Abstract
Articular cartilage can withstand substantial compressive and shear forces within the joint and also reduces friction during motion. The exceptional mechanical properties of articular cartilage stem from its highly organized extracellular matrix (ECM). The ECM is composed mainly of collagen type II and is pivotal in conferring mechanical durability to the tissue within its proteoglycan-rich matrix. Articular cartilage is prone to injury and degeneration, and current treatments often fail to restore the mechanical function of this tissue. A key challenge is replicating the intricate collagen–proteoglycan network, which is essential for the long-lasting restoration and mechanical durability of the tissue. Understanding articular cartilage development, which arises between late embryonic and early juvenile development, is vital for the creation of durable therapeutic strategies. The development of the articular ECM involves the biosynthesis, fibrillogenesis and self-assembly of the collagen type II network, which, along with proteoglycans and minor ECM components, shapes the architecture of adult articular cartilage. A deeper understanding of these processes could inform biomaterial-based therapies aimed at improving therapeutic outcomes. Emerging biofabrication technologies offer new opportunities to integrate developmental principles into the creation of durable articular cartilage implants. Bridging fundamental biology with innovative engineering offers novel approaches to generating more-durable 3D implants for articular cartilage restoration.
Key points
-
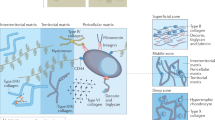
Articular cartilage is a highly anisotropic tissue that is characterized by depth-dependent collagen fibre orientation, which provides the tissue with its unique biomechanical properties.
-
The collagen architecture of the articular cartilage forms during late fetal and postnatal development in response to biomechanical stimuli and tissue growth.
-
Collagen interacts with the surrounding extracellular matrix (ECM), including aggrecan, by limiting the water-swelling pressure caused by negatively charged glycosaminoglycans, thereby enhancing cartilage resilience.
-
Small proteoglycans and (glyco)proteins promote tissue cohesion and stability by regulating fibrillogenesis and connecting various ECM components together.
-
Achieving native-like collagen anisotropy in engineered cartilage remains a challenge, as current scaffolds cannot replicate the depth-dependent alignment of collagen and its ECM interactions.
-
Insights gained from cartilage development and its anisotropic organization could inspire novel strategies for achieving long-term cartilage regeneration.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Shepherd, D. E. T. & Seedhom, B. B. Thickness of human articular cartilage in joints of the lower limb. Ann. Rheum. Dis. 58, 27–34 (1999).
Eckstein, F. et al. Correlation and sex differences between ankle and knee cartilage morphology determined by quantitative magnetic resonance imaging. Ann. Rheum. Dis. 63, 1490–1495 (2004).
Kurz, B., Lange, T., Voelker, M., Hart, M. L. & Rolauffs, B. Articular cartilage — from basic science structural imaging to non-invasive clinical quantitative molecular functional information for AI classification and prediction. Int. J. Mol. Sci. 24, 14974 (2023).
Lawless, B. M. et al. Viscoelasticity of articular cartilage: analysing the effect of induced stress and the restraint of bone in a dynamic environment. J. Mech. Behav. Biomed. Mater. 75, 293–301 (2017).
Malda, J. et al. Comparative study of depth-dependent characteristics of equine and human osteochondral tissue from the medial and lateral femoral condyles. Osteoarthritis Cartilage 20, 1147–1151 (2012).
Alcaide-Ruggiero, L., Molina-Hernández, V., Granados, M. M. & Domínguez, J. M. Main and minor types of collagens in the articular cartilage: the role of collagens in repair tissue evaluation in chondral defects. Int. J. Mol. Sci. 22, 13329 (2021).
Holmes, D. F. & Kadler, K. E. The 10 + 4 microfibril structure of thin cartilage fibrils. Proc. Natl Acad. Sci. USA 103, 17249–17254 (2006).
Hagg, R., Bruckner, P. & Hedbom, E. Cartilage fibrils of mammals are biochemically heterogeneous: differential distribution of decorin and collagen IX. J. Cell Biol. 142, 285–294 (1998).
Benninghoff, V. A. Form und Bau der Gelenkknorpel in ihren Beziehungen zur Funktion. Z. Zellforsch. Mik. Ana. 2, 783–862 (1925).
Watanabe, H., Yamada, Y. & Kimata, K. Roles of aggrecan, a large chondroitin sulfate proteoglycan, in cartilage structure and function. J. Biochem. 124, 687–693 (1998).
Kiani, C., Chen, L., Wu, Y. J., Yee, A. J. & Yang, B. B. Structure and function of aggrecan. Cell Res. 12, 19–32 (2002).
Askew, M. J. & Mow, V. C. The biomechanical function of the collagen fibril ultrastructure of articular cartilage. J. Biomech. Eng. 100, 105–115 (1978).
Wang, Y., Wei, L., Zeng, L., He, D. & Wei, X. Nutrition and degeneration of articular cartilage. Knee Surg. Sports Traumatol. Arthrosc. 21, 1751–1762 (2013).
Fox, A. J. S., Bedi, A. & Rodeo, S. A. The basic science of articular cartilage: structure, composition, and function. Sports Health 1, 461–468 (2009).
Quinn, T. M., Häuselmann, H. J., Shintani, N. & Hunziker, E. B. Cell and matrix morphology in articular cartilage from adult human knee and ankle joints suggests depth-associated adaptations to biomechanical and anatomical roles. Osteoarthritis Cartilage 21, 1904–1912 (2013).
Loeser, R. F. Aging processes and the development of osteoarthritis. Curr. Opin. Rheumatol. 25, 108–113 (2013).
Maroudas, A., Palla, G. & Gilav, E. Racemization of aspartic acid in human articular cartilage. Connect. Tissue Res. 28, 161–169 (1992).
Silver, F. H. & Glasgold, A. I. Cartilage wound healing: an overview. Otolaryngol. Clin. North. Am. 28, 847–864 (1995).
Nuelle, C. W., Laprade, C. M. & Sherman, S. L. in Advances in Knee Ligament and Knee Preservation Surgery (eds Nakamura, N. et al.) 379–394 (Springer, 2022).
Minas, T., Ogura, T. & Bryant, T. Autologous chondrocyte implantation. JBJS Essent. Surg. Tech. 6, 1–11 (2016).
Wasyłeczko, M., Sikorska, W. & Chwojnowski, A. Review of synthetic and hybrid scaffolds in cartilage tissue engineering. Membranes 10, 1–28 (2020).
Wu, Z., Korntner, S. H., Mullen, A. M. & Zeugolis, D. I. Collagen type II: from biosynthesis to advanced biomaterials for cartilage engineering. Biomater. Biosyst. 4, 100030 (2021).
Liu, G. et al. 3D printed osteochondral scaffolds: design strategies, present applications and future perspectives. Front. Bioeng. Biotechnol. 12, 1339916 (2024).
Groen, W. M., Diloksumpan, P., van Weeren, P. R., Levato, R. & Malda, J. From intricate to integrated: biofabrication of articulating joints. J. Orthop. Res. 35, 2089–2097 (2017).
Malda, J., Groll, J. & van Weeren, P. R. Rethinking articular cartilage regeneration based on a 250-year-old statement. Nat. Rev. Rheumatol. 15, 571–572 (2019).
Decker, R. S. Articular cartilage and joint development from embryogenesis to adulthood. Semin. Cell Dev. Biol. 62, 50–56 (2017).
Mancini, I. A. D. et al. Effects of body mass on microstructural features of the osteochondral unit: a comparative analysis of 37 mammalian species. Bone 127, 664–673 (2019).
Mansfield, J. C., Bell, J. S. & Winlove, C. P. The micromechanics of the superficial zone of articular cartilage. Osteoarthritis Cartilage 23, 1806–1816 (2015).
Hossain, M. J. et al. Anisotropic properties of articular cartilage in an accelerated in vitro wear test. J. Mech. Behav. Biomed. Mater. 109, 103834 (2020).
Below, S., Arnoczky, S. P., Dodds, J., Kooima, C. & Walter, N. The split-line pattern of the distal femur: a consideration in the orientation of autologous cartilage grafts. Arthroscopy 18, 613–617 (2002).
Qiu, C. Coefficients of friction of human joints. The Physics Factbook (ed. Elert, G.) https://hypertextbook.com/facts/2007/ConnieQiu.shtml (Hypertextbook, 2007).
Li, L. et al. Superficial cells are self-renewing chondrocyte progenitors, which form the articular cartilage in juvenile mice. FASEB J. 31, 1067–1084 (2017).
Decker, R. S. et al. Cell origin, volume and arrangement are drivers of articular cartilage formation, morphogenesis and response to injury in mouse limbs. Dev. Biol. 426, 56–68 (2017).
Kozhemyakina, E. et al. Identification of a Prg4-expressing articular cartilage progenitor cell population in mice. Arthritis Rheumatol. 67, 1261–1273 (2015).
Levato, R. et al. The bio in the ink: cartilage regeneration with bioprintable hydrogels and articular cartilage-derived progenitor cells. Acta Biomater. 61, 41 (2017).
Ustunel, I. et al. The immunohistochemical localization of notch receptors and ligands in human articular cartilage, chondroprogenitor culture and ultrastructural characteristics of these progenitor cells. Acta Histochem. 110, 397–407 (2008).
Williams, R. et al. Identification and clonal characterisation of a progenitor cell sub-population in normal human articular cartilage. PLoS ONE 5, e13246 (2010).
Dowthwaite, G. P. et al. The surface of articular cartilage contains a progenitor cell population. J. Cell Sci. 117, 889–897 (2004).
Rikkers, M., Korpershoek, J. V., Levato, R., Malda, J. & Vonk, L. A. The clinical potential of articular cartilage-derived progenitor cells: a systematic review. NPJ Regen. Med. 7, 1–20 (2022).
Seol, D. et al. Chondrogenic progenitor cells respond to cartilage injury. Arthritis Rheum. 64, 3626–3637 (2012).
Melero Martin, J. M., Smith, M. & Al-Rubeai, M. Cryopreservation and in vitro expansion of chondroprogenitor cells isolated from the superficial zone of articular cartilage. Biotechnol. Prog. 21, 168–177 (2005).
Melero-Martin, J. M., Dowling, M. A., Smith, M. & Al-Rubeai, M. Expansion of chondroprogenitor cells on macroporous microcarriers as an alternative to conventional monolayer systems. Biomaterials 27, 2970–2979 (2006).
Melero-Martin, J. M., Dowling, M. A., Smith, M. & Al-Rubeai, M. Optimal in-vitro expansion of chondroprogenitor cells in monolayer culture. Biotechnol. Bioeng. 93, 519–533 (2006).
Poole, C. A., Flint, M. H. & Beaumont, B. W. Chondrons in cartilage: ultrastructural analysis of the pericellular microenvironment in adult human articular cartilages. J. Orthop. Res. 5, 509–522 (1987).
Gottardi, R. et al. Supramolecular organization of collagen fibrils in healthy and osteoarthritic human knee and hip joint cartilage. PLoS ONE 11, e0163552 (2016).
Wang, F. et al. Histomorphometric analysis of adult articular calcified cartilage zone. J. Struct. Biol. 168, 359–365 (2009).
Mark, V. Der et al. Type X collagen, a natural component of mouse articular cartilage. Arthritis Rheum. 41, 1287–1295 (1998).
Malda, J. et al. Of mice, men and elephants: the relation between articular cartilage thickness and body mass. PLoS ONE 8, e57683 (2013).
Mancini, I. A. D. et al. Microstructural differences in the osteochondral unit of terrestrial and aquatic mammals. eLife 12, e80936 (2023).
Zhang, Z. Chondrons and the pericellular matrix of chondrocytes. Tissue Eng. Part. B Rev. 21, 267–277 (2015).
Wilusz, R. E., DeFrate, L. E. & Guilak, F. A biomechanical role for perlecan in the pericellular matrix of articular cartilage. Matrix Biol. 31, 320–327 (2012).
Zelenski, N. A. et al. Collagen VI regulates pericellular matrix properties, chondrocyte swelling, and mechanotransduction in articular cartilage. Arthritis Rheumatol. 67, 1286–1294 (2015).
Glant, T. T., Hadházy, C., Mikecz, K. & Sipos, A. Appearance and persistence of fibronectin in cartilage — specific interaction of fibronectin with collagen type II. Histochemistry 82, 149–158 (1985).
Loeser, R. F. Integrins and chondrocyte-matrix interactions in articular cartilage. Matrix Biol. 39, 11–16 (2014).
Mansfield, J. C., Mandalia, V., Toms, A., Peter Winlove, C. & Brasselet, S. Collagen reorganization in cartilage under strain probed by polarization sensitive second harmonic generation microscopy. J. R. Soc. Interface 16, 20180611 (2019).
Decker, R. S., Koyama, E. & Pacifici, M. Genesis and morphogenesis of limb synovial joints and articular cartilage. Matrix Biol. 39, 5–10 (2014).
Bian, Q. et al. A single cell transcriptional atlas of early synovial joint development. Development 147, dev185777 (2020).
Chijimatsu, R. & Saito, T. Mechanisms of synovial joint and articular cartilage development. Cell Mol. Life Sci. 76, 3939–3952 (2019).
Shwartz, Y., Viukov, S., Krief, S. & Zelzer, E. Joint development involves a continuous influx of Gdf5-positive cells. Cell Rep. 15, 2577–2587 (2016).
Chen, H. et al. Heads, shoulders, elbows, knees, and toes: modular Gdf5 enhancers control different joints in the vertebrate skeleton. PLoS Genet. 12, e1006454 (2016).
Sun, K., Guo, J., Yao, X., Guo, Z. & Guo, F. Growth differentiation factor 5 in cartilage and osteoarthritis: a possible therapeutic candidate. Cell Prolif. 54, e12998 (2021).
Pitsillides, A. A., Archer, C. W., Prehm, P., Bayliss, M. T. & Edwards, J. C. W. Alterations in hyaluronan synthesis during developing joint cavitation. J. Histochem. Cytochem. 43, 263–273 (1995).
Drachman, D. B. & Sokoloff, L. The role of movement in embryonic joint development. Dev. Biol. 14, 401–420 (1966).
Pacifici, M., Koyama, E. & Iwamoto, M. Mechanisms of synovial joint and articular cartilage formation: recent advances, but many lingering mysteries. Birth Defects Res. C. Embryo Today 75, 237–248 (2005).
Ignatyeva, N., Gavrilov, N., Timashev, P. S. & Medvedeva, E. V. Prg4-expressing chondroprogenitor cells in the superficial zone of articular cartilage. Int. J. Mol. Sci. 25, 5605 (2024).
Yang, L., Tsang, K. Y., Tang, H. C., Chan, D. & Cheah, K. S. E. Hypertrophic chondrocytes can become osteoblasts and osteocytes in endochondral bone formation. Proc. Natl Acad. Sci. USA 111, 12097–12102 (2014).
Li, J. & Dong, S. The signaling pathways involved in chondrocyte differentiation and hypertrophic differentiation. Stem Cell Int. 2016, 1–12 (2016).
Wu, M., Wu, S., Chen, W. & Li, Y. P. The roles and regulatory mechanisms of TGF-β and BMP signaling in bone and cartilage development, homeostasis and disease. Cell Res. 34, 101–123 (2024).
Hoemann, C. D., Lafantaisie-Favreau, C.-H., Lascau-Coman, V., Chen, G. & Guzmán-Morales, J. The cartilage-bone interface. J. Knee Surg. 25, 85–97 (2012).
Luo, Y. et al. The minor collagens in articular cartilage. Protein Cell 8, 560–572 (2017).
Ni, G. X., Li, Z. & Zhou, Y. Z. The role of small leucine-rich proteoglycans in osteoarthritis pathogenesis. Osteoarthritis Cartilage 22, 896–903 (2014).
Responte, D. J., Natoli, R. M. & Athanasiou, K. A. Collagens of articular cartilage: structure, function, and importance in tissue engineering. Crit. Rev. Biomed. Eng. 35, 363–411 (2007).
Perrier-Groult, E. et al. Presence of type IIB procollagen in mouse articular cartilage and growth plate is revealed by immuno-histochemical analysis with a novel specific antibody. Matrix Biol. 18, 100130 (2023).
Mcalinden, A. Alternative splicing of type II procollagen: IIB or not IIB? Connect. Tissue Res. 55, 165–176 (2014).
Shoulders, M. D. & Raines, R. T. Modulating collagen triple-helix stability with 4-chloro, 4-fluoro, and 4-methylprolines. Adv. Exp. Med. Biol. 611, 251–252 (2009).
Canty, E. G. & Kadler, K. E. Procollagen trafficking, processing and fibrillogenesis. J. Cell Sci. 118, 1341–1353 (2005).
Antipova, O. & Orgel, J. P. R. O. In situ D-periodic molecular structure of type II collagen. J. Biol. Chem. 285, 7087–7096 (2010).
Kadler, K. E., Hill, A. & Canty-Laird, E. G. Collagen fibrillogenesis: fibronectin, integrins, and minor collagens as organizers and nucleators. Curr. Opin. Cell Biol. 20, 495–501 (2008).
Wu, J. J., Woods, P. E. & Eyre, D. R. Identification of cross-linking sites in bovine cartilage type IX collagen reveals an antiparallel type II-type IX molecular relationship and type IX to type IX bonding. J. Biol. Chem. 267, 23007–23014 (1992).
Chen, C. H. et al. Interactions between collagen IX and biglycan measured by atomic force microscopy. Biochem. Biophys. Res. Commun. 339, 204–208 (2006).
Parsons, P. et al. Type IX collagen interacts with fibronectin providing an important molecular bridge in articular cartilage. J. Biol. Chem. 286, 34986–34997 (2011).
Vasios, G. et al. Cartilage type IX collagen-proteoglycan contains a large amino-terminal globular domain encoded by multiple exons. J. Biol. Chem. 263, 2324–2329 (1988).
Mendler, M., Eich-Bender, S. G., Vaughan, L., Winterhalter, K. H. & Bruckner, P. Cartilage contains mixed fibrils of collagen types II, IX, and XI. J. Cell Biol. 108, 191–197 (1989).
Gannon, A. R., Nagel, T., Bell, A. P., Avery, N. C. & Kelly, D. J. Postnatal changes to the mechanical properties of articular cartilage are driven by the evolution of its collagen network. Eur. Cell Mater. 29, 105–123 (2015).
Douglas, T., Heinemann, S., Bierbaum, S., Scharnweber, D. & Worch, H. Fibrillogenesis of collagen types I, II, and III with small leucine-rich proteoglycans decorin and biglycan. Biomacromolecules 7, 2388–2393 (2006).
Wiberg, C. et al. Complexes of matrilin-1 and biglycan or decorin connect collagen VI microfibrils to both collagen II and aggrecan. J. Biol. Chem. 278, 37698–37704 (2003).
Saeidi, N. et al. Molecular crowding of collagen: a pathway to produce highly-organized collagenous structures. Biomaterials 33, 7366–7374 (2012).
Siegel, R. C. Collagen cross-linking. Synthesis of collagen cross-links in vitro with highly purified lysyl oxidase. J. Biol. Chem. 251, 5786–5792 (1976).
Eyre, D. R., Weis, M. A. & Wu, J. J. Maturation of collagen ketoimine cross-links by an alternative mechanism to pyridinoline formation in cartilage. J. Biol. Chem. 285, 16675–16682 (2010).
Eyre, D. R., Dickson, I. R. & Van Ness, K. Collagen cross-linking in human bone and articular cartilage. Age-related changes in the content of mature hydroxypyridinium residues. Biochem. J. 252, 495–500 (1988).
Ito, K. & Tepic, S. in Osteoarthritis (eds Grifka, J. & Ogilvie-Harris, D. J.) 36–53 (Springer, 2000).
Van Turnhout, M. C. et al. Postnatal development of depth-dependent collagen density in ovine articular cartilage. BMC Dev. Biol. 10, 1–16 (2010).
Van Turnhout, M. C. et al. Quantitative description of collagen structure in the articular cartilage of the young and adult equine distal metacarpus. Anim. Biol. 58, 353–370 (2008).
Hunziker, E. B., Kapfinger, E. & Geiss, J. The structural architecture of adult mammalian articular cartilage evolves by a synchronized process of tissue resorption and neoformation during postnatal development. Osteoarthritis Cartilage 15, 403–413 (2007).
Morrison, E. H., Ferguson, M. W., Bayliss, M. T. & Archer, C. W. The development of articular cartilage: I. The spatial and temporal patterns of collagen types. J. Anat. 189, 9–22 (1996).
Archer’, C. W., Morrison’, E. H., Bayliss, M. T. & Ferguson, M. W. J. The development of articular cartilage: II. The spatial and temporal patterns of glycosaminoglycans and small leucine-rich proteoglycans. J. Anat. 189, 23 (1996).
Jia, Y. et al. Double-edged role of mechanical stimuli and underlying mechanisms in cartilage tissue engineering. Front. Bioeng. Biotechnol. 11, 1271762 (2023).
Canty, E. G. et al. Coalignment of plasma membrane channels and protrusions (fibripositors) specifies the parallelism of tendon. J. Cell Biol. 165, 553–563 (2004).
Cui, P. et al. Advanced review on type II collagen and peptide: preparation, functional activities and food industry application. Crit. Rev. Food Sci. Nutr. 64, 11302–11319 (2023).
Peters, J. R. et al. Tissue growth as a mechanism for collagen fiber alignment in articular cartilage. Sci. Rep. 14, 1–12 (2024).
Hayes, A. J., Hall, A., Brown, L., Tubo, R. & Caterson, B. Macromolecular organization and in vitro growth characteristics of scaffold-free neocartilage grafts. J. Histochem. Cytochem. 55, 853–866 (2007).
Castilho, M., Mouser, V., Chen, M., Malda, J. & Ito, K. Bi-layered micro-fibre reinforced hydrogels for articular cartilage regeneration. Acta Biomater. 95, 297–305 (2019).
Lecocq, M. et al. Cartilage matrix changes in the developing epiphysis: early events on the pathway to equine osteochondrosis? Equine Vet. J. 40, 442–454 (2008).
Nesic, D. et al. Cartilage tissue engineering for degenerative joint disease. Adv. Drug Deliv. Rev. 58, 300–322 (2006).
De Rooij, P. P., Siebrecht, M. A. N., Tägil, M. & Aspenberg, P. The fate of mechanically induced cartilage in an unloaded environment. J. Biomech. 34, 961–966 (2001).
Arokoski, J. P. A., Jurvelin, J. S., Väätäinen, U. & Helminen, H. J. Normal and pathological adaptations of articular cartilage to joint loading. Scand. J. Med. Sci. Sports 10, 186–198 (2000).
Visser, J. D. Pediatric Orthopedics: Symptoms, Differential Diagnosis, Supplementary Assessment and Treatment (Springer, 2017).
Nowlan, N. C., Chandaria, V. & Sharpe, J. Immobilized chicks as a model system for early-onset developmental dysplasia of the hip. J. Orthopaedic Res. 32, 777–785 (2014).
Brunt, L. H. et al. Differential effects of altered patterns of movement and strain on joint cell behaviour and skeletal morphogenesis. Osteoarthritis Cartilage 24, 1940–1950 (2016).
Ford, C. A., Nowlan, N. C., Thomopoulos, S. & Killian, M. L. Effects of imbalanced muscle loading on hip joint development and maturation. J. Orthop. Res. 35, 1128–1136 (2017).
Felsenthal, N. & Zelzer, E. Mechanical regulation of musculoskeletal system development. Development 144, 4271–4283 (2017).
Khoshgoftar, M., van Donkelaar, C. C. & Ito, K. Mechanical stimulation to stimulate formation of a physiological collagen architecture in tissue-engineered cartilage: a numerical study. Comput. Methods Biomech. Biomed. Eng. 14, 135–144 (2011).
Wilson, W., Huyghe, J. M. & van Donkelaar, C. C. A composition-based cartilage model for the assessment of compositional changes during cartilage damage and adaptation. Osteoarthritis Cartilage 14, 554–560 (2006).
Driessen, N. J. B., Boerboom, R. A., Huyghe, J. M., Bouten, C. V. C. & Baaijens, F. P. T. Computational analyses of mechanically induced collagen fiber remodeling in the aortic heart valve. J. Biomech. Eng. 125, 549–557 (2003).
Iijima, H. et al. Immature articular cartilage and subchondral bone covered by menisci are potentially susceptive to mechanical load. BMC Musculoskelet. Disord. 15, 1–12 (2014).
O’Connor, P., Bland, C. & Gardner, D. L. Fine structure of artificial splits in femoral condylar cartilage of the rat: a scanning electron microscopic study. J. Pathol. 132, 169–179 (1980).
Kelly, T. A. N., Ng, K. W., Wang, C. C. B., Ateshian, G. A. & Hung, C. T. Spatial and temporal development of chondrocyte-seeded agarose constructs in free-swelling and dynamically loaded cultures. J. Biomech. 39, 1489–1497 (2006).
Tepic, S. Dynamics of and Entropy Production in the Cartilage Layers of the Synovial Joint. Thesis, MIT (1982).
Rieppo, J. et al. Changes in spatial collagen content and collagen network architecture in porcine articular cartilage during growth and maturation. Osteoarthritis Cartilage 17, 448–455 (2009).
Brama, P. A. J., Tekoppele, J. M., Bank, R. A., Barneveld, A. & Van Weeren, P. R. Functional adaptation of equine articular cartilage: the formation of regional biochemical characteristics up to age one year. Equine Vet. J. 32, 217–221 (2000).
Hyttinen, M. M. et al. Changes in collagen fibril network organization and proteoglycan distribution in equine articular cartilage during maturation and growth. J. Anat. 215, 584–591 (2009).
Julkunen, P. et al. Maturation of collagen fibril network structure in tibial and femoral cartilage of rabbits. Osteoarthritis Cartilage 18, 406–415 (2010).
Torzilli, P. A., Dethmers, D. A., Rose, D. E. & Schryuer, H. F. Movement of interstitial water through loaded articular cartilage. J. Biomech. 16, 169–171 (1983).
Mow, V. C. et al. The influence of link protein stabilization on the viscometric properties of proteoglycan aggregate solutions. Biochim. Biophys. Acta 992, 201–208 (1989).
Chahine, N. O., Wang, C. C. B., Hung, C. T. & Ateshian, G. A. Anisotropic strain-dependent material properties of bovine articular cartilage in the transitional range from tension to compression. J. Biomech. 37, 1251–1261 (2004).
Inamdar, S. R., Prévost, S., Terrill, N. J., Knight, M. M. & Gupta, H. S. Reversible changes in the 3D collagen fibril architecture during cyclic loading of healthy and degraded cartilage. Acta Biomater. 136, 314–326 (2021).
Inamdar, S. R., Barbieri, E., Terrill, N. J., Knight, M. M. & Gupta, H. S. Proteoglycan degradation mimics static compression by altering the natural gradients in fibrillar organisation in cartilage. Acta Biomater. 97, 437–450 (2019).
Amuasi, H. E. Multiscale Structure and Mechanics of Collagen. Thesis, Technische Universiteit Eindhoven (2012).
Rojas, F. P. et al. Molecular adhesion between cartilage extracellular matrix macromolecules. Biomacromolecules 15, 772–780 (2014).
Paulsson, M. et al. Extended and globular protein domains in cartilage proteoglycans. Biochem. J. 245, 763–772 (1987).
Wiedemann, H., Paulsson, M., Timpl, R., Engel, J. & Heinegård, D. Domain structure of cartilage proteoglycans revealed by rotary shadowing of intact and fragmented molecules. Biochem. J. 224, 331–333 (1984).
Wei, Q., Zhang, X., Zhou, C., Ren, Q. & Zhang, Y. Roles of large aggregating proteoglycans in human intervertebral disc degeneration. Connect. Tissue Res. 60, 209–218 (2018).
Vynios, D. H. Metabolism of cartilage proteoglycans in health and disease. Biomed. Res. Int. 2014, 452315 (2014).
Plaas, A. H. K., Moran, M. M., Sandy, J. D. & Hascall, V. C. Aggrecan and hyaluronan: the infamous cartilage polyelectrolytes—then and now. Adv. Exp. Med. Biol. 1402, 3–29 (2023).
Alcaide-Ruggiero, L., Cugat, R. & Domínguez, J. M. Proteoglycans in articular cartilage and their contribution to chondral injury and repair mechanisms. Int. J. Mol. Sci. 24, 10824 (2023).
Paganini, C., Costantini, R., Superti-Furga, A. & Rossi, A. Bone and connective tissue disorders caused by defects in glycosaminoglycan biosynthesis: a panoramic view. FEBS J. 286, 3008–3032 (2019).
Vanderploeg, E. J., Wilson, C. G., Levenston, M. E. & Woodruff, G. W. Articular chondrocytes derived from distinct tissue zones differentially respond to in vitro oscillatory tensile loading. Osteoarthritis Cartilage 16, 1228–1236 (2008).
Aydelotte, M. B. & Kuettner, K. E. Differences between sub-populations of cultured bovine articular chondrocytes. I. Morphology and cartilage matrix production. Connect. Tissue Res. 18, 205–222 (1988).
Lee, H. Y., Han, L., Roughley, P. J. & Grodzinsky, A. J. & Ortiz, C. Age-related nanostructural and nanomechanical changes of individual human cartilage aggrecan monomers and their glycosaminoglycan side chains. J. Struct. Biol. 181, 264 (2013).
Wang, C., Kahle, E. R., Li, Q. & Han, L. Nanomechanics of aggrecan: a new perspective on cartilage biomechanics, disease and regeneration. Adv. Exp. Med. Biol. 1402, 69–82 (2023).
Plaas, A. H. K., West, L. A., Wong-Palms, S. & Nelson, F. R. T. Glycosaminoglycan sulfation in human osteoarthritis: disease-related alterations at the non-reducing termini of chondroitin and dermatan sulfate. J. Biol. Chem. 273, 12642–12649 (1998).
Zaucke, F. Cartilage Glycoproteins. Cartilage: Volume 1: Physiology and Development (Springer, 2016).
Hildebrand, A. et al. Interaction of the small interstitial proteoglycans biglycan, decorin and fibromodulin with transforming growth factor β. Biochem. J. 302, 527–534 (1994).
Heinemeier, K. M. et al. Radiocarbon dating reveals minimal collagen turnover in both healthy and osteoarthritic human cartilage. Sci. Transl. Med. 8, 346ra90 (2016).
Wagner, K. R. et al. Osteochondral allograft transplantation for focal cartilage defects of the femoral condyles. JBJS Essent. Surg. Tech. 12, e21.00037 (2022).
Makris, E. A., Gomoll, A. H., Malizos, K. N., Hu, J. C. & Athanasiou, K. A. Repair and tissue engineering techniques for articular cartilage. Nat. Rev. Rheumatol. 11, 21–34 (2015).
Ekser, B. et al. Clinical xenotransplantation: the next medical revolution? Lancet 379, 672–683 (2012).
Merrild, N. G. et al. Local depletion of proteoglycans mediates cartilage tissue repair in an ex vivo integration model. Acta Biomater. 149, 179–188 (2022).
Hulme, C. H. et al. Cell therapy for cartilage repair. Emerg. Top Life Sci. 5, 575–589 (2021).
Dunkin, B. S. & Lattermann, C. New and emerging techniques in cartilage repair: MACI. Oper. Tech. Sports Med. 21, 100 (2013).
Zhao, X. et al. Applications of biocompatible scaffold materials in stem cell-based cartilage tissue engineering. Front. Bioeng. Biotechnol. 9, 603444 (2021).
Hafezi, M., Khorasani, S. N., Zare, M., Neisiany, R. E. & Davoodi, P. Advanced hydrogels for cartilage tissue engineering: recent progress and future directions. Polymers 13, 4199 (2021).
Gao, Y. et al. Injectable and self-crosslinkable hydrogels based on collagen type II and activated chondroitin sulfate for cell delivery. Int. J. Biol. Macromol. 118, 2014–2020 (2018).
Chen, W. C., Wei, Y. H., Chu, I. M. & Yao, C. L. Effect of chondroitin sulphate C on the in vitro and in vivo chondrogenesis of mesenchymal stem cells in crosslinked type II collagen scaffolds. J. Tissue Eng. Regen. Med. 7, 665–672 (2013).
Hsu, S. H. et al. Evaluation of biodegradable polyesters modified by type II collagen and Arg-Gly-Asp as tissue engineering scaffolding materials for cartilage regeneration. Artif. Organs 30, 42–55 (2006).
Groll, J. et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 11, 013001 (2018).
Malda, J. et al. 25th anniversary article: Engineering hydrogels for biofabrication. Adv. Mater. 25, 5011–5028 (2013).
Sbirkov, Y., Redzheb, M., Forraz, N., McGuckin, C. & Sarafian, V. High hopes for the biofabrication of articular cartilage — what lies beyond the horizon of tissue engineering and 3D bioprinting? Biomedicines 12, 665 (2024).
Mouser, V. H. M. et al. Three-dimensional bioprinting and its potential in the field of articular cartilage regeneration. Cartilage 8, 327 (2017).
Levato, R. et al. From shape to function: the next step in bioprinting. Adv. Mater. 32, 1906423 (2020).
Visser, J. et al. Biofabrication of multi-material anatomically shaped tissue constructs. Biofabrication 5, 035007 (2013).
3D extrusion bioprinting. Nat. Rev. Methods Primers 1, 76 (2021).
Gibney, R. & Ferraris, E. Bioprinting of collagen type I and II via aerosol jet printing for the replication of dense collagenous tissues. Front. Bioeng. Biotechnol. 9, 786945 (2021).
Prendergast, M. E. et al. Hybrid printing of mechanically and biologically improved constructs for cartilage tissue engineering applications. Biofabrication 5, 015001 (2012).
Nuñez Bernal, P. et al. Volumetric bioprinting of complex living-tissue constructs within seconds. Adv. Mater. 31, 1904209 (2019).
Murphy, S. V., Skardal, A. & Atala, A. Evaluation of hydrogels for bio-printing applications. J. Biomed. Mater. Res. A 101, 272–284 (2013).
de Ruijter, M. et al. Orthotopic equine study confirms the pivotal importance of structural reinforcement over the pre-culture of cartilage implants. Bioeng. Transl. Med. 9, e10614 (2024).
Dalton, P. D. Melt electrowriting with additive manufacturing principles. Curr. Opin. Biomed. Eng. 2, 49–57 (2017).
Ainsworth, M. J. et al. Convergence of melt electrowriting and extrusion-based bioprinting for vascular patterning of a myocardial construct. Biofabrication 15, 035025 (2023).
Dufour, A. et al. Integrating melt electrowriting and inkjet bioprinting for engineering structurally organized articular cartilage. Biomaterials 283, 121405 (2022).
Größbacher, G. et al. Volumetric printing across melt electrowritten scaffolds fabricates multi-material living constructs with tunable architecture and mechanics. Adv. Mater. 35, 2300756 (2023).
Haigh, J. N., Dargaville, T. R. & Dalton, P. D. Additive manufacturing with polypropylene microfibers. Mater. Sci. Eng. C 77, 883–887 (2017).
Arden, N. & Nevitt, M. C. Osteoarthritis: epidemiology. Best. Pract. Res. Clin. Rheumatol. 20, 3–25 (2006).
Katz, J. N. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J. Bone Jt. Surg. 88, 21–24 (2006).
Ruberti, J. W. & Zieske, J. D. Prelude to corneal tissue engineering — gaining control of collagen organization. Prog. Retin. Eye Res. 27, 549–577 (2008).
Engelmayr, G. C. et al. Accordion-like honeycombs for tissue engineering of cardiac anisotropy. Nat. Mater. 7, 1003–1010 (2008).
Kim, Y. T., Haftel, V. K., Kumar, S. & Bellamkonda, R. V. The role of aligned polymer fiber-based constructs in the bridging of long peripheral nerve gaps. Biomaterials 29, 3117–3127 (2008).
Jia, S. et al. Oriented cartilage extracellular matrix-derived scaffold for cartilage tissue engineering. J. Biosci. Bioeng. 113, 647–653 (2012).
Yang, S. et al. Oriented collagen fiber membranes formed through counter-rotating extrusion and their application in tendon regeneration. Biomaterials 207, 61–75 (2019).
Lee, P., Lin, R., Moon, J. & Lee, L. P. Microfluidic alignment of collagen fibers for in vitro cell culture. Biomed. Microdevices 8, 35–41 (2006).
Torbet, J. et al. Orthogonal scaffold of magnetically aligned collagen lamellae for corneal stroma reconstruction. Biomaterials 28, 4268–4276 (2007).
Hoogenkamp, H. R. et al. Directing collagen fibers using counter-rotating cone extrusion. Acta Biomater. 12, 113–121 (2015).
Lai, E. S., Anderson, C. M. & Fuller, G. G. Designing a tubular matrix of oriented collagen fibrils for tissue engineering. Acta Biomater. 7, 2448–2456 (2011).
Saeidi, N., Sander, E. A., Zareian, R. & Ruberti, J. W. Production of highly aligned collagen lamellae by combining shear force and thin film confinement. Acta Biomater. 7, 2437–2447 (2011).
Saeidi, N., Sander, E. A. & Ruberti, J. W. Dynamic shear-influenced collagen self-assembly. Biomaterials 30, 6581–6592 (2009).
Lanfer, B. et al. Aligned fibrillar collagen matrices obtained by shear flow deposition. Biomaterials 29, 3888–3895 (2008).
Voge, C. M., Kariolis, M., Macdonald, R. A. & Stegemann, J. P. Directional conductivity in SWNT-collagen-fibrin composite biomaterials through strain-induced matrix alignment. J. Biomed. Mater. Res. A 86, 269–277 (2008).
Vader, D., Kabla, A., Weitz, D. & Mahadevan, L. Strain-induced alignment in collagen gels. PLoS ONE 4, 5902 (2009).
Wilson, D. L. et al. Surface organization and nanopatterning of collagen by dip-pen nanolithography. Proc. Natl Acad. Sci. USA 98, 13660 (2001).
Lin, J. et al. Mechanical roles in formation of oriented collagen fibers. Tissue Eng. Part B Rev. 26, 116–128 (2020).
Wakuda, Y., Nishimoto, S., Suye, S. I. & Fujita, S. Native collagen hydrogel nanofibres with anisotropic structure using core-shell electrospinning. Sci. Rep. 8, 6248 (2018).
Matthews, J. A., Wnek, G. E., Simpson, D. G. & Bowlin, G. L. Electrospinning of collagen nanofibers. Biomacromolecules 3, 232–238 (2002).
Caves, J. M. et al. Fibrillogenesis in continuously spun synthetic collagen fiber. J. Biomed. Mater. Res. B Appl. Biomater. 93, 24–38 (2010).
Betsch, M. et al. Incorporating 4D into bioprinting: real-time magnetically directed collagen fiber alignment for generating complex multilayered tissues. Adv. Healthc. Mater. 7, 1800894 (2018).
Cheng, X. et al. An electrochemical fabrication process for the assembly of anisotropically oriented collagen bundles. Biomaterials 29, 3278–3288 (2008).
Denis, F. A., Pallandre, A., Nysten, B., Jonas, A. M. & Dupont-Gillain, C. C. Alignment and assembly of adsorbed collagen molecules induced by anisotropic chemical nanopatterns. Small 1, 984–991 (2005).
Puetzer, J. L., Ma, T., Sallent, I., Gelmi, A. & Stevens, M. M. Driving hierarchical collagen fiber formation for functional tendon, ligament, and meniscus replacement. Biomaterials 269, 120527 (2021).
Bates, M. E., Troop, L., Brown, M. E. & Puetzer, J. L. Temporal application of lysyl oxidase during hierarchical collagen fiber formation differentially effects tissue mechanics. Acta Biomater. 160, 98–111 (2023).
Gonzalez-Leon, E. A., Bielajew, B. J., Hu, J. C. & Athanasiou, K. A. Engineering self-assembled neomenisci through combination of matrix augmentation and directional remodeling. Acta Biomater. 109, 73–81 (2020).
Blaschke, U. K., Eikenberry, E. F., Hulmes, D. J. S., Galla, H. J. & Bruckner, P. Collagen XI nucleates self-assembly and limits lateral growth of cartilage fibrils. J. Biol. Chem. 275, 10370–10378 (2000).
Ciferri, A. On collagen II fibrillogenesis. Liq. Cryst. 34, 693–696 (2007).
Kock, L., van Donkelaar, C. C. & Ito, K. Tissue engineering of functional articular cartilage: the current status. Cell Tissue Res. 347, 613–627 (2012).
Elder, B. D. & Athanasiou, K. A. Effects of confinement on the mechanical properties of self-assembled articular cartilage constructs in the direction orthogonal to the confinement surface. J. Orthop. Res. 26, 238–246 (2008).
Kock, L. M. et al. Tuning the differentiation of periosteum-derived cartilage using biochemical and mechanical stimulations. Osteoarthritis Cartilage 18, 1528–1535 (2010).
Lee, J. K. et al. Tension stimulation drives tissue formation in scaffold-free systems. Nat. Mater. 16, 864–873 (2017).
Visser, J. et al. Reinforcement of hydrogels using three-dimensionally printed microfibres. Nat. Commun. 6, 6933 (2015).
Pilipchuk, S. P. et al. Integration of 3D printed and micropatterned polycaprolactone scaffolds for guidance of oriented collagenous tissue formation in vivo. Adv. Healthc. Mater. 5, 676–687 (2016).
Daly, A. C. & Kelly, D. J. Biofabrication of spatially organised tissues by directing the growth of cellular spheroids within 3D printed polymeric microchambers. Biomaterials 197, 194–206 (2019).
Liu, H. et al. Filamented light (FLight) biofabrication of highly aligned tissue-engineered constructs. Adv. Mater. 34, e2204301 (2022).
Puiggalí-Jou, A. et al. FLight biofabrication supports maturation of articular cartilage with anisotropic properties. Adv. Healthc. Mater. 13, 2302179 (2024).
Braunmiller, D. L. et al. Pre-programmed rod-shaped microgels to create multi-directional anisogels for 3D tissue engineering. Adv. Funct. Mater. 32, 2202430 (2022).
Wang, B., Chariyev-Prinz, F., Burdis, R., Eichholz, K. & Kelly, D. J. Additive manufacturing of cartilage-mimetic scaffolds as off-the-shelf implants for joint regeneration. Biofabrication 14, 024101 (2022).
Browe, D. C. et al. Bilayered extracellular matrix derived scaffolds with anisotropic pore architecture guide tissue organization during osteochondral defect repair. Acta Biomater. 143, 266–281 (2022).
Nordberg, R. C. et al. Recent advancements in cartilage tissue engineering innovation and translation. Nat. Rev. Rheumatol. 20, 323–346 (2024).
Zhang, Y., Pizzute, T. & Pei, M. Anti-inflammatory strategies in cartilage repair. Tissue Eng. Part. B Rev. 20, 655–668 (2014).
Goldring, M. B., Otero, M., Tsuchimochi, K., Ijiri, K. & Li, Y. Defining the roles of inflammatory and anabolic cytokines in cartilage metabolism. Ann. Rheum. Dis. 67, iii75–iii82 (2008).
Chen, D. et al. Versican binds collagen via its G3 domain and regulates the organization and mechanics of collagenous matrices. J. Biol. Chem. 300, 107968 (2024).
Choocheep, K. et al. Versican facilitates chondrocyte differentiation and regulates joint morphogenesis. J. Biol. Chem. 285, 21114 (2010).
Peng, Z. et al. The regulation of cartilage extracellular matrix homeostasis in joint cartilage degeneration and regeneration. Biomaterials 268, 120555 (2021).
Guilak, F., Hayes, A. J. & Melrose, J. Perlecan in pericellular mechanosensory cell-matrix communication, extracellular matrix stabilisation and mechanoregulation of load-bearing connective tissues. Int. J. Mol. Sci. 22, 1–20 (2021).
Xu, X., Li, Z., Leng, Y., Neu, C. P. & Calve, S. Knockdown of the pericellular matrix molecule perlecan lowers in situ cell and matrix stiffness in developing cartilage. Dev. Biol. 418, 242–247 (2016).
Flowers, S. A. et al. Lubricin binds cartilage proteins, cartilage oligomeric matrix protein, fibronectin and collagen II at the cartilage surface. Sci Rep 7, 13149 (2017).
Jay, G. D. & Waller, K. A. The biology of lubricin: near frictionless joint motion. Matrix Biol. 39, 17–24 (2014).
Zappone, B., Ruths, M., Greene, G. W., Jay, G. D. & Israelachvili, J. N. Adsorption, lubrication, and wear of lubricin on model surfaces: polymer brush-like behavior of a glycoprotein. Biophys. J. 92, 1693–1708 (2007).
Rhee, D. K. et al. The secreted glycoprotein lubricin protects cartilage surfaces and inhibits synovial cell overgrowth. J. Clin. Invest. 115, 622–631 (2005).
Kalamajski, S., Bihan, D., Bonna, A., Rubin, K. & Farndale, R. W. Fibromodulin interacts with collagen cross-linking sites and activates lysyl oxidase. J. Biol. Chem. 291, 7951–7960 (2016).
Li, C. et al. Fibromodulin — a new target of osteoarthritis management? Front. Pharmacol. 10, 492043 (2019).
Barreto, G. et al. Lumican is upregulated in osteoarthritis and contributes to TLR4-induced pro-inflammatory activation of cartilage degradation and macrophage polarization. Osteoarthritis Cartilage 28, 92–101 (2020).
Kafienah, W. et al. Lumican inhibits collagen deposition in tissue engineered cartilage. Matrix Biol. 27, 526–534 (2008).
Haglund, L. et al. Identification and characterization of the integrin alpha2beta1 binding motif in chondroadherin mediating cell attachment. J. Biol. Chem. 286, 3925–3934 (2011).
Hessle, L. et al. The skeletal phenotype of chondroadherin deficient mice. PLoS ONE 8, 63080 (2013).
Bengtsson, E. et al. The leucine-rich repeat protein PRELP binds perlecan and collagens and may function as a basement membrane anchor. J. Biol. Chem. 277, 15061–15068 (2002).
Klatt, A. R., Becker, A. K. A., Neacsu, C. D., Paulsson, M. & Wagener, R. The matrilins: modulators of extracellular matrix assembly. Int. J. Biochem. Cell Biol. 43, 320–330 (2011).
Calabro, N. E. et al. Thrombospondin-2 regulates extracellular matrix production, LOX levels, and cross-linking via downregulation of miR-29. Matrix Biol. 82, 71 (2019).
Halász, K., Kassner, A., Mörgelin, M. & Heinegård, D. COMP acts as a catalyst in collagen fibrillogenesis. J. Biol. Chem. 282, 31166–31173 (2007).
Rosenberg, K., Olsson, H., Mörgelin, M. & Heinegård, D. Cartilage oligomeric matrix protein shows high affinity zinc-dependent interaction with triple helical collagen. J. Biol. Chem. 273, 20397–20403 (1998).
Danalache, M., Erler, A. L., Wolfgart, J. M., Schwitalle, M. & Hofmann, U. K. Biochemical changes of the pericellular matrix and spatial chondrocyte organization — two highly interconnected hallmarks of osteoarthritis. J. Orthop. Res. 38, 2170–2180 (2020).
Yu, J. & Urban, J. P. G. The elastic network of articular cartilage: an immunohistochemical study of elastin fibres and microfibrils. J. Anat. 216, 533 (2010).
Ramanayake, W. et al. Fibrillin-1 expression, which regulates of TGF-B bioavailability, is modified during osteoarthritis and mutations lead to osteoarthritis. Osteoarthritis Cartilage 22, S141 (2014).
He, B., Wu, J. P., Chen, H. H., Kirk, T. B. & Xu, J. Elastin fibers display a versatile microfibril network in articular cartilage depending on the mechanical microenvironments. J. Orthop. Res. 31, 1345–1353 (2013).
Mansfield, J. et al. The elastin network: its relationship with collagen and cells in articular cartilage as visualized by multiphoton microscopy. J. Anat. 215, 682–691 (2009).
Pacifici, M. Tenascin-C and the development of articular cartilage. Matrix Biol. 14, 689–698 (1995).
Leiss, M., Beckmann, K., Girós, A., Costell, M. & Fässler, R. The role of integrin binding sites in fibronectin matrix assembly in vivo. Curr. Opin. Cell Biol. 20, 502–507 (2008).
Maylin, A. B. et al. Genetic abrogation of the fibronectin-α5β1 integrin interaction in articular cartilage aggravates osteoarthritis in mice. PLoS ONE 13, e0198559 (2018).
Sun, Y., Wang, T. L., Toh, W. S. & Pei, M. The role of laminins in cartilaginous tissues: from development to regeneration. Eur. Cell Mater. 34, 40 (2017).
Schminke, B., Frese, J., Bode, C., Goldring, M. B. & Miosge, N. Laminins and nidogens in the pericellular matrix of chondrocytes: their role in osteoarthritis and chondrogenic differentiation. Am. J. Pathol. 186, 410–418 (2016).
Batsalova, T. & Dzhambazov, B. Significance of type II collagen posttranslational modifications: from autoantigenesis to improved diagnosis and treatment of rheumatoid arthritis. Int. J. Mol. Sci. 24, 9884 (2023).
Acknowledgements
A.P.M., M.d.R., K.I. and J.M. would like to acknowledge the support of the Dutch Research Council (NWO), project LS-NeoCarE (NWA.1389.20.192) and the Gravitation Program “Materials Driven Regeneration” (024.003.013). J.M. would like to acknowledge support from the European Research Council (ERC) under the European Union’s Horizon Europe research and innovation programme (Re-COLL: 101142063). F.Z. would like to acknowledge funding from the Deutsche Forschungsgemeinschaft (ZA 561/3-2, project number 407168728) within the research unit FOR2722. D.J.K. would like to acknowledge support from Science Foundation Ireland (22/FFP-A/11042) and the European Research Council (ERC) under the European Union’s Horizon Europe research and innovation programme (4D BOUNDARIES: 101019344). J.M. and D.J.K. would like to acknowledge the European Commission (m2M: 101191729).
Author information
Authors and Affiliations
Contributions
A.P.M. researched data for the article. A.P.M., J.M. and M.d.R. wrote the article. All authors contributed substantially to discussion of the content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks Daniel Grande and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Anisotropic
-
Having direction-dependent properties.
- Arcade-like structure
-
A collagen fibre arrangement in articular cartilage whereby fibres curve from the surface to the deep zone, anchoring to the subchondral bone.
- Beaded microfibrillar network
-
A structural arrangement of thin, bead-like fibrils connected in a network.
- Fibrillogenesis
-
The process of collagen fibril formation.
- Heterotypic fibres
-
Collagen fibres composed of more than one type of collagen molecule.
- Hypertrophy
-
The process by which cells undergo significant enlargement owing to volumetric increase and distinct metabolic and molecular changes.
- Isotropic
-
Having uniform properties in all directions.
- Joint cavitation
-
The formation of the joint cavity during embryonic development, separating the joint into two articular surfaces.
- Liquid crystallinity
-
A state of matter with properties between those of a liquid and a solid crystal, characterized by ordered molecular alignment.
- Molecular crowding
-
A high concentration of macromolecules in a confined space that influences molecular interactions and assembly.
- Nascent
-
Newly formed or immature.
- Non-reducible crosslinks
-
A non-reversible chemical bond that permanently stabilizes collagen fibrils.
- Nucleator
-
A molecule or structure that promotes the initiation of fibrillogenesis.
- Procollagen
-
The precursor molecule of collagen that undergoes enzymatic processing to form tropocollagen.
- Reducible crosslinks
-
A reversible chemical bond that stabilizes collagen fibrils.
- Resistance to the positive principal strain
-
The ability of a material to withstand tensile forces along the direction of the highest principal strain.
- Split-line orientation pattern
-
A pattern observed on the surface of cartilage that reflects the underlying collagen fibre alignment.
- Suprafibrillar assemblies
-
Higher-order structures formed by the organization of fibrils into complex networks.
- Tidemark
-
Boundary between the calcified and non-calcified zones of articular cartilage.
- Tropocollagen
-
The basic triple-helical collagen molecule that assembles into fibrils.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pueyo Moliner, A., Ito, K., Zaucke, F. et al. Restoring articular cartilage: insights from structure, composition and development. Nat Rev Rheumatol 21, 291–308 (2025). https://doi.org/10.1038/s41584-025-01236-7
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41584-025-01236-7
This article is cited by
-
Analysis of cartilage creep-recovery of human osteoarthritic cartilage
European Journal of Medical Research (2026)
-
Smart biomaterials for skeletal aging repair and regeneration
Bone Research (2026)
-
Zone-inspired hydrogel constructs promote spatially controlled chondrogenesis for osteochondral regeneration
Scientific Reports (2025)