022).
Stringer, C. et al. Spontaneous behaviors drive multidimensional, brainwide activity. Science 364, eaav7893 (2019).
Musall, S., Kaufman, M. T., Juavinett, A. L., Gluf, S. & Churchland, A. K. Single-trial neural dynamics are dominated by richly varied movements. Nat. Neurosci. 22, 1677–1686 (2019).
Salkoff, D. B., Zagha, E., McCarthy, E. & McCormick, D. A. Movement and performance explain widespread cortical activity in a visual detection task. Cereb. Cortex 30, 421–437 (2020).
Allen, W. E. et al. Thirst regulates motivated behavior through modulation of brainwide neural population dynamics. Science 364, eaav3932 (2019).
Flavell, S. W., Gogolla, N., Lovett-Barron, M. & Zelikowsky, M. The emergence and influence of internal states. Neuron 110, 2545–2570 (2022).
Hayden, B. Y. The dangers of cortical brain maps. J. Cogn. Neurosci. 35, 372–375 (2023).
Steinmetz, N. A., Zatka-Haas, P., Carandini, M. & Harris, K. D. Distributed coding of choice, action and engagement across the mouse brain. Nature 576, 266–273 (2019).
International Brain Laboratory et al. A brain-wide map of neural activity during complex behaviour. Nature 645, 177–191 (2025).
Chen, S. et al. Brain-wide neural activity underlying memory-guided movement. Cell 187, 676–691.e16 (2024).
Khilkevich, A. et al. Brain-wide dynamics linking sensation to action during decision-making. Nature 634, 890–900 (2024).
O’Shea, D. J. et al. The need for calcium imaging in nonhuman primates: new motor neuroscience and brain-machine interfaces. Exp. Neurol. 287, 437–451 (2017).
Bondy, A. G. et al. Coordinated cross-brain activity during accumulation of sensory evidence and decision commitment. Preprint at bioRxiv https://doi.org/10.1101/2024.08.21.609044 (2024).
Zagha, E. et al. The importance of accounting for movement when relating neuronal activity to sensory and cognitive processes. J. Neurosci. 42, 1375–1382 (2022).
Posani, L., Wang, S., Muscinelli, S. P., Paninski, L. & Fusi, S. Rarely categorical, always high-dimensional: how the neural code changes along the cortical hierarchy. Preprint at bioRxiv https://doi.org/10.1101/2024.11.15.623878 (2025).
Van Essen, D. C. et al. Cerebral cortical folding, parcellation, and connectivity in humans, nonhuman primates, and mice. Proc. Natl Acad. Sci. USA 116, 26173–26180 (2019).
Gămănuţ, R. et al. The mouse cortical connectome, characterized by an ultra-dense cortical graph, maintains specificity by distinct connectivity profiles. Neuron 97, 698–715.e10 (2018).
Markov, N. T. et al. A weighted and directed interareal connectivity matrix for macaque cerebral cortex. Cereb. Cortex 24, 17–36 (2014).
Theodoni, P. et al. Structural attributes and principles of the neocortical connectome in the marmoset monkey. Cereb. Cortex 32, 15–28 (2022).
Magrou, L. et al. The meso-connectomes of mouse, marmoset, and macaque: network organization and the emergence of higher cognition. Cereb. Cortex 34, bhae174 (2024).
Newsome, W. T., Britten, K. H. & Movshon, J. A. Neuronal correlates of a perceptual decision. Nature 341, 52–54 (1989).
Shadlen, M. N. & Newsome, W. T. Motion perception: seeing and deciding. Proc. Natl Acad. Sci. USA 93, 628–633 (1996).
Kim, J.-N. & Shadlen, M. N. Neural correlates of a decision in the dorsolateral prefrontal cortex of the macaque. Nat. Neurosci. 2, 176–185 (1999).
Horwitz, G. D. & Newsome, W. T. Separate signals for target selection and movement specification in the superior colliculus. Science 284, 1158–1161 (1999).
Roitman, J. D. & Shadlen, M. N. Response of neurons in the lateral intraparietal area during a combined visual discrimination reaction time task. J. Neurosci. 22, 9475–9489 (2002).
Shadlen, M. N., Kiani, R., Hanks, T. D. & Churchland, A. K. in Better than Conscious? Decision Making, the Human Mind, and Implications for Institutions 71–101 (MIT Press, 2008).
Ding, L. & Gold, J. I. Caudate encodes multiple computations for perceptual decisions. J. Neurosci. 30, 15747–15759 (2010).
Stine, G. M., Trautmann, E. M., Jeurissen, D. & Shadlen, M. N. A neural mechanism for terminating decisions. Neuron 111, 2601–2613.e5 (2023).
Branam, K., Gold, J. I. & Ding, L. The subthalamic nucleus contributes causally to perceptual decision-making in monkeys. eLife 13, RP98345 (2024).
Steinemann, N. et al. Direct observation of the neural computations underlying a single decision. eLife 12, RP90859 (2024).
Britten, K. H., Newsome, W. T., Shadlen, M. N., Celebrini, S. & Movshon, J. A. A relationship between behavioral choice and the visual responses of neurons in macaque MT. Vis. Neurosci. 13, 87–100 (1996).
Celebrini, S. & Newsome, W. T. Microstimulation of extrastriate area MST influences performance on a direction discrimination task. J. Neurophysiol. 73, 437–448 (1995).
Jun, E. J. et al. Causal role for the primate superior colliculus in the computation of evidence for perceptual decisions. Nat. Neurosci. 24, 1121–1131 (2021).
Hernández, A., Salinas, E., Garcı́a, R. & Romo, R. Discrimination in the sense of flutter: new psychophysical measurements in monkeys. J. Neurosci. 17, 6391–6400 (1997).
Romo, R., Brody, C. D., Hernández, A. & Lemus, L. Neuronal correlates of parametric working memory in the prefrontal cortex. Nature 399, 470–473 (1999).
Hernández, A., Zainos, A. & Romo, R. Neuronal correlates of sensory discrimination in the somatosensory cortex. Proc. Natl Acad. Sci. USA 97, 6191–6196 (2000).
Salinas, E., Hernández, A., Zainos, A. & Romo, R. Periodicity and firing rate as candidate neural codes for the frequency of vibrotactile stimuli. J. Neurosci. 20, 5503–5515 (2000).
Hernández, A., Zainos, A. & Romo, R. Temporal evolution of a decision-making process in medial premotor cortex. Neuron 33, 959–972 (2002).
Romo, R., Hernández, A., Zainos, A., Lemus, L. & Brody, C. D. Neuronal correlates of decision-making in secondary somatosensory cortex. Nat. Neurosci. 5, 1217–1225 (2002).
Brody, C. D., Hernández, A., Zainos, A. & Romo, R. Timing and neural encoding of somatosensory parametric working memory in macaque prefrontal cortex. Cereb. Cortex 13, 1196–1207 (2003).
Romo, R., Hernández, A. & Zainos, A. Neuronal correlates of a perceptual decision in ventral premotor cortex. Neuron 41, 165–173 (2004).
Hernández, A. et al. Decoding a perceptual decision process across cortex. Neuron 66, 300–314 (2010).
Fassihi, A., Akrami, A., Esmaeili, V. & Diamond, M. E. Tactile perception and working memory in rats and humans. Proc. Natl Acad. Sci. USA 111, 2331–2336 (2014).
Fassihi, A., Akrami, A., Pulecchi, F., Schönfelder, V. & Diamond, M. E. Transformation of perception from sensory to motor cortex. Curr. Biol. 27, 1585–1596.e6 (2017).
Akrami, A., Kopec, C. D., Diamond, M. E. & Brody, C. D. Posterior parietal cortex represents sensory history and mediates its effects on behaviour. Nature 554, 368–372 (2018).
Esmaeili, V. & Diamond, M. E. Neuronal correlates of tactile working memory in prefrontal and vibrissal somatosensory cortex. Cell Rep. 27, 3167–3181.e5 (2019).
Freedman, D. J. & Assad, J. A. Experience-dependent representation of visual categories in parietal cortex. Nature 443, 85–88 (2006).
Fitzgerald, J. K., Freedman, D. J. & Assad, J. A. Generalized associative representations in parietal cortex. Nat. Neurosci. 14, 1075–1079 (2011).
Swaminathan, S. K. & Freedman, D. J. Preferential encoding of visual categories in parietal cortex compared with prefrontal cortex. Nat. Neurosci. 15, 315–320 (2012).
Swaminathan, S. K., Masse, N. Y. & Freedman, D. J. A comparison of lateral and medial intraparietal areas during a visual categorization task. J. Neurosci. 33, 13157–13170 (2013).
Rishel, C. A., Huang, G. & Freedman, D. J. Independent category and spatial encoding in parietal cortex. Neuron 77, 969–979 (2013).
Sarma, A., Masse, N. Y., Wang, X.-J. & Freedman, D. J. Task-specific versus generalized mnemonic representations in parietal and prefrontal cortices. Nat. Neurosci. 19, 143–149 (2016).
Mohan, K., Zhu, O. & Freedman, D. J. Interaction between neuronal encoding and population dynamics during categorization task switching in parietal cortex. Neuron 109, 700–712.e4 (2021).
Zhou, Y., Mohan, K. & Freedman, D. J. Abstract encoding of categorical decisions in medial superior temporal and lateral intraparietal cortices. J. Neurosci. 42, 9069–9081 (2022).
Peysakhovich, B. et al. Primate superior colliculus is causally engaged in abstract higher-order cognition. Nat. Neurosci. https://doi.org/10.1038/s41593-024-01744-x (2024).
Zhou, Y., Zhu, O. & Freedman, D. J. Posterior parietal cortex plays a causal role in abstract memory-based visual categorical decisions. J. Neurosci. 43, 4315–4328 (2023).
Thura, D. & Cisek, P. Deliberation and commitment in the premotor and primary motor cortex during dynamic decision making. Neuron 81, 1401–1416 (2014).
Kaufman, M. T., Churchland, M. M., Ryu, S. I. & Shenoy, K. V. Vacillation, indecision and hesitation in moment-by-moment decoding of monkey motor cortex. eLife 4, e04677 (2015).
Mione, V., Brunamonti, E., Pani, P., Genovesio, A. & Ferraina, S. Dorsal premotor cortex neurons signal the level of choice difficulty during logical decisions. Cell Rep. 32, 107961 (2020).
Diaz-deLeon, G. et al. An abstract categorical decision code in dorsal premotor cortex. Proc. Natl Acad. Sci. USA 119, e2214562119 (2022).
Maisson, D. J.-N. et al. Widespread coding of navigational variables in prefrontal cortex. Curr. Biol. 33, 3478–3488.e3 (2023).
Ferrera, V. P., Yanike, M. & Cassanello, C. Frontal eye field neurons signal changes in decision criteria. Nat. Neurosci. 12, 1458–1462 (2009).
Mante, V., Sussillo, D., Shenoy, K. V. & Newsome, W. T. Context-dependent computation by recurrent dynamics in prefrontal cortex. Nature 503, 78–84 (2013).
Siegel, M., Buschman, T. J. & Miller, E. K. Cortical information flow during flexible sensorimotor decisions. Science 348, 1352–1355 (2015).
Charlton, J. A. & Goris, R. L. T. Abstract deliberation by visuomotor neurons in prefrontal cortex. Nat. Neurosci. 27, 1167–1175 (2024).
Ding, L. & Gold, J. I. Separate, causal roles of the caudate in saccadic choice and execution in a perceptual decision task. Neuron 75, 865–874 (2012).
Doi, T., Fan, Y., Gold, J. I. & Ding, L. The caudate nucleus contributes causally to decisions that balance reward and uncertain visual information. eLife 9, e56694 (2020).
Noel, J.-P. et al. Coding of latent variables in sensory, parietal, and frontal cortices during closed-loop virtual navigation. eLife 11, e80280 (2022).
Kaufman, M. T., Churchland, M. M., Ryu, S. I. & Shenoy, K. V. Cortical activity in the null space: permitting preparation without movement. Nat. Neurosci. 17, 440–448 (2014).
Hasnain, M. A. et al. Separating cognitive and motor processes in the behaving mouse. Nat. Neurosci. 28, 640–653 (2025).
Rosen, M. C. & Freedman, D. J. Multiplexing of cognitive encoding by oculomotor networks leads to incidental gaze shifts. Proc. Natl Acad. Sci. USA 122, e2422331122 (2025).
Erlich, J. C., Bialek, M. & Brody, C. D. A cortical substrate for memory-guided orienting in the rat. Neuron 72, 330–343 (2011).
Mangin, E. N., Chen, J., Lin, J. & Li, N. Behavioral measurements of motor readiness in mice. Curr. Biol. 33, 3610–3624.e4 (2023).
Cazettes, F. et al. Facial expressions in mice reveal latent cognitive variables and their neural correlates. Nat. Neurosci. https://doi.org/10.1038/s41593-025-02071-5 (2025).
Yin, C. et al. Spontaneous movements and their relationship to neural activity fluctuate with latent engagement states. Neuron 113, 3048–3063.e5 (2025).
Dotson, N. M., Hoffman, S. J., Goodell, B. & Gray, C. M. Feature-based visual short-term memory is widely distributed and hierarchically organized. Neuron 99, 215–226.e4 (2018).
Lakshminarasimhan, K. J. et al. Tracking the mind’s eye: primate gaze behavior during virtual visuomotor navigation reflects belief dynamics. Neuron 106, 662–674.e5 (2020).
Stavropoulos, A., Lakshminarasimhan, K. J. & Angelaki, D. E. Belief embodiment through eye movements facilitates memory-guided navigation. Nat. Commun. https://doi.org/10.1038/s41467-025-66080-5 (2025).
Caron, A. & Ester, E. F. Human gaze behaviors track stimulus meaning. Preprint at bioRxiv https://doi.org/10.1101/2025.08.08.669367 (2025).
van Ede, F., Chekroud, S. R. & Nobre, A. C. Human gaze tracks attentional focusing in memorized visual space. Nat. Hum. Behav. 3, 462–470 (2019).
Linde-Domingo, J. & Spitzer, B. Geometry of visuospatial working memory information in miniature gaze patterns. Nat. Hum. Behav. 8, 336–348 (2024).
Zokaei, N., Board, A. G., Manohar, S. G. & Nobre, A. C. Modulation of the pupillary response by the content of visual working memory. Proc. Natl Acad. Sci. USA 116, 22802–22810 (2019).
Prut, Y. & Fetz, E. E. Primate spinal interneurons show pre-movement instructed delay activity. Nature 401, 590–594 (1999).
Tremblay, S., Testard, C., DiTullio, R. W., Inchauspé, J. & Petrides, M. Neural cognitive signals during spontaneous movements in the macaque. Nat. Neurosci. 26, 295–305 (2023).
Yang, S., Dong, Y. & Kiyonaga, A. Flexible working memory in the peripheral nervous system. Preprint at bioRxiv https://doi.org/10.1101/2025.09.26.678884 (2025).
Guo, Z. V. et al. Flow of cortical activity underlying a tactile decision in mice. Neuron 81, 179–194 (2014).
Komiyama, T. et al. Learning-related fine-scale specificity imaged in motor cortex circuits of behaving mice. Nature 464, 1182–1186 (2010).
Zatka-Haas, P., Steinmetz, N. A., Carandini, M. & Harris, K. D. Sensory coding and the causal impact of mouse cortex in a visual decision. eLife 10, e63163 (2021).
Pinto, L. et al. Task-dependent changes in the large-scale dynamics and necessity of cortical regions. Neuron 104, 810–824.e9 (2019).
Arlt, C. et al. Cognitive experience alters cortical involvement in goal-directed navigation. eLife 11, e76051 (2022).
Johnston, W. J., Tetrick, S. M. & Freedman, D. J. The lateral intraparietal area preferentially supports stimulus selection in directed tasks compared to undirected free behavior. Preprint at bioRxiv https://doi.org/10.1101/2022.03.09.483625 (2022).
McKee, J. L., Riesenhuber, M., Miller, E. K. & Freedman, D. J. Task dependence of visual and category representations in prefrontal and inferior temporal cortices. J. Neurosci. 34, 16065–16075 (2014).
Kar, K., Kubilius, J., Schmidt, K., Issa, E. B. & DiCarlo, J. J. Evidence that recurrent circuits are critical to the ventral stream’s execution of core object recognition behavior. Nat. Neurosci. 22, 974–983 (2019).
Latimer, K. W. & Freedman, D. J. Low-dimensional encoding of decisions in parietal cortex reflects long-term training history. Nat. Commun. 14, 1010 (2023).
Chowdhury, S. A. & DeAngelis, G. C. Fine discrimination training alters the causal contribution of macaque area MT to depth perception. Neuron 60, 367–377 (2008).
Liu, L. D. & Pack, C. C. The contribution of area MT to visual motion perception depends on training. Neuron 95, 436–446.e3 (2017).
Zerbi, V. et al. Rapid reconfiguration of the functional connectome after chemogenetic locus coeruleus activation. Neuron 103, 702–718.e5 (2019).
Goodwin, S. J., Blackman, R. K., Sakellaridi, S. & Chafee, M. V. Executive control over cognition: stronger and earlier rule-based modulation of spatial category signals in prefrontal cortex relative to parietal cortex. J. Neurosci. 32, 3499–3515 (2012).
Crowe, D. A. et al. Prefrontal neurons transmit signals to parietal neurons that reflect executive control of cognition. Nat. Neurosci. 16, 1484–1491 (2013).
Kawai, R. et al. Motor cortex is required for learning but not for executing a motor skill. Neuron 86, 800–812 (2015).
Mizes, K. G. C., Lindsey, J., Escola, G. S. & Ölveczky, B. P. The role of motor cortex in motor sequence execution depends on demands for flexibility. Nat. Neurosci. 27, 2466–2475 (2024).
International Brain Laboratory et al. Reproducibility of in vivo electrophysiological measurements in mice. eLife 13, RP100840 (2025).
Kriegeskorte, N. & Kievit, R. A. Representational geometry: integrating cognition, computation, and the brain. Trends Cogn. Sci. 17, 401–412 (2013).
Bernardi, S. et al. The geometry of abstraction in the hippocampus and prefrontal cortex. Cell 183, 954–967.e21 (2020).
Kriegeskorte, N. & Wei, X.-X. Neural tuning and representational geometry. Nat. Rev. Neurosci. 22, 703–718 (2021).
Trautmann, E. M. et al. Large-scale high-density brain-wide neural recording in nonhuman primates. Nat. Neurosci. 28, 1562–1575 (2025).
Trepka, E. B., Zhu, S., Xia, R., Chen, X. & Moore, T. Functional interactions among neurons within single columns of macaque V1. eLife 11, e79322 (2022).
Shi, Y. et al. Rapid, concerted switching of the neural code in inferotemporal cortex. Preprint at bioRxiv https://doi.org/10.1101/2023.12.06.570341 (2023).
Panichello, M. F. et al. Intermittent rate coding and cue-specific ensembles support working memory. Nature 636, 422–429 (2024).
Chung, J. E. et al. High-density single-unit human cortical recordings using the Neuropixels probe. Neuron 110, 2409–2421.e3 (2022).
Khanna, A. R. et al. Single-neuronal elements of speech production in humans. Nature 626, 603–610 (2024).
Leonard, M. K. et al. Large-scale single-neuron speech sound encoding across the depth of human cortex. Nature 626, 593–602 (2024).
Michaels, J. A. et al. Sensory expectations shape neural population dynamics in motor circuits. Nature https://doi.org/10.1038/s41586-025-09690-9 (2025).
Namima, T., Kempkes, E., Zamarashkina, P., Owen, N. & Pasupathy, A. High-density recording reveals sparse clusters (but not columns) for shape and texture encoding in macaque V4. J. Neurosci. 45, e1893232024 (2025).
Liu, Y. et al. A high-density 1,024-channel probe for brain-wide recordings in non-human primates. Nat. Neurosci. 27, 1620–1631 (2024).
Clark, A. M. et al. An optrode array for spatiotemporally-precise large-scale optogenetic stimulation of deep cortical layers in non-human primates. Commun. Biol. 7, 329 (2024).
Sadakane, O. et al. Long-term two-photon calcium imaging of neuronal populations with subcellular resolution in adult non-human primates. Cell Rep. 13, 1989–1999 (2015).
Miller, C. T. et al. Marmosets: a neuroscientific model of human social behavior. Neuron 90, 219–233 (2016).
Mitchell, J. F. & Leopold, D. A. The marmoset monkey as a model for visual neuroscience. Neurosci. Res. 93, 20–46 (2015).
Findling, C. et al. Brain-wide representations of prior information in mouse decision-making. Nature 645, 192–200 (2025).
Burgess, C. P. et al. High-yield methods for accurate two-alternative visual psychophysics in head-fixed mice. Cell Rep. 20, 2513–2524 (2017).
Abbott, L. F. et al. An international laboratory for systems and computational neuroscience. Neuron 96, 1213–1218 (2017).
International Brain Laboratory et al. Standardized and reproducible measurement of decision-making in mice. eLife 10, e63711 (2021).
Acknowledgements
The authors acknowledge funding support from the Margot and Tom Pritzker Foundation, the NSF-Simons National Institute for Theory and Mathematics in Biology, NIH R01EY019041 and NIH R01EY037119.
Author information
Authors and Affiliations
Contributions
M.C.R. researched data for the article. Both authors provided substantial contributions to the discussion of its content, wrote the article, and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neuroscience thanks Anne Churchland, who co-reviewed with Gabriel Rojas-Bowe; Hendrikje Nienborg, who co-reviewed with Bharath Chandra Talluri; and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rosen, M.C., Freedman, D.J. How distributed is the brain-wide network that is recruited for cognition?. Nat. Rev. Neurosci. 27, 138–150 (2026). https://doi.org/10.1038/s41583-025-00992-5
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41583-025-00992-5