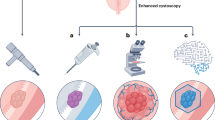
Abstract
Bladder cancer remains a major global health challenge, characterized by diagnostic uncertainty, substantial treatment costs and high recurrence rates. Current diagnostic and treatment modalities, including cystoscopy, transurethral resection of bladder tumour and standard histopathology, have limitations, including the inability to detect flat lesions, frequent understaging and interobserver variability, highlighting a crucial need for improved approaches. Advances in artificial intelligence (AI), blue-light cystoscopy, narrow-band imaging, cytology and urinary markers show promise in enhancing early detection and diagnosis. Developments in multiparametric MRI, radiomics, genomics and AI-driven algorithms for histopathological analyses have demonstrated considerable improvements in staging and risk stratification of bladder tumours, enabling personalized therapy selection and prognostication. Despite these promising developments, challenges remain regarding standardization, external validation, cost-effectiveness and ethical considerations in clinical implementation. Future research should prioritize addressing these barriers through collaborative, multi-institutional studies and robust validation frameworks. Ultimately, adopting a comprehensive multimodal strategy, such as proposed, novel, multimodal decision-making frameworks in which these advances and technologies are integrated, promises to considerably advance precision oncology in bladder cancer, improving patient outcomes and reducing health care burdens.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 71, 209–249 (2021).
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer statistics, 2022. CA. Cancer J. Clin. 72, 7–33 (2022).
van Osch, F. H. M., Jochems, S. H., Schooten, F.-J., Bryan, R. T. & Zeegers, M. P. Quantified relations between exposure to tobacco smoking and bladder cancer risk: a meta-analysis of 89 observational studies. Int. J. Epidemiol. 45, 857–870 (2016).
Cumberbatch, M. G. K., Cox, A., Teare, D. & Catto, J. W. F. Contemporary occupational carcinogen exposure and bladder cancer. JAMA Oncol. 1, 1282–1290 (2015).
Huang, Y.-K. et al. Arsenic exposure, urinary arsenic speciation, and the incidence of urothelial carcinoma: a twelve-year follow-up study. Cancer Causes Control. 19, 829–839 (2008).
Saginala, K. et al. Epidemiology of bladder cancer. Med. Sci. 8, 15 (2020).
Kamat, A. M. et al. Bladder cancer. Lancet 388, 2796–2810 (2016).
Flaig, T. W. et al. NCCN Guidelines® insights: bladder cancer, version 2.2022. J. Natl. Compr. Cancer Netw. 20, 866–878 (2022).
Dyrskjøt, L. et al. Bladder cancer. Nat. Rev. Dis. Primer 9, 58 (2023).
Lobo, N. et al. Epidemiology, screening, and prevention of bladder cancer. Eur. Urol. Oncol. 5, 628–639 (2022).
Cheng, L. et al. Bladder Pathology. (Wiley-Blackwell,2012).
Babjuk, M. et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (Ta, T1, and Carcinoma in Situ). Eur. Urol. 81, 75–94 (2022).
Holzbeierlein, J. M. et al. Diagnosis and treatment of non-muscle invasive bladder cancer: AUA/SUO guideline: 2024 amendment. J. Urol. 211, 533–538 (2024).
Mohanty, S. K., Lobo, A., Mishra, S. K. & Cheng, L. Precision medicine in bladder cancer: present challenges and future directions. J. Pers. Med. 13, 756 (2023).
Stein, J. P. et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J. Clin. Oncol. 19, 666–675 (2001).
Lenis, A. T., Lec, P. M., Chamie, K. & Mshs, M. D. Bladder cancer: a review. JAMA 324, 1980–1991 (2020).
Botteman, M. F., Pashos, C. L., Redaelli, A., Laskin, B. & Hauser, R. The health economics of bladder cancer: a comprehensive review of the published literature. Pharmacoeconomics 21, 1315–1330 (2003).
Svatek, R. S. et al. The economics of bladder cancer: costs and considerations of caring for this disease. Eur. Urol. 66, 253–262 (2014).
Yeung, C., Dinh, T. & Lee, J. The health economics of bladder cancer: an updated review of the published literature. Pharmacoeconomics 32, 1093–1104 (2014).
Lopez-Beltran, A., Cookson, M. S., Guercio, B. J. & Cheng, L. Advances in diagnosis and treatment of bladder cancer. BMJ 384, e076743 (2024).
Witjes, J. A. Management of BCG failures in superficial bladder cancer: a review. Eur. Urol. 49, 790–797 (2006).
Pfail, J. L., Small, A. C., Cumarasamy, S. & Galsky, M. D. Real world outcomes of patients with bladder cancer: effectiveness versus efficacy of modern treatment paradigms. Hematol. Oncol. Clin. North. Am. 35, 597–612 (2021).
Konieczkowski, D. J., Efstathiou, J. A. & Mouw, K. W. Contemporary and emerging approaches to bladder-preserving trimodality therapy for muscle-invasive bladder cancer. Hematol. Oncol. Clin. North. Am. 35, 567–584 (2021).
Venkatramani, V. & Parekh, D. J. Surgery for bladder and upper tract urothelial cancer. Hematol. Oncol. Clin. North. Am. 35, 543–566 (2021).
Sonpavde, G. P., Mouw, K. W. & Mossanen, M. Therapy for muscle-invasive urothelial carcinoma: controversies and dilemmas. J. Clin. Oncol. 40, 1275–1280 (2022).
Knoedler, J. J. et al. Does partial cystectomy compromise oncologic outcomes for patients with bladder cancer compared to radical cystectomy? A matched case-control analysis. J. Urol. 188, 1115–1119 (2012).
Holzbeierlein, J. et al. Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/SUO guideline (2017; Amended 2020, 2024). J. Urol. 212, 3–10 (2024).
Alfred Witjes, J. et al. European Association of Urology guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2023 guidelines. Eur. Urol. 85, 17–31 (2024).
Grossman, H. B. et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N. Engl. J. Med. 349, 859–866 (2003).
Flaig, T. W. et al. NCCN Guidelines® insights: bladder cancer, version 3.2024. J. Natl. Compr. Cancer Netw. 22, 216–225 (2024).
Powles, T. et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. N. Engl. J. Med. 390, 875–888 (2024).
Powles, T. et al. Avelumab maintenance therapy for advanced or metastatic urothelial carcinoma. N. Engl. J. Med. 383, 1218–1230 (2020).
FDA approves nivolumab in combination with cisplatin and gemcitabine for unresectable or metastatic urothelial carcinoma. FDA https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-nivolumab-combination-cisplatin-and-gemcitabine-unresectable-or-metastatic-urothelial (2024).
van der Heijden, M. S. et al. Nivolumab plus gemcitabine-cisplatin in advanced urothelial carcinoma. N. Engl. J. Med. 389, 1778–1789 (2023).
Mowatt, G. et al. Photodynamic diagnosis of bladder cancer compared with white light cystoscopy: systematic review and meta-analysis. Int. J. Technol. Assess. Health Care 27, 3–10 (2011).
Burger, M. et al. Photodynamic diagnosis of non-muscle-invasive bladder cancer with hexaminolevulinate cystoscopy: a meta-analysis of detection and recurrence based on raw data. Eur. Urol. 64, 846–854 (2013).
Jocham, D. et al. Improved detection and treatment of bladder cancer using hexaminolevulinate imaging: a prospective, phase III multicenter study. J. Urol. 174, 862–866 (2005). discussion 866.
LeCun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Shkolyar, E. et al. Optimizing cystoscopy and TURBT: enhanced imaging and artificial intelligence. Nat. Rev. Urol. 22, 46–54 (2025).
Borhani, S., Borhani, R. & Kajdacsy-Balla, A. Artificial intelligence: a promising frontier in bladder cancer diagnosis and outcome prediction. Crit. Rev. Oncol. Hematol. 171, 103601 (2022).
Ma, X. et al. Artificial intelligence application in the diagnosis and treatment of bladder cancer: advance, challenges, and opportunities. Front. Oncol. 14, 1487676 (2024).
Bera, K., Schalper, K. A., Rimm, D. L., Velcheti, V. & Madabhushi, A. Artificial intelligence in digital pathology — new tools for diagnosis and precision oncology. Nat. Rev. Clin. Oncol. 16, 703–715 (2019).
Lotter, W. et al. Artificial intelligence in oncology: current landscape, challenges, and future directions. Cancer Discov. 14, 711–726 (2024).
Haug, C. J. & Drazen, J. M. Artificial intelligence and machine learning in clinical medicine, 2023. N. Engl. J. Med. 388, 1201–1208 (2023).
Ascione, C. M. et al. Role of FGFR3 in bladder cancer: treatment landscape and future challenges. Cancer Treat. Rev. 115, 102530 (2023).
Guancial, E. A. & Rosenberg, J. E. The role of genomics in the management of advanced bladder cancer. Curr. Treat. Options Oncol. 16, 319 (2015).
Jia, E. & Agarwal, G. Clinical utility of liquid biopsy in bladder cancer: the beginning of a new era. J. Liq. Biopsy 6, 100271 (2024).
de Jong, F. C., Rutten, V. C., Zuiverloon, T. C. M. & Theodorescu, D. Improving anti-PD-1/PD-L1 therapy for localized bladder cancer. Int. J. Mol. Sci. 22, 2800 (2021).
Boca, B. et al. MRI-based radiomics in bladder cancer: a systematic review and radiomics quality score assessment. Diagnostics 13, 2300 (2023).
Huang, S. et al. Accuracy of the vesical imaging–reporting and data system (VIRADS) for pre-treatment staging of bladder cancer in an Australian cohort. J. Med. Imaging Radiat. Oncol. 66, 370–376 (2022).
Panebianco, V. et al. Multiparametric magnetic resonance imaging for bladder cancer: development of VI-RADS (Vesical Imaging-Reporting And Data System). Eur. Urol. 74, 294–306 (2018).
Messing, E. M. et al. Long-term outcome of hematuria home screening for bladder cancer in men. Cancer 107, 2173–2179 (2006).
Moyer, V. A. & on behalf of the U.S. preventive services task force. screening for bladder cancer: U.S. preventive services task force recommendation statement. Ann. Intern. Med. 155, 246–251 (2011).
Larré, S. et al. Screening for bladder cancer: rationale, limitations, whom to target, and perspectives. Eur. Urol. 63, 1049–1058 (2013).
Devlies, W. et al. The diagnostic accuracy of cystoscopy for detecting bladder cancer in adults presenting with haematuria: a systematic review from the European Association of Urology guidelines office. Eur. Urol. Focus. 10, 115–122 (2024).
Jubber, I. et al. Non-visible haematuria for the detection of bladder, upper tract, and kidney cancer: an updated systematic review and meta-analysis. Eur. Urol. 77, 583–598 (2020).
Khadhouri, S. et al. Developing a diagnostic multivariable prediction model for urinary tract cancer in patients referred with haematuria: results from the identify collaborative study. Eur. Urol. Focus. 8, 1673–1682 (2022).
Lander, E. S. et al. Initial sequencing and analysis of the human genome. Nature 409, 860–921 (2001).
Zhao, E. Y., Jones, M. & Jones, S. J. M. Whole-genome sequencing in cancer. Cold Spring Harb. Perspect. Med. 9, a034579 (2019).
Yu, E. Y.-W. et al. Genome-wide exploration of genetic interactions for bladder cancer risk. Int. J. Cancer 154, 81–93 (2024).
Selinski, S. Discovering urinary bladder cancer risk variants: status quo after almost ten years of genome-wide association studies. EXCLI J. 16, 1288–1296 (2017).
Lai, C. et al. Screening model for bladder cancer early detection with serum miRNAs based on machine learning: a mixed-cohort study based on 16,189 participants. Cancer Med. 13, e70338 (2024).
Demir, R. et al. Artificial intelligence assisted patient blood and urine droplet pattern analysis for non-invasive and accurate diagnosis of bladder cancer. Sci. Rep. 14, 2488 (2024).
Shkolyar, E. et al. Augmented bladder tumor detection using deep learning. Eur. Urol. 76, 714–718 (2019).
Wu, S. et al. An artificial intelligence system for the detection of bladder cancer via cystoscopy: a multicenter diagnostic study. J. Natl. Cancer Inst. 114, 220–227 (2022).
Ikeda, A. et al. Support system of cystoscopic diagnosis for bladder cancer based on artificial intelligence. J. Endourol. 34, 352–358 (2020).
Du, Y. et al. A deep learning network-assisted bladder tumour recognition under cystoscopy based on caffe deep learning framework and easyDL platform. Int. J. Med. Robot. Comput. Assist. Surg. 17, 1–8 (2021).
Freitas, N. R. et al. Detection of bladder cancer with feature fusion, transfer learning and CapsNets. Artif. Intell. Med. 126, 102275 (2022).
Eminaga, O., Eminaga, N., Semjonow, A. & Breil, B. Diagnostic classification of cystoscopic images using deep convolutional neural networks. JCO Clin. Cancer Inform. 2, 1–8 (2018).
Lorencin, I., Anđelić, N., Španjol, J. & Car, Z. Using multi-layer perceptron with Laplacian edge detector for bladder cancer diagnosis. Artif. Intell. Med. 102, 101746 (2020).
Yang, R. et al. Automatic recognition of bladder tumours using deep learning technology and its clinical application. Int. J. Med. Robot. Comput. Assist. Surg. 17, e2194 (2021).
Lebret, T. et al. Artificial intelligence to improve cytology performance in urothelial carcinoma diagnosis: results from validation phase of the French, multicenter, prospective VISIOCYT1 trial. World J. Urol. 41, 2381–2388 (2023).
Eminaga, O. et al. An efficient framework for video documentation of bladder lesions for cystoscopy: a proof-of-concept study. J. Med. Syst. 46, 73 (2022).
Eminaga, O. et al. Conceptual framework and documentation standards of cystoscopic media content for artificial intelligence. J. Biomed. Inform. 142, 104369 (2023).
Kriegmair, M. C. et al. Digital mapping of the urinary bladder: potential for standardized cystoscopy reports. Urology 104, 235–241 (2017).
Soper, T. D., Porter, M. P. & Seibel, E. J. Surface mosaics of the bladder reconstructed from endoscopic video for automated surveillance. IEEE Trans. Biomed. Eng. 59, 1670–1680 (2012).
Lurie, K. L., Angst, R., Zlatev, D. V., Liao, J. C. & Ellerbee Bowden, A. K. 3D reconstruction of cystoscopy videos for comprehensive bladder records. Biomed. Opt. Express 8, 2106–2123 (2017).
Ben-Hamadou, A., Daul, C. & Soussen, C. Construction of extended 3D field of views of the internal bladder wall surface: a proof of concept. 3D Res. 7, 19 (2016).
Mutaguchi, J. et al. Artificial intelligence for segmentation of bladder tumor cystoscopic images performed by U-Net with dilated convolution. J. Endourol 36, 827–834 (2022).
Ronneberger, O., Fischer, P., Brox, T. U-Net: convolutional Networks for biomedical image segmentation. in Medical Image Computing and Computer-Assisted Intervention — MICCI 2015 (eds Navab, N. et al.) 234–241 (Springer, 2015).
Yoo, J. W. et al. Deep learning diagnostics for bladder tumor identification and grade prediction using RGB method. Sci. Rep. 12, 17699 (2022).
Varnyú, D. & Szirmay-Kalos, L. A comparative study of deep neural networks for real-time semantic segmentation during the transurethral resection of bladder tumours. Diagnostics 12, 2849 (2022).
Zhang, Q. et al. A comparative study of attention mechanism based deep learning methods for bladder tumor segmentation. Int. J. Med. Inf. 171, 104984 (2023).
Jia, X. et al. Tumor detection under cystoscopy with transformer-augmented deep learning algorithm. Phys. Med. Biol. 68, 165013 (2023).
Hwang, W. K. et al. Artificial intelligence-based classification and segmentation of bladder cancer in cystoscope images. Cancers 17, 57 (2025).
Lazo, J. F. et al. Semi-supervised bladder tissue classification in multi-domain endoscopic images. IEEE Trans. Biomed. Eng. 70, 2822–2833 (2023).
Dilmaghani, S. & Coelho-Prabhu, N. Role of artificial intelligence in colonoscopy: a literature review of the past, present, and future directions. Tech. Innov. Gastrointest. Endosc. 25, 399–412 (2023).
Chadebecq, F., Lovat, L. B. & Stoyanov, D. Artificial intelligence and automation in endoscopy and surgery. Nat. Rev. Gastroenterol. Hepatol. 20, 171–182 (2023).
Urban, G. et al. Deep learning localizes and identifies polyps in real time with 96% accuracy in screening colonoscopy. Gastroenterology 155, 1069–1078.e8 (2018).
Wang, P. et al. Real-time automatic detection system increases colonoscopic polyp and adenoma detection rates: a prospective randomised controlled study. Gut 68, 1813–1819 (2019).
Liu, X., Cruz Rivera, S., Moher, D., Calvert, M. J. & Denniston, A. K. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Nat. Med. 26, 1364–1374 (2020).
Messmann, H. et al. Expected value of artificial intelligence in gastrointestinal endoscopy: European Society of Gastrointestinal Endoscopy (ESGE) position statement. Endoscopy 54, 1211–1231 (2022).
Rivera, S. C. et al. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Nat. Med. 26, 1351–1363 (2020).
Borgli, H. et al. HyperKvasir, a comprehensive multi-class image and video dataset for gastrointestinal endoscopy. Sci. Data 7, 283 (2020).
US Department of Health and Human Services. Premarket Approval (PMA) https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpma/pma.cfm?id=P050027S028 (2002).
Krieg, R. C., Messmann, H., Rauch, J., Seeger, S. & Knuechel, R. Metabolic characterization of tumor cell–specific protoporphyrin IX accumulation after exposure to 5-aminolevulinic acid in human colonic cells. Photochem. Photobiol. 76, 518–525 (2002).
Lange, N. et al. Photodetection of early human bladder cancer based on the fluorescence of 5-aminolaevulinic acid hexylester-induced protoporphyrin IX: a pilot study. Br. J. Cancer 80, 185–193 (1999).
Daneshmand, S. et al. Blue light cystoscopy for the diagnosis of bladder cancer: results from the US prospective multicenter registry. Urol. Oncol. 36, 361.e1–361.e6 (2018).
Chappidi, M. R. et al. Utility of blue light cystoscopy for post-bacillus Calmette-Guérin bladder cancer recurrence detection: implications for clinical trial recruitment and study comparisons. J. Urol. 207, 534–540 (2022).
Kriegmair, M. et al. Detection of early bladder cancer by 5-aminolevulinic acid induced porphyrin fluorescence. J. Urol. 155, 105–109 (1996).
Grossman, H. B. et al. Long-term decrease in bladder cancer recurrence with hexaminolevulinate enabled fluorescence cystoscopy. J. Urol. 188, 58–62 (2012).
Veeratterapillay, R. et al. Time to turn on the blue lights: a systematic review and meta-analysis of photodynamic diagnosis for bladder cancer. Eur. Urol. Open. Sci. 31, 17–27 (2021).
Maisch, P. et al. Blue versus white light for transurethral resection of non-muscle invasive bladder cancer. Cochrane Database Syst. Rev. 12, CD013776 (2021).
Ontario Health (Quality) Enhanced visualization methods for first transurethral resection of bladder tumour in suspected non-muscle-invasive bladder cancer: recommendation. Ont. Health Technol. Assess. Ser. 21, 1–123 (2021).
Sari Motlagh, R. et al. Impact of enhanced optical techniques at time of transurethral resection of bladder tumour, with or without single immediate intravesical chemotherapy, on recurrence rate of non-muscle-invasive bladder cancer: a systematic review and network meta-analysis of randomized trials. BJU Int. 128, 280–289 (2021).
Zhao, H. et al. Comparison of hexaminolevulinate (HAL) -guided versus white light transurethral resection for NMIBC: a systematic review and meta-analysis of randomized controlled trials. Photodiagnosis Photodyn. Ther. 41, 103220 (2023).
Maisch, P. et al. Blue vs white light for transurethral resection of non-muscle-invasive bladder cancer: an abridged Cochrane review. BJU Int. 130, 730–740 (2022).
Heer, R. et al. A randomized trial of PHOTOdynamic surgery in non–muscle-invasive bladder cancer. NEJM Evid. 1, EVIDoa2200092 (2022).
Garfield, S. S., Gavaghan, M. B., Armstrong, S. O. & Jones, J. S. The cost-effectiveness of blue light cystoscopy in bladder cancer detection: United States projections based on clinical data showing 4.5 years of follow up after a single hexaminolevulinate hydrochloride instillation. Can. J. Urol. 20, 6682–6689 (2013).
Witjes, J. A. et al. Clinical and cost effectiveness of hexaminolevulinate-guided blue-light cystoscopy: evidence review and updated expert recommendations. Eur. Urol. 66, 863–871 (2014).
Babjuk, M. et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (TaT1 and Carcinoma In Situ) — 2019 update. Eur. Urol. 76, 639–657 (2019).
Klaassen, Z. et al. Contemporary cost-consequence analysis of blue light cystoscopy with hexaminolevulinate in non-muscle-invasive bladder cancer. Can. Urol. Assoc. J. 11, 173–181 (2017).
Williams, S. B., Gavaghan, M. B., Fernandez, A., Daneshmand, S. & Kamat, A. M. Macro and microeconomics of blue light cystoscopy with CYSVIEW® in non-muscle invasive bladder cancer. Urol. Oncol. 40, 10.e7–10.e12 (2022).
Klaassen, Z. SUO 2021: Economic outcomes of hexaminolevulinate blue-light cystoscopy compared with white light cystoscopy for diagnosis and monitoring of non-muscle invasive bladder cancer: a 5-year, Medicare-based model. UroToday https://www.urotoday.com/conference-highlights/suo-2021/suo-2021-bladder-cancer/134174-suo-2021-economic-outcomes-of-hexaminolevulinate-blue-light-cystoscopy-compared-with-white-light-cystoscopy-for-diagnosis-and-monitoring-of-non-muscle-invasive-bladder-cancer-a-5-year-medicare-based-model.html (2021).
Ali, N. Deep learning-based classification of blue light cystoscopy imaging during transurethral resection of bladder tumors. Sci. Rep. 11, 11629 (2021).
Gono, K. et al. Appearance of enhanced tissue features in narrow-band endoscopic imaging. J. Biomed. Opt. 9, 568–577 (2004).
Naselli, A. et al. A randomized prospective trial to assess the impact of transurethral resection in narrow band imaging modality on non–muscle-invasive bladder cancer recurrence. Eur. Urol. 61, 908–913 (2012).
Li, K., Lin, T., Fan, X., Duan, Y. & Huang, J. Diagnosis of narrow-band imaging in non-muscle-invasive bladder cancer: a systematic review and meta-analysis. Int. J. Urol. 20, 602–609 (2013).
Naito, S. et al. The clinical research office of the endourological society (CROES) multicentre randomised trial of narrow band imaging-assisted transurethral resection of bladder tumour (TURBT) versus conventional white light imaging-assisted TURBT in primary non-muscle-invasive bladder cancer patients: trial protocol and 1-year results. Eur. Urol. 70, 506–515 (2016).
Ikeda, A. et al. PD26-02 real-time bladder tumor detection at clinics in flexible cystoscopy with white light and narrow band imaging using deep learning. J. Urol. 207 (2022).
Kamphuis, G. M. et al. Storz professional image enhancement system: a new technique to improve endoscopic bladder imaging. J. Cancer Sci. Ther. https://doi.org/10.4172/1948-5956.1000394 (2016).
Howard, J. M., Woldu, S. L., Daneshmand, S. & Lotan, Y. Enhanced endoscopy with IMAGE1 S CHROMA improves detection of nonmuscle invasive bladder cancer during transurethral resection. J. Endourol. 35, 647–651 (2021).
Trelles Guzmán, C. R. et al. Randomized clinical trial on the use of IMAGE1 S LIGHT (SPIES) vs. white light in the prevention of recurrence during transurethral resection of bladder tumors: analysis after 12-month follow-up. Actas Urol. Esp. (Engl. Ed.) 48, 512–520 (2024).
Tomiyama, E., Fujita, K., Hashimoto, M., Uemura, H. & Nonomura, N. Urinary markers for bladder cancer diagnosis: a review of current status and future challenges. Int. J. Urol. 31, 208–219 (2024).
Yafi, F. A. et al. Prospective analysis of sensitivity and specificity of urinary cytology and other urinary biomarkers for bladder cancer. Urol. Oncol. 33, 66.e25–e31 (2015).
Reid, M. D., Osunkoya, A. O., Siddiqui, M. T. & Looney, S. W. Accuracy of grading of urothelial carcinoma on urine cytology: an analysis of interobserver and intraobserver agreement. Int. J. Clin. Exp. Pathol. 5, 882–891 (2012).
Sullivan, P. S., Chan, J. B., Levin, M. R. & Rao, J. Urine cytology and adjunct markers for detection and surveillance of bladder cancer. Am. J. Transl. Res. 2, 412–440 (2010).
Wojcik, E. M., Kurtycz, D. F. I. & Rosenthal, D. L. (eds) The Paris System for Reporting Urinary Cytology 2nd edn (Springer, 2022).
Meilleroux, J. et al. One year of experience using the Paris system for reporting urinary cytology. Cancer Cytopathol. 126, 430–436 (2018).
Raitanen, M.-P. et al. Differences between local and review urinary cytology in diagnosis of bladder cancer. an interobserver multicenter analysis. Eur. Urol. 41, 284–289 (2002).
Santoni, G., Morelli, M. B., Amantini, C. & Battelli, N. Urinary markers in bladder cancer: an update. Front. Oncol. 8, 362 (2018).
Dreyer, T. et al. Use of the Xpert Bladder Cancer Monitor urinary biomarker test for guiding cystoscopy in high-grade non-muscle-invasive bladder cancer: results from the randomized controlled DaBlaCa-15 trial. Eur. Urol. 88, 23–30 (2025).
Silva-Ferreira, M. et al. Diagnostic test accuracy of urinary DNA methylation-based biomarkers for the detection of primary and recurrent bladder cancer: a systematic review and meta-analysis. Eur. Urol. Focus 10, 922–934 (2024).
Yang, Z., Song, F. & Zhong, J. Urinary biomarkers in bladder cancer: FDA-approved tests and emerging tools for diagnosis and surveillance. Cancers 17, 3425 (2025).
Chou, R. et al. Urinary biomarkers for diagnosis of bladder cancer: a systematic review and meta-analysis. Ann. Intern. Med. 163, 922–931 (2015).
Harvey, J. C. et al. Analytical validation of Cxbladder® Detect, Triage, and Monitor: assays for detection and management of urothelial carcinoma. Diagnostics 14, 2061 (2024).
O’Sullivan, P. et al. A multigene urine test for the detection and stratification of bladder cancer in patients presenting with hematuria. J. Urol. 188, 741–747 (2012).
Kavalieris, L. et al. A segregation index combining phenotypic (clinical characteristics) and genotypic (gene expression) biomarkers from a urine sample to triage out patients presenting with hematuria who have a low probability of urothelial carcinoma. BMC Urol. 15, 23 (2015).
Kavalieris, L. et al. Performance characteristics of a multigene urine biomarker test for monitoring for recurrent urothelial carcinoma in a multicenter study. J. Urol. 197, 1419–1426 (2017).
Lotan, Y. et al. A multicenter prospective randomized controlled trial comparing Cxbladder triage to cystoscopy in patients with microhematuria: the safe testing of risk for asymptomatic microhematuria trial. J. Urol. 212, 41–51 (2024).
Barocas, D. A. et al. Microhematuria: AUA/SUFU Guideline. J. Urol. 204, 778–786 (2020).
Sanghvi, A. B., Allen, E. Z., Callenberg, K. M. & Pantanowitz, L. Performance of an artificial intelligence algorithm for reporting urine cytopathology. Cancer Cytopathol. 127, 658–666 (2019).
Shao, C.-H. et al. Metabolite marker discovery for the detection of bladder cancer by comparative metabolomics. Oncotarget 8, 38802–38810 (2017).
Lebret, T. et al. Artificial intelligence to improve cytology performances in bladder carcinoma detection: results of the VisioCyt test. BJU Int. 129, 356–363 (2022).
Sokolov, I. et al. Noninvasive diagnostic imaging using machine-learning analysis of nanoresolution images of cell surfaces: detection of bladder cancer. Proc. Natl. Acad. Sci. 115, 12920–12925 (2018).
Compérat, E. & van der Kwast, T. H. Pathological staging of bladder cancer. Diagn. Histopathol. 19, 366–375 (2013).
Tosoni, I. et al. Clinical significance of interobserver differences in the staging and grading of superficial bladder cancer. BJU Int. 85, 48–53 (2000).
Chen, S. et al. Clinical use of machine learning-based pathomics signature for diagnosis and survival prediction of bladder cancer. Cancer Sci. 112, 2905–2914 (2021).
Pan, J. et al. An artificial intelligence model for the pathological diagnosis of invasion depth and histologic grade in bladder cancer. J. Transl. Med. 21, 42 (2023).
Yin, P.-N. et al. Histopathological distinction of non-invasive and invasive bladder cancers using machine learning approaches. BMC Med. Inform. Decis. Mak. 20, 162 (2020).
Shalata, A. T. et al. Precise grading of non-muscle invasive bladder cancer with multi-scale pyramidal CNN. Sci. Rep. 14, 25131 (2024).
Niazi, M. K. K. et al. Semantic segmentation to identify bladder layers from H&E images. Diagn. Pathol. 15, 87 (2020).
Khoraminia, F. et al. Artificial intelligence in digital pathology for bladder cancer: hype or hope? A systematic review. Cancers 15, 4518 (2023).
Pereira, J. F. et al. The perioperative morbidity of transurethral resection of bladder tumor: implications for quality improvement. Urology 125, 131–137 (2019).
Iqbal, U. et al. Upstaging and survival outcomes for non-muscle invasive bladder cancer after radical cystectomy: results from the international robotic cystectomy consortium. J. Endourol. 35, 1541–1547 (2021).
Balbay, M. D. et al. The actual incidence of bladder perforation following transurethral bladder surgery. J. Urol. 174, 2260–2262 (2005).
Breda, A. et al. The DEpth of Endoscopic Perforation scale to assess intraoperative perforations during transurethral resection of bladder tumor: subgroup analysis of a randomized controlled trial. World J. Urol. 41, 2583–2589 (2023).
Nayyar, R., Saini, S., Sharma, A., Kurra, S. & Dogra, P. N. Systemic dissemination of tumor cells during transurethral resection in patients with bladder tumor and its clinical relevance: a follow up study. Urol. Oncol. 39, 497.e17–497.e22 (2021).
Lämmle, M. et al. Reliability of MR imaging-based virtual cystoscopy in the diagnosis of cancer of the urinary bladder. AJR Am. J. Roentgenol. 178, 1483–1488 (2002).
Kulkarni, G. S. et al. Cost-effectiveness analysis of immediate radical cystectomy versus intravesical Bacillus Calmette-Guerin therapy for high-risk, high-grade (T1G3) bladder cancer. Cancer 115, 5450–5459 (2009).
Zhang, N. et al. Diagnostic accuracy of multi-parametric magnetic resonance imaging for tumor staging of bladder cancer: meta-analysis. Front. Oncol. 9, 981 (2019).
Yoshida, S. et al. Diffusion-weighted magnetic resonance imaging in management of bladder cancer, particularly with multimodal bladder-sparing strategy. World J. Radiol. 6, 344–354 (2014).
Wang, H. et al. Multiparametric 3-T MRI for differentiating low-versus high-grade and category T1 versus T2 bladder urothelial carcinoma. AJR Am. J. Roentgenol. 204, 330–334 (2015).
Tekes, A. et al. Dynamic MRI of bladder cancer: evaluation of staging accuracy. AJR Am. J. Roentgenol. 184, 121–127 (2005).
Lai, A. L. & Law, Y. M. VI-RADS in bladder cancer: overview, pearls and pitfalls. Eur. J. Radiol. 160, 110666 (2023).
Del Giudice, F. et al. The accuracy of Vesical Imaging-Reporting and Data System (VI-RADS): an updated comprehensive multi-institutional, multi-readers systematic review and meta-analysis from diagnostic evidence into future clinical recommendations. World J. Urol. 40, 1617–1628 (2022).
Panebianco, V. et al. Clinical application of bladder MRI and the vesical imaging-reporting and data system. Nat. Rev. Urol. 21, 243–251 (2024).
Bryan, R. T. et al. Randomized comparison of magnetic resonance imaging versus transurethral resection for staging new bladder cancers: results from the prospective Bladderpath trial. J. Clin. Oncol. 43, 1417–1418 (2025).
Herr, H. W. Routine CT scan in cystectomy patients: does it change management? Urology 47, 324–325 (1996).
Fuletra, J. G. et al. Can preoperative imaging characteristics predict pT3 bladder cancer following cystectomy? World J. Urol. 39, 1941–1945 (2021).
Eldred-Evans, D. et al. The rapid assessment for prostate imaging and diagnosis (RAPID) prostate cancer diagnostic pathway. BJU Int. 131, 461–470 (2023).
Zhang, X. et al. MRI evaluation of vesical imaging reporting and data system for bladder cancer after neoadjuvant chemotherapy. Cancer Imaging 24, 49 (2024).
Garapati, S. S. et al. Urinary bladder cancer staging in CT urography using machine learning. Med. Phys. 44, 5814–5823 (2017).
Yang, Y., Zou, X., Wang, Y. & Ma, X. Application of deep learning as a noninvasive tool to differentiate muscle-invasive bladder cancer and non-muscle-invasive bladder cancer with CT. Eur. J. Radiol. 139, 109666 (2021).
Zhang, G. et al. Deep learning on enhanced CT images can predict the muscular invasiveness of bladder cancer. Front. Oncol. 11, 654685 (2021).
Ye, Y. et al. Radiomics prediction of muscle invasion in bladder cancer using semi-automatic lesion segmentation of MRI compared with manual segmentation. Bioeng. Basel Switz. 10, 1355 (2023).
Li, J. et al. Predicting muscle invasion in bladder cancer based on MRI: A comparison of radiomics, and single-task and multi-task deep learning. Comput. Methods Prog. Biomed. 233, 107466 (2023).
Gresser, E. et al. Radiomics signature using manual versus automated segmentation for lymph node staging of bladder cancer. Eur. Urol. Focus. 9, 145–153 (2023).
Xu, X. et al. Quantitative identification of nonmuscle-invasive and muscle-invasive bladder carcinomas: a multiparametric MRI radiomics analysis. J. Magn. Reson. Imaging 49, 1489–1498 (2019).
Gharibvand, M. M., Kazemi, M., Motamedfar, A., Sametzadeh, M. & Sahraeizadeh, A. The role of ultrasound in diagnosis and evaluation of bladder tumors. J. Fam. Med. Prim. Care 6, 840–843 (2017).
Islam, M. T. et al. Non-invasive imaging of Young’s modulus and Poisson’s ratio in cancers in vivo. Sci. Rep. 10, 7266 (2020).
Gao, R.-Z. et al. Radiomics analysis based on ultrasound images to distinguish the tumor stage and pathological grade of bladder cancer. J. Ultrasound Med. 40, 2685–2697 (2021).
Weinstein, J. N. et al. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 507, 315–322 (2014).
Sanli, O. et al. Bladder cancer. Nat. Rev. Dis. Primer 3, 1–19 (2017).
Guo, C. C. et al. Molecular profile of bladder cancer progression to clinically aggressive subtypes. Nat. Rev. Urol. 21, 391–405 (2024).
Olislagers, M. et al. Molecular biomarkers of progression in non-muscle-invasive bladder cancer — beyond conventional risk stratification. Nat. Rev. Urol. 22, 75–91 (2025).
Zhang, J., Chen, J., Xu, M. & Zhu, T. Exploring prognostic DNA methylation genes in bladder cancer: a comprehensive analysis. Discov. Oncol. 15, 331 (2024).
Yu, L., Zheng, Y. & Gao, L. MiRNA–disease association prediction based on meta-paths. Brief. Bioinform. 23, bbab571 (2022).
Kluth, L. A. et al. Prognostic and prediction tools in bladder cancer: a comprehensive review of the literature. Eur. Urol. 68, 238–253 (2015).
Fuster, S., Kiraz, U., Eftestøl, T., Janssen, E. A. M. & Engan, K. NMGrad: advancing histopathological bladder cancer grading with weakly supervised deep learning. Bioengineering 11, 909 (2024).
Narayan, V. M. et al. Evaluation of a natural language processing model to identify and characterize patients in the united states with high-risk non-muscle-invasive bladder cancer. JCO Clin. Cancer Inform. 7, e2300096 (2023).
Yang, G. et al. Enhancing recurrence risk prediction for bladder cancer using multi-sequence MRI radiomics. Insights Imaging 15, 88 (2024).
Wang, G.-Y. et al. Prediction of non-muscle invasive bladder cancer recurrence using deep learning of pathology image. Sci. Rep. 14, 18931 (2024).
Schmitz-Dräger, B. J. et al. Results of the prospective randomized UroFollow trial comparing marker-guided versus cystoscopy-based surveillance in patients with low/intermediate-risk bladder cancer. Eur. Urol. Oncol. 8, 1041–1049 (2025).
Reyes, R. M. et al. A randomized feasibility trial comparing surveillance regimens for patients with low and low-intermediate risk non-muscle invasive bladder cancer. Bladder Cancer 7, 285–295 (2021).
Kulkarni, G. S. et al. Propensity score analysis of radical cystectomy versus bladder-sparing trimodal therapy in the setting of a multidisciplinary bladder cancer clinic. J. Clin. Oncol. 35, 2299–2305 (2017).
Woźnicki, P. et al. Radiomics for the prediction of overall survival in patients with bladder cancer prior to radical cystectomy. Cancers 14, 4449 (2022).
Wei, Z. et al. A CT-based deep learning model predicts overall survival in patients with muscle invasive bladder cancer after radical cystectomy: a multicenter retrospective cohort study. Int. J. Surg. 110, 2922–2932 (2024).
Wang, Z. et al. Machine learning-based autophagy-related prognostic signature for personalized risk stratification and therapeutic approaches in bladder cancer. Int. Immunopharmacol. 138, 112623 (2024).
Zhang, Z., Zhang, G. & Kong, C. Targeted inhibition of polo-like kinase 1 by a novel small-molecule inhibitor induces mitotic catastrophe and apoptosis in human bladder cancer cells. J. Cell. Mol. Med. 21, 758–767 (2017).
Li, M., Jiang, Z., Shen, W. & Liu, H. Deep learning in bladder cancer imaging: a review. Front. Oncol. 12, 930917 (2022).
Wei, Z. et al. Development and validation of a CT-based deep learning radiomics nomogram to predict muscle invasion in bladder cancer. Heliyon 10, e24878 (2024).
Moore, N. S., McWilliam, A. & Aneja, S. Bladder cancer radiation oncology of the future: prognostic modelling, radiomics, and treatment planning with artificial intelligence. Semin. Radiat. Oncol. 33, 70–75 (2023).
Thurman, S. A. & DeWeese, T. L. Multimodality therapy for the treatment of muscle-invasive bladder cancer. Semin. Urol. Oncol. 18, 313–322 (2000).
Filon, M. & Schmidt, B. New treatment options for non-muscle-invasive bladder cancer. Am. Soc. Clin. Oncol. 45, e471942 (2025).
Lemiński, A. et al. Combined modality bladder-sparing therapy for muscle-invasive bladder cancer: how (should) we do it? A narrative review. J. Clin. Med. 12, 1560 (2023).
Sylvester, R. J. et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur. Urol. 49, 466–465 (2006). discussion 475–477.
Teoh, J. Y.-C. et al. Recurrence mechanisms of non-muscle-invasive bladder cancer — a clinical perspective. Nat. Rev. Urol. 19, 280–294 (2022).
Chan, V. W.-S. et al. The impact of transurethral en bloc resection of bladder tumour on pathological and oncological outcomes. AME Med. J. 5, 29 (2020).
Lochhead, P. et al. Etiologic field effect: reappraisal of the field effect concept in cancer predisposition and progression. Mod. Pathol. 28, 14–29 (2015).
Raviv, S. et al. Long-term survival after “drop metastases” of renal cell carcinoma to the bladder. Urology 60, 697 (2002).
Yuen-Chun Teoh, J. et al. Transurethral en bloc resection versus standard resection of bladder tumour: a randomised, multicentre, phase 3 trial. Eur. Urol. 86, 103–111 (2024).
Cumberbatch, M. G. K. et al. Repeat transurethral resection in non-muscle-invasive bladder cancer: a systematic review. Eur. Urol. 73, 925–933 (2018).
US Food and Drug Administration. Bacillus Calmette-Guérin-Unresponsive Nonmuscle Invasive Bladder Cancer: Developing Drugs and Biological Products for Treatment. FDA https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bacillus-calmette-guerin-unresponsive-nonmuscle-invasive-bladder-cancer-developing-drugs-and (2024).
Balar, A. V. et al. Pembrolizumab monotherapy for the treatment of high-risk non-muscle-invasive bladder cancer unresponsive to BCG (KEYNOTE-057): an open-label, single-arm, multicentre, phase 2 study. Lancet Oncol. 22, 919–930 (2021).
Tan, W. S. et al. Radiofrequency-induced thermo-chemotherapy effect versus a second course of Bacillus Calmette-Guérin or institutional standard in patients with recurrence of non-muscle-invasive bladder cancer following induction or maintenance Bacillus Calmette-Guérin therapy (HYMN): a phase III, open-label, randomised controlled trial. Eur. Urol. 75, 63–71 (2019).
Pignot, G. et al. Efficacy of hyperthermic intravesical chemotherapy (HIVEC) in patients with non-muscle invasive bladder cancer after BCG failure. World J. Urol. 41, 3195–3203 (2023).
Prip, F. et al. Comprehensive genomic characterization of early-stage bladder cancer. Nat. Genet. 57, 115–125 (2025).
US Food and Drug Administration. Adstiladrin. FDA https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/adstiladrin (2024).
Wong, C. H.-M., Leung, D. K.-W., Gontero, P. & Teoh, J. Y.-C. Gene-mediated therapy for BCG-unresponsive nonmuscle-invasive bladder cancer: mechanisms, clinical evidence, and practical implementation. Curr. Opin. Urol. 35, 666 (2025).
Boorjian, S. A. et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 22, 107–117 (2021).
Narayan, V. M. et al. Efficacy of intravesical nadofaragene firadenovec for patients with Bacillus Calmette-Guérin-unresponsive nonmuscle-invasive bladder cancer: 5-year follow-up from a phase 3 trial. J. Urol. 212, 74–86 (2024).
Daneshmand, S. et al. TAR-200 for Bacillus Calmette-Guérin-unresponsive high-risk non-muscle-invasive bladder cancer: results from the phase IIb SunRISe-1 study. J. Clin. Oncol. 43, 3578–3588 (2025).
U.S. FDA approval of INLEXZOTM (gemcitabine intravesical system) set to transform how certain bladder cancers are treated. JNJ.com https://www.jnj.com/media-center/press-releases/u-s-fda-approval-of-inlexzo-gemcitabine-intravesical-system-set-to-transform-how-certain-bladder-cancers-are-treated (2025).
Kamoun, A. et al. A consensus molecular classification of muscle-invasive bladder cancer. Eur. Urol. 77, 420–433 (2020).
van Ginkel, N. et al. Survival outcomes of patients with muscle-invasive bladder cancer according to pathological response at radical cystectomy with or without neo-adjuvant chemotherapy: a case-control matching study. Int. Urol. Nephrol. 54, 3145–3152 (2022).
Li, D. et al. Efficiency of transurethral en-bloc resection vs. conventional transurethral resection for non-muscle-invasive bladder cancer: an umbrella review. Cancer Med. 13, e7323 (2024).
Bandini, M. et al. Does the administration of preoperative pembrolizumab lead to sustained remission post-cystectomy? First survival outcomes from the PURE-01 study☆. Ann. Oncol. 31, 1755–1763 (2020).
Necchi, A. et al. Updated results of PURE-01 with preliminary activity of neoadjuvant pembrolizumab in patients with muscle-invasive bladder carcinoma with variant histologies. Eur. Urol. 77, 439–446 (2020).
Basile, G. et al. Neoadjuvant pembrolizumab and radical cystectomy in patients with muscle-invasive urothelial bladder cancer: 3-year median follow-up update of PURE-01 trial. Clin. Cancer Res. 28, 5107–5114 (2022).
Miyagi, H. et al. Predicting complete response to neoadjuvant chemotherapy in muscle-invasive bladder cancer. Cancers 15, 168 (2022).
Apolo, A. B. et al. Adjuvant pembrolizumab versus observation in muscle-invasive urothelial carcinoma. N. Engl. J. Med. 392, 45–55 (2025).
Galsky, M. D. et al. Adjuvant nivolumab in high-risk muscle-invasive urothelial carcinoma: expanded efficacy from checkmate 274. J. Clin. Oncol. 43, 15–21 (2025).
Jackson-Spence, F. et al. IMvigor011: a study of adjuvant atezolizumab in patients with high-risk MIBC who are ctDNA+ post-surgery. Future Oncol. 19, 509–515 (2023).
Powles, T. et al. ctDNA-guided adjuvant atezolizumab in muscle-invasive bladder cancer. N. Engl. J. Med. 393, 2395–2408 (2025).
Lemberger, U. et al. Alterations in DNA damage repair genes before and after neoadjuvant cisplatin-based chemotherapy in muscle-invasive bladder cancer. Eur. Urol. Open. Sci. 71, 38–48 (2025).
Iyer, G. et al. DNA damage response alterations predict for neoadjuvant chemotherapy sensitivity in muscle-invasive bladder cancer: a correlative analysis of the SWOG S1314 trial. JCO Precis. Oncol. 8, e2400287 (2024).
von der Maase, H. et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J. Clin. Oncol. 18, 3068–3077 (2000).
Bellmunt, J. et al. Prognostic factors in patients with advanced transitional cell carcinoma of the urothelial tract experiencing treatment failure with platinum-containing regimens. J. Clin. Oncol. 28, 1850–1855 (2010).
Powles, T. et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet 391, 748–757 (2018).
Rosenberg, J. E. et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387, 1909–1920 (2016).
Nadal, R., Valderrama, B. P. & Bellmunt, J. Progress in systemic therapy for advanced-stage urothelial carcinoma. Nat. Rev. Clin. Oncol. 21, 8–27 (2024).
Klümper, N. et al. Membranous NECTIN-4 expression frequently decreases during metastatic spread of urothelial carcinoma and is associated with enfortumab vedotin resistance. Clin. Cancer Res. 29, 1496–1505 (2023).
Duan, X. et al. First-in-human study of the radioligand 68Ga-N188 targeting nectin-4 for PET/CT imaging of advanced urothelial carcinoma. Clin. Cancer Res. 29, 3395–3407 (2023).
Aslanova, M., Yu, E.-M. & Aragon-Ching, J. B. The rapidly changing treatment landscape of first-line advanced urothelial cancer (aUC) or metastatic urothelial cancer (mUC). Explor. Target. Antitumor Ther. 5, 971–980 (2024).
Balar, A. V. et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): a multicentre, single-arm, phase 2 study. Lancet Oncol. 18, 1483–1492 (2017).
Chou, J. et al. TROP2 expression across molecular subtypes of urothelial carcinoma and enfortumab vedotin-resistant cells. Eur. Urol. Oncol. 5, 714–718 (2022).
Powles, T. et al. Sacituzumab govitecan in advanced urothelial carcinoma: TROPiCS-04, a phase III randomized trial. Ann. Oncol. 36, 561–571 (2025).
FDA grants accelerated approval to sacituzumab govitecan for advanced urothelial cancer. FDA https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-sacituzumab-govitecan-advanced-urothelial-cancer (2024).
Funda, M.-B. et al. Datopotamab deruxtecan (Dato-DXd) in locally advanced/metastatic urothelial cancer: updated results from the phase 1 TROPIONPanTumor01 study. J. Clin. Oncol. https://doi.org/10.1200/JCO.2025.43.5_suppl.663 (2025).
Sheng, X. et al. Open-label, multicenter, phase II study of RC48-ADC, a HER2-targeting antibody-drug conjugate, in patients with locally advanced or metastatic urothelial carcinoma. Clin. Cancer Res. 27, 43–51 (2021).
Meric-Bernstam, F. et al. Efficacy and safety of trastuzumab deruxtecan in patients with HER2-expressing solid tumors: primary results from the DESTINY-PanTumor02 phase II trial. J. Clin. Oncol. 42, 47–58 (2024).
Rebuzzi, S. E. et al. Prognostic and predictive factors in advanced urothelial carcinoma treated with immune checkpoint inhibitors: a review of the current evidence. Cancers 13, 5517 (2021).
Fornarini, G. et al. A novel immunotherapy prognostic score for patients with pretreated advanced urinary TrAct CArcinoma from the subgroup analysis of the SAUL study: the ITACA Score. Minerva Urol. Nephrol. 75, 308–318 (2023).
Loriot, Y. et al. Erdafitinib in locally advanced or metastatic urothelial carcinoma. N. Engl. J. Med. 381, 338–348 (2019).
Loriot, Y. et al. Erdafitinib or chemotherapy in advanced or metastatic urothelial carcinoma. N. Engl. J. Med. 389, 1961–1971 (2023).
Wu, S. et al. Liquid biopsy in urothelial carcinoma: detection techniques and clinical applications. Biomed. Pharmacother. Biomed. Pharmacother. 165, 115027 (2023).
Crabb, S. J. et al. A randomized, double-blind, biomarker-selected, phase ii clinical trial of maintenance poly ADP-ribose polymerase inhibition with rucaparib following chemotherapy for metastatic urothelial carcinoma. J. Clin. Oncol. 41, 54–64 (2023).
Laurie, M. A. et al. Bladder cancer and artificial intelligence: emerging applications. Urol. Clin. North. Am. 51, 63–75 (2024).
Kwong, J. C. C. et al. Predicting non-muscle invasive bladder cancer outcomes using artificial intelligence: a systematic review using APPRAISE-AI. NPJ Digit. Med. 7, 1–11 (2024).
Cha, K. H. et al. Urinary bladder segmentation in CT urography using deep-learning convolutional neural network and level sets. Med. Phys. 43, 1882–1896 (2016).
Kwong, J. C. C. et al. APPRAISE-AI tool for quantitative evaluation of AI studies for clinical decision support. JAMA Netw. Open. 6, e2335377 (2023).
He, C. et al. The accuracy and quality of image-based artificial intelligence for muscle-invasive bladder cancer prediction. Insights Imaging 15, 185 (2024).
Wei, J. et al. A semi-supervised multi-region segmentation framework of bladder wall and tumor with wall-enhanced self-supervised pre-training. Bioengineering 11, 1225 (2024).
Wadden, J. J. Defining the undefinable: the black box problem in healthcare artificial intelligence. J. Med. Ethics 48, 764–768 (2022).
Truhn, D. et al. Encrypted federated learning for secure decentralized collaboration in cancer image analysis. Med. Image Anal. 92, 103059 (2024).
Del Giudice, F. et al. Prospective assessment of vesical imaging reporting and data system (VI-RADS) and its clinical impact on the management of high-risk non-muscle-invasive bladder cancer patients candidate for repeated transurethral resection. Eur. Urol. 77, 101–109 (2020).
Powles, T. et al. Pembrolizumab for advanced urothelial carcinoma: exploratory ctDNA biomarker analyses of the KEYNOTE-361 phase 3 trial. Nat. Med. 30, 2508–2516 (2024).
Kamat, A. M. et al. Evidence-based assessment of current and emerging bladder-sparing therapies for non-muscle-invasive bladder cancer after Bacillus Calmette-Guerin therapy: a systematic review and meta-analysis. Eur. Urol. Oncol. 3, 318–340 (2020).
Gaba, F. et al. Tumour factors and the variation in non-muscle invasive bladder cancer recurrence after transurethral resection surgery between sites: results from the RESECT study. J. Clin. Oncol. 41, 4510–4510 (2023).
Brancato, V. et al. Standardizing digital biobanks: integrating imaging, genomic, and clinical data for precision medicine. J. Transl. Med. 22, 136 (2024).
Daneshmand, S. et al. Efficacy and safety of blue light flexible cystoscopy with hexaminolevulinate in the surveillance of bladder cancer: a phase III, comparative, multicenter study. J. Urol. 199, 1158–1165 (2018).
Chou, R. et al. Urinary biomarkers for diagnosis of bladder cancer. Ann. Intern. Med. 163, 922–931 (2015).
Sharma, G., Sharma, A., Krishna, M., Ahluwalia, P. & Gautam, G. Diagnostic performance of minichromosome maintenance 5 (MCM5) in bladder cancer: a systematic review and meta-analysis. Urol. Oncol. Semin. Orig. Investig. 40, 235–242 (2022).
Cai, Q. et al. Urine BLCA-4 exerts potential role in detecting patients with bladder cancers: a pooled analysis of individual studies. Oncotarget 6, 37500–37510 (2015).
Hirasawa, Y. et al. Diagnostic performance of Oncuria™, a urinalysis test for bladder cancer. J. Transl. Med. 19, 141 (2021).
Babu, S. et al. Keratin 17 is a sensitive and specific biomarker of urothelial neoplasia. Mod. Pathol. 32, 717–724 (2019).
Vasdev, N. et al. The role of URO17™ biomarker to enhance diagnosis of urothelial cancer in new hematuria patients — first European data. BJUI Compass 2, 46–52 (2021).
Davis, N. et al. A novel urine-based assay for bladder cancer diagnosis: multi-institutional validation study. Eur. Urol. Focus. 4, 388–394 (2018).
Shang, D., Liu, Y., Xu, X., Chen, Z. & Wang, D. Diagnostic value comparison of CellDetect, fluorescent in situ hybridization (FISH), and cytology in urothelial carcinoma. Cancer Cell Int. 21, 1–7 (2021).
Cowan, B. et al. Longitudinal follow-up and performance validation of an mRNA-based urine test (Xpert® Bladder Cancer Monitor) for surveillance in patients with non-muscle-invasive bladder cancer. BJU Int. 128, 713–721 (2021).
Witjes, J. A. et al. Performance of the bladder EpiCheck™ methylation test for patients under surveillance for non-muscle-invasive bladder cancer: results of a multicenter, prospective, blinded clinical trial. Eur. Urol. Oncol. 1, 307–313 (2018).
van Kessel, K. E. M. et al. Validation of a DNA methylation-mutation urine assay to select patients with hematuria for cystoscopy. J. Urol. 197, 590–595 (2017).
Springer, S. U. et al. Non-invasive detection of urothelial cancer through the analysis of driver gene mutations and aneuploidy. eLife 7, e32143 (2018).
Wong, R. & Rosser, C. J. UroSEEK gene panel for bladder cancer surveillance. Transl. Androl. Urol. 8, S546–S54S549 (2019).
Salari, K. et al. Development and multicenter case–control validation of urinary comprehensive genomic profiling for urothelial carcinoma diagnosis, surveillance, and risk-prediction. Clin. Cancer Res. 29, 3668–3680 (2023).
Chamie, K. et al. Final clinical results of pivotal trial of IL-15RαFc superagonist N-803 with BCG in BCG-unresponsive CIS and papillary nonmuscle-invasive bladder cancer (NMIBC). J. Clin. Oncol. 40, 4508–4508 (2022).
Tyson, M. D. et al. P2-02 pivotal results from bond-003: a phase 3, single-arm study of intravesical cretostimogene grenadenorepvec for the treatment of high risk, BCG-unresponsive non-muscle invasive bladder cancer with carcinoma in situ. J. Urol. https://doi.org/10.1097/01.JU.0001015816.87470.c9.02 (2024).
Author information
Authors and Affiliations
Contributions
H.A.-S., H.D., O.O., A.G., G.L.B., S.W., J.W., J.T., N.V. and S.A. researched data for the article. H.A.-S., H.D., O.O., S.O., G.L.B., J.T. and S.A. contributed substantially to discussion of the content. H.A.-S., H.D., O.O., S.O., A.G., G.L.B., J.W., J.T. and S.A. wrote the article H.A.-S., H.D., S.O., A.G., G.L.B., S.W., A.H., J.W., J.T. N.V., E.E. S.B. and S.A. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
G.L.B. reports receiving personal fees from advisory boards for Accord, AstraZeneca, Amgen and Merck; and from speaker bureaus for Astellas, AstraZeneca, Amgen, Bayer, Merck, Novartis and Pfizer. G.L.B. holds four patents with ST Microelectronics and has received travel and accommodation support for scientific conferences from Accord, Merck and Janssen. J.T. reports receiving honoraria for lectures from Astellas, Boston Scientific, Combat Medical, Ferring, Ipsen, Janssen, Olympus and Sanofi; consultancy or advisory board fees from Astellas, Aulea Medical, CMR Robotics, Combat Medical, EQT, Ferring, Illumicell AI, Janssen, MRI PRO, MedTech Syndicates, Merck Sharp & Dohme, Phase Scientific and Procept BioRobotics; and research grant support from Baxter, Bristol-Myers Squibb, Ferring, Janssen, Merck Sharp & Dohme, Karl Storz and Olympus. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Urology thanks Shahrokh F. Shariat, Morgan Roupret and Benjamin Pradere for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Al-Sattar, H., Ding, H., Okoli, O. et al. A multi-modal approach for decision making in bladder cancer. Nat Rev Urol (2026). https://doi.org/10.1038/s41585-025-01122-7
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41585-025-01122-7